Abstract
Cellular reprogramming is a promising strategy to generate neural stem cells (NSCs) or desired subtype-specific neurons for cell-based therapeutic intervention. By far, the intricate cell event like reprogramming of non-neural cells to desired cell types can be achieved by forced expression of lineage-related transcription factors (TFs), nuclear transfer, a defined set of factors, and via non-coding microRNAs (miRNAs), as well as other precisely defined conditions. In addition, scientists have been trying to develop better approaches for reprogramming, either by using distinct combinations of a set of small molecules and certain TFs or delivery of appropriate small molecules and miRNAs. The miRNA-mediated approach is fascinating because of its potential to rapidly generate a variety of therapeutically desired cell types from other cell lineages. Recent studies have made great progress in miRNA-mediated neural reprogramming of somatic cells to various specific neuronal subtypes with more efficiency even though the exact mechanisms remain to be further explored. Based on key roles of miRNAs in neural reprogramming across differentiated cell lineages, it is of vital interest to summarize the recent knowledge regarding the instructive role of miRNAs in direct conversion of somatic cells into neural lineages. This precise review mainly focuses on recent discoveries of miRNAs functions in initiating cell reprogramming and fate specification of the neuronal subtype. Moreover, we discuss most recent findings about some miRNAs’ activity in regulating various developmental stages of neurons, which is helpful for understanding the event network between miRNAs and their targets.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
MicroRNAs (miRNAs) are a class of small, endogenous short non-coding RNAs 19-23 nucleotides in length that regulate gene expression at the post-transcriptional level in wide variety of cellular processes including cell proliferation, cell fate determination and differentiation, and metabolism [1–6]. MiRNAs specifically recognize and regulate expression of mRNAs by sequence complementarity with the mRNAs 3′ untranslated region. Importantly, a single miRNA can have several target mRNAs to regulate a process; and conversely, a unique mRNA can be targeted by more than one miRNA. Thereby, by degrading target mRNAs or inhibiting translation, miRNAs exert a potential impact on the transcriptome, influencing various biological events [7, 8].
Since the discovery of the first miRNA in 1993, numerous miRNAs have been identified in various species from plants to mammals [9]. Furthermore, miRNA characteristics and regulatory roles in many important cellular processes were revealed. Strikingly, specific miRNAs were employed in the regulation of de novo DNA methylation which is responsible for the direct or indirect cell reprogramming event [10, 11]. Since Takahashi and Yamanaka discovered that the simple combination of a few transcription factors (TFs) can initiate the reprogramming toward a pluripotent state [12, 13], direct cell reprogramming into a variety of target cells has sparked great interest in the field of regenerative medicine and opened several new research avenues due to the facts that ethical controversies can be avoid and immune rejection reduced. In the most studied cellular reprogramming approaches, different TFs and signaling pathways have been described as crucial players within the intricate gene expression regulatory networks during cell reprogramming processes [14, 15]. On basis of Yamanaka and colleagues’ strategy for conversion of fibroblasts to their desirable cell types, increasing studies have demonstrated that numerous transcriptional regulators play critical roles in neurogenesis and cell reprogramming, and that several TFs such as Oct4, Sox2, Nanog, and kif4 are sufficient to robustly induce neuroblasts [16–23], suggesting that these TFs possess powerful reprogramming activity. Although TFs in cell fate conversion has been widely documented during this reprogramming process, epigenetic and gene expression regulation by non-coding RNAs (ncRNAs) as additional and essential regulatory mechanisms for the reprogramming process must be precisely regulated to accomplish this cell event at the same time, and maintain the reprogrammed cell profiles. Of note, some miRNAs are sufficient to induce reprogramming of mammalian somatic cells such as fibroblasts without forced expression of other TFs. Thus, miRNAs may be a valuable means to regulate the distinct stages of a stepwise cell reprogramming. Here, we will review the recent findings about miRNAs as post-transcriptional regulators in regulating and orchestrating the cellular event.
miRNA Biogenesis
miRNAs were initially discovered in Caenorhabditis elegans during a genetic screen to uncover molecules involved in regulating nematode development [9, 24]. The majority of the characterized miRNA genes were intergenic or antisense oriented to neighboring genes and therefore, suspected to be transcribed as independent units that consist of one or more hairpin structure, each composed of a stem and terminal loop [1]. Generation of miRNAs from long double-stranded RNAs is a stepwise process that can be canonical (Drosha/Dgcr8-dependent) or non-canonical (Drosha/Dgcr8-independent) [25]. The canonical miRNA biogenesis in animals undergo the following processes: (1) miRNA loci transcription and further editing to precursor transcripts known as pri-miRNA by RNA polymerase II and some related proteins factors; (2) The conversion of pri-miRNA to precursor miRNA (pre-miRNA) in the nucleus by type III RNase Drosha complex [26]; (3) Transportation of pre-miRNA to the cytoplasm by the nucleo-cytoplasmic transport factor Exportin5, in a Ran-GTP-dependent manner [27]; and (4) Generation of an approximately 21-22 nt RNA duplex structure (miRNA/miRNA*) by a second type III RNase endonuclease called Dicer [2]. Subsequently, one guide strand of miRNA duplex is preferentially incorporated into the RNA-induced silencing complex (RISC), which guides the binding of the miRNA response elements (MREs) to specific mRNA transcripts by partial base pairing between the miRNA and the target mRNA [28, 29]. For better understanding of the miRNA biogenesis process, a schematic diagram of the possibilities of miRNA biogenesis is illustrated in Fig. 1. Besides, miRNA*, also called passenger strand, is typically assumed to be merely a carrier strand, which is finally degraded by several exoribonucleases and is therefore non-functional [30, 31]. Furthermore, pre-miRNAs can be generated by non-canonical mirtron pathway [32, 33]. Mirtrons are generated when their host genes are transcribed and, then, short introns with potential hairpin enter the mirtron pathway. After lariat debranching enzyme (Ldbr) gives rise to shorter pre-miRNAs that abutted intron-exon boundaries, this pre-miRNA is actively transported from the nucleus to the cytoplasm by Ran-GTP and the export receptor Exportin5 [34]. Once pre-miRNAs are cleaved to 22-nucleotides by the Drosha cleavage site of Dicer, an imperfect duplex consisting of the mature miRNA and its corresponding complementary sequence derived from the other arm of pre-miRNA results [1]. Intragenic miRNAs are generated from transcripts located within protein-coding regions of genes (Fig. 1).
Diverse Functions for miRNAs in Reprogramming
Since substantial evidence has shown that somatic cells could be transformed to iPSCs through epigenetic reprogramming by forced overexpression of the defined TFs, miRNA-based cellular processes including cell proliferation, differentiation, and reprogramming as well as maintenance of stemness, have been intensively studied to evaluate therapeutic applications of this technique. Although traditional DNA-based reprogramming for generation of iPSCs has made progress, there is still the major drawback of possible random insertion of the reprogramming factors into the genome, which could lead to genome disruption. Therefore, studies have been conducted to modify the approaches to improve safety and efficiency of the iPSCs using different combinations of TFs, or non-genetic strategies, such as employing various small molecules and specific miRNAs to induce pluripotency or reprogramming. In this respect, microRNA analysis defined that stem cells from diverse origins have a distinct miRNA expression pattern as compared to differentiated somatic cells [35]. This has prompted the researchers using miRNAs for cellular reprogramming. Notably, no matter what kind of patterns for somatic cell reprogramming was used, a prerequisite condition is the participation of some specific TFs. Recent studies revealed that the combinations of TFs including Oct4, Sox2, Nanog, Klf4, and c-Myc can convert fibroblasts or other somatic cells to pluripotent cells, including neural stem cells (NSCs) [12, 15, 36, 37]. Likewise, some chemicals in combination with growth factors are shown to induce somatic cells to convert into distinct lineage cells [38–40]. In transdifferentiation or cell reprogramming events, miRNAs were also reported to target multiple molecules, i.e., TFs and some regulatory factors which are actively involved in cell reprogramming, and enhance efficiency of defined TF-mediated reprogramming [41–47]. Recently, several studies showed that miRNAs can reprogram primary somatic cells toward pluripotent cells. This may mainly be attributed to involvement of miRNA in control of pluripotency-related factors. For example, miRNA302 regulates the expression of pluripotent markers Oct-4, Sox2, Nanog, and SSEA3/4 [48, 49], which further result in DNA demethylation and downregulation of Dnmt [48]. These cascade events eventually trigger somatic cell reprogramming. More strikingly, some small molecules such as SB431542, BIX-01291, and CHIR 99021 were confirmed to function as TFs [39, 40, 50, 51]. Recently, a combination of analytical techniques showed that numerous molecule-mediated cell reprogramming events are intimately linked to miRNA regulation [51]. Generally, miRNAs possess a unique ability to modulate an immense and complex regulatory network of gene expression [52] in a broad spectrum of developmental and cellular processes. They regulate gene expression via association with effector complexes (called “micro-ribonucleoprotein” or “miRNP”) and sequence-specific recognition of target sites [52]. The biological outcome of miRNA-mRNA interaction can result in binding strength and repressive effect of a potential target site [53], leading to repression of transcription or translational initiation. Thus, miRNAs targeting to directly or indirectly control the expression of reprogramming genes will suppress the cellular reprogramming. For instance, miRNA-34 targets P53 and, thus, contributes to p53 repression of iPSCs generation by cell reprogramming [54]. Also, the let-7 family members have been reported to inhibit reprogramming [55, 56]. In contrast to the general assumption, some miRNAs can upregulate gene expression and translation in specific cell types and conditions. This miRNA-mediated gene upregulation could promote cellular reprogramming [57]. In this sense, either miRNA-mediated gene regulation by promoting or inhibiting special mRNA expressions could have distinct consequences in cell reprogramming [58]. Therefore, miRNAs either facilitate or inhibit cell reprogramming in generation of induced stem cells. Although several approaches like virus-mediated TF transduction, induction of small molecules or growth factors, and delivery of chemicals could promote cell reprogramming, the miRNA-mediated cell reprogramming technique was claimed to be more efficient than the standard defined factor overexpression methods [45]. Apart from no concerns of genome integration of miRNAs, miRNA activity as a whole regulates reprogramming and efficiency, and miRNA-mediated cell reprogramming may provide an alternative and be likely a safer approach for generation of reprogrammed stem cells as compared to the traditional DNA-based cell reprogramming methods.
