Abstract
Bakery industries are thriving to augment the diverse properties of Saccharomyces cerevisiae to increase its flavor, texture and nutritional parameters to attract the more consumers. The improved technologies adopted for quality improvement of baker’s yeast are attracting the attention of industry and it is playing a pivotal role in redesigning the quality parameters. Modern yeast strain improvement tactics revolve around the use of several advanced technologies such as evolutionary engineering, systems biology, metabolic engineering, genome editing. The review mainly deals with the technologies for improving S. cerevisiae, with the objective of broadening the range of its industrial applications.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Baking is a very old process, which uses extended dry heat for cooking [1]. Generally, yeast, Saccharomyces cerevisiae has been used for the baking process commonly known as baker’s yeast. It is a unicellular microorganism classified under fungi (Ascomycota and Fungi imperfecti). Baker’s yeast is used as a primary leavening agent for bread production and other products such as bread, cookies, crackers, cakes, pretzels, pastries, pies, tarts, quiches and more, collectively known as baking goods [2, 3]. It uses the sugar present in dough for its metabolic processes and generates carbon dioxide (CO2) and ethanol, which causes the dough leavening during fermentation and oven rises [4]. Besides these, some other metabolites like organic acids, glycerol and aroma compounds are also produced [5]. These metabolites play significant role in fine quality, texture and rheological properties of bread. Although, yeast have been engaged in bakery industries since long times, the performance is limited due to various industrial constraints and requirements [6]. At industrial scale, baker’s yeast gets exposed to several multiple and fluctuating environmental stresses, which ultimately diminish the product yield and also negatively affect the quality of bakery products [7]. Additionally, these environmental constraints largely affect cellular metabolism and viability.
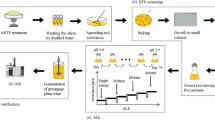
Therefore, in order to overcome these constraints, it is very essential to improve the conventional baker’s yeast using advanced techniques such as systems biology, bioinformatics tools, metabolic engineering, genome editing using CRISPR-Cas, etc. [8,9,10,11], along with the conventional approach like evolutionary engineering and genetic engineering. A schematic diagram of various technologies used for improvement of baker’s yeast is described in Fig. 1. The approaches used for obtaining genetically engineered host depends on the target to be customized and on a phylogentically distant source of molecular determinants [12]. Many research reports have been published for improvement of baker’s yeast for stress tolerance, enhancing fermentation and leavening abilities and improving aroma of bakery products [13]. It has been noticed that most of the commercial strains of baker’s yeast are polyploids in nature with the lacking any mating type behavior, a low level of sporulation or very poor spore viability. The advances in genetic engineering and protoplast fusion technologies to the yeast genetics have solved the problems at certain levels [14, 15].
Considering the importance of baker’s yeast, in this review, we highlighted the major bakery industries manufacturing the baker’s yeast and its various products worldwide. Further, we describe the baker’s yeast strain improvement for increased flavor induction, various industrial stress tolerances, and enhanced leavening ability. Moreover, we have illustrated different advance technologies such as systems biology, metabolic engineering, gene editing used for strain microbes improvement along with conventional techniques like recombinant DNA technologies, evolutionary engineering that can play very important role in further desired trait improvement.
Bakery Industry
Baker’s products have been used as a vital component of the balanced diet for many centuries. Today, the baker’s industry is one of the largest sectors of the food industry worldwide, which creates thousands of employments and generates billions of dollars in revenue. As the data available for the year 2016, the U.K. bread and morning goods are worth almost £4 billion. While in India, estimated turnover of the bakery products is US $ 1.3 billion and is also one of the largest manufacturing sectors. This constant growth has been driven by demands of consumer for appropriate and premium baked goods which are fresh, shelf-stable, nutritious and conveniently packaged. A large numbers of yeast manufacturing industries as described in Table 1 are situated around the globe. These industries are involved in production of different types of baker’s yeast and other bakery products.
Improvement of Baker’s Yeast and Flavor Induction
Initially, the wild strain of baker’s yeast was not very effective for the production of bakery products. Thus, several industrial initiative to improve conventional baker’s yeast strains for (1) pleasant flavor, (2) increased resistance to various stresses like high/low temperature, pH, osmotic pressure, oxidation, high sugar and salt tolerances, (3) increased fermentation, and (4) good leavening ability.
