Abstract
A simple and fast method for the detection of aldehydes by headspace solid-phase microextraction (HS-SPME) based on nanostructure polypyrrole film coupled to ion mobility spectrometery (IMS) is described. The detection of aldehydes (pentanal, hexanal and heptanal) has been successfully accomplished using in situ chemical derivatization with dibutylamine as the derivatization reagent and IMS. The simultaneous and rapid detection of aldehydes is important, since elevated level of aldehydes is considered as the biomarker of different diseases. The highlight of this method was that it involved an amine nucleophilic addition reaction (which was often considered as Mannich reaction) in gas phase for enhancing IMS sensitivity of aldehydes. Dibutylamine was used as the derivatization reagent driven into the cell by using a syringe pump. The calibration graphs were linear in the range of 2.0–50.0 μg mL−1 with R 2 ≥ 0.99 in aqueous solutions and limit of detections were determined ≤ 1.8 μg mL−1. The RSD% values of the aldehydes determination was ≤ 8 %. Here we have demonstrated that other amine types (butylamine and tributylamine) can derivatize aldehydes and significantly improve the IMS sensitivity of tagged analytes. However, IMS spectrum is complicated by presence of the different product ions in the process.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Aldehydes receive an increasingly considerable concern due to their potential adverse health effects and environmental prevalence; various aldehydes can be recognized as biomarkers of cancer diseases [1]. The toxicity of low molecular aldehydes to humans and animals is well known as a result [2]. There are many possible aldehydes sources for humans, such as aldehydes of environmental or occupational concerns, dietary and formed endogenously aldehydes by intermediary metabolism, and drugs that are aldehydes or that form reactive aldehyde metabolites that cause side-effect toxicity [3]. The analysis of exhaled breath has been used to noninvasively obtain information about inflammatory processes within the lung. The exhaled breath contains a complex mixture of volatile organic compounds (VOCs), which can be detected using different methods. The development of highly sensitive and selective techniques with trace-level detection and unequivocal analyte identification capabilities is becoming increasingly desirable in analytical chemistry [4]. In recent years, a range of spectrometric techniques have been deployed to investigate human breath with studies being initiated into different diseases, infections, cancer and more general point of care and intensive care applications [5–12].
As a result, the analysis of aldehydes in biological samples (mainly blood) has obtained much attention due to their directly being related to the internal activities [1]. Many methods for the detection of aldehyde in samples have been reported, such as fluorometry, spectrophotometry, high-performance liquid chromatography (HPLC), gas chromatography, headspace-solid phase microextraction gas chromatography–mass spectrometry (HS-SPME- GC–MS) [2, 13–17] and ion mobility spectrometry (IMS) [18]. Rapid determination of different compounds by IMS devices has nowadays become a routine technique [19]. This fast and low-cost method has become an ideal technique for the fast and sensitive detection of trace quantities of volatile organic and inorganic compounds such as: explosives [20–22], drugs [23, 24], metal [25], biological compound [26–29], and volatile environmental pollutants[30–32], VOCs obtained from bronchoscopic sampling of breath [33]. Breath samples taken within the lung during bronchoscopy and to search for significant differences between signals arising from the tumor site compared to those from the collateral lung [33]. Two different types of IMS, such coupled to multi-capillary columns (MCC/IMS) [34–37] and differential mobility spectrometers [12, 38, 39] were reported.
Headspace solid phase microextraction (HS-SPME) with sampling from the headspace above the sample can especially offer a high potential in biomedical analysis, for producing neat spectra and protecting the fiber from irreversible damages by non-volatile concomitants present in the biological matrix [40].
In our study, we were able to extract aldehydes from headspace of solution by nanostructure polypyrrole as solid phase microextraction fiber and detected with IMS at atmospheric pressure after in situ chemical derivatization with dibutylamine as the derivatization reagent in gas phase. Dibutylamine was driven into the cell by using a syringe pump. Experimental conditions, like extraction temperature and time, solution pH, pump speed were optimized firstly.
Experimental
Chemicals and reagents
Pyrrole (Fluka, Switzerland) was distillated and stored in a refrigerator in dark prior to use. Sodium dodecylbenzenesulfonate (SDBS) was used as dopant from Aldrich. The stock solution of aldehydes and amines from Merck (Darmstadt, Germany) at a concentration of 1000 mg L−1 was prepared by dissolving appropriate amounts of each reagent in methanol (analytical grade). These standards were stored at 4 °C and were used for the preparation of working standard solutions. All other chemicals and reagents were of analytical grade purchased from Merck (Darmstadt, Germany).
