Abstract
Background
The vascular wall tension (WT) of small cerebral vessels can be quantitatively estimated through the concept of critical closing pressure (CrCP), which denotes the lower limit of arterial blood pressure (ABP), below which small cerebral arterial vessels collapse and blood flow ceases. WT can be expressed as the difference between CrCP and intracranial pressure (ICP) and represent active vasomotor tone. In this study, we investigated the association of WT and CrCP with autoregulation and outcome of a large group of patients after traumatic brain injury (TBI).
Methods
We retrospectively analysed recordings of ABP, ICP and transcranial Doppler (TCD) blood flow velocity from 280 TBI patients (median age: 29 years; interquartile range: 20–43). CrCP and WT were calculated using the cerebrovascular impedance methodology. Autoregulation was assessed based on TCD-based indices, Mx and ARI.
Results
Low values of WT were found to be associated with an impaired autoregulatory capacity, signified by its correlation to FV-based indices Mx (R = −0.138; p = 0.021) and ARI (R = 0.118; p = 0.048). No relationship could be established between CrCP and any of the autoregulatory indices. Neither CrCP nor WT was found to correlate with outcome.
Conclusions
Impaired autoregulation was found to be associated with a lower WT supporting the role of vasoparalysis in the loss of autoregulatory capacity. In contrast, no links between CrCP and autoregulation could be identified.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The outcome of a patient following traumatic brain injury (TBI) has been known to be significantly affected by exposures to secondary insults, such as systemic hypotension or intracranial hypertension, which may result in cerebral ischaemia [1–4]. Reaching this condition would be indicative of a loss of cerebral autoregulation, with the small cerebral vessels becoming maximally dilated and no longer pressure-reactive, hence being unable to control cerebral blood flow (CBF) [1, 2, 5, 6]. Even though many indices exist in order to assess the pressure reactivity status of the cerebrovascular bed, these can only be used at a low frequency range where slow waves are present, a range which is suitable for assessing autoregulation [7, 8]. A way to quantitatively describe the vasomotor tone of the arterial bed at a higher frequency is through estimation of vascular wall tension (WT), a parameter which is based on the concepts of critical closing pressure (CrCP) and cerebrovascular impedance.
CrCP denotes the lower threshold of arterial blood pressure (ABP), below which the intraluminal pressure in small cerebral vessels is no longer adequate for keeping them open. Mean ABP becoming lower than CrCP would then result in a collapse of these vessels and cessation of blood flow, characterising then a theoretical situation where a cerebral circulatory arrest can be expected [9]. As CrCP is known to be expressed as a sum of intracranial pressure (ICP) and arterial WT [10], the estimation of WT can be based on the difference CrCP-ICP.
However, the use of CrCP and therefore of WT in clinical applications has been restricted, mainly due to methodological drawbacks, which have been rendering non-physiological negative values [11–14], particularly during vasodilatation (hyperaemia, hypercapnia). We have recently addressed this issue, by creating a new method for estimating CrCP and subsequently WT, based on a model of cerebrovascular impedance [15, 16]. The newly developed methodology has been demonstrated in previous studies to correlate with the traditional methods in various scenarios, from experimental [16, 17] to clinical studies [18]. Therefore, the impedance method has allowed a reliable estimation of WT, which can express the behaviour of the vascular tone quantitatively, with units of pressure (mmHg).
In the present study, we sought to investigate the relationship between WT, CrCP and indices of autoregulation like Mx and ARI in a large sample of patients following head injury. Although the relationship of CrCP and WT with ICP and ABP has been studied previously, both experimentally [16] and during plateau waves of ICP [18], we explored these relationships again in TBI patients, in order to assess the links between WT, CrCP and outcome post head injury.
Materials and Methods
Patients
This study was a retrospective analysis of prospectively collected data from 280 sedated and ventilated TBI patients [median age: 29 years (interquartile range 20–43)], hospitalised in the Neurocritical Care Unit of Addenbrooke’s Hospital (Cambridge, UK) from 2002 to 2011 with 78.2 % being male. Baseline assessment of patients termed an overall median Glasgow coma scale (GCS) of 6 (interquartile range 4–8), with GCS being categorised as severe (GCS: 3–8), moderate (9–12) or mild/minor (13–15) [19]. The patients’ outcome was assessed 6 months after injury, with Glasgow outcome score (GOS) varying from good recovery to mortality, with most of the cases (51.4 %) having a favourable outcome (defined as GOS 4 and 5). Patients remaining in a persistent vegetative state (n = 9 or 3.2 % of total number of patients) were excluded from the study.