The pleasing and appealing flavor of yeast raised products gives them universal acceptance and public preference. The characteristics flavor of yeast raised products arises from yeast fermentation and subsequent reactions of the by-products with other dough compounds during baking [16]. Fermentation by yeast produced large number of volatile compounds that provide different aroma to bakery products. Any single component cannot be responsible for the bread aroma while they act in synergistically [17]. Moreover, it is not necessary that specific component present during dough can induce specific flavor in the bread. Generally it is assumed that many secondary metabolites like aldehydes, ketoses, organic acids, higher alcohols, esters and various enzymes involved in the biochemical reactions as listed in Table 2 during dough fermentation are responsible for bearing of characteristics flavor [18,19,20,21]. As we know that, vanilla is most pleasing and a high demanding flavor produced from vanillin compound among the population. So to meet the demands, various chemical methods were developed for the synthesis of vanillin. But vanillin synthesized by chemical route limits its usability [22]. Thus biotechnological approaches have been developed for vanillin production such as biocatalytic transformation of lignin biopolymer to vanillin catalyzed by genetically modified Rhodococcus jostii [23]. Hansen et al. [17], has also been de novo synthesized the vanillin in the presence of glucose by metabolically engineered yeast. However, new non-conventional yeast strains Torulaspora delbrueckii and S. bayanus have been identified which provides satisfactory dough fermentation with an interesting flavor profile to the bread [24].
Stress Tolerance
In most of the bakery process, yeast strain is exposed to the various environmental conditions such as temperatures, pressure, pH, water content, osmotic, oxidation and various chemical compounds [30, 31]. These harsh conditions cause drastic damage to cellular organelles and membranes, which ultimately leads to growth inhibition or cell death [32]. So, it is necessary to acquire or induces different stress-adaptation cellular mechanisms such as stress protein induction, favorable changes in membrane structure, up and down-regulation of corresponding gene expression to survive under these stresses [33, 34]. Stress tolerance properties can significantly enhance the growth of yeast strain during fermentation conditions which ultimately increase the product yield. Many studies have been conducted for improvement of wild strain of baker’s yeast in order to reduce these stresses [32, 35,36,37]. Recently, AtTIL gene encoding temperature-induced lipocalins, TIL protein from Arabidopsis thaliana was expressed in S. cerevisiae using genetic engineering approach. The recombinant strain conferred a high tolerance to the stresses i.e. oxidative agents, heat shock, freezing and exposure to organic acids [38]. Similarly in another research, the recombinant strain of S. cerevisiae has been created by over expression of MAL62 gene. This gene is responsible for trehalose accumulation in the cytoplasm which improve the cell for freezing tolerance [36]. Furthermore, some of the recent examples are also described in Table 3.
Fermentation and Leavening Ability
The higher fermentation and leavening ability of S. cerevisiae is essential for the manufacturing of various bakery goods. As we discussed above, during baking process, baker’s yeast exposed to several environmental stresses which significantly lowers the cell survival and leavening ability [39, 40]. Several other key factors such as salt, carbohydrate sources, dough textures are also significantly affected the fermentation and leavening abilities [41]. In yeast, Glycogen is the main source of carbon and energy reserve. It is involved in many metabolic processes. Regulation of glycogen metabolism directly influences the fermentation ability [42]. Recently, S. cerevisiae was engineered for increasing the fermentation performance by inserting the promoter of a gluconeogenic gene, PCK1, into the upstream side of RIM 15 gene to achieve its repression in glucose rich medium. RIM 15 gene encode a protein kinase and down regulation of this gene increases the fermentation ability [43]. Many studies have been conducted in order to improve the leavening ability of bakers’ yeast. In a study, the leavening property of S. cerevisiae has been enhanced by reducing the sucrase activity in sweet dough. The yeast strain of varying sucrose activity were constructed by SUC2 gene deletion which encodes for sucrase enzyme [44]. Along with these, some other studies are enlisted in Table 3.
