Abstract
Differentiated thyroid cancer (DTC) is an important clinical entity in our population (Novara, Piedmont, Italy) which is characterized by important environmental influences, as iodine deficiency (ID) and subsequent supplementation, thyroiditis and occupational exposure. To evaluate the features of DTC in our population 20 years after the iodine-prophylaxis pondering the effects of the introduction of the new guidelines for diagnosis and management of DTC after 2005. 322 patients [244 females, age: mean (±SD) 53.8 ± 15.8 years] treated for DTC in a tertiary care center between 1997 and 2010 were retrospectively evaluated. Medical history, demographics, and pathological features were considered. Patients were subdivided into two groups: A (n = 139, diagnosis 1997–2005) and B (n = 183, diagnosis 2006–2010). The population of group A showed a mild ID, while normal iodine status was recorded in group B. A significant increase in histological tumor-associated thyroiditis was found from group A to B (p = 0.021). Recurrent or persistent diseases were found to be correlated with lymph nodes metastases and/or a distant disease at diagnosis, stimulated thyroglobulin levels at the first follow-up and an additional radioiodine therapy. Twenty percent of our patients were females employed in textile industries. The tumor-related inflammation and the occupational exposure should be considered as important factors in the pathogenesis of DTC. Further studies are required in order to confirm our findings.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The incidence of the differentiated thyroid carcinoma (DTC) has been increasing in many countries over the last 30 years. In United States, the incidence of thyroid cancer rose up from 4.9/100,000/year people in 1975 to 11.0/100,000/year people in 2006, with a 2.3-fold increase across all patients [1]. The same trend has been reported in Europe from 1973–1977 to 1998–2002, with an increase in the incidence rate ranging between 5.3% in Switzerland and 155.6% in France [2]. Data from the Italian Network of Cancer Registries report nearly 5.2 new thyroid cancers/100,000 males and 15.5/100,000 females each year. Incidence varies significantly across different Italian Regions, being the highest incidence in Emilia Romagna and the lowest documented in Trentino Alto Adige [3].
Data extrapolated from the urban Turin Cancer Registry show that the annual age-standardized incidence rate was 5.1/100,000/year in males and 15.4/100,000/year in females, from 2004 to 2006 [4].
On the other hand, mortality rates associated with DTC has been slowly decreasing in all countries. The coexistent increase in the incidence rate could therefore imply significant improvements in a better recognition of the smallest papillary thyroid cancers (PTC), as a result of the improved diagnostic accuracy achieved by neck ultrasound (US) and US-guided fine needle aspiration biopsy (US-FNAB). Currently, nearly 60–80% of thyroid carcinomas detected are micropapillary (<1 cm in size) and carry an excellent long-term prognosis [1, 5]. Despite the favorable prognosis of these tumors, with a 10-year survival rate exceeding 90.0%, rates of loco-regional and distant recurrences are 5.9 and 1.5%, respectively, and the cancer-related mortality is 1.0–2.0% [1, 6].
Considering the risk factors for DTC, literature data show the importance of environmental factors in the pathogenesis of this tumor [7–12]. In particular, cohort studies [8, 9] have investigated the correlation between iodine supplementation (with the exception of radioactive iodine) and the incidence of new PTCs in goiter endemic areas, with controversial results.
Based on the foregoing, aim of our study was to investigate the clinical and pathological trend of DTC followed in an Italian tertiary care center from 1997 to 2010 (University Hospital “Maggiore della Carità,” Novara). Our center may be representative for an Italian iodine-deficient population 20 years after the start of iodine prophylaxis and exposed to undetectable radiation after the Chernobyl accident in 1986.
Patients and methods
Patients
We retrospectively selected from the hospital chart all patients with thyroid cancer treated and followed at our institution (University Hospital “Maggiore della Carità,” Novara, Italy) between July 1997 and July 2010, following as inclusion criteria: (1) patients who had an histological diagnosis of PTC or follicular thyroid carcinoma (FTC); (2) available information for age at diagnosis, gender, coexisting thyroid disease, and median urinary iodine concentration (UIC); (3) patients resident from birth in a iodine-deficient geographic area and were invited to adopt a iodized salt prophylaxis program in the last years [13]. Environmental and occupational exposures were identified through a direct interview.
Patients with history of neck irradiation, poorly differentiated, anaplastic, medullary carcinomas and secondary tumors were excluded from the database.