Apparatus
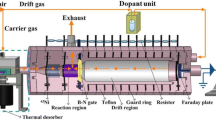
The SPME holder for manual sampling was obtained from Azar Electrode (Ourumieh, Iran). A magnetic stirrer (Heidolph MR 3001 K) and a 2 mm × 5 mm stirring bar were used to stir the solution. A laboratory made circulating cell was used to keep stable the temperature of solutions and SPME fiber, respectively. All pH measurements were performed at 25 ± 0.1 °C by use of a pH meter (Metrohm 713, Switzerland) with a standard uncertainty of 0.1 mV. In all experiments, amine solutions (in methanol) directly infused into the injection port of IMS using a syringe pump at a steady state pumping speed (40 μL min−1) high enough to meet optimum conditions. Electrochemical polymerization of pyrrole was carried out using Behpajuh potentiostat (BHP 2061-C model, Isfahan, Iran). Detection and quantification of aldehydes were carried out using an ion mobility spectrometer (IMS). The IMS (model 1000) used in this study was constructed in Isfahan University of Technology, Iran. The scanning electron micrographs of the fiber surface were obtained using a model S4160C Hitachi (Japan).
Fiber coating
Polypyrrole film doped with DBS (PPy-DBS) as solid phase was prepared electrochemically using a three-electrode system. PPy-DBS film was directly electrodeposited on the surface of a platinum wire (as working electrode) from an aqueous solution containing pyrrole (0.1 mol L−1) and SDBS (7 × 10−3 mol L−1) by the application of a constant potential of +1.1 V versus Ag/AgCl electrode for 300 s. Prior to electropolymerization, the solution was deoxygenated by purging with N2 for 10 min, and the platinum wire surface was cleaned in acetone and methanol using ultrasonic bath for 10 min and was subsequently washed with distilled water.
Results and discussion
Sampling and spectral behavior of aldehydes
The surface characteristics of the polymer film were investigated by scanning electron microscopy (SEM). Figure 1 shows the micrographs of the PPy-DBS fiber at 35000 fold magnification. One clearly observes that the deposited polymers containing the spherical shape with particle size between 90 to 350 nm.
Figure 2 shows IMS spectra of aldehydes sampling by PPy-DBS fiber from the headspace above the sample solution. In the IMS spectral analysis of aldehydes, the instability of protonated aldehyd groups can pose significant problems in IMS analysis, affording a large number of pseudomolecular ions having different drift time values, resulting in highly complex spectra and decreased sensitivity (Fig. 2).
The presence of dibutylamine as derivatization reagent resulted in improvement in ion intensity and the substantial spectral simplification possibly by weakening of ion interactions and the subsequent stability of analyte ions in IMS cell. The detection of aldehydes (pentanal, hexanal and heptanal) has been successfully accomplished using in situ chemical derivatization with dibutylamine. Dibutylamine was used as the derivatization reagent driven into the cell by using a syringe pump. The enhancement sensitivity of IMS in positive ion mode toward compounds including nitrogen functional groups is due to high proton affinity of these groups. One of simple reaction that can be useful for aldehyde derivatization is amine nucleophilic addition reactions. Most reactions of aldehydes involve the carbonyl oxygen and are, in the first instance, addition reactions. Primary and secondary amines are especially suited to the study of the addition reactions of aldehydes, since they contain easily detachable (“active”) hydrogen atoms. In most cases the first recognizable step is addition of the amine to the carbonyl group of the aldehyde, in the sense of the first phase of the amine nucleophilic addition reactions to aldehydes (1):
In the majority of cases, the hydroxy amine reacts further, in one of several ways. It may, for example, lose water, to give an imine (Schiff base); this corresponds to the “crotonaldehyde stage” of the Mannich condensation (2):
Alternately, the hydroxy amine may react further with one or the other of the reagents or with itself; or the Schiff base may be involved in the various subsequent reactions which are encountered, and which often lead to the formation of complex reaction products. The nature of the amine is the determining factor, even more frequently than the experimental conditions, for example tertiary amines bear no “active” hydrogen; they seldom enter into reaction with aldehydes [41].