Techniques Used for Strain Improvement
Evolutionary Engineering
Evolutionary engineering is most widely used and very cost-effective technique for improvement of strains that includes mutagenesis and recombination of genes, pathways or sometimes whole cells followed by screening of improved strains developing desired phenotype [45]. This can be natural or induced by providing specific conditions to the cells. In this approach first cells are exposed to specific stress conditions which induce the mutation (addition, subtraction and substitution) in the genome. These mutations may be at a single site or at multiple sites. Treated cells are then screened for particular phenotype. This approach requires multiple generation cycles of random genetic changes and selections which finally give needful phenotype [46]. In this connection, several techniques of strain improvement like (1) classical mating, (2) protoplast fusion i.e. hybridization, (3) UV-induced mutations, and (4) stress-induced mutation are frequently used [14, 15, 47]. Many recent examples have demonstrated the satisfactory performance of this approach for strain improvement [45, 48, 49]. Moreover, this conventional approach has significantly improved S. cerevisiae for stress tolerance to various environmental conditions. Recently, S. cerevisiae has been engineered for improving ethanol tolerance using this approach. The strain was cultivated in the presence of gradually increasing ethanol concentration and selected mutants showed tolerance up to 12% (v/v) of ethanol. Along with this, the improved strain has also been shown the diplodization of haploid cells under ethanol stress [31]. Some of the recent examples are listed in Table 3. Although, this approach has been widely exploited for strain improvement, but it has several limitations such as lack of suitable screening methods for most of the traits, less chances of getting desired phenotype, randomness, hit and trial method, slow, labor intensive and require multiple generation cycles for a particular trait. Moreover, it is very difficult to determine which genetic modification is responsible for the improvement. Additionally, it is not possible to transfer the improved characteristics to other strains [31].
Systems Biology
The rapid growth of high throughput systems biology approaches in diversified fields, including strain improvement made this approach very important. It provides complete information of all cellular responses in different conditions which helps in determination of major changes at cellular levels or community levels and their interactions under specified conditions [10, 50]. The ‘omics’ techniques, i.e. genomics, transcriptomics, proteomics, metabolomics and phenomics are used alone or in combinations for the complete study of systems biology [51, 52]. The huge amount of data generated using systems biology tools is compiled, processed using several computational tools which provides valuable information about the systems. Further, complete genome of S. cerevisiae has already been sequenced and stored in the yeast genome database (YGD) developed by Stanford University, Stanford. In addition to this, complete information on all metabolic pathways of S. cerevisiae is present in YGD. Another database, Yeast metabolome Database (YMDB 2.0) having complete information of yeast metabolome are also developed [53]. The behavior of yeast varies according to their ambient and systems biology approach provides complete information about deferential behavior of yeast in varied environments. This data provide preliminary information for designing of strategies for further improvement. Recently, proteomics data has been analysis of two baker’s yeast strains exposed to near freezing point (4 °C). From this data, it has been deduced that glycolytic protein expression up regulated, increased intracellular accumulation of glycerol and trials to prevent severe cold and freeze injury. At 4 °C, total of 16 hyper-expressed proteins were identified which takes part in energy-metabolism, translation and redox homeostasis [54]. In another research study, comparative transcriptomic and metabolic analysis was performed during fermentation of four different types of S. cerevisiae strains for rational identification of new targets for improving aroma production. The advance technologies (HPLC, GC-MS and microarrays) was used for comparative analysis [55].
Metabolic Engineering
Metabolic engineering is a powerful approach which has been successfully used to improve the various microbial strains for production of metabolites which cannot be formed naturally [56]. Moreover, this approach provides the ability to microorganisms to survive under stress conditions. This technique directly modifies the metabolic fluxes [57]. Generally, metabolic engineering involves manipulation of cellular pathways by altering the enzymatic, transport and regulatory functions of the cell by using recombinant DNA technology [58]. Basically, for metabolic engineering, it is necessary to have complete knowledge of metabolic pathways and thanks to the next generation sequencing, which provides complete information of genome sequences of baker’s yeast [59]. Along with this, many bioinformatics tools and models have been developed which decode these genome information and provide full information about all metabolic pathways. This information is further used for construction of new metabolic networks, which will be executed in real by various molecular techniques such as genetic engineering and genome engineering, etc. A number of reviews and research work have been published for successful improvement of strains using metabolic engineering [60,61,62]. Recently, the metabolic pathways of S. cerevisiae have been engineered for the production of ethanol and its isomers which are used for biofuel production [76]. In this approach, a combination of valine synthesis and degradation and complete re-localization of cytosolic and mitochondrial pathways has been done [63]. Generally, recombinant DNA approach is used for metabolic engineering, but recently the emergence of gene editing or genome engineering tools using CRISPR/Cas9 made the metabolic engineering more uncomplicated [8, 9, 64, 65].