The population sample (n = 322) was subsequently divided into two groups: group A, patients with DTC diagnosed between 1997 and 2005; group B, patients with DTC diagnosed between 2006 and 2010. The rationale for the division into two groups was based on the introduction in the clinical practice of the new guidelines for the diagnosis and management of thyroid carcinoma after 2005 [14, 15]. The last date for the enrolment was July 30, 2010.
Moreover, in 103/322 patients we reported the short-term follow-up (defined as the first 5 years after diagnosis) data, because all the patients had the following inclusion criteria:
-
thyroglobulin (Tg) evaluation, obtained after endogenous or exogenous stimulation with recombinant human thyroid stimulating hormone (rhTSH);
-
neck US evaluation;
-
information about the radioiodine therapy when performed;
-
information about 18F-fluoro-deoxyglucose-positron emission tomography (FDG-PET) uptake performing when patients showed detectable Tg levels, but negative US and total body scintigraphy (TBS) with I131 (TBS).
Patients were thus considered as disease free when Tg and antithyroglobulin antibodies (TgAb) were undetectable, and imaging studies were negative, during the endogenous or exogenous TSH stimulation. Recurrence was diagnosed on the evidence of disease status, documented at least 6 months after the previous definition of a disease free condition. Persistence condition was defined when Tg levels persisted at detectable levels and/or regional lymph nodes or distant metastases were present.
Methods
Cytology and histology
The cytology specimens were evaluated and classified according to the international guidelines [16].
Histological slides were reviewed by two independent pathologists for the purposes of this study. For all cases, tumor-associated thyroiditis was assessed. The tumor size, number of foci, focality, extension, presence of loco-regional, and/or distant metastases were reported and classified according to the 2002 tumor-node-metastases (TNM) system [17].
Thyroid function test and urinary iodine
Serum TSH, Tg, TgAb, and thyroid peroxidase (TPOAb) were measured with the amplified chemiluminescence method (Immunolite 2000, DPC, Los Angeles, USA). Tg assays had a functional sensitivity of 0.6 ng/ml.
All the analyses were automatically performed using “ADVIA Centaur” (Siemens) in the Hospital’s chemistry laboratory.
Urine samples were kept covered at 4°C in deiodized tubes and analyzed within 30 days from the collection. UIC was determined by using methods recommended by World Health Organization (WHO) and International Council for Control of Iodine Deficiency Disorders (ICCIDD). Criteria used for assessing iodine deficiency (ID) were as follows [18]:
-
Severe ID: UIC < 20 μg/l;
-
Moderate ID: UIC = 20–9 μg/l;
-
Mild ID: UIC = 50–99 μg/l;
-
No deficiency: UIC ≥ 100 μg/l
Neck ultrasound
US was routinely performed in all patients.
The study was performed with a “Color Doppler Toshiba Apparatus” equipped with a linear transducer of 7.5 MHz. The hypoechogenicity, in the presence of an irregular peripheral halo, microcalcifications and an intranodular blood flow, were classified as US risk factors [19]. Cervical lymphadenopathies and their US features were also evaluated.
I131 total body scintigraphy
Therapeutic TBS was performed in other referral tertiary care centers using a total body scanner (E. CAM 180 dual detector γ camera, Siemens Medical System). A therapeutic dose of I131 (3,700–5,500 MBq) was administered, and repeated until post-therapeutic TBS, FDG-PET or US were negative, and until Tg levels were undetectable.
Diagnostic TBS was performed at the Service of Nuclear Medicine of our center, 2–3 days after the administration of rhTSH, using a diagnostic dose of I131 (74–185 MBq).
Scans were performed in anterior and posterior projection 5–7 days after administration of the radionuclide.
18F-fluoro-deoxyglucose-positron emission tomography
FDG-PET/CT scans were obtained by Biograph Hirez (pico-3D) PET/CT scanner (Siemens Medical Solutions). Patients were intravenously injected with 3–4 MBq/kg of 18F-FDG according to standard protocols.
The examination was only performed if fasting blood glucose levels were <140 mg/dl. According to the body mass index of each patient, the average duration of the FDG-PET/CT image acquisition was about 15 min, followed by emission tomographic images reconstruction.