The data included continuous recordings of ABP, ICP and intermittent daily transcranial Doppler (TCD) measurements (up to 1-h sessions daily per patient), recorded as part of the standard clinical brain monitoring protocol on our Neurocritical care unit [20]. This material has been, in part presented before, used for various other studies describing cerebral haemodynamics [15, 21–24]. The analysis was performed as part of an anonymous clinical audit, with approval of Neurocritical Care Users Committee at Addenbrooke’s Hospital, Cambridge, UK.
Monitoring and Data Analysis
Monitored signals included ABP, ICP and TCD FV. A pressure monitoring kit (Baxter Healthcare CA, USA; Sidcup, UK) at radial artery was used in order to monitor ABP invasively, zeroed at the level of the heart. An intraparenchymal probe (Codman & Shurtleff, MA, USA or Camino Laboratories, CA, USA) was used in order to monitor ICP. These two transducers have comparable accuracy and frequency response [25]. FV was monitored from the middle cerebral artery with Doppler Box (DWL Compumedics, Germany) or Neuroguard (Medasonic, CA, USA). Acquisition of TCD recordings included monitoring on a daily basis for periods ranging from 10 min up to 1 h starting from the day of initiation of invasive monitoring. The end of the monitoring period was decided on clinical grounds.
The raw data signals were digitised at a sampling frequency of 50 Hz with an analogue–digital converter (DT2814 or DT9801, Data Translation, Marlboro, Mass, USA) and recorded by using either WREC (Warsaw University of Technology), BioSAn (University of Cambridge, UK) or ICM+ (Cambridge Enterprise, Cambridge, UK, http://www.neurosurg.cam.ac.uk/icmplus/) software. Heart rate was calculated using spectral position of the peak associated with the first harmonic of ABP. Cerebral perfusion pressure (CPP) was approximated as ABP-ICP. All calculations, including mean values of ABP, ICP, FV and CPP, were performed over a 10-s long-sliding window.
Calculation of CrCP and WT
For estimation of CrCP, we have used a recently introduced model, which is based on the concept of cerebrovascular impedance [16]. The model input requires signals of ABP, FV and ICP:
In the above formula, CPP refers to mean CPP, CVR denotes cerebrovascular resistance, Ca stands for arterial compliance of the cerebral bed, while HR is the heart rate (beats/s). CVR and Ca can be estimated through the use of an algorithm based on measurements of TCD blood flow velocity and ABP, CPP waveforms, presented in previous studies [23, 26, 27].
Vascular WT was estimated as [10] WT = CrCP − ICP.
The ratio of WT/CrCP describes what part of CrCP can be attributed to active WT.
Autoregulation Indices
In order to associate WT and CrCP to cerebral autoregulation, we used two TCD-based indices: Mx and ARI.
Mx is calculated as a Pearson’s moving correlation coefficient between 30 consecutive samples of averaged (over 10 s) CPP and FV. A positive Mx indicates passive dependence of blood flow on CPP and impaired autoregulation. A zero or negative Mx signifies none or inverse association between CPP and FV, implying the presence of active cerebrovascular responses to changes in CPP and preserved autoregulation [8, 28, 29]. Mx has been demonstrated to be significantly higher, indicating worse autoregulation, in patients with unfavourable outcome [28].
ARI stands for autoregulation index and represents a model for assessing autoregulation based on responses of CBF to changes in ABP [30, 31]. Calculation of ARI is possible through spectral analysis of input/output of hypothetical linear high-pass filter linking changes in FV to ABP, where the impulse response takes ABP as input and FV as output. Possible CBF responses, signifying the status of autoregulation, are presented by an ARI of 0 (loss of autoregulation) to 9 (over reactive autoregulation). It has been shown [32] that ARI is associated with outcome after TBI, with ARI being significantly lower in cases of mortality.
Statistical Analysis
The IBM SPSS Statistics 20 package (Armonk, NY, USA) was used for statistical analysis of the data, which consisted of recording averages. The analysis included bivariate correlations with standardised parameters following normalisation, with R being the Pearson correlation coefficient and the level of significance (p value) set at 0.05. Results in Tables 1 and 2 are presented in a mean value ± standard deviation (SD) format. Normal distribution was established with the Shapiro–Wilk test. One-way Anova tests were used to test the distribution of parameters across different types of categories.