Possible Mechanism for Somatic Cell Reprogramming
Because miRNAs typically mediate gene silencing, they are traditionally regarded as fine tuners of gene expression. Consistent with this view, most published papers on cell fate switches used TF-based cocktails, rather than miRNAs. In spite of this, recent reports revealed the potential of miRNAs in cell reprogramming and further influencing neuronal cell fates. One report demonstrated that miRNA-9/9* and miRNA-124 alone promote the conversion of fibroblasts toward NSCs and neurons in the presence of as few as one TF [59, 60], suggesting a critical role of these miRNAs in cell reprogramming and induction of neural cell fates. Moreover, there is a growing number of literatures about other miRNAs, including miRNA132, miRNA137, miRNA184, miRNA-302/367, and Let7 family that participate in mediating cell reprogramming [42, 61–63]. Thus, the prevailing orthodoxy regarding cell reprogramming mainly mediated by TFs has been challenged. Although the critical roles of specific factors, signaling molecules, and miRNAs in cell reprogramming have intensively been investigated, the mechanism for miRNA-mediated cell reprogramming remains to be further explored.
It is well known that DNA methylation determines the specific expression pattern in cells and plays an essential role in mammalian development. A substantial condition for somatic cell reprogramming is the removal of DNA methylation at promoter regions of crucial stem cell TFs [64]. Once some miRNAs target the epigenetic regulators that are responsible for different types of DNA methylation, the transcription machinery can gain access to these genes and further activate their expression to initiate the reprogramming process [64]. Due to the importance of histone H3K4 and H3K9 demethylase for the de novo DNA methylation, their deficiency caused by miRNA usually induces DNA demethylation, thereby, leading to the resetting of genomic DNA methylation pattern which results in somatic cell reprogramming [42, 65–67]. This alteration in methylation causes the gene expression profiles to resemble that of stem cells by granting TFs [42]. Generally, 3′ untranslated regions (3′-UTRs) of messenger RNAs often contain regulatory sequences that post-transcriptionally cause RNA interference [68]. Theoretically, such 3′-UTRs often contain both binding sites for miRNAs as well as for regulatory proteins [69]. By binding to specific sites within the 3′-UTR, miRNAs can decrease gene expression of various mRNAs by either inhibiting translation or directly causing degradation of the transcript [70]. The 3′-UTR also may have silencer regions that bind repressor proteins that inhibit the expression of an mRNA, further causing downregulation of the relative protein translation [71, 72], which is considered as a crucial step for reprogramming and maintaining stemness. As reported previously, miRNA-302 directly target NR2F2, a member of a nuclear receptor subfamily that negatively regulates Oct-4 [73]. A reduction of NR2F2 together with the removal of genomic methylation site on the Oct-4 promoter due to global DNA demethylation causes an increase in Oct-4 expression [44, 73, 74]. Although miRNAs exert a pivotal role in inducing cell reprogramming, the interaction of miRNAs with IFs and other regulators is are also instructive to orchestrate reprogramming, and actively contributes to molecular mechanisms of cell reprogramming. For example, Lin28, an ESC-specific RNA binding protein, has been demonstrated to facilitate reprogramming by suppressing the activity of Let7 [75, 76]. Thus, it is possible that other regulators other than miRNAs also participate in this biological process.
Since somatic cell reprogramming is a complex and dynamic process involving many different transcriptional and epigenetic changes, it is insufficient for somatic cells to regain an alternative biochemical phenotype and morphological properties just by the central role of miRNA in reprogramming [77]. Accordingly, there also must be other molecule participation in the reprogramming biological process. Along with specific TFs, reprogramming factors such as SV40 large antigen and telomerase reverse transcriptase also contribute to the mechanisms for reprogramming. More strikingly, a recent report revealed that vitamin C enhances the efficiency of somatic cell reprogramming [78]. Consequently, future studies to identify new regulators and target genes of miRNAs will provide a better understanding of mechanisms underlying miRNA-mediated cellular reprogramming.
Main miRNAs Involved in Neural Reprogramming of Somatic Cells
miRNA-124
miRNA-124 is highly conserved through diverse species. It has been documented as one of the best characterized and most abundant miRNA within the CNS, and accounts for 25–48 % of all brain miRNA [79–81]. MiRNA-124 is expressed in neurons but not in other cells in the CNS such as glial cells and NSCs while also found to be expressed in microglial cells and downregulated in activated microglial cells [82–84]. For NSCs, miRNA-124 expression just begins during the transition from NSCs to neural progenitor cells [85]. Several studies have revealed that within the temporal progression of neurogenesis in subventricular zone (SVZ), miRNA-124 promotes the cell cycle exit of neuroblasts, and its expression at highest level occurs during the terminal neuronal differentiation processes such as neurite outgrowth. Delivery of miRNA-124 by a lentiviral vector into glioblastoma cells inhibits proliferation and induces differentiation [60, 85–87]. Consistently, overexpression of miR-124 in cultured NSCs and embryonic cortical neural progenitors (NPs) induced a neuronal phenotype [88]. On the contrary, inhibiting miR-124 expression in vitro by administrating antisence 2′-O-methyl AMO prevented the commitment for a neuronal fate while the proliferation of NSCs was promoted [86]. Together, these experiments indicate that miRNA-124 plays a pivotal role in regulating neurogenesis in neuron development. Interestingly, Cao and his colleagues found that miRNA-124 is less important for spinal cord neurogenesis during development [89]. Thus, the in vivo role of miRNA-124 remains unclear and controversial.
Considering the importance of miRNA-124 in neurogenesis, numerous miRNA-124 target genes have been found and substantiated. REI silencing transcription factor (REST) has been found as an important target of miRNA-124, and mainly functions as a regulator of neuronal phenotype [90, 91]. MiRNA-124 promotes expression of neural genes by repression of REST. In contrast, REST also downregulates miRNA-124 in non-neural cells, which results in inhibition of expression of neural genes [92]. Another miRNA-124 target is the polypyrimidine tract-binding protein 1 (Ptbp1), a repressor of alternative splicing in non-neural cells. MiRNA-124 promotes a neuronal protranscriptome by targeting Ptbp1, leading to dramatic inhibition of non-neuronal genes [93]. Zhou et al. found that overexpression of pluripotency stem cell-specific miRNA-302/367 cluster, together with two other neuron-specific miRNAs (miRNA-9/9* and miRNA-124) can convert fibroblasts into neurons [63]. In this report, fibroblasts reprogramming and further acquisition of neuronal fate is largely due to miRNA-124 regulating Ptbp1-mediated alternative splicing. Other targets include Jagged1, a ligand of Notch and Sox9. Jagged1 is critical for the maintenance of the self-renewal capacity of NSCs and for inhibiting their differentiation [86, 94, 95]. Liu et al. revealed that introduction of miRNA-124a in neural progenitor cells significantly reduced Jagged1 transcript and protein levels, consequently leading to inactivation of Notch signals, which finally results in cell cycle exit and neuronal differentiation [29]. The report demonstrated the roles of miR-124a-mediated Jagged1 regulation. Sox9, a high mobility group (HMG)-box transcription factor which functions as a pleiotropic actor in numerous terminal differentiation processes, including heart development, sex determination, chondrogenesis, neural crest differentiation, gliogenesis, hair follicle function, pancreas development, prostate development, and retina development [96–98]. Besides, a crucial function of Sox9 is participation in proliferation control. Once miRNA-124 is elevated in somatic cells such as fibroblasts and neuroblasts, cell growth could be significantly suppressed while blocking miRNA-124 could promote cell proliferation, revealing that miRNA-124 can directly regulate the expression of Sox9. In SVZ, miR-124 mediated repression of Sox9 also plays an important role for progression along the SVZ stem cell lineage to neurons. In this sense, Sox9 is involved in regulating neuronal differentiation besides controlling cell proliferation [86].