Statistics
Data were expressed as mean ± SD or percentages. The distributions of dichotomous variables were evaluated using the Chi-square test. The association between the factors for the variables significantly associated in the univariate analysis was assessed with the multinomial logistic regression using the stepwise method with 95% confidence intervals. p values <0.05 were considered significant. Computations have been performed using SPSS 17.0 (SPSS Inc., Chicago, IL, USA).
Results
Clinical features
139 patients with DTC were included in group A and 183 in group B. The females gender was the most represented among the two groups; the mean age at diagnosis was 53.5 ± 15.8 years with any difference between the two groups.
Table 1 summarizes patients’ epidemiological and clinical–pathological features.
Median UIC was 90.0 μg/l in group A, and 136.0 μg/l in group B, indicating mild and absence of deficiency, respectively.
Regarding patients’ occupational exposures, a predominance of patients was females employed in textile industries (20.0%).
Goiter was the most common coexisting benign thyroid disease associated with DTC. It was found in 82.7% patients in group A and in 74.9% in group B. Detectable TPOAb levels were found in 14.4% patients in group A and 24.0% in group B.
Cytological examination was the first suspicion of malignancy (THY 4 and 5) then confirmed by histology in 28.0 and 36.6% of patients in group A and B, respectively; 7.3% underwent surgery for an indeterminate cytological diagnosis (THY 3) (5.7% group A, 8.7% group B).
In the 61.5% (66.3% group A, 54.7% group B) of patients the diagnosis of malignancy was made after surgery. In these cases, we reviewed the missing pre-operative diagnosis. In group A, FNAC was not performed in the 35.5% of patients, while the 30.8% of cases showed a non-diagnostic results by FNAC (THY1) because performed only by palpation without US help. In group B, FNAC was not performed in the 16.9% of patients undergoing surgical therapy for multinodular compressive goiter (defined as “incidentalomas”); the rate of non-diagnostic result (THY 1) is reduced to a 19.3% when US-guided FNAC was performed. In these cases, one of the main reasons for non-diagnostic specimens is the inclusion of aspirates arising from cysts. False negative (THY2) in these group was 8.3%. Again, 10.2% of malignancy found in the gland outside the nodule analyzed by FNAC, consisting of papillary microcarcinoma (again defined as “incidentalomas”). Interestingly, lymphocytic thyroiditis was mainly observed in these patients.
Total thyroidectomy was performed in 92.6% of cases, with any difference between the two groups, even in the presence of malignant cytological diagnosis in nodule <1 cm.
Histo-pathological features
The histo-pathological features of the 322 cases of DTC are shown in Table 2.
PTC was the most frequent diagnosis in both groups (89.9% in group A and 86.9% in group B); in particular, the classical variant was the most represented histological type, with 42.4% in group A and 42.8% in group B. Relatively to the tumor size, microcarcinomas (<1 cm) represented the majority of cases in both groups (42.4% in group A, 43.2% in group B); small cancers (<2 cm) were more represented in group B than in group A (31.1 and 25.9%, respectively), but the increase in frequency was not significant.
We also found the lymphocytic infiltration associated to DTC, with a typical focal and peripheral zone distribution. This co-staining showed an interesting increase over years (14.4% in group A and 24.0% in group B, p = 0.021).
Short-term follow-up
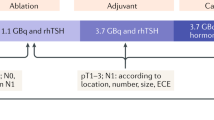
After the initial surgery, all patients of group A and 60.0% of group B, considered at a moderate-high risk of recurrence, underwent radioiodine therapy (p = 0.08).
219/322 patients were lost at follow-up because they were followed in other centers or did not need a strict follow-up. Thus, 103 patients out of 322 (23 in group A and 80 in group B) are currently followed-up in our care center (months of follow-up: 37.3 ± 33.1; group A 90.0 ± 26.1 months; group B 22.4 ± 14.3 months). Detectable TgAb levels were found in 5.2% of patients in group A and 22.5% in group B (p = 0.079).
Considering the change in the follow-up management after 2005, the main differences between group A and group B concern:
-
(1)
the method of choice for Tg stimulation at first follow-up. In fact, in group B in all patients were performed rhTSH stimulation vs 17.4% of group A (p = 0.0001);
-
(2)
the progressive reduction in the use of diagnostic TBS during Tg stimulation (26.2% group B vs 91.3% group A) over the years (p = 0.0001). In particular, in group B, diagnostic TBS was performed for the evaluation of patients with AbTg positive (22.5%);
-
(3)
an increase, though not significant, of new diagnostic techniques such as 18FDG-PET (group B = 9.7% vs group A = 3.8%) in order to detect metastatic patients (p = 0.382);
-
(4)
moreover since 2005, the preparation to perform radioiodine ablation is based on the administration of recombinant human TSH (rhTSH) while patients were on L-T4 therapy.