Conclusions and Perspectives
Bakery products have been used as an essential part of a balanced diet for many centuries. A wide variety of morning fast foods are found on the supermarket shelves. There is vast competition in the baking industries for the above-said bakery products in terms of premium quality, increased shelf-life, good flavor, high nutritional contents and pleasing appearance with cosmetic touch [2]. Yeast plays an important role in dough maturation due to production of CO2 during fermentation. Along with this, different types of metabolites such as organic acids, alcohols, aldehydes, ketones, esters and enzymes responsible for flavor induction. The textures of bread are also dependent upon all these metabolites. There are lots of industrial stress, such as temperature, pH, salt, sugar, freezing and thawing, lower growth of yeast, less leavening ability, increase the CO2 production, etc., which limits the use of native yeast strain for the production of bakery products and that ultimately negatively affect the overall yield [5]. So, in order to overcome these limitations, several approaches have been exploited for further strain improvement [35, 66, 67, 69, 73]. Many conventional techniques including genetic engineering and evolutionary engineering have been used successfully worldwide, but still there is some limitation with conventional yeast strain, i.e. S. cerevisiae. Moreover, most of the traits of industrial importance like stress tolerance and increase dough raising power have been polygenic in nature with multi-step regulations at many sites, difficult to be dealt with recombinant DNA technology and evolutionary engineering. Unfortunately, even after putting much effort towards the production of genetically modified microorganisms, these are still not acceptable for human consumption. Besides this, the rheology and leavening properties of dough and bread characteristics are not yet fully tested for the newly created genetically modified strains derived from Baker’s yeast. The emergence of some advanced technologies such as systems biology, genome engineering, metabolic engineering, bioinformatics tools and computational biology provide better understanding and analysis of intracellular metabolic networks and metabolites produced under varying conditions. Moreover, the future perspective might be in the field of isolation and screening of non-conventional yeast strains that can grow in high stress conditions, produce different flavors and cost-effective.
References
Papasidero D, Pierucci S, Manenti F (2016) Energy optimization of bread baking process undergoing quality constraints. Energy 116:1417–1422. doi:10.1016/j.energy.2016.06.046
Gobbetti M, Rizzello CG, Di Cagno R, De Angelis M (2014) How the sourdough may affect the functional features of leavened baked goods. Food Microbiol 37:30–40. doi:10.1016/j.fm.2013.04.012
Lai HM, Lin TC (2007) Bakery products: science and technology. In: Hui YH (ed) Bakery products: science and technology. Blackwell Publishing, Ames, pp 3–68. doi:10.1002/9780470277553.ch1
Joseph R, Bachhawat AK (2014) Yeasts: production and commercial uses. In: Batt CA, Tortorello ML (eds) Encyclopedia of food microbiology. Academic Press, Oxford, pp 823–830. doi:10.1016/B978-0-12-384730-0.00361-X
Birch AN, Petersen MA, Arneborg N, Hansen AS (2013) Influence of commercial baker’s yeasts on bread aroma profiles. Food Res Int 52:160–166. doi:10.1016/j.foodres.2013.03.011
Struyf N, Maelen E, Hemdane S, Verspreet J, Verstrepen KJ, Courtin CM (2017) Bread dough and baker’s yeast: an uplifting synergy. Compr Rev Food Sci Food Saf. doi:10.1111/1541-4337.12282
Gibson BR, Lawrence SJ, Leclaire JP, Powell CD, Smart KA (2007) Yeast responses to stresses associated with industrial brewery handling. FEMS Microbiol Rev 31:535–569. doi:10.1111/j.1574-6976.2007.00076.x