Importantly, miRNA-124 target genes together with miRNA-124 and other molecules can induce a neuronal program, suggesting that miRNA-124 exerts critical role in establishing and developing a neuronal transcription network within somatic cell reprogramming (Table 1). In the light of these reports regarding biofunctions of miRNA-124 and its target molecules, it is hopeful to develop an approach for cell reprogramming into neural cells using molecular technology.
miRNA-9
miR-9 is also an enriched miRNA within brain [79] and it is evolutionarily conserved [99]. miR-9 usually plays an important role in neuronal proliferation, subtype specification, differentiation, and migration [5, 100, 101]. Notably, miRNA-9 also maintains the balance of NPs proliferation and migration to complete successful maturation process during CNS development [5]. Along with diverse roles in different species, miRNA-9 has been shown to have region diversity in brain axis [102]. MiRNA-9 inhibits neural precursor migration by targeting stathmin (Stmn1), resulting in a more mature NP fate [5]. Other miRNA targets include Gsh2 that plays an early role in the differentiation and/or migration of the ventral telencephalon known as the medial ganglionic eminence (MGE) and the lateral ganglionic eminence (LGE) and TLX that regulate stem cell fate [103]. TLX repression accelerates stem cell differentiation [104, 105]. In addition, a recent report showed that miR-9 controls NPs proliferation by targeting Cyclin D1 mRNA [106].
Due to its high degree of conservation, miR-9 in the zebrafish also keeps the balance between maintenance and differentiation of NSCs and NPs by targeting various genes of the FGF signaling and the anti-neurogenic genes her5 and her9, leading to the establishment of the midbrain to hindbrain boundary [107]. Hairy1 has also been suggested as a miR-9 target on mediating cell proliferation [102]. In accordance to Gsh2, Forkhead box g1 (Foxg1) in vertebrates is a nuclear-cytosolic transcription factor essential for the development of telencephalon, cell migration, and cerebral cortex patterning and layering [108, 109]. During early phases of cortical development, Foxg1 controls the rate of neurogenesis by keeping progenitor cells in a proliferative state and by inhibiting their differentiation into neurons [110]. By overexpression and knockdown of miRNA-9, it has been demonstrated that apart from cortical and bulb development, miRNA-9 also participates in regulating development of motor neurons in spinal cord and pre-ganglionic autonomic motor neurons by targeting Foxg1 [110]. Strikingly, miRNA-9 has reciprocal actions with REST. REST suppresses neuronal genes in non-neuronal cells by binding a conserved repressor element (RE1) in neuronal gene loci and recruiting the corepressor complex containing histone deacetylases and methyl CpG-binding protein MeCP2 [111, 112]. Overexpression of miRNA-9 promotes transition of progenitors to post-mitotic neurons. Thus, downregulation of REST during cell reprogramming enhances neuronal gene expression. In addition to REST and co-REST, the expression of BAF53a in human fibroblasts can be repressed by miR-9/9*-124 [112, 113]. Blocking BAF53a results in neuronal conversion of fibroblasts, which showed similar results as repressing the expression of REST, Co-REST, or PTBP1 [112, 114]. This report suggests that these neuron-specific (n)BAFs (BAF53a BAF45a) targeted by miR-9/9* are essential for post-mitotic functions [112]. Consequently, it has been validated that miR-9/9* operates programmatically on multiple targets. However, the functions of miR-9 distinct target genes still depend on the temporality of the CNS development, different neural cells differentiation programs and even between different organisms.
Let-7
Let-7 is one of the first miRNA discovered in C. elegans and it is conserved in human and other species [115]. It has been documented that Let7 is the miRNA family with the highest expression in NSCs/NPs [83]. There are distinct mature forms of the miRNA encoding from Let7a to Let7i [116]. Generally, Let7 mainly participates in early neurogenesis, neural differentiation and fate determination, albeit the different roles of Let7 in the CNS were characterized (Table 1). Let7a mainly maintains NSCs self-renewal capacity by targeting lin-28 which inhibits pre-let-7 processing by Dicer in embryonic stem cells (ESCs) [117]. Let7b exerts over different genes involved in cell cycle control such as CycleD1 and TLX [117]. Overexpression of Let7b in NSCs causes reduced proliferation and an increase in neural differentiation [117]. In somatic cells, Let7c and Let7d have similar roles in regulating genes involved in self-renewal. Strikingly, downregulation of TLX promotes NSC proliferation and inhibits neural differentiation and migration [33, 118]. The bioprocess can be mediated by let-7d. Despite several let-7 target genes were unveiled, further studies are required to investigate the let-7 signaling pathways for regulation of cell reprogramming. Based on these reports, it is likely that by using miRNA sponge technology, somatic cells can be induced into neuronal lineage or NSCs commitment [117].
miRNA-25
MiRNA-25 belongs to part of the evolutionary conserved miR-106b∼25 cluster. This cluster is located within the intronic region of the Mcm7 gene and codes for three different mature miRNA species: miRNA-106b, miRNA-93, and miRNA-25 [119, 120]. The miR-106b∼25 cluster has been reported to have proliferative and anti-apoptotic effects. Emerging data have indicated that the miRNA-106b∼25 cluster plays a critical role in adult NSC proliferation [121, 122]. The effect of miRNA-25 on proliferation results from direct regulation of the cell cycle inhibitor p57, and p57 was indeed identified as miRNA-25 target gene. As is known, p57 is a Cip/Kip family member of cyclin-dependent kinase (Cdk) inhibitors that block the cycle progression through all stages of G1/S, thereby functioning as a brake on the cell cycle [123]. In addition, miRNA-25 was also shown to participate in reprogramming, stem cell self-renewal, and maintenance of pluripotency [122, 123]. Moreover, bioinformatics analysis and relative experiments revealed that miRNA-25 directly regulates Wwp2, an E3 ubiquitin ligase that targets Oct4 for ubiquitination, Fbxw7, an important regulator of c-Myc, Klf5, and other important factors [124]. Consistent with this, p53-, TGFβ-, insulin/IGF-, and nitric oxide-signaling as promising putative target genes for miRNA-25 were thought as important regulators for neural differentiation and maintenance of stemness [122]. Nevertheless, further experiments are required to substantiate these targets’ biofunctions and their interplays in cell reprogramming.
miRNA-137
MiRNA-137 is a short non-coding RNA molecule that functions to regulate expression levels of other genes by various mechanisms. MiR-137 is located on human chromosome 1p22 and has been implicated to act as a tumor suppressor in several cancer types. In nervous system, apart from the adult NSCs, the expression of miRNA-137 was found in different brain regions including amygdala, hippocampus, cerebral cortex, and hypothalamus [125, 126]. Recently, it has been demonstrated that miRNA-137 regulates NSC proliferation and differentiation in mouse embryonic stem cells, and neuronal maturation, including promotion of dendrite morphogenesis and spine density in hippocampus neuronal development and maturation [85, 127]. To date, several target genes of miR-137 have been documented and shown to exert crucial roles in various human cancers, cell cycle signaling, and mouse embryonic stem cell development (Table 1) [128, 129]. Balaguer et al. identified a list of 32 genes targeted by miRNA-137 [130, 131]. Among these targets, LSD1 (lysine-specific histone demethylase 1A) has been shown to be directly downregulated by miRNA-137 via binding to its 3′-UTR. Interestingly, LSD1 has been demonstrated as a transcription repressor of TLX, implying the role of miRNA-137 in maintaining the undifferentiated phenotype [126, 131]. In addition, several studies have identified Cdc42 (cell division cycle 42), a well-known member of the Rho GTPase family, as direct miRNA-137 target gene, which is associated with the induction of G1 cell cycle arrest resulting in neuronal differentiation of NSCs and decreased cell growth and/or proliferation of glioblastoma and colorectal cancer cells [126]. Thus, by inhibiting the Cdc42/PAK signaling pathway, miRNA-137 can decrease proliferation, invasion, and G0/G1 cell cycle progression of tumor cells such as colorectal cancer cells [132]. In adult, miR-137NA also directly inhibits cyclin-dependent kinase 6 (CDK6) expression and decreases the level of phosphorylated RB, a known CDK6 downstream target. This is proposed to be the mechanism whereby miR-137 promotes differentiation and inhibits proliferation of adult mouse NSCs, oligodendroma-derived SCs, as well as human glioblastoma multiforme-derived SCs [85]. Besides, miR-137 targets Mib1 (Mind Bomb-1), an ubiquitin ligase known to be important for neurogenesis and neurodevelopment [133]. In ESCs, Jarid1b (also known as KDM5b, a histone H3 Lysine 4 demethylase) has recently been shown to be another direct target of miR-137. Jarid1b is frequently expressed during mouse embryonic development and maintains ESC undifferentiation state. By suppressing Jarid1b protein level, miR-137 is believed to play a role in inhibiting the differentiation of ESCs [134]. Consequently, miRNA-137 expression must be highly regulated to maintain the correct proliferation without losing the differentiation potential of NSCs. In order to maintain the balance of the proliferation and differentiation of NSCs, a feedback loop was formed among miRNA-137 and LSD1. The feedback loop, thus, controls dynamics between the proliferation and differentiation during CNS development.