At the last follow-up, 79 patients (76.7%) were considered as disease free, 15 patients (14.6%) showed signs of recurrence, and 9 (8.7%) had a persistent disease (Fig. 1). Interestingly, all relapsed patients of group A had a micro or small cancers. In group B, among those who had relapsed/persisted in loco-regional lymph nodes, 14/17 patients (82.3%) presented a micro or small cancer.
No patients died because of thyroid cancer.
Correlation analysis
Tables 3 and 4 summarize correlation analysis.
Recurrence and/or persistence of the disease was correlated with the presence of lymph node metastases and/or a distant disease at diagnosis, as well as with stimulated Tg values at the first follow-up and following radioiodine therapy. No correlation was found between a recurrent disease and a tumor-associated thyroiditis.
In the multivariate analysis, both tumor relapse and/or persistence were independently predicted by Tg values after stimulation at the first follow-up, also when weighted for the time of follow-up.
Discussion
The first objective of our study was to analyze features of DTC in a homogeneous population over years time-frame analysis. Then, we subdivided these consecutive series of DTC into two groups according to the year of the diagnosis; the rationale for choosing the year 2005 as a cut-off was related to the introduction into clinical practice of the new guidelines for the diagnosis and management of thyroid cancers [14, 15].
Our study has been conducted in an industrial area (Piedmont) known to be iodine-deficient [21], where population has undergone a food education campaign [13] in order to promote the use of iodized salt during the last 20 years. Despite being related only to a single clinical center, our data confirm the trend toward an increase in the incidence of the thyroid cancer as reported in recent years [1]. This evidence may represent a real increase in the pathology, although a greater diagnostic accuracy may also play a role [22]. However, despite the high clinical and scientific interest for the growing number of thyroid cancer diagnosed, few is known about recognized risk factors for thyroid cancer, apart from ionizing radiation both in childhood and in adulthood and a medical history of goiter or thyroid nodules.
In our population, we found that goiter was the most common coexisting benign thyroid disease in the whole population, but its frequency decreased from group A to group B though not significantly; conversely, the evidence of thyroid autoimmune disease increased over the years with a clear significativity; as a matter of fact, positive serum TPOAb were greatly detected in group B than in A. Some authors simply report that this trend of rising is tied up to the increased attitude to measure serum thyroid autoantibodies during the work-up of a thyroid disease [23]. Instead, other studies [24, 25] show a real increase in the annual incidence of Hashimoto’s thyroiditis in Italy, proposing a number of environmental factors as endocrine disrupters. Regarding this, we suspect, in our geographic area, a possible role of iodine supplementation in exacerbating thyroid autoimmunity. This hypothesis is supported by some literature data indicating an evolution of the thyroid autoimmune markers during the course of iodine prophylaxis [26]. However, a limitation of our study is that we defined the ID only on the basis of median UIC such as an extemporaneous evaluation of the iodine intake, while effects of an ID over time may be determined [27]. Further studies methodologically closer to ours will be required to confirm these hypotheses.
Interestingly, we also observed a significant increase of lymphocytic infiltration, mostly focal, associated to DTC from group A to group B at histological analysis.
A role for the lymphocytic infiltration co-expressed with thyroid cancer was hypothesized after the Chernobyl accident [28]; this interpretation is in keeping with the observation that thyroid autoimmune reactions may be related to radiation exposure. Moreover, after the radioactive event (April 26, 1986), the occurrence of thyroid carcinoma in children reached its peak in 1993, with a trend to a plateau in the following years [28]; however, this evidence does not exclude the possibility of a second peak occurring in following decades and having a longer latency period, similarly to that observed after external radiation [29]; in fact the majority of our patients was older than 45 years and was affected by a classical variant of PTC with a mean latency period exposure to Chernobyl accident and diagnosis of 16.0 ± 2.5 years and 22.5 ± 1.3 years in group A and B, respectively. Regarding other environmental exposures, except ionizing radiation, no occupational risk factors for thyroid cancers have been consistently identified so far, with the exception of a Chinese study [12] documenting an association between thyroid cancer and a long-term employment in textile industry, where an exposure to formaldehyde and benzene can be considered. Interestingly our data seems to confirm these findings. In fact, when we reviewed occupational status of our cohort of patients, the majority of our cohort was employed in textile industries; in particular, female sex was found to be an important variable, though not statistically significant, for development of malignant thyroid pathologies in exposed groups.