Results
Mean values and standard deviations (mean ± SD) of monitored signals (ICP, ABP, CPP, FV) and derived parameters CrCP and WT for the full cohort of patients are presented in Table 1. On average, WT amounts to 59 % of total CrCP, assessed through the ratio of WT/CrCP.
Relationship with Autoregulation
Overall, WT was found to be inversely correlated to Mx (R = −0.138; p = 0.021), meaning that an increased Mx—or impaired autoregulation—was associated with a decreased WT (vasodilatation). No relationship could be established, however, between Mx and CrCP (p = 0.543).
WT was also found to be correlated to the second TCD-based autoregulation index, ARI (R = 0.118; p = 0.048): a low ARI (impaired autoregulation) was associated with a lower WT, thus confirming the character of the relationship of WT with autoregulation as assessed with Mx. Similarly, as with Mx, no relationship could be established between ARI and CrCP (p = 0.164).
Dichotomising outcome groups, the described overall relationship between Mx and WT is sustained in cases of favourable outcome (R = −0.168; p = 0.044; N = 144) but lost in unfavourable outcome (R = −0.086; p = 0.317; N = 136). Similarly, this was also the case for non-fatal (R = −0.136; p = 0.045; N = 217) and fatal outcomes (R = −0.127; p = 0.323; N = 63).
Relationship with Clinically Monitored Physiological Parameters
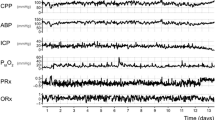
WT and CrCP were found to be directly related to ABP (R = 0.428 and R = 0.645, respectively; p < 0.000001 for both), with a rise in ABP resulting in compensatory vasoconstriction, and hence in increases in both WT and CrCP. A decrease in ABP would have an opposite effect, presented in the clinical example of arterial hypotension of Fig. 1: a decrement of ABP caused a vasodilatatory response, depicted as a decrease in WT, which subsequently resulted in a decreased CrCP, with ICP remaining relatively unchanged.
Monitoring session of a patient, with recordings of arterial blood pressure (ABP), intracranial pressure (ICP) and blood flow velocity (FV). A decrease in ABP, the start of which is depicted with an arrow, results in a decrease in critical closing pressure (CrCP), which can be explained by a decreasing vascular wall tension (WT) denoting compensatory vasodilatation. Whereas ICP remained unchanged, FV is shown to decrease, indicating that vasodilatation was inadequate for compensating for the decrement of ABP
CrCP was positively correlated to ICP (R = 0.635; p < 0.000001), whereas in the same instance, WT was shown to be inversely associated to ICP (R = −0.248; p < 0.000001; Fig. 2). The latter relationship demonstrates a link between raised ICP and vasodilatation, expressed as a decrease in vasomotor tone and thereby a decrease in WT, as depicted in the clinical scenario of Fig. 3.
Recording from a single patient with monitored signals of intracranial pressure (ICP), arterial blood pressure (ABP) and blood flow velocity (FV). This example demonstrates the behaviour of critical closing pressure (CrCP) and vascular wall tension (WT) during a case of increasing ICP, the start of which is depicted with an arrow. Rising ICP leads to an increase of CrCP and a decrease in WT due to compensating vasodilatation
CPP demonstrated a positive relationship to WT (R = 0.608; p < 0.000001; Fig. 4), and a positive but weaker relationship to CrCP (R = 0.170; p = 0.004268).
In order to examine further the effect that each of clinically monitored parameters independently have on WT and CrCP, we also used partial correlations, where each variable was correlated to WT and CrCP, whilst the others were controlled. All the results confirmed the above statements reached through bivariate analysis.
The relationship between WT and TCD-assessed vertebral blood flow velocity can be demonstrated through changes in PaCO2, illustrated through the example of hypocapnia of Fig. 5. Reduced PaCO2 lead to constriction of vessels and increased WT, resulting in a subsequent increase in CrCP and a decrease in FV.
Recording from a single patient with signals of cerebral blood flow velocity (FV), arterial blood pressure (ABP) and intracranial pressure (ICP) during hypocapnia, resulting in a decrease of FV. The black arrow indicates the beginning of hypocapnia, with baseline PaCO2 starting from 5.36 kPa and then reduced to 4.50 kPa. Gaps left after artefacts removal have been interpolated graphically. Thanks to Dr L. Steiner for sharing PaCO2 data with us
Relationship with Demographics and Outcome
We found no significant differences across different categories of GCS (severe, moderate, mild/minor) for either WT (One-way Anova test: F = 0.955; p = 0.386) or CrCP (F = 2.858; p = 0.059). In the same instance, no relationship of either WT or CrCP with age could be established (R = 0.112; p = 0.073 and R = 0.023; p = 0.709, respectively).