miRNA-134
miRNA-134 is a family of microRNA precursors found in mammals, belongs to the miRNA379-410 cluster and it is specifically expressed in brain tissues [79, 135]. In rats, it is localized specifically in hippocampal neurons and may indirectly regulate synaptic development [136, 137]. High expression of miRNA-134 could result in the apoptosis of neuronal cells, and the expression level of miRNA-134 has been shown to be significantly elevated in the rat brain after ischemia/reperfusion [138]. Depending on the stage of neural differentiation process, this miRNA has different targets and effects. Overexpression of miRNA-134 in cultured cortical neural precursor cells enhances their proliferation and counteracts apoptosis through regulating Chrdl-1 and Dcx [139]. HSPA12B was validated to be a target of miR-134, downregulation of miRNA-134 could induce neuroprotection against ischemic injury in vitro and in vivo by negatively upregulating HSPA12B protein expression [140]. Additionally, other members of miRNA379-410 cluster miR-369-3p, miR-496, and miR-543 have been shown to regulate cell proliferation in the developing CNS [135]. These miRNAs negatively regulates N-cadherin, leading to premature neuronal migration [141]. This trio of miRNA fine-tunes their target levels to control cell proliferation and achieve other cell type-specific functions in the biological process. The summarized functions and targets of miRNA-134 are shown in Table 1.
miRNA-200
The miR-200 family consists of miR-200a, miR-200b, miR-200c, miR-141, and miR-429, which form two different clusters located in two genomic regions, respectively. Among them miR-200a, miR-200b, and miR-429 are found in one cluster, and miR-200c and miR-141 in another cluster [142, 143]. Members of the miR-200 family are highly enriched in epithelial tissues, play an essential role in tumorigenesis, development, progression, and intravasation through targeting a variety of important proteins. MiRNA-200 targets the E-cadherin transcriptional repressors zinc finger e-box bind homeobox 1 (Zeb1) and 2 (Zeb2), increasing cell motility and inducing EMT [142, 144, 145]. In nerve system, miR-200 family (in particular, miR-200c) has been shown to promote cell cycle exit and neuronal differentiation of ventral midbrain/hindbrain (vMH) neural progenitors by downregulating Sox2 and E2F3 expression and that miR-200c directly targets the Sox2 and E2F3 mRNAs via specific binding sequences (BSs) in their 3′-UTRs [146]. Sox2 is required in a dose-dependent manner for maintaining the multipotency of neural stem/progenitor cells and inhibiting their cell cycle exit and differentiation into neurons or glial cells (Table 1) [147–149]. Moreover, Choi and colleagues found that the miR-200 family, which is among the most highly and most specifically miRNA subset expressed in the developing olfactory system, has potential functions mediating differentiation and cell fate determinant of progenitor cells in the olfactory system [150]. Knocking down the expression of mature miR-200 family members led to impairment of mature olfactory marker expression and expansion of the early marker foxg1 in the olfactory primordium, suggesting that the loss of miR-200 family function disrupts terminal differentiation of olfactory progenitor cells [150]. In addition, the expression of the miR-200 family initiates neuronal differentiation by downregulating Sox2 and Klf4 genes [151], and regulates the reprogramming of neuroepithelial cells into NSCs [152]. More interestingly, the miR-200 family, by targeting Zebs, affects ESC differentiation choices between ectodermal fate and meso-endodermal fate at an early stage. These reports show complexity of miRNAs regulatory network for modulating neural specification and/or reprogramming.
miRNA-302/367
MiRNA-302/367 is highly-richened in early embryonic development and rapidly declines after differentiation [153], and several lines of evidence demonstrated that miRNA-302/367 serves as upstream pluripotency regulator to modulate the expression of Oct4, Sox2, Nanog, and other embryonic TFs [64, 154, 155]. MiRNA-302/367 targets multiple epigenetic factors, leading to global demethylation. Global DNA demethylation occurs at the promoter binding site of several ESC-specific TFs during the 1–8 cell stages of early zygotes. MiRNA-302 silences lysine-specific histone demethylases 1 and 2 (AOF1 and AOF2) and methyl CpG-binding proteins 1 and 2 (MECP1-p66 and MECP2), leading to co-activation of pluripotency-promoting genes [48, 68]. Of note, miR-302/367 also directly targets NR2F2, a member of the nuclear orphan receptor family of TFs and a negative regulator of Oct4 [73]. Remarkably, a number of studies have also shown that Oct4, Nanog, and Sox2 bind to the promoter regions of miR-302/367 and increase its expression level [74]. miRNA-302/367 expression induces global demethylation and suppresses NR2F2, two events that indirectly activate Oct4 expression, which in turn elevates miR-302/367 levels. This reciprocal cycle increases cellular levels of miRNA-302/367 and Oct4, which leads to the co-activation of other transcription regulators, such as Sox2 and Nanog [44, 156]. Overexpression of miRNA-302/367 can result in global demethylation and coexpression of Oct4, Sox-2, and Nanog contributing to human iPSCs (Table 1). Consistently, overexpression of pluripotency stem cell-specific miRNA-302/367 cluster, together with two other neuron-specific miRNAs (miRNA-9/9* and miRNA-124) induced conversion of fibroblasts into neurons [157]. Likewise, Ghasemi-Kasman found that adult human astrocytes could be reprogrammed to neuroblasts by miRNA-302/367, both in vivo and in vitro. In the presence of valproic acid (VPA), reprogramming via miRNA-302/367 converts adult astrocytes to neuroblasts by targeting epigenetic factor Oct4 [158]. Additionally, targets of miRNA-302b and miRNA-372 include transforming growth factor beta receptor II (TGFBR2) and ras homolog gene family member C (RHOC), which correlates with increased efficiency of iPSC induction [159]. Also, miRNA-302/367 targets cell cycle regulators cyclin E-CDK and cyclin D-CDK4/6, leading to an inhibition of the G1 to S phase transition [160]. Taken together, miRNA-302/367 plays a pivotal role in self-renewal and multiple differentiation potential maintenance in pluripotent stem cells, and also exert crucial role in somatic cell reprogramming (Table 1). Moreover, given that each miRNA of miRNA-302/367 owns a relative conserved region, miRNA-302/367 also promote mesenchymal epithelial transition (MET) through inhibiting target gene translation, inhibiting cell cycle, regulating differentiation-associated gene expression and epigenetic modification, finally acts well in somatic cell reprogramming [161].
Future Perspectives
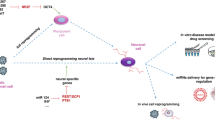
Rapidly accumulating data significantly broaden our knowledge on miRNA regulation and function in somatic cell reprogramming. Based on evidence of the successful generation of NSCs or neurons from somatic cells using a miRNA-mediated strategy, it seems that the numerous diverse miRNA families, especially neural cell-specific miRNAs, can effectively induce reprogramming events similar to those of Yamanaka factors or other small molecules. In comparison, the primary advantage of miRNAs is that they directly and efficiently alter the adult transcriptome and proteome, leading to increased efficiency and decreased time-consumption for cell reprogramming. More interestingly, NSC-specific miRNAs target various cell cycle regulators and effectors that not only induce the alteration of cell cycle, but also alter cell growth and biochemical phenotypical features, leading to cell reprogramming. Although the molecular mechanism by which NSC-specific miRNAs re-direct somatic cells to acquire pluripotency needs to be intensively explored, miRNA-based reprogramming could prove useful for the refinement of current reprogramming techniques and may provide new strategies for future NSCs and neural cell generation to treat neurodegenerative diseases and CNS injury. Considering the importance of miRNAs in neuroregenerative medicine, miRNAs have thereby become the focus in the exploration of regulating the spatial and temporal expression of genes crucial for fine-tuning of signaling pathways during reprogramming and/or development. Nevertheless, due to the complexity of reprogramming process, miRNA-mediated conversion of cell phenotype by targeting repression must rely on coordination with other molecules such as TFs and reprogramming factors. Meanwhile, a regulatory feedback loop exists among miRNAs, target molecules, and TFs to orchestrate the reprogramming. Efficient reprogramming of somatic cells into NSCs including neurons may be performed via several mechanisms (Fig. 2) but there is still a long way to go for comprehensive understanding of miRNAs regulatory mechanism as well as their applications in clinical therapy. In summary, there is little doubt that the strategy of miRNA-mediated reprogramming may provide a basis for future neural cell generation and drug screening for cell-based therapies for SCI and neurodegenerative disorders.