However, further epidemiological studies in larger and detailed cohorts are required in order to characterize and confirm the latter evidence. All together, these data could hypothesize that environment features create an inflammatory trigger that facilitates tumor promotion.
In our cohort of patients, we found an higher percentage of tumors not diagnosed before surgery with FNAC, than literature show [30, 31]. Most of these patients were in group A, the older diagnosed, when false negative and non-diagnostic FNAC and a clinical practice not focused on guidelines where evidenced in our institution.
However, the real meaning of high prevalence of incidentalomas, and of its link with lymphocytic thyroiditis, in the occupational exposed group requires a longer observation of these subjects with larger database.
Moreover, we found an increase in the diagnosis of micro or small intrathyroidal carcinoma over the years. This pattern has been reported in other series recently published [23, 32], where some authors raising the question whether the reduced tumors size could be due to an early diagnosis, or to an identification of persistently small thyroid carcinomas, that would have never become clinically evident. In agreement with the first hypothesis, we found that most of recurrences in group B occurred in micro/small, often incidental, carcinomas during the first years after initial treatments, though they were initially considered at low risk.
Notably, we found an increased prevalence of cancers N0 and N1 in group B, as a consequence of the reduction of Nx stage, as well as a greater number of patients with metastatic disease. This evidence can be correctly interpreted as the results of an improvement in the diagnostic accuracy (rhTSH introduction in clinical practice, FNAB, US and other imaging techniques as 18FDG-PET) in the last years, more than a worse clinical profile in group B. Our data fit with others in scientific literature [33, 34].
When we consider the changes in the follow-up and outcome management in our patients, we found that, in agreement with other data in literature [35–37], the presence of loco-regional or distant metastases at the time of diagnosis were principal predictors of recurrence or persistence of the disease. Particularly, Tg levels at the first postsurgical evaluation, after L-tiroxine withdrawal or exogenous rhTSH stimulation, were the strongest predictor of the outcome, as reported by Castagna et al. [38]. In our experience, radioiodine therapy showed to be related to a better outcome, also in microcarcinomas. However, the benefit to perform radioiodine therapy even in low risk patients remains unclear, given that the disease-specific mortality rate on DTC patients is very low. Speculations on alternative outcome markers in a longer follow-up should be considered. This concept has been recently validated by Tuttle [39] that proposed a dynamic risk assessment that can be used to tailor more effectively, ongoing follow-up recommendation.
In conclusion, our data show that after the introduction of the new guidelines in 2006 earlier diagnosis and more effective treatments for DTC was present in our patients’ population. We speculate that an early identification of the tumor (and its recurrences) is the reason of a better outcomes together with the introduction of the new guidelines in clinical practice with a better stratification through the new TNM staging. In our population, we can speculate that the inflammation induced by the iodine prophylaxis might accelerate the tumor progression in patients submitted to an iodine supplementation in adulthood. Other variables, such as occupational factors and the fallout influence of Chernobyl, could help to individualize the follow-up and indentify populations needing more aggressive treatments.