In order to explore the relationship of WT to outcome, we performed examination of its distribution across outcome categories ranging from good recovery to mortality (One-way Anova test). The result was that WT did not present any significant differences across outcome categories (F = 0.288, p = 0.834). CrCP followed the same pattern, presenting no differentiation with outcome (F = 1.280, p = 0.282).
As a further investigation, we dichotomised outcome categories in groups, and we compared differences in WT and CrCP across groups of favourable (good recovery, moderate disability) versus unfavourable (severe disability and fatal outcome) and non-fatal (good recovery to severe disability) versus fatal outcome (Table 2). We found no significant difference for WT or CrCP parameters in either unfavourable or fatal outcome. However, the ratio of WT/CrCP was found to be significantly lower in cases of unfavourable outcome (p = 0.018; Table 2) and presented a non-significant tendency to decrease in fatal outcome (p = 0.084). In the same instance, both ABP and ICP were shown to be significantly increased in patients with worse outcomes (Table 2), resulting in CPP not being differentiated in either unfavourable (p = 0.765) or fatal outcome (p = 0.740).
Discussion
The concept of CrCP has been introduced in the early 1950s and is well known since to be a descriptor of cerebral dynamics [9, 10, 12, 13]. A very important aspect of CrCP is that it has allowed assessment of the behaviour of vasomotor tone, through its association to vascular WT. However, the presence of unreliable, negative values through calculation with past methods has been a serious limitation, discouraging the clinical uptake of both CrCP and WT. The impedance methodology for calculating CrCP has addressed those issues, hence reopening the use of WT estimation to further physiological and clinical scrutiny. On that basis, this study was focused on exploring the association of WT to autoregulation and outcome following head trauma.
Links between Impaired Autoregulation and Low Vascular Tone
Two TCD-based indices were used to prove the significant correlation between WT and autoregulation. The relationship with both Mx and ARI signified impaired autoregulation to be associated with low values of WT, painting a picture of highly decreased vasomotor tone associated with vasodilatation or indeed vasoparalysis. Vessels at this kind of dilated state have lost then their pressure reactivity or else their ability to produce a compensatory response to pressure changes threatening to reduce CBF. This situation has been expressed with a high Mx and worse autoregulation in TBI patients presenting an unfavourable outcome [28]. In our study, the relationship between Mx and WT was lost in cases of worse outcome, therefore further underlying the paralysed state of vessels with impaired autoregulation.
Many authors have discussed the complex relationship between cerebrovascular reactivity and autoregulation, arguing that these two expressions are not completely equivalent and should not be used interchangeably, as vascular responses can occur outside of the autoregulatory limits [29, 33–35]. Our results quantitatively support the grounds of this theory, with the smooth muscle tone, depicted as vascular WT, being shown to correlate significantly but weakly to autoregulation, confirming that vascular reactivity does represent a key element of cerebral autoregulation [33, 34], without, however, the two modules being identical.
Furthermore, the nature of the significant but weak correlation of WT to autoregulation can be better understood through the fact that the autoregulatory indices and WT both describe the status of the cerebrovascular bed, albeit in different frequency bandwidths. The descriptive role of Mx refers to a very low frequency range, where slow waves associated to autoregulation are present [8], and the relationship between pressure and flow is non-linear, as has been demonstrated with the Lassen curve [5]. In contrast, the concept of impedance WT methodology denotes a high-pass filter, working at a higher frequency range where the system is linear, and autoregulation does not react to very fast changes in pressure and flow [36]. Therefore, the autoregulatory and WT parameters do not overlap but rather complement each other, with their significant relationship providing a link that covers a wide frequency spectrum.
Contrary to WT, no association between CrCP and either of the autoregulatory indices could be established. This difference between CrCP and WT could be explained by the fact that the latter describes, by definition, and in a direct way, the behaviour of the vascular wall of distal circulation, which is responsible for autoregulatory responses. On the other hand, CrCP is equally governed by WT as well as ICP, as it represents the sum of the two [10]. The addition of ICP to WT seems to be responsible for ‘diluting’ the presented association between WT and autoregulation. This can be illustrated through the contrasting effect of ICP on WT and CrCP: rising ICP causes CrCP and WT to change in opposite directions, with CrCP increasing and WT decreasing due to compensatory to decreasing CPP vasodilatation.