The mobility of an ion in electric fields in IMS drift tubes depends on the inverse of both its collision cross section and the square root of its reduced mass. Eiceman et al. reported that mobility resolution of ions in IMS and IMS/MS experiments can be enhanced through chemical modification of the supporting gas atmosphere [42, 43]. For example, protonated ammonia and hydrazines form complexes with ketones and the differences in masses and mobilities of the resulting ions are sufficient for separation in an ion mobility spectrometer at ambient pressure. Morrison and Howard also reported, that a protonated hydrazone is observed under chemical ionization conditions in a drift tube attached to a mass spectrometer in the reaction of protonated hydrazone with aldehydes and ketones [44].
The effect of butylamine (primary amine) as derivatization reagent on the sensitivity and spectra simplification of selected aldehydes was also studied. The results showed that sensitivities were practically constant at the same experimental conditions with dibutylamine, whereas IMS spectra complicated due to the formation of unwanted side products. Clear, sensitive and simplified spectra were generated by using dibutylamine as derivatization reagent.
The concentration dibutylamine in IMS cell are directly proportional to the derivatization solution flow rate. By increasing the pumping speed, the concentration of protonated amine in the IMS is increased until proton exchange reaction between reactant ions and analyte reach to equilibrium in the ionization source at a steady state condition under a constant temperature. At high pump speeds may lead to blackout corona in the ionization chamber. So we obtained 40 μL min−1 as the optimized flow rate of dibutylamine (Fig. 3A). At this flow rate aldehyed-amine have maximum intensities. Ion mobility spectrum of different aldehyed-amine prodcts is shown in Fig. 3B.
A Effect of pump speed on the HS-SPME for 2 μg mL−1 aldehyde IMS peak height after in situ chemical derivatization with dibutylamine in gas phase; sample volume: 5 mL, stirring rate: 300 rpm, extraction time: 20 min, extraction temperature: 40 °C, NaCl: 2.0 mol L−1 , dibutylamine concentration: 150 μg mL−1 in methanol, pump speed 40: μL min−1, pentanal-amine (○), hexanal-amine (Δ) and heptanal-amine (□). B The ion mobility spectrum obtained by HS-SPME using a PPy-DBS film from headspace of 2 μg mL−1 of pentanal (a), hexanal(b), heptanal (c) and mixed solution (d) after in situ chemical derivatization with dibutylamine in gas phase
For identification purposes, reduced mobility (K0) is often used instead of drift time. Reduced mobilities are calculated for positive ion mode using ammonium as the calibrant. The reduced mobility is normalized to a standard pressure (760 Torr) and temperature (273 K). Reduced mobility can be calculated using Eq. (3), where K0 is the reduced mobility with the units of (cm2 V−1 s−1), and td is the drift time. The regression equation of the mass-to-mobility correlation curve for corona discharge IMS was shown in Eq. (4):
The peaks observed in the corona discharge IMS spectra can be assigned to ions formed by hydrogen abstraction and clustering with water [45, 46]. Therefore, in Eq. (4) m is the mass of product ions. The optimized instrumental conditions for obtaining the IMS spectra of the aldehydes and its products are listed in Table 1. The drift times and reduced mobility ions together with their corresponding mass-to-mobility are given in Table 2.
Optimization of HS-SPME conditions for aldehydes determination
To optimize the method all the extractions were performed from the headspace of 5 mL water solution spiked with appropriate amount of standard aldehydes solution (2 μg mL−1). The effects of the main extraction parameters, such as the temperature, sample pH, extraction time and ionic strength of sample were studied and optimized. The maximum of peak height of analyte was used as the IMS response to evaluate the extraction efficiency under different conditions. The optimum extraction time was determined by varying the exposure time of the fiber in the headspace of 2 μg mL−1 aldehydes in aqueous solution from 10 to 45 min. It is generally accepted that the reduction of the diffusion layer is essential in order to reach equilibrium faster, which is easily achieved by sample agitation. Therefore, magnetic stirring was applied during the extraction step; Fig. 4a illustrates the extraction time profile.
Effect of extraction time (a), extraction temperature (b) and sample pH (c) on the HS-SPME-IMS for 2 μg mL−1 of pentanal (○), hexanal (Δ) and heptanal (□) after in situ chemical derivatization with dibutylamine in gas phase; sample volume: 5 mL; stirring rate: 300 rpm, extraction temperatures: 40 °C (in a, c), extraction time: 20 min (in b, c), extraction pH: 7 (in a, b) NaCl: 2 mol L−1 (a, b, c)
In HS-SPME approach temperature has a profound effect on extraction efficiency. It influences the rates of mass transfer and the partition coefficients of the analyte by increasing the Henry’s constant and vapor pressure of analyte. Consequently, the concentration of analyte in the headspace and fiber coating would be increased. But temperature has two effect, one of them is increase concentration of sample in head space and another decries adsorption of sample in high temperature because of adsorption is exothermic processes, then we should optimized it in point that first phenomenon’s be in high and second be in low amount. The extraction ability enhances with increasing temperature (Fig. 4b). Thus, the sample temperature was held at 40 °C for subsequent analysis.