References
Bartel DP (2004) MicroRNAs: genomics, biogenesis, mechanism, and function. Cell 116(2):281–297
Carthew RW, Sontheimer EJ (2009) Origins and mechanisms of miRNAs and siRNAs. Cell 136(4):642–655
Singh SK (2007) miRNAs: from neurogeneration to neurodegeneration. Pharmacogenomics 8(8):971–978
Li X, Jin P (2010) Roles of small regulatory RNAs in determining neuronal identity. Nat Rev Neurosci 11(5):329–338
Delaloy C, Liu L, Lee JA, Su H, Shen F, Yang GY, Young WL, Ivey KN et al (2010) MicroRNA-9 coordinates proliferation and migration of human embryonic stem cell-derived neural progenitors. Cell Stem Cell 6(4):323–335
He L, Hannon GJ (2004) MicroRNAs: small RNAs with a big role in gene regulation. Nat Rev Genet 5(7):522–531
Lim LP, Lau NC, Garrett-Engele P, Grimson A, Schelter JM, Castle J, Bartel DP, Linsley PS et al (2005) Microarray analysis shows that some microRNAs downregulate large numbers of target mRNAs. Nature 433(7027):769–773
Bartel DP (2009) MicroRNAs: target recognition and regulatory functions. Cell 136(2):215–233
Lee RC, Feinbaum RL, Ambros V (1993) The C. elegans heterochronic gene lin-4 encodes small RNAs with antisense complementarity to lin-14. Cell 75(5):843–854
Almeida R, Allshire RC (2005) RNA silencing and genome regulation. Trends Cell Biol 15(5):251–258
Sinkkonen L, Hugenschmidt T, Berninger P, Gaidatzis D, Mohn F, Artus-Revel CG, Zavolan M, Svoboda P et al (2008) MicroRNAs control de novo DNA methylation through regulation of transcriptional repressors in mouse embryonic stem cells. Nat Struct Mol Biol 15(3):259–267
Takahashi K, Yamanaka S (2006) Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 126(4):663–676
Takahashi K, Tanabe K, Ohnuki M, Narita M, Ichisaka T, Tomoda K, Yamanaka S (2007) Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell 131(5):861–872
Okita K, Nakagawa M, Hyenjong H, Ichisaka T, Yamanaka S (2008) Generation of mouse induced pluripotent stem cells without viral vectors. Science 322(5903):949–953
Takahashi K, Yamanaka S (2016) A decade of transcription factor-mediated reprogramming to pluripotency. Nat Rev Mol Cell Biol 17(3):183–193
Kim J, Efe JA, Zhu S, Talantova M, Yuan X, Wang S, Lipton SA, Zhang K et al (2011) Direct reprogramming of mouse fibroblasts to neural progenitors. Proc Natl Acad Sci U S A 108(19):7838–7843
Han DW, Tapia N, Hermann A, Hemmer K, Höing S, Araúzo-Bravo MJ, Zaehres H, Wu G et al (2012) Direct reprogramming of fibroblasts into neural stem cells by defined factors. Cell Stem Cell 10(4):465–472
Ring KL, Tong LM, Balestra ME, Javier R, Andrews-Zwilling Y, Li G, Walker D, Zhang WR et al (2012) Direct reprogramming of mouse and human fibroblasts into multipotent neural stem cells with a single factor. Cell Stem Cell 11(1):100–109
Zou Q, Yan Q, Zhong J, Wang K, Sun H, Yi X, Lai L (2014) Direct conversion of human fibroblasts into neuronal restricted progenitors. J Biol Chem 289(8):5250–5260
Niu W, Zang T, Smith DK, Vue TY, Zou Y, Bachoo R, Johnson JE, Zhang CL (2015) SOX2 reprograms resident astrocytes into neural progenitors in the adult brain. Stem Cell Reports 4(5):780–794
Kim YJ, Lim H, Li Z, Oh Y, Kovlyagina I, Choi IY, Dong X, Lee G (2014) Generation of multipotent induced neural crest by direct reprogramming of human postnatal fibroblasts with a single transcription factor. Cell Stem Cell 15(4):497–506
Nakajima-Koyama M, Lee J, Ohta S, Yamamoto T, Nishida E (2015) Induction of pluripotency in astrocytes through a neural stem cell-like state. J Biol Chem 290(52):31173–31188
Kim SM, Flaßkamp H, Hermann A, Araúzo-Bravo MJ, Lee SC, Lee SH, Seo EH, Lee SH et al (2014) Direct conversion of mouse fibroblasts into induced neural stem cells. Nat Protoc 9(4):871–881
Lim LP, Lau NC, Weinstein EG, Abdelhakim A, Yekta S, Rhoades MW, Burge CB, Bartel DP (2003) The microRNAs of Caenorhabditis elegans. Genes Dev 17(8):991–1008
Lee Y, Jeon K, Lee JT, Kim S, Kim VN (2002) MicroRNA maturation: stepwise processing and subcellular localization. EMBO J21(17):4663–4670
Lee Y, Ahn C, Han J, Choi H, Kim J, Yim J, Lee J, Provost P et al (2003) The nuclear RNase III Drosha initiates microRNA processing. Nature 425(6956):415–419
Zeng Y, Yi R, Cullen BR (2005) Recognition and cleavage of primary microRNA precursors by the nuclear processing enzyme Drosha. EMBO J 24(1):138–148
Grimson A, Farh KK, Johnston WK, Garrett-Engele P, Lim LP, Bartel DP (2007) MicroRNA targeting specificity in mammals: determinants beyond seed pairing. Mol Cell 27(1):91–105
Liu J, Valencia-Sanchez MA, Hannon GJ, Parker R (2005) MicroRNA-dependent localization of targeted mRNAs to mammalian P-bodies. Nat Cell Biol 7(7):719–723
Zeng Y, Cullen BR (2004) Structural requirements for pre-microRNA binding and nuclear export by exportin 5. Nucleic Acids Res 32(16):4776–4785
Guo L, Lu Z (2010) The fate of miRNA* strand through evolutionary analysis: implication for degradation as merely carrier strand or potential regulatory molecule? PLoS One 5(6):e11387
Westholm JO, Lai EC (2011) Mirtrons: microRNA biogenesis via splicing. Biochimie 93(11):1897–1904
Meza-Sosa KF, Pedraza-Alva G, Pérez-Martínez L (2014) microRNAs: key triggers of neuronal cell fate. Front Cell Neurosci 8:175
Yi R, Qin Y, Macara IG, Cullen BR (2003) Exportin-5 mediates the nuclear export of pre-microRNAs and short hairpin RNAs. Genes Dev 17(24):3011–3016
Razak SR, Ueno K, Takayama N, Nariai N, Nagasaki M, Saito R, Koso H, Lai CY et al (2013) Profiling of microRNA in human and mouse ES and iPS cells reveals overlapping but distinct microRNA expression patterns. PLoS One 8(9):e73532
Pang ZP, Yang N, Vierbuchen T, Ostermeier A, Fuentes DR, Yang TQ, Citri A, Sebastiano V et al (2011) Induction of human neuronal cells by defined transcription factors. Nature 476(7359):220–223
Son EY, Ichida JK, Wainger BJ, Toma JS, Rafuse VF, Woolf CJ, Eggan K (2011) Conversion of mouse and human fibroblasts into functional spinal motor neurons. Cell Stem Cell 9(3):205–218
Ichida JK, Blanchard J, Lam K, Son EY, Chung JE, Egli D, Loh KM, Carter AC et al (2009) A small-molecule inhibitor of tgf-Beta signaling replaces sox2 in reprogramming by inducing nanog. Cell Stem Cell 5(5):491–503
Li K, Zhu S, Russ HA, Xu S, Xu T, Zhang Y, Ma T, Hebrok M et al (2014) Small molecules facilitate the reprogramming of mouse fibroblasts into pancreatic lineages. Cell Stem Cell 14(2):228–236
Yuan X, Wan H, Zhao X, Zhu S, Zhou Q, Ding S (2011) Brief report: combined chemical treatment enables Oct4-induced reprogramming from mouse embryonic fibroblasts. Stem Cells 29(3):549–553
Wang G, Guo X, Hong W, Liu Q, Wei T, Lu C, Gao L, Ye D et al (2013) Critical regulation of miR-200/ZEB2 pathway in Oct4/Sox2-induced mesenchymal-to-epithelial transition and induced pluripotent stem cell generation. Proc Natl Acad Sci U S A 110(8):2858–2863
Lüningschrör P, Hauser S, Kaltschmidt B, Kaltschmidt C (2013) MicroRNAs in pluripotency, reprogramming and cell fate induction. Biochim Biophys Acta 1833(8):1894–1903
Li Z, Yang CS, Nakashima K, Rana TM (2011) Small RNA-mediated regulation of iPS cell generation. EMBO J 30(5):823–834
Card DA, Hebbar PB, Li L, Trotter KW, Komatsu Y, Mishina Y, Archer TK (2008) Oct4/Sox2-regulated miR-302 targets cyclin D1 in human embryonic stem cells. Mol Cell Biol 28(20):6426–6438