Outcome Analysis
The association of WT to outcome was evaluated in two ways, with both of them denoting a lack of differentiation for vascular WT between patients of different outcome groups. This finding could be explained based on the changes in physiological parameters presented in worse outcome groups, with both ABP and ICP found to be significantly increased in unfavourable and fatal cases, as seen in earlier studies [22, 37]. As ABP and ICP increase in union, the changes in CPP, estimated as the difference between the two parameters, are insignificant. The association of WT to CPP is strong (Fig. 4), with their relationship displaying a highly significant linear character, demonstrated with a strong correlation between the two, as has been also shown in a recent experimental study [16]. Considering then this direct relationship, the lack of CPP difference across outcome groups could possibly explain the insignificant changes in WT.
This lack of relationship between WT and outcome was presented despite the association of WT to autoregulatory indices, which are known to be related to outcome [28, 32]. This contradiction can be explained by worse outcome cases known to be correlated with impaired autoregulation [28, 32], a condition which was shown with WT to be associated with vasoparalysis. As the relationship between Mx and WT was lost in worse outcome cases, governed by the vessels’ dilated and paralysed state, WT did not follow the association of Mx to outcome.
Subsequently, the behaviour of WT produced a non-significant relationship of CrCP with outcome (confirming the results of an earlier study [38]), even though CrCP presented a tendency to increase, albeit not significantly, in both unfavourable and fatal outcome possibly reflecting the increased ICP in those cases. The interplay between WT, CrCP and ICP in regards to outcome could also be demonstrated through the WT/CrCP ratio, which was shown to be significantly associated with outcome, being lower in worse cases (Table 2). This could reflect that in these conditions where vasoparalysis is present, WT amounts less for CrCP, pronouncing therefore the role of ICP.
How can the WT Parameter be Useful in Clinical Practice?
Calculation of the WT parameter through impedance CrCP can be used for assessing the behaviour of vasomotor tone quantitatively, expressed in millimetres of mercury. The fact that WT has pressure units represents its physiological substance, aiding in its interpretation in clinical reality where pressure parameters like arterial or ICP are being used. Examination of its relationship with clinically monitored parameters revealed a strong association of WT with both ABP and ICP. These findings can be explained by small vessel diameter adjustments in response to pressure changes, pronouncing the role of WT as a surrogate marker of vascular behaviour. Monitoring then of WT in clinical scenarios like arterial hypotension or intracranial hypertension can demonstrate the compensatory vasodilatation through a decrease in the autoregulation-associated WT. More importantly, the use of the reliable impedance WT could aid in a confirmation of a response of the cerebral microvasculature to therapeutical manoeuvres i.e. in cases of rising ICP, hyperventilation would cause the vessels to constrict, a condition which would be depicted quantitatively as an increase in WT—see Fig. 5.
Limitations
Unlike measurements of ABP and ICP, the TCD blood flow velocity signals were intermittent with limited monitoring duration. This comes as a direct consequence of limitations of the current TCD technology, regarding the limited capability of probe holders, which are not well suited for prolonged continuous recording. Continuous TCD measurements were further limited due to requirements of managing head-injured patients in a neurointensive care environment: the need for further assessment of their condition i.e. with transferring of the patient for a CT scan would ultimately result in an unavoidable interruption of TCD monitoring. Under these TCD undersampling circumstances, it is possible that various phenomena related to CrCP parameters-like a significantly low blood flow during diastole for a period of time—may have not been captured and thus may have been excluded from the analysis, therefore potentially affecting the relationship of CrCP with outcome.
Specifically for CrCP, the treatment of the patients itself might have posed a limitation on the scopes of this study. By definition, CrCP is associated to a terminal condition of the cerebral circulation, which would ultimately result in a cease of blood flow. This was not seen in our cohort of patients as ABP never fell below CrCP during the recording sessions. Moreover, in cases of fatal outcome, no terminal state regarding CrCP could be seen, as TCD measurements were not performed in terminal stages.
Conclusion
Impedance CrCP has allowed a reliable estimation of vascular WT which can quantitatively present changes of the cerebral vasculature, expressed in units of pressure. Based on its relationship with two autoregulation indices, a lower vascular WT, signifying vasodilatation, was found to be associated with impaired autoregulation. Neither of CrCP or WT was able to differentiate between outcome groups post TBI, although WT/CrCP ratio did.