To consider the effect of pH on the extraction, the PPy-DBS fiber was exposed to the headspace over the stirring liquid samples for 20 min with the same concentration in the 2 μg mL−1 level at different pH values (2.0–10.0). Observed values in Fig. 4c have revealed that sample pH has an important influence on the form of the extracted analyte on the PPy-DBS fiber, and have the best extraction efficiency in pH 7. SPME methods can be optimized by altering the ionic strength of the matrix. Typically, analyte solubility decreases as ionic strength increases (salting-out effect). A decrease in analyte solubility improve sensitivity by promote analyte partitioning into the fiber. The influence of ionic strength of the sample solution on the HS-SPME effectiveness was evaluated in the range of 0–4 mol L−1 of NaCl concentration. The best results obtained for an aqueous sample containing 2 mol L−1 NaCl, therefore, all further extractions were conducted with 2 mol L−1 NaCl added.
The spiked water sample was employed to investigate the linearity, repeatability and limit of detection under the optima experimentally conditions. Main figures of merit were established under optimized conditions for HS-SPME of aldehydes in water samples. Best analytical characteristics were obtained with pH 7, extraction time of 20 min and sample containing 2 mol L−1 of NaCl at temperature of 40 °C, respectively. In order to assess the repeatability, peak height of three replicate analysis for 2 μg mL−1 adehydes were used and expressed as relative standard deviation (RSD%). The RSD% for aldehyde determination was ≤ 8 %. The calibration graphs was linear in the range of 2–50 μg mL−1 with R 2 ≥ 0.99 in aqueous solution. The limit of detections was determined ≤ 1.8 μg mL−1 at a signal-to-noise ratio (S/N) of 3. Result of HS-SPME–IMS analysis for spiked water samples is displayed in Table 3.
Conclusion
In this work, a new method for the analysis of aldehydes was proposed using dibutylamine as the derivatization reagent for simultaneous determination of pentanal, hexanal and heptanal by HS-SPME-IMS. This method represents several advantages such as, in situ chemical derivatization in gas phase, enhancing sensitivity and successfully simplified IMS spectra. Regarding sensitivity, repeatability, time of analysis among other characteristics, the proposed method was shown to be adequate to evaluate aldehyde levels in water samples without any further pre-treatment.
References
Ma J, Xiao R, Li J, Shi B, Liang Y, Lu W, Chen L (2011) Headspace solid-phase microextraction withon-fiber derivatization for the determination of aldehydes in algae by gas chromatography–mass spectrometry. J Sep Sci 34:1477–1483
Kim H, Shin H (2011) Simple derivatization of aldehydes with d-cysteine and their determination in beverages by liquid chromatography–tandem mass spectrometry. Anal Chim Acta 702:225–232
Poli D, Goldonia M, Corradi M, Acampaa O, Carbognani P, e Internullo E, Casalini A, Mutti A (2010) Determination of aldehydes in exhaled breath of patients with lung cancer by means of on-fiber-derivatisation SPME–GC/MS. J Chromatogr B 878:2643–2651
Garrido-Delgado R, Mercader-Trejo F, Arce L, Valcrcel M (2011) Enhancing sensitivity and selectivity in the determination of aldehydes in olive oil by use of a Tenax TA trap coupled to a UV-ion mobility spectrometer. J Chromatogr A 1218:7543–7549
Corradi M, Rubinstein I, Andreoli R, Manini P, Caglieri A, Poli D, Alinovi R, Mutti A (2003) Aldehydes in exhaled breath condensate of patients with chronic obstructive pulmonary disease. Am J Respir Crit Care Med 167:1380–1386
Ruzsanyi V et al (2005) Detection of human metabolites using multi-capillary columns coupled to ion mobility spectrometers. J Chromatogr A 1084:145–151
Phillips M et al (2007) Prediction of lung cancer using volatile biomarkers in breath. Canc Biomarkers 3:95–109
Davies AN, Baumbach JI (2008) Early lung cancer diagnostics by ion mobility spectrometry data handling. Spectrosc Eur 20:18–21