Yadav R, Kumar V, Baweja M, Shukla P (2016) Gene editing and genetic engineering approaches for advanced probiotics: a review. Crit Rev Food Sci Nutr. doi:10.1080/10408398.2016.1274877
Gupta SK, Shukla P (2017) Gene editing for cell engineering: trends and applications. Crit Rev Biotechnol 37:672–684. doi:10.1080/07388551.2016.1214557
Kumar PS, Singh PK, Shukla P (2014) Systems biology as an approach for deciphering microbial interactions. Brief Funct Genomics 14:23–25. doi:10.1093/bfgp/elu023
Kumar V, Baweja M, Singh PK, Shukla P (2016) Recent developments in systems biology and metabolic engineering of plant–microbe interactions. Front Plant Sci 7:1–12. doi:10.3389/fpls.2016.01421
Pandey B, Saini M, Sharma P (2016) Molecular phylogenetic and sequence variation analysis of dimeric α-amylase inhibitor genes in wheat and its wild relative species. Plant Gene 6:48–58. doi:10.1016/j.plgene.2016.03.004
Chopda VR, Rathore AS, Gomes J (2015) Maximizing biomass concentration in baker’s yeast process by using a decoupled geometric controller for substrate and dissolved oxygen. Bioresour Technol 196:160–168. doi:10.1016/j.biortech.2015.07.050
Chambers PJ, Bellon JR, Schmidt SA, Varela C, Pretorius IS (2009) Non-genetic engineering approaches for isolating and generating novel yeasts for industrial applications. In: Satyanarayana T, Kunze G (eds) Yeast biotechnology: diversity and applications. Springer, Dordrecht, pp 433–457. doi:10.1007/978-1-4020-8292-4_20
Steensels J, Snoek T, Meersman E, Nicolino MP, Voordeckers K, Verstrepen KJ (2014) Improving industrial yeast strains: exploiting natural and artificial diversity. FEMS Microbiol Rev 38:947–995. doi:10.1111/1574-6976.12073
Brochado AR, Matos C, Møller BL, Hansen J, Mortensen UH, Patil KR (2010) Improved vanillin production in baker’s yeast through in silico design. Microb Cell Fact 9:84. doi:10.1186/1475-2859-9-84
Hansen EH, Møller BL, Kock GR, Bünner CM, Kristensen C, Jensen OR, Okkels FT, Olsen CE, Motawia MS, Hansen J (2009) De novo biosynthesis of vanillin in fission yeast (Schizosaccharomyces pombe) and baker’s yeast (Saccharomyces cerevisiae). Appl Environ Microbiol 75:2765–2774. doi:10.1128/AEM.02681-08
Donalies UEB, Nguyen HTT, Stahl U, Nevoigt E (2008) Improvement of Saccharomyces yeast strains used in brewing, wine making and baking. Adv Biochem Eng Biotechnol 111:67–98. doi:10.1007/10_2008_099
Smit BA, Engels WJM, Smit G (2009) Branched chain aldehydes: production and breakdown pathways and relevance for flavour in foods. Appl Microbiol Biotechnol 81:987–999. doi:10.1007/s00253-008-1758-x
Zhang C, Zhang H, Wang L, Gao H, Guo XN, Yao HY (2007) Improvement of texture properties and flavor of frozen dough by carrot (Daucus carota) antifreeze protein supplementation. J Agric Food Chem 55:9620–9626. doi:10.1021/jf0717034
Pozo-Bayón MA, Guichard E, Cayot N (2006) Flavor control in baked cereal products. Food Rev Int 22:335–379. doi:10.1080/87559120600864829
Carlquist M, Gibson B, Karagul YY, Paraskevopoulou A, Sandell M, Angelov AI, Gotcheva V, Angelov AD, Etschmann M, Billerbeck GM, Lidén G (2015) Process engineering for bioflavour production with metabolically active yeasts—a mini-review. Yeast 32:123–143. doi:10.1002/yea.3058
Sainsbury PD, Hardiman EM, Ahmad M, Otani H, Seghezzi N, Eltis LD, Bugg TD (2013) Breaking down lignin to high-value chemicals: the conversion of lignocellulose to vanillin in a gene deletion mutant of Rhodococcus jostii RHA1. ACS Chem Biol 8:2151–2156. doi:10.1021/cb400505a
Aslankoohi E, Herrera-Malaver B, Rezaei MN, Steensels J, Courtin CM, Verstrepen KJ (2016) Non-onventional yeast strains increase the aroma complexity of bread. PLoS ONE 11:1–18. doi:10.1371/journal.pone.0165126
Serra S, Fuganti C, Brenna E (2005) Biocatalytic preparation of natural flavours and fragrances. Trends Biotechnol 23:193–198. doi:10.1016/j.tibtech.2005.02.003