References
Overgaard J, Tweed WA. Cerebral circulation after head injury. Cerebral blood flow and its regulation after closed head injury with emphasis on clinical correlations. J Neurosurg. 1974;41:531–41.
Bouma GJ, Muizelaar JP. Cerebral blood flow, cerebral blood volume, and cerebrovascular reactivity after severe head injury. J Neurotrauma. 1992;9(Suppl):S333–48.
Aaslid R. Cerebral hemodynamics. In: Newell DW, Aaslid R, editors. Transcranial doppler. New York: Raven Press; 1992. p. 49.
Manley G, Knudson MM, Morabito D, Damron S, Erickson V, Pitts L. Hypotension, hypoxia, and head injury: frequency, duration, and consequences. Arch Surg. 2001;136:1118–23.
Lassen NA. Autoregulation of cerebral blood flow. Circ Res. 1964;15:201–4.
Kontos HA, Wei EP, Navari RM, Levasseur JE, Rosenblum WI, Patterson JL Jr. Responses of cerebral arteries and arterioles to acute hypotension and hypertension. Am J Physiol. 1978;234:H371–83.
Czosnyka M, Price JD, Williamson M. Monitoring of cerebrospinal dynamics using continuous analysis of intracranial pressure and cerebral perfusion pressure in head injury. Acta Neurochir (Wien). 1994;126(2–4):113–9.
Czosnyka M, Smielewski P, Kirkpatrick P, Menon DK, Pickard JD. Monitoring of cerebral autoregulation in head-injured patients. Stroke. 1996;27:829–34.
Burton AC. Fundamental instability of the small blood vessels and critical closing pressure in vascular beds. Am J Physiol. 1951;164:330–1.
Dewey RC, Pierer HP, Hunt WE. Experimental cerebral hemodynamics-vasomotor tone, critical closing pressure, and vascular bed resistance. J Neurosurg. 1974;41(5):597–606.
Michel E, Hillebrand S, von Twickel J, Zernikow B, Jorch G. Frequency dependence of cerebrovascular impedance in preterm neonates: a different view on critical closing pressure. J Cereb Blood Flow Metab. 1997;17:1127–31.
Panerai RB. The critical closing pressure of the cerebral circulation. Med Eng Phys. 2003;25:621–32.
Kumar A, Schmidt E, Hiler M, Smielewski P, Pickard JD, Czosnyka M. Asymmetry of critical closing pressure following head injury. J Neurol Neurosurg Psychiatry. 2005;76:1570–3.
Puppo C, Camacho J, Yelicich B, Moraes L, Biestro A, Gomez H. Bedside study of cerebral critical closing pressure in patients with severe traumatic brain injury: a transcranial Doppler study. Acta Neurosurg Suppl. 2012;114:283–8.
de Riva N, Budohoski KP, Smielewski P, et al. Transcranial Doppler pulsatility index: what it is and what it isn’t. Neurocrit Care. 2012;17:58–66.
Varsos GV, Richards H, Kasprowicz M, et al. Critical closing pressure determined with a model of cerebrovascular impedance. J Cereb Blood Flow Metab. 2012;33:235–43.
Varsos GV, Richards HK, Kasprowicz M, et al. Cessation of diastolic cerebral blood flow velocity: the role of critical closing pressure. Neurocrit Care. 2014;20(1):40–8.
Varsos GV, de Riva N, Smielewski P, et al. Critical closing pressure during intracranial pressure plateau waves. Neurocrit Care. 2013;18:341–8.
Kolias AG, Guilfoyle MR, Helmy A, Allanson J, Hutchinson PJ. Traumatic brain injury in adults. Pract Neurol. 2013;13:228–35.
Menon DK, Wheeler DW. Neuronal injury and neuroprotection. Anaesth Intens Care. 2005;6:184–8.
Czosnyka M, Matta BF, Smielewski P, Kirkpatrick PJ, Pickard JD. Cerebral perfusion pressure in head-injured patients: a noninvasive assessment using transcranial Doppler ultrasonography. J Neurosurg. 1998;88:802–8.
Czosnyka M, Balestreri M, Steiner L, et al. Age, intracranial pressure, autoregulation, and outcome after brain trauma. J Neurosurg. 2005;102:450–4.