Westhoff M et al (2009) Ion mobility spectrometry for the detection of volatile organic compounds in exhaled breath of patients with lung cancer: results of a pilot study. Thorax 64:744–748
Ligor M et al (2009) Determination of volatile organic compounds in exhaled breath of patients with lung cancer using solid phase microextraction and gas chromatography mass spectrometry. Clin Chem Lab Med 47:550–560
Fuchs P, Loeseken C, Schubert JK, Miekisch W (2010) Breath gas aldehydes as biomarkers of lung cancer. Int J Cancer 126:2663–2670
Basanta M et al (2010) Non-invasive metabolomic analysis of breath using differential mobility spectrometry in patients withchronic obstructive pulmonary disease and healthy smokers. Analyst 135:315–320
Hyun-Ji K, Ho-Sang S (2011) Simple and automatic determination of aldehydes and acetone in water by headspace solid-phase microextraction and gas chromatography–mass spectrometry. J Sep Sci 34:693–699
Lopez-Vazquez C, Orriols I, Perello M, de Revel G (2011) Determination of aldehydes as pentafluorobenzyl derivatives in grape pomace distillates by HS-SPME-GC/MS. Food Chem 130:1127–1133
Catherine Tessinia C, Niels Müllerb N, Claudia Mardonesa C, Dietrich Meierc D, Alex Bergb A, Baera D (2012) Chromatographic approaches for determination of low-molecular massaldehydes in bio-oil. J Chromatogr A 1219:154–160
Eugenia Banos C, Silva M (2010) Liquid chromatography–tandem mass spectrometry for the determination of low-molecular mass aldehydes in human urine. J Chromatogr B 878:653–658
Ma J, Xiao R, Li J, Zhu S, Liang Y, Lv L (2011) Determination of Aldehydes in Diatoms by Headspace Solid-Phase Microextraction Coupled with GC–MS. J Chromatogr Sci 49:15–19
Ruzsanyi V, Sielemann S, Baumbach JI (2002) determination of VOC in human breath using IMS. I J IMS 3:45–48
Mohammadi A, Ameli A, Alizadeh N (2009) Headspace solid-phase microextraction using a dodecylsulfate-doped polypyrrole film coupled to ion mobility spectrometry for the simultaneous determination of atrazine and ametryn in soil and water samples. Talanta 78:1107–1114
Tabrizchi M, Ilbeigi V (2010) Detection of Explosives by Positive Corona Discharge Ion Mobility Spectrometry. J Hazard Mater 176:692–696
Guerra P, Lai H, Almirall JR (2008) Analysis of the volatile chemical markers of Explosives using novel solid phase microextractioncoupled to ion mobility spectrometry. J Sep Sci 31:2891–2898
Lai H, Leung A, Magee M, Almirall JR (2010) Identification of volatile chemical ignatures from plastic explosives by SPME-GC/MS and detection by ion mobility spectrometry. J Anal Bioanal Chem 396:2997–3007
Ameli A, Alizadeh N (2011) Nanostructured conducting molecularly imprinted polymer for selective extraction of salicylate from urine and serum samples by electrochemically controlled solid-phase micro-extraction. Anal Chim Acta 707:62–68
Gura S, Guerra P, Lai H, Almirall JR (2009) Enhancement in sample collection for the Detection of MDMA using a novel planar SPME (PSPME) device coupled to ion mobility spectrometry (IMS). Drug Test Anal 1:355–363
Shahdousti P, Alizadeh N (2011) Headspace-solid phase microextraction of selenium (IV) from human blood and water samples using polypyrrole film and analysis with ion mobility Spectrometry. Anal Chim Acta 684:67–71
Colgrave ML, Bramwell CJ, Creaser CS (2003) Nanoelectrospray ion mobility spectrometry and ion trap massspectrometry studies of the non-covalent complexes of amino acids and peptides with polyethers. Int J Mass Spectrom 229:209–216
Beegle LW, Kanik I (2001) Electrospray ionization high-resolution ion mobility spectrometry for the detection of organic compounds, 1. amino acids. Anal Chem 73:3028–3034
Steiner WE, Clowers BH, Hill HH Jr (2003) Rapid separation of phenylthiohydantoin amino acids:ambient pressure ion-mobility mass spectrometry (IMMS). Anal Bioanal Chem 375:99–102
Hashemian Z, Mardihallaj A, Khayamian T (2010) analysis of biogenic amines using corona discharge ion mobility spectrometry. Talanta 81:1081–1087