Anokye-Danso F, Trivedi CM, Juhr D, Gupta M, Cui Z, Tian Y, Zhang Y, Yang W et al (2011) Highly efficient miRNA-mediated reprogramming of mouse and human somatic cells to pluripotency. Cell Stem Cell 8(4):376–388
Miyoshi N, Ishii H, Nagano H, Haraguchi N, Dewi DL, Kano Y, Nishikawa S, Tanemura M et al (2011) Reprogramming of mouse and human cells to pluripotency using mature microRNAs. Cell Stem Cell 8(6):633–638
Kim BM, Thier MC, Oh S, Sherwood R, Kanellopoulou C, Edenhofer F, Choi MY (2012) MicroRNAs are indispensable for reprogramming mouse embryonic fibroblasts into induced stem cell-like cells. PLoS One 7(6):e39239
Lin SL, Chang DC, Lin CH, Ying SY, Leu D, DT W (2011) Regulation of somatic cell reprogramming through inducible mir-302 expression. Nucleic Acids Res 39(3):1054–1065
Sandmaier SE, Telugu BP (2015) MicroRNA-mediated reprogramming of somatic cells into induced pluripotent stem cells. Methods Mol Biol 1330:29–36
Kubicek S, O’Sullivan RJ, August EM, Hickey ER, Zhang Q, Teodoro ML, Rea S, Mechtler K et al (2007) Reversal of H3K9me2 by a small-molecule inhibitor for the G9a histone methyltransferase. Mol Cell 25(3):473–481
Bar-Nur O, Brumbaugh J, Verheul C, Apostolou E, Pruteanu-Malinici I, Walsh RM, Ramaswamy S, Hochedlinger K (2014) Small molecules facilitate rapid and synchronous iPSC generation. Nat Methods 11(11):1170–1176
Ambros V (2004) The functions of animal microRNAs. Nature 431(7006):350–355
Carroll AP, Goodall GJ, Liu B (2014) Understanding principles of miRNA target recognition and function through integrated biological and bioinformatics approaches. Wiley Interdiscip Rev RNA 5(3):361–379
Choi YJ, Lin CP, Ho JJ, He X, Okada N, Bu P, Zhong Y, Kim SY et al (2011) miR-34 miRNAs provide a barrier for somatic cell reprogramming. Nat Cell Biol 13(11):1353–1360
Melton C, Judson RL, Blelloch R (2010) Opposing microRNA families regulate self-renewal in mouse embryonic stem cells. Nature 463(7281):621–626
Unternaehrer JJ, Zhao R, Kim K, Cesana M, Powers JT, Ratanasirintrawoot S, Onder T, Shibue T et al (2014) The epithelial-mesenchymal transition factor SNAIL paradoxically enhances reprogramming. Stem Cell Reports 3(5):691–698
Kim VN, Nam JW (2006) Genomics of microRNA. Trends Genet 22(3):165–173
Valinezhad Orang A, Safaralizadeh R, Kazemzadeh-Bavili M (2014) Mechanisms of miRNA-mediated gene regulation from common downregulation to mRNA-specific upregulation. Int J Genomics 2014:970607
Ambasudhan R, Talantova M, Coleman R, Yuan X, Zhu S, Lipton SA, Ding S (2011) Direct reprogramming of adult human fibroblasts to functional neurons under defined conditions. Cell Stem Cell 9(2):113–118
Yoo AS, Sun AX, Li L, Shcheglovitov A, Portmann T, Li Y, Lee-Messer C, Dolmetsch RE et al (2011) MicroRNA-mediated conversion of human fibroblasts to neurons. Nature 476(7359):228–231
Li MA, He L (2012) microRNAs as novel regulators of stem cell pluripotency and somatic cell reprogramming. BioEssays 34(8):670–680
Wang T, Shi SB, Sha HY (2013) MicroRNAs in regulation of pluripotency and somatic cell reprogramming: small molecule with big impact. RNA Biol 10(8):1255–1261
Zhou C, Gu H, Fan R, Wang B, Lou J (2015) MicroRNA 302/367 cluster effectively facilitates direct reprogramming from human fibroblasts into functional neurons. Stem Cells Dev 24(23):2746–2755
Kuo CH, Ying SY (2012) Advances in microRNA-mediated reprogramming technology. Stem Cells Int 2012:823709
Ciccone DN, Su H, Hevi S, Gay F, Lei H, Bajko J, Xu G, Li E et al (2009) KDM1B is a histone H3K4 demethylase required to establish maternal genomic imprints. Nature 461(7262):415–418
Wang T, Warren ST, Jin P (2013) Toward pluripotency by reprogramming: mechanisms and application. Protein Cell 4(11):820–832
Gruber AJ, Zavolan M (2013) Modulation of epigenetic regulators and cell fate decisions by miRNAs. Epigenomics 5(6):671–683
Reik W, Dean W, Walter J (2001) Epigenetic reprogramming in mammalian development. Science 293(5532):1089–1093
Thomas M, Lieberman J, Lal A (2010) Desperately seeking microRNA targets. Nat Struct Mol Biol 17(10):1169–1174
Majoros WH, Ohler U (2007) Spatial preferences of microRNA targets in 3′ untranslated regions. BMC Genomics 8:152
Lee I, Ajay SS, Yook JI, Kim HS, Hong SH, Kim NH, Dhanasekaran SM, Chinnaiyan AM et al (2009) New class of microRNA targets containing simultaneous 5′-UTR and 3′-UTR interaction sites. Genome Res 19(7):1175–1183
Brümmer A, Hausser J (2014) MicroRNA binding sites in the coding region of mRNAs: extending the repertoire of post-transcriptional gene regulation. BioEssays 36(6):617–626
Rosa A, Brivanlou AH (2011) A regulatory circuitry comprised of miR-302 and the transcription factors OCT4 and NR2F2 regulates human embryonic stem cell differentiation. EMBO J 30(2):237–248
Marson A, Levine SS, Cole MF, Frampton GM, Brambrink T, Johnstone S, Guenther MG, Johnston WK et al (2008) Connecting microRNA genes to the core transcriptional regulatory circuitry of embryonic stem cells. Cell 134(3):521–533
Tsialikas J, Romer-Seibert J (2015) LIN28: roles and regulation in development and beyond. Development 142(14):2397–2404
Nam Y, Chen C, Gregory RI, Chou JJ, Sliz P (2011) Molecular basis for interaction of let-7 microRNAs with Lin28. Cell 147(5):1080–1091
Park IH, Lerou PH, Zhao R, Huo H, Daley GQ (2008) Generation of human-induced pluripotent stem cells. Nat Protoc 3(7):1180–1186
Esteban MA, Pei D (2012) Vitamin C improves the quality of somatic cell reprogramming. Nat Genet 44(4):366–367
Landgraf P, Rusu M, Sheridan R, Sewer A, Iovino N, Aravin A, Pfeffer S, Rice A et al (2007) A mammalian microRNA expression atlas based on small RNA library sequencing. Cell 129(7):1401–1414
Meza-Sosa KF, Valle-García D, Pedraza-Alva G, Pérez-Martínez L (2012) Role of microRNAs in central nervous system development and pathology. J Neurosci Res 90(1):1–12
Lagos-Quintana M, Rauhut R, Yalcin A, Meyer J, Lendeckel W, Tuschl T (2002) Identification of tissue-specific microRNAs from mouse. Curr Biol 12(9):735–739
Smirnova L, Gräfe A, Seiler A, Schumacher S, Nitsch R, Wulczyn FG (2005) Regulation of miRNA expression during neural cell specification. Eur J Neurosci 21(6):1469–1477
Akerblom M, Jakobsson J (2013) MicroRNAs as neuronal fate determinants. Neuroscientist 20(3):235–242
Visvanathan J, Lee S, Lee B, Lee JW, Lee SK (2007) The microRNA miR-124 antagonizes the anti-neural REST/SCP1 pathway during embryonic CNS development. Genes Dev 21(7):744–749
Silber J, Lim DA, Petritsch C, Persson AI, Maunakea AK, Yu M, Vandenberg SR, Ginzinger DG et al (2008) miR-124 and miR-137 inhibit proliferation of glioblastoma multiforme cells and induce differentiation of brain tumor stem cells. BMC Med 6:14
Cheng LC, Pastrana E, Tavazoie M, Doetsch F (2009) miR-124 regulates adult neurogenesis in the subventricular zone stem cell niche. Nat Neurosci 12(4):399–408
Yu JY, Chung KH, Deo M, Thompson RC, Turner DL (2008) MicroRNA miR-124 regulates neurite outgrowth during neuronal differentiation. Exp Cell Res 314(14):2618–2633
Maiorano NA, Mallamaci A (2009) Promotion of embryonic cortico-cerebral neuronogenesis by miR-124. Neural Dev 4:40
Cao X, Pfaff SL, Gage FH (2007) A functional study of miR-124 in the developing neural tube. Genes Dev 21(5):531–536
Wu J, Xie X (2006) Comparative sequence analysis reveals an intricate network among REST, CREB and miRNA in mediating neuronal gene expression. Genome Biol 7(9):R85
Lunyak VV, Rosenfeld MG (2005) No rest for REST: REST/NRSF regulation of neurogenesis. Cell 121(4):499–501