Kim DJ, Kasprowicz M, Carrera E, et al. The monitoring of relative changes in compartmental compliances of brain. Physiol Meas. 2009;30:647–59.
Budohoski KP, Czosnyka M, de Riva N, et al. The relationship between cerebral blood flow autoregulation and cerebrovascular pressure reactivity after traumatic brain injury. Neurosurgery. 2012;71:652–60.
Czosnyka M, Czosnyka Z, Pickard JD. Laboratory testing of three intracranial pressure microtransducers: technical report. Neurosurgery. 1996;38:219–24.
Carrera E, Kim DJ, Castellani G, et al. Cerebral arterial compliance in patients with internal carotid artery disease. Eur J Neurol. 2010;18:711–8.
Kasprowicz M, Czosnyka M, Soehle M, et al. Vasospasm shortens cerebral arterial time constant. Neurocrit Care. 2011;16:213–8.
Czosnyka M, Smielewski P, Piechnik S, Steiner LA, Pickard JD. Cerebral autoregulation following head injury. J Neurosurg. 2001;95:756–63.
Zweifel C, Lavinio A, Steiner LA, et al. Continuous monitoring of cerebrovascular pressure reactivity in patients with head injury. Neurosurg Focus. 2008;25:E2.
Tiecks FP, Lam AM, Aaslid R, Newell DW. Comparison of static and dynamic cerebral autoregulation measurements. Stroke. 1995;26:1014–9.
Panerai RB, Dawson SL, Potter JF. Linear and nonlinear analysis of human dynamic cerebral autoregulation. Am J Physiol. 1999;277:H1089–99.
Panerai RB, Kerins V, Fan L, Yeoman PM, Hope T, Evans DH. Association between dynamic cerebral autoregulation and mortality in severe head injury. Br J Neurosurg. 2004;18:471–9.
Cold GE, Jensen FT. Cerebral autoregulation in unconscious patients with brain injury. Acta Anaesthesiol Scand. 1978;22:270–80.
Paulson OB, Strandgaard S, Edvinsson L. Cerebral autoregulation. Cerebrovasc Brain Metab Rev. 1990;2:161–92.
Czosnyka M, Smielewski P, Kirkpatrick P, Laing RJ, Menon D, Pickard JD. Continuous assessment of the cerebral vasomotor reactivity in head injury. Neurosurgery. 1997;41:11–7.
O’Rourke MF, Taylor MG. Input impedance of the systemic circulation. Circ Res. 1967;20:365–80.
Czosnyka M, Hutchinson P, Balesteri M, Hiler M, Smielewski P, Pickard JD. Monitoring and interpretation of intracranial pressure after head injury. Acta Neurochir. 2006;96:114–8.
Czosnyka M, Smielewski P, Piechnik S, et al. Critical closing pressure in cerebrovascular circulation. J Neurol Neurosurg Psychiatry. 1999;66:606–11.
Acknowledgments
We would like to thank Gordon Stevenson, PhD, from Evelyn Perinatal Imaging Center, Rosie Hospital, Cambridge, for proof reading of this article. GVV is supported by an A. G. Leventis Foundation Scholarship, and a Charter Studentship from St Edmund’s College, Cambridge. AGK is supported by a Royal College of Surgeons of England Research Fellowship, a National Institute for Health Research (NIHR) Academic Clinical Fellowship, and a Raymond and Beverly Sackler Studentship. PJH is supported by an NIHR Research Professorship, the NIHR Cambridge Biomedical Research Centre and has been appointed as the Surgical Specialty Lead for Neurosurgery, Royal College of Surgeons of England Clinical Research Initiative. JDP is supported by the NIHR Cambridge Biomedical Research Centre and an NIHR Senior Investigator Award. MC is NIHR Cambridge Biomedical Research Centre principal investigator on cerebrospinal dynamics.
Conflict of interest
ICM+ Software is licensed by Cambridge Enterprise, Cambridge, UK, http://www.neurosurg.cam.ac.uk/icmplus/. MC and PS have a financial interest in a fraction of the licensing fee. The corresponding author and the rest of the co-authors do not have any conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Varsos, G.V., Budohoski, K.P., Kolias, A.G. et al. Relationship of Vascular Wall Tension and Autoregulation Following Traumatic Brain Injury. Neurocrit Care 21, 266–274 (2014). https://doi.org/10.1007/s12028-014-9971-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12028-014-9971-1