Petel C, Onno B, Prost C (2017) Sourdough volatile compounds and their contribution to bread: a review. Trends Food Sci Technol 59:105–123. doi:10.1016/j.tifs.2016.10.015
Salim-ur-Rehman Paterson A, Piggott JR (2006) Flavour in sourdough breads: a review. Trends Food Sci Technol 17:557–566. doi:10.1016/j.tifs.2006.03.006
O’Shea N, Kilcawley KN, Gallagher E (2016) Influence of α-amylase and xylanase on the chemical, physical and volatile compound properties of wheat bread supplemented with wholegrain barley flour. Eur Food Res Technol 242:1503–1514. doi:10.1007/s0021
Kulshrestha S, Tyagi P, Sindhi V, Yadavilli KS (2013) Invertase and its applications—a brief review. J Pharm Res 7:792–797. doi:10.1016/j.jopr.2013.07.014
Takagi H (2017) Construction of baker’s yeast strains with enhanced tolerance to baking-associated stresses. In: Sibirny A (ed) Biotechnology of yeasts and filamentous fungi. Springer, Cham, pp 63–85. doi:10.1007/978-3-319-58829-2_3
Turanlı-Yıldız B, Benbadis L, Alkım C, Sezgin T, Akşit A, Gökçe A, Öztürk Y, Baykal AT, Çakar ZP, François JM (2017) In vivo evolutionary engineering for ethanol-tolerance of Saccharomyces cerevisiae haploid cells triggers diploidization. J Biosci Bioeng 124:309–318. doi:10.1016/j.jbiosc.2017.04.012
Takagi H, Shima J (2015) Stress tolerance of baker’s yeast during bread-making processes. In: Takagi H, Kitagaki H (eds) Stress biology of yeasts and fungi. Springer, Tokyo, pp 23–42. doi:10.1007/978-4-431-55248-2_2
Takagi H (2008) Proline as a stress protectant in yeast: physiological functions, metabolic regulations, and biotechnological applications. Appl Microbiol Biotechnol 81:211. doi:10.1007/s00253-008-1698-5
Yoshiyama Y, Tanaka K, Yoshiyama K, Hibi M, Ogawa J, Shima J (2015) Trehalose accumulation enhances tolerance of Saccharomyces cerevisiae to acetic acid. J Biosci Bioeng 119:172–175. doi:10.1016/j.jbiosc.2014.06.021
Tsolmonbaatar A, Hashida K, Sugimoto Y, Watanabe D, Furukawa S, Takagi H (2016) Isolation of baker’s yeast mutants with proline accumulation that showed enhanced tolerance to baking-associated stresses. Int J Food Microbiol 238:233–240. doi:10.1016/j.ijfoodmicro.2016.09.015
Sun X, Zhang CY, Wu MY, Fan ZH, Liu SN, Zhu WB, Xiao DG (2016) MAL62 overexpression and NTH1 deletion enhance the freezing tolerance and fermentation capacity of the baker’s yeast in lean dough. Microb Cell Fact 15:54. doi:10.1186/s12934-016-0453-3
Sasano Y, Haitani Y, Hashida K, Oshiro S, Shima J, Takagi H (2013) Improvement of fermentation ability under baking-associated stress conditions by altering the POG1 gene expression in baker’s yeast. Int J Food Microbiol 165:241–245. doi:10.1016/j.ijfoodmicro.2013.05.015
Berterame NM, Bertagnoli S, Codazzi V, Porro D, Branduardi P (2017) Temperature-induced lipocalin (TIL): a shield against stress-inducing environmental shocks in Saccharomyces cerevisiae. FEMS Yeast Res 17:fox056. doi:10.1093/femsyr/fox056
Lin X, Zhang CY, Bai XW, Song HY, Xiao DG (2014) Effects of MIG1, TUP1 and SSN6 deletion on maltose metabolism and leavening ability of baker’s yeast in lean dough. Microb Cell Fact 13:93. doi:10.1186/s12934-014-0093-4
Lin X, Zhang CY, Bai XW, Xiao DG (2015) Enhanced leavening ability of baker’s yeast by overexpression of SNR84 with PGM2 deletion. J Ind Microbiol Biotechnol 42:939–948. doi:10.1007/s10295-015-1618-5
Ilowefah M, Chinma C, Bakar J, Ghazali HM, Muhammad K, Makeri M (2014) Fermented brown rice flour as functional food ingredient. Foods 3:149–159. doi:10.3390/foods3010149
Pérez-Torrado R, Matallana E (2015) Enhanced fermentative capacity of yeasts engineered in storage carbohydrate metabolism. Biotechnol Prog 31:20–24. doi:10.1002/btpr.1993
Watanabe D, Kaneko A, Sugimoto Y, Ohnuki S, Takagi H, Ohya Y (2017) Promoter engineering of the Saccharomyces cerevisiae RIM15 gene for improvement of alcoholic fermentation rates under stress conditions. J Biosci Bioeng 123:183–189. doi:10.1016/j.jbiosc.2016.08.004