Conaco C, Otto S, Han JJ, Mandel G (2006) Reciprocal actions of REST and a microRNA promote neuronal identity. Proc Natl Acad Sci U S A 103(7):2422–2427
Makeyev EV, Zhang J, Carrasco MA, Maniatis T (2007) The MicroRNA miR-124 promotes neuronal differentiation by triggering brain-specific alternative pre-mRNA splicing. Mol Cell 27(3):435–448
Liu XS, Chopp M, Zhang RL, Tao T, Wang XL, Kassis H, Hozeska-Solgot A, Zhang L et al (2011) MicroRNA profiling in subventricular zone after stroke: MiR-124a regulates proliferation of neural progenitor cells through notch signaling pathway. PLoS One 6(8):e23461
Farrell BC, Power EM, Mc Dermott KW (2011) Developmentally regulated expression of Sox9 and microRNAs 124, 128 and 23 in neuroepithelial stem cells in the developing spinal cord. Int J Dev Neurosci 29(1):31–36
Lefebvre V, Dumitriu B, Penzo-Méndez A, Han Y, Pallavi B (2007) Control of cell fate and differentiation by Sry-related high-mobility-group box (Sox) transcription factors. Int J Biochem Cell Biol 39(12):2195–2214
Poché RA, Furuta Y, Chaboissier MC, Schedl A, Behringer RR (2008) Sox9 is expressed in mouse multipotent retinal progenitor cells and functions in Müller glial cell development. J Comp Neurol 510(3):237–250
Thomsen MK, Francis JC, Swain A (2008) The role of Sox9 in prostate development. Differentiation 76(6):728–735
Yuva-Aydemir Y, Simkin A, Gascon E, Gao FB (2011) MicroRNA-9: functional evolution of a conserved small regulatory RNA. RNA Biol 8(4):557–564
Tan SL, Ohtsuka T, González A, Kageyama R (2012) MicroRNA9 regulates neural stem cell differentiation by controlling Hes1 expression dynamics in the developing brain. Genes Cells 17(12):952–961
Coolen M, Katz S, Bally-Cuif L (2013) miR-9: a versatile regulator of neurogenesis. Front Cell Neurosci 7:220
Bonev B, Pisco A, Papalopulu N (2011) MicroRNA-9 reveals regional diversity of neural progenitors along the anterior-posterior axis. Dev Cell 20(1):19–32
Shibata M, Nakao H, Kiyonari H, Abe T, Aizawa S (2011) MicroRNA-9 regulates neurogenesis in mouse telencephalon by targeting multiple transcription factors. J Neurosci 31(9):3407–3422
Zhao C, Sun G, Li S, Shi Y (2009) A feedback regulatory loop involving microRNA-9 and nuclear receptor TLX in neural stem cell fate determination. Nat Struct Mol Biol 16(4):365–371
Denli AM, Cao X, Gage FH (2009) miR-9 and TLX: chasing tails in neural stem cells. Nat Struct Mol Biol 16(4):346–347
Jiang JQ, Zhou Z (2013) Removal of pharmaceutical residues by ferrate(VI. PLoS One 8(2):e55729
Leucht C, Stigloher C, Wizenmann A, Klafke R, Folchert A, Bally-Cuif L (2008) MicroRNA-9 directs late organizer activity of the midbrain-hindbrain boundary. Nat Neurosci 11(6):641–648
Otaegi G, Pollock A, Hong J, Sun T (2011) MicroRNA miR-9 modifies motor neuron columns by a tuning regulation of FoxP1 levels in developing spinal cords. J Neurosci 31(3):809–818
Garaffo G, Conte D, Provero P, Tomaiuolo D, Luo Z, Pinciroli P, Peano C, D’Atri I et al (2015) The Dlx5 and Foxg1 transcription factors, linked via miRNA-9 and −200, are required for the development of the olfactory and GnRH system. Mol Cell Neurosci 68:103–119
Clovis YM, Enard W, Marinaro F, Huttner WB, De Pietri Tonelli D (2012) Convergent repression of Foxp2 3ʹUTR by miR-9 and miR-132 in embryonic mouse neocortex: implications for radial migration of neurons. Development 139(18):3332–3342
Laneve P, Gioia U, Andriotto A, Moretti F, Bozzoni I, Caffarelli E (2010) A minicircuitry involving REST and CREB controls miR-9-2 expression during human neuronal differentiation. Nucleic Acids Res 38(20):6895–6905
Packer AN, Xing Y, Harper SQ, Jones L, Davidson BL (2008) The bifunctional microRNA miR-9/miR-9* regulates REST and CoREST and is downregulated in Huntington’s disease. J Neurosci 28(53):14341–14346
Yoo AS, Staahl BT, Chen L, Crabtree GR (2009) MicroRNA-mediated switching of chromatin-remodelling complexes in neural development. Nature 460(7255):642–646
Giusti SA, Vogl AM, Brockmann MM, Vercelli CA, Rein ML, Trümbach D, Wurst W et al (2014) MicroRNA-9 controls dendritic development by targeting REST. Elife 3. doi:10.7554/eLife.02755
Rougvie AE (2001) Control of developmental timing in animals. Nat Rev Genet 2(9):690–701
Wulczyn FG, Smirnova L, Rybak A, Brandt C, Kwidzinski E, Ninnemann O, Strehle M, Seiler A et al (2007) Post-transcriptional regulation of the let-7 microRNA during neural cell specification. FASEB J 21(2):415–426
Rybak A, Fuchs H, Smirnova L, Brandt C, Pohl EE, Nitsch R, Wulczyn FG (2008) A feedback loop comprising lin-28 and let-7 controls pre-let-7 maturation during neural stem-cell commitment. Nat Cell Biol 10(8):987–993
Zhao H, Li M, Li L, Yang X, Lan G, Zhang Y (2013) MiR-133b is down-regulated in human osteosarcoma and inhibits osteosarcoma cells proliferation, migration and invasion, and promotes apoptosis. PLoS One 8(12):e83571
Tanzer A, Stadler PF (2004) Molecular evolution of a microRNA cluster. J Mol Biol 339(2):327–335
Renault VM, Rafalski VA, Morgan AA, Salih DA, Brett JO, Webb AE, Villeda SA, Thekkat PU et al (2009) FoxO3 regulates neural stem cell homeostasis. Cell Stem Cell 5(5):527–539
Kan T, Sato F, Ito T, Matsumura N, David S, Cheng Y, Agarwal R, Paun BC et al (2009) The miR-106b-25 polycistron, activated by genomic amplification, functions as an oncogene by suppressing p21 and Bim. Gastroenterol 136(5):1689–1700
Brett JO, Renault VM, Rafalski VA, Webb AE, Brunet A (2011) The microRNA cluster miR-106b∼25 regulates adult neural stem/progenitor cell proliferation and neuronal differentiation. Aging (Albany NY) 3(2):108–124
Rodríguez-Aznar E, Barrallo-Gimeno A, Nieto MA (2013) Scratch2 prevents cell cycle re-entry by repressing miR-25 in postmitotic primary neurons. J Neurosci 33(12):5095–5105
Lu D, Davis MP, Abreu-Goodger C, Wang W, Campos LS, Siede J, Vigorito E, Skarnes WC et al (2012) MiR-25 regulates Wwp2 and Fbxw7 and promotes reprogramming of mouse fibroblast cells to iPSCs. PLoS One 7(8):e40938
Herzer S, Silahtaroglu A, Meister B (2012) Locked nucleic acid-based in situ hybridisation reveals miR-7a as a hypothalamus-enriched microRNA with a distinct expression pattern. J Neuroendocrinol 24(12):1492–1504
Sun G, Ye P, Murai K, Lang MF, Li S, Zhang H, Li W, Fu C et al (2011) miR-137 forms a regulatory loop with nuclear receptor TLX and LSD1 in neural stem cells. Nat Commun 2:529
Szulwach KE, Li X, Smrt RD, Li Y, Luo Y, Lin L, Santistevan NJ, Li W et al (2010) Cross talk between microRNA and epigenetic regulation in adult neurogenesis. J Cell Biol 189(1):127–141
Bier A, Giladi N, Kronfeld N, Lee HK, Cazacu S, Finniss S, Xiang C, Poisson L et al (2013) MicroRNA-137 is downregulated in glioblastoma and inhibits the stemness of glioma stem cells by targeting RTVP-1. Oncotarget 4(5):665–676
Althoff K, Beckers A, Odersky A, Mestdagh P, Köster J, Bray IM, Bryan K, Vandesompele J et al (2013) MiR-137 functions as a tumor suppressor in neuroblastoma by downregulating KDM1A. Int J Cancer 133(5):1064–1073
Balaguer F, Link A, Lozano JJ, Cuatrecasas M, Nagasaka T, Boland CR, Goel A (2010) Epigenetic silencing of miR-137 is an early event in colorectal carcinogenesis. Cancer Res 70(16):6609–6618
Bemis LT, Chen R, Amato CM, Classen EH, Robinson SE, Coffey DG, Erickson PF, Shellman YG et al (2008) MicroRNA-137 targets microphthalmia-associated transcription factor in melanoma cell lines. Cancer Res 68(5):1362–1368