Tiebe C, Miessner H, Koch B, Hübert T (2009) Detection of microbial volatile organic compounds (MVOCs) by ion-mobility spectrometry. J Anal Bioanal Chem 395:2313–2323
Sabo M, Matejcik S (2012) Corona discharge ion mobility spectrometry with orthogonal acceleration time of flight mass spectrometry for monitoring of volatile organic compounds. Anal Chem 84:5327–5334
Alizadeh N, Jafari M, Mohammadi A (2009) Headspace-solid-phase microextraction using a dodecylsulfate-doped polypyrrole film coupled to ion mobility spectrometry for analysis methyl tert-butyl ether in water and gasoline. J Hazard Mater 169:861–867
Baumbach JI, Maddula S, Sommerwerck U, Besa V, Kurth I, Bödeker B, Teschler H, Freitag L, Darwiche K (2011) Significant different volatile biomarker during bronchoscopic ion mobility spectrometry investigation of patients suffering lung carcinoma. Int J Ion Mobil Spec 14:159–166
Bessa V, Darwiche K, Teschler H, Sommerwerck U, Rabis T, Baumbach JI, Freitag L (2011) Detection of volatile organic compounds (VOCs) in exhaled breath of patients with chronic obstructive pulmonary disease (COPD) by ionmobility spectrometry. Int J Ion Mobil Spec 14:7–13
Westhoff M, Litterst P, Maddula S, Bödecker B, Rahmann S, Davies AN, Baumbach JI (2010) Differentiation of chronic obstructive pulmonary disease (COPD) including lung cancer from healthy control group by breath analysis using ion mobility spectrometry. Int J Ion Mobil Spec 13:131–139
Jünger M, Bödeker B, Baumbach JI (2010) Peak assignment in multi-capillary column ion mobility spectrometry using comparative studies with gas chromatography—mass spectrometry for exhaled breath analysis. Anal Bioanal Chem 396:471–482
Bödecker B, Davies AN, Maddula S, Baumbach JI (2010) Biomarker validation—room air variation during human breath investigations. Int J Ion Mobil Spec 13:177–184
Koczulla R, Hattesohl A, Schmid S, Bödeker B, Maddula S, Baumbach JI (2011) MCC/IMS as potential noninvasive technique in the diagnosis of patients with COPD with and without alpha 1-antitrypsin deficiency. Int J Ion Mobil Spec 14:177–185
Basanta M, Koimtzis T, Thomas CLP (2006) Sampling and analysis of exhaled breath on human subjects with thermal desorption gas chromatography differential mobility spectrometry. Int J Ion Mobil Spec 9:45–49
Ulrich S (2000) Solid-phase microextraction in biomedical analysis. J Chromatogr A 902:167–194
Sprung M (1939) Research Laboratory. General Electric Company, Schenectady
Bollan HR, Stone JA, Brokenshire JL, Rodriguez JE, Eiceman GA (2007) Mobility resolution and mass analysis of ions from ammonia and hydrazine complexes with ketones formed in air at ambient pressure. J Am Soc Mass Spectrom 18:940–951
Eiceman GA, Salazar MR, Rodriguez MR, Limero TF, Beck SW, Cross JH, Young R, James JT (1993) Ion mobility spectrometry of hydrazine, monomethylhydrazine, and ammonia in air with 5-nonanone reagent gas. Anal Chem 65:1696–1702
Morrison GC, Howard CJ (2001) Selective detection of gas-phase aldehydes and ketones using protonated hydrazine. Int J Mass Spectrom 210(211):503–509
Bell S, Ewing RG, Eiceman GA, Karpas Z (1994) Atmospheric pressure chemical ionization of alkanes, alkenes, and cycloalkanes. J Am Soc Mass Spect 3:177–185
Karimi A, Alizadeh N (2009) Rapid analysis of captopril in human plasma and pharmaceutical preparations by headspace solid phase microextraction based on polypyrrole film coupled to ion mobility spectrometry. Talanta 79:479–485
Acknowledgments
This work was supported with grants from Tarbiat Modares University Research Council.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kalhor, H., Alizadeh, N. Enhancing sensitivity of ion mobility spectrometry determination of aldehydes by in situ gas phase derivatization with dibutylamine. Int. J. Ion Mobil. Spec. 16, 199–205 (2013). https://doi.org/10.1007/s12127-013-0119-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12127-013-0119-3