Zhang CY, Lin X, Feng B, Liu XE, Bai XW, Xu J, Pi L, Xiao DG (2016) Enhanced leavening properties of baker’s yeast by reducing sucrase activity in sweet dough. Appl Microbiol Biotechnol 100:6375–6383. doi:10.1007/s00253-016-7449-0
Winkler JD, Kao KC (2014) Recent advances in the evolutionary engineering of industrial biocatalysts. Genomics 104:406–411. doi:10.1016/j.ygeno.2014.09.006
Nevoigt E (2008) Progress in metabolic engineering of Saccharomyces cerevisiae. Microbiol Mol Biol Rev 72:379–412. doi:10.1128/MMBR.00025-07
Zhang M, Shukla P, Ayyachamy M, Permaul K, Singh S (2010) Improved bioethanol production through simultaneous saccharification and fermentation of lignocellulosic agricultural wastes by Kluyveromyces marxianus 6556. World J Microbiol Biotechnol 26:1041–1046. doi:10.1007/s11274-009-0267-0
Cakar ZP, Turanli-Yildiz B, Alkim C, Yilmaz U (2012) Evolutionary engineering of Saccharomyces cerevisiae for improved industrially important properties. FEMS Yeast Res 12:171–182. doi:10.1111/j.1567-1364.2011.00775.x
Young E, Lee SM, Alper H (2010) Optimizing pentose utilization in yeast: the need for novel tools and approaches. Biotechnol Biofuels 3:24. doi:10.1186/1754-6834-3-24
Pavlopoulos GA, Malliarakis D, Papanikolaou N, Theodosiou T, Enright AJ, Iliopoulos I (2015) Visualizing genome and systems biology: technologies, tools, implementation techniques and trends, past, present and future. Gigascience 4:38. doi:10.1186/s13742-015-0077-2
Vervoort Y, Linares AG, Roncoroni M, Liu C, Steensels J, Verstrepen KJ (2017) High-throughput system-wide engineering and screening for microbial biotechnology. Curr Opin Biotechnol 46:120–125. doi:10.1016/j.copbio.2017.02.011
Gandhi A, Shah NP (2016) Integrating omics to unravel the stress response mechanisms in probiotic bacteria: approaches, challenges, and prospects. Crit Rev Food Sci Nutr 57:3464–3471. doi:10.1080/10408398.2015.1136805
Ramirez-Gaona M, Marcu A, Pon A, Guo AC, Sajed T, Wishart NA, Karu N, Djoumbou FY, Arndt D, Wishart DS (2017) YMDB 2.0: a significantly expanded version of the yeast metabolome database. Nucleic Acids Res 45:D440–D445. doi:10.1093/nar/gkw1058
Ballester-Tomas L, Perez-Torrado R, Rodríguez-Vargas S, Prieto JA, Randez-Gil F (2016) Near-freezing effects on the proteome of industrial yeast strains of Saccharomyces cerevisiae. J Biotechnol 221:70–77. doi:10.1016/j.jbiotec.2016.01.029
Mendes I, Sanchez I, Franco-Duarte R, Camarasa C, Schuller D, Dequin S, Sousa MJ (2017) Integrating transcriptomics and metabolomics for the analysis of the aroma profiles of Saccharomyces cerevisiae strains from diverse origins. BMC Genom 18:455. doi:10.1186/s12864-017-3816-1
Nakashima N, Miyazaki K (2014) Bacterial cellular engineering by genome editing and gene silencing. Int J Mol Sci 15:2773–2793. doi:10.3390/ijms15022773
Pereira R, Nielsen J, Rocha I (2016) Improving the flux distributions simulated with genome-scale metabolic models of Saccharomyces cerevisiae. Metab Eng Commun 3:153–163. doi:10.1016/j.meteno.2016.05.002
Kim SR, Park YC, Jin YS, Seo JH (2013) Strain engineering of Saccharomyces cerevisiae for enhanced xylose metabolism. Biotechnol Adv 31:851–861. doi:10.1016/j.biotechadv.2013.03.004
McIlwain SJ, Peris D, Sardi M, Moskvin OV, Zhan F, Myers KS, Riley NM, Buzzell A, Parreiras LS, Ong IM, Landick R (2016) Genome sequence and analysis of a stress-tolerant, wild-derived strain of Saccharomyces cerevisiae used in biofuels research. G3 Genes Genomes Genet 6:1757–1766. doi:10.1534/g3.116.029389
Papapetridis I, Dijk M, Maris AJA, Pronk JT (2017) Metabolic engineering strategies for optimizing acetate reduction, ethanol yield and osmotolerance in Saccharomyces cerevisiae. Biotechnol Biofuels 10:107. doi:10.1186/s13068-017-0791-3
Krivoruchko A, Nielsen J (2015) Production of natural products through metabolic engineering of Saccharomyces cerevisiae. Curr Opin Biotechnol 35:7–15. doi:10.1016/j.copbio.2014.12.004