Liu M, Lang N, Qiu M, Xu F, Li Q, Tang Q, Chen J, Chen X et al (2011) miR-137 targets Cdc42 expression, induces cell cycle G1 arrest and inhibits invasion in colorectal cancer cells. Int J Cancer 128(6):1269–1279
Smrt RD, Szulwach KE, Pfeiffer RL, Li X, Guo W, Pathania M, Teng ZQ, Luo Y et al (2010) MicroRNA miR-137 regulates neuronal maturation by targeting ubiquitin ligase mind bomb-1. Stem Cells 28(6):1060–1070
Tarantino C, Paolella G, Cozzuto L, Minopoli G, Pastore L, Parisi S, Russo T (2010) miRNA 34a, 100, and 137 modulate differentiation of mouse embryonic stem cells. FASEB J 24(9):3255–3263
Rago L, Beattie R, Taylor V, Winter J (2014) miR379-410 cluster miRNAs regulate neurogenesis and neuronal migration by fine-tuning N-cadherin. EMBO J 33(8):906–920
Schratt GM, Tuebing F, Nigh EA, Kane CG, Sabatini ME, Kiebler M, Greenberg ME (2006) A brain-specific microRNA regulates dendritic spine development. Nature 439(7074):283–289
Tai HC, Schuman EM (2006) MicroRNA: microRNAs reach out into dendrites. Curr Biol 16(4):R121–R123
Huang W, Liu X, Cao J, Meng F, Li M, Chen B, Zhang J (2015) miR-134 regulates ischemia/reperfusion injury-induced neuronal cell death by regulating CREB signaling. J Mol Neurosci 55(4):821–829
Gaughwin P, Ciesla M, Yang H, Lim B, Brundin P (2011) Stage-specific modulation of cortical neuronal development by Mmu-miR-134. Cereb Cortex 21(8):1857–1869
Chi W, Meng F, Li Y, Wang Q, Wang G, Han S, Wang P, Li J (2014) Downregulation of miRNA-134 protects neural cells against ischemic injury in N2A cells and mouse brain with ischemic stroke by targeting HSPA12B. Neurosci 277:111–122
Shikanai M, Nakajima K, Kawauchi T (2011) N-cadherin regulates radial glial fiber-dependent migration of cortical locomoting neurons. Commun Integr Biol 4(3):326–330
Gregory PA, Bert AG, Paterson EL, Barry SC, Tsykin A, Farshid G, Vadas MA, Khew-Goodall Y et al (2008) The miR-200 family and miR-205 regulate epithelial to mesenchymal transition by targeting ZEB1 and SIP1. Nat Cell Biol 10(5):593–601
Boese AS, Saba R, Campbell K, Majer A, Medina S, Burton L, Booth TF, Chong P et al (2016) MicroRNA abundance is altered in synaptoneurosomes during prion disease. Mol Cell Neurosci 71:13–24
Park SM, Gaur AB, Lengyel E, Peter ME (2008) The miR-200 family determines the epithelial phenotype of cancer cells by targeting the E-cadherin repressors ZEB1 and ZEB2. Genes Dev 22(7):894–907
Zheng M, Jiang YP, Chen W, Li KD, Liu X, Gao SY, Feng H, Wang SS et al (2015) Snail and slug collaborate on EMT and tumor metastasis through miR-101-mediated EZH2 axis in oral tongue squamous cell carcinoma. Oncotarget 6(9):6797–6810
Peng C, Li N, Ng YK, Zhang J, Meier F, Theis FJ, Merkenschlager M, Chen W et al (2012) A unilateral negative feedback loop between miR-200 microRNAs and Sox2/E2F3 controls neural progenitor cell-cycle exit and differentiation. J Neurosci 32(38):13292–13308
Avilion AA, Nicolis SK, Pevny LH, Perez L, Vivian N, Lovell-Badge R (2003) Multipotent cell lineages in early mouse development depend on SOX2 function. Genes Dev 17(1):126–140
Graham V, Khudyakov J, Ellis P, Pevny L (2003) SOX2 functions to maintain neural progenitor identity. Neuron 39(5):749–765
Pevny LH, Nicolis SK (2010) Sox2 roles in neural stem cells. Int J Biochem Cell Biol 42(3):421–424
Choi PS, Zakhary L, Choi WY, Caron S, Alvarez-Saavedra E, Miska EA, McManus M, Harfe B et al (2008) Members of the miRNA-200 family regulate olfactory neurogenesis. Neuron 57(1):41–55
Pandey A, Singh P, Jauhari A, Singh T, Khan F, Pant AB, Parmar D, Yadav S (2015) Critical role of the miR-200 family in regulating differentiation and proliferation of neurons. J Neurochem 133(5):640–652
Morante J, Vallejo DM, Desplan C, Dominguez M (2013) Conserved miR-8/miR-200 defines a glial niche that controls neuroepithelial expansion and neuroblast transition. Dev Cell 27(2):174–178
Zhang Z, Hong Y, Xiang D, Zhu P, Wu E, Li W, Mosenson J, Wu WS (2015) MicroRNA-302/367 cluster governs hESC self-renewal by dually regulating cell cycle and apoptosis pathways. Stem Cell Reports 4(4):645–657
Ren J, Jin P, Wang E, Marincola FM, Stroncek DF (2009) MicroRNA and gene expression patterns in the differentiation of human embryonic stem cells. J Transl Med 7:20
Kuo CH, Deng JH, Deng Q, Ying SY (2012) A novel role of miR-302/367 in reprogramming. Biochem Biophys Res Commun 417(1):11–16
Rosa A, Spagnoli FM, Brivanlou AH (2009) The miR-430/427/302 family controls mesendodermal fate specification via species-specific target selection. Dev Cell 16(4):517–527
Kuo CH, Ying SY (2013) MicroRNA-mediated somatic cell reprogramming. J Cell Biochem 114(2):275–281
Ghasemi-Kasman M, Hajikaram M, Baharvand H, Javan M (2015) MicroRNA-mediated in vitro and in vivo direct conversion of astrocytes to neuroblasts. PLoS One 10(6):e0127878
Subramanyam D, Lamouille S, Judson RL, Liu JY, Bucay N, Derynck R, Blelloch R (2011) Multiple targets of miR-302 and miR-372 promote reprogramming of human fibroblasts to induced pluripotent stem cells. Nat Biotechnol 29(5):443–448
Lin SL, Chang DC, Ying SY, Leu D, Wu DT (2010) MicroRNA miR-302 inhibits the tumorigenecity of human pluripotent stem cells by coordinate suppression of the CDK2 and CDK4/6 cell cycle pathways. Cancer Res 70(22):9473–9482
Liao B, Bao X, Liu L, Feng S, Zovoilis A, Liu W, Xue Y, Cai J et al (2011) MicroRNA cluster 302-367 enhances somatic cell reprogramming by accelerating a mesenchymal-to-epithelial transition. J Biol Chem 286(19):17359–17364
Akerblom M, Sachdeva R, Barde I, Verp S, Gentner B, Trono D, Jakobsson J (2012) MicroRNA-124 is a subventricular zone neuronal fate determinant. J Neurosci 32(26):8879–8889
Zhao C, Sun G, Li S, Lang MF, Yang S, Li W, Shi Y (2010) MicroRNA let-7b regulates neural stem cell proliferation and differentiation by targeting nuclear receptor TLX signaling. Proc Natl Acad Sci U S A 107(5):1876–1881
de Chevigny A, Coré N, Follert P, Gaudin M, Barbry P, Béclin C, Cremer H (2012) miR-7a regulation of Pax6 controls spatial origin of forebrain dopaminergic neurons. Nat Neurosci 15(8):1120–1126
Tay YM, Tam WL, Ang YS, Gaughwin PM, Yang H, Wang W, Liu R, George J et al (2008) MicroRNA-134 modulates the differentiation of mouse embryonic stem cells, where it causes post-transcriptional attenuation of nanog and LRH1. Stem Cells 26(1):17–29
Niu CS, Yang Y, Cheng CD (2013) MiR-134 regulates the proliferation and invasion of glioblastoma cells by reducing nanog expression. Int J Oncol 42(5):1533–1540
Acknowledgments
We thank Dr. Sebastian Schmull for the critical reading of the manuscript. This work was supported by the Natural Science Foundation of China (grant nos. 81472098, 81371411, 81571208 YG2014MS45, and YJ2014001). The authors confirm that there has been no financial support for this research that could have influenced its outcome.
Grant information
Grant sponsor: Natural Science Foundation of China; Grant numbers 81371411, 81472098, 81571208, YG2014MS45, YJ2014001.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Yang, H., Zhang, L., An, J. et al. MicroRNA-Mediated Reprogramming of Somatic Cells into Neural Stem Cells or Neurons. Mol Neurobiol 54, 1587–1600 (2017). https://doi.org/10.1007/s12035-016-0115-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-016-0115-9