Miskovic L, Alff-Tuomala S, Soh KC, Barth D, Salusjärvi L, Pitkänen JP, Ruohonen L, Penttilä M, Hatzimanikatis V (2017) A design–build–test cycle using modeling and experiments reveals interdependencies between upper glycolysis and xylose uptake in recombinant Saccharomyces cerevisiae and improves predictive capabilities of large-scale kinetic models. Biotechnol Biofuels 10:166. doi:10.1186/s13068-017-0838-5
Generoso WC, Schadeweg V, Oreb M, Boles E (2015) Metabolic engineering of Saccharomyces cerevisiae for production of butanol isomers. Curr Opin Biotechnol 33:1–7. doi:10.1016/j.copbio.2014.09.004
Jakociunas T, Bonde I, Herrgård M, Harrison SJ, Kristensen M, Pedersen LE, Jensen MK, Keasling JD (2015) Multiplex metabolic pathway engineering using CRISPR/Cas9 in Saccharomyces cerevisiae. Metab Eng 28:213–222. doi:10.1016/j.ymben.2015.01.008
Ledford H (2015) CRISPR, the disruptor. Nature 522:20–24. doi:10.1038/522020a
Gottardi M, Knudsen JD, Prado L, Oreb M, Branduardi P, Boles E (2017) De novo biosynthesis of trans-cinnamic acid derivatives in Saccharomyces cerevisiae. Appl Microbiol Biotechnol 101:4883–4893. doi:10.1007/s00253-017-8220-x
Zha M, Yin S, Sun B, Wang X, Wang C (2017) STR3 and CYS3 contribute to 2-furfurylthiol biosynthesis in Chinese sesame-flavored baijiu yeast. J Agric Food Chem 65:5503–5511. doi:10.1021/acs.jafc.7b01359
Chen X, Nielsen KF, Borodina I, Kielland-Brandt MC, Karhumaa K (2011) Increased isobutanol production in Saccharomyces cerevisiae by overexpression of genes in valine metabolism. Biotechnol Biofuels 4:21. doi:10.1186/1754-6834-4-21
Nakagawa Y, Ogihara H, Mochizuki C, Yamamura H, Iimura Y, Hayakawa M (2017) Development of intra-strain self-cloning procedure for breeding baker’s yeast strains. J Biosci Bioeng 123:319–326. doi:10.1016/j.jbiosc.2016.10.008
Dong J, Chen D, Wang G, Zhang C, Du L, Liu S, Zhao Y, Xiao D (2016) Improving freeze-tolerance of baker’s yeast through seamless gene deletion of NTH1. J Ind Microbiol Biotechnol 43:817–828. doi:10.1007/s10295-016-1753-7
Ballester-Tomás L, Randez-Gil F, Pérez-Torrado R, Prieto JA (2015) Redox engineering by ectopic expression of glutamate dehydrogenase genes links NADPH availability and NADH oxidation with cold growth in Saccharomyces cerevisiae. Microb Cell Fact 14:100. doi:10.1186/s12934-015-0289-2
Dong J, Wang G, Zhang C, Tan H, Sun X, Wu M, Xiao D (2013) A two-step integration method for seamless gene deletion in baker’s yeast. Anal Biochem 439:30–36. doi:10.1016/j.ab.2013.04.005
Ando A, Nakamura T (2016) Prevention of GABA reduction during dough fermentation using a baker’s yeast dal81 mutant. J Biosci Bioeng 122:441–445. doi:10.1016/j.jbiosc.2016.03.006
Zhang CY, Lin X, Song HY, Xiao DG (2015) Effects of MAL61 and MAL62 overexpression on maltose fermentation of baker’s yeast in lean dough. World J Microbiol Biotechnol 31:1241–1249. doi:10.1007/s11274-015-1874-6
Zhang CY, Bai XW, Lin X, Liu XE, Xiao DG (2015) Effects of SNF1 on maltose metabolism and leavening ability of baker’s yeast in lean dough. J Food Sci 80:M2879–M2885. doi:10.1111/1750-3841.13137
Jun H, Jiayi C (2012) Metabolic engineering of Saccharomyces cerevisiae for increased bioconversion of lignocellulose to ethanol. Ind J Microbiol 52:442–448. doi:10.1007/s12088-012-0259-x
Acknowledgements
The author acknowledges Maharshi Dayanand University, Rohtak for providing infrastructure and lab facility for compilation of this interesting and informative review.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Dangi, A.K., Dubey, K.K. & Shukla, P. Strategies to Improve Saccharomyces cerevisiae: Technological Advancements and Evolutionary Engineering. Indian J Microbiol 57, 378–386 (2017). https://doi.org/10.1007/s12088-017-0679-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12088-017-0679-8