References
J.A. Sipos, E.L. Mazzaferri, Thyroid cancer epidemiology and prognostic variables. Clin. Oncol. 22, 395–404 (2010)
B.A. Kilfoy, T. Zheng, T.R. Holford et al., International patterns and trends in thyroid cancer incidence, 1973–2002. Cancer Causes Control 20(5), 525–531 (2009)
Associazione Italiana Registro Tumori (AIRTUM) Available from http://www.registri-tumori.it/cms/?q=sede_tiroide (2011). Accessed 14 October 2011
Centro di Riferimento per l’Epidemiologia e la Prevenzione Oncologica in Piemonte (CPO-Piemonte). Available from http://www.cpo.it/dationcologici/registro_tum2011.htm (2011). Accessed 14 October 2011
F. Pacini, M.G. Castagna, L. Brilli et al., Differentiated thyroid cancer: ESMOClinical reccomendations for diagnosis, treatment and follow up. Ann. Oncol. 19(2), 99–101 (2008)
E.L. Mazzaferri, Managing low risk thyroid cancer. Endocr. Pract. 13(5), 499 (2007)
D.E. Thompson, K. Mabuchi, E. Ron et al., Cancer incidence in atomic bomb survivors, part II: solid tumors, 1958–1987. Radiat. Res. 137, S17–S67 (1994)
v. Gomez Segovia, H.J. Gallowitsch, E. Kresnik et al., Descriptive epidemiology of thyroid carcinoma in Carinthia, Austria: 1984–2001. Histopathologic features and tumor classification of 734 cases under elevated general iodination of table salt since 1990: population-based age-stratified analysis on thyroid carcinoma incidence. Thyroid 14(4), 277–286 (2004)
A. Belfiore, G.L. La Rosa, G. Padova et al., The frequency of cold thyroid nodules and thyroid malignancies in patients from an iodine-deficient area. Cancer 60, 3096–3102 (1987)
R.A. Ott, A.R. McCall, H. Jarosz et al., The incidence of thyroid carcinoma in Hashimoto’s thyroiditis. Am. Surg. 53, 442–445 (1987)
G. Pellegriti, F. De Vathaire, C. Scollo et al., Papillary thyroid cancer incidence in the volcanic area of Sicily. J. Natl. Cancer Inst. 101, 1575–1583 (2009)
E.Y. Wong, R. Ray, D.L. Gao et al., Reproductive history, occupational exposures, and thyroid cancer risk among women textile workers in Shangai, China. Int. Arch. Occup. Environ. Health 79, 251–258 (2006)
WHO. Prevention and control of iodine deficiency disorders. Fifty-second world healthy assembly. Provisonal agenda item 13. Geneva; World Health Organization (1999)
F. Pacini, M. Schlumberger, H. Dralle et al., European consensus for the management of patients with differentiated thyroid carcinoma of the follicular epithelium. Eur. J. Endocrinol. 154, 787–803 (2006)
D.S. Cooper, G.M. Doherty, B.R. Haugen et al., Management guidelines for patients with thyroid nodules and differentiated thyroid cancer. Thyroid 16(2), 109–141 (2006)
H. Gharib, E. Papini, R. Paschke, for the AACE/AME/ETA Task Force on Thyroid Nodules et al., American Association of Clinical Endocrinologists, Associazione Medici Endocrinologi, and European Thyroid Association. Medical Guidelines for Clinical Practice for the Diagnosis and Management of Thyroid Nodules. Endocr. Pract. 16(1), 1–43 (2010)
American Joint Committee on Cancer: Chapter 8: Thyroid in: AJCC cancer staging handbook. 6th edition. Spinger, New York, pp 89–98 (2002)
WHO, UNICEF, ICCIDD, Indicators for assessing iodine deficiency disorders and their control through salt iodination (World Health Organization, Geneva, 1992)
American Thyroid Association (ATA) Guidelines Taskforce on Thyroid Nodules and Differentiated Thyroid Cancer, Cooper, D.S., Doherty, G.M., Haugen, B.R. et al. Revised American Thyroid Association Management Guidelines for patients with thyroid nodules and differentiated thyroid cancer. Thyroid 19(11): 1167-1214 (2009)
AJCC Seventh Edition Staging Series: Staging Head and Neck Cancers—transitioning to the seventh edition of the AJCC Cancer Staging Manual. American Joint Committee on Cancer Web site. Available from http://www.cancerstaging.org/products/ajccguide.pdf. Accessed 14 October 2011
S. Lonati, A. Rapa, G. Di Dio et al., Iodine status in historically iodine deficiency area. Minerva Pediatr. 58(3), 255–256 (2006)
L. Enewold, K. Zhu, E. Ron et al., Rising thyroid cancer incidence in the United States by demographic and tumor characteristics, 1980–2005. Cancer Epidemiol. Biomarkers Prev. 18, 784–791 (2009)
R. Elisei, E. Molinaro, L. Agate et al., Are the clinical and pathological features of differentiated thyroid carcinoma really changed over last 35 years? Study on 4187 patients from a single Italian Institution to answer to this question. J. Clin. Endocrinol. Metab. 95(4), 1516–1527 (2010)
M. Rizzo, R.T. Rossi, O. Bonaffini et al., Increased annual frequency of Hashimoto’s thyroiditis between years 1988 and 2007 at a cytological unit of Sicily. Ann. Endocrinol. (Paris) 71(6), 525–534 (2010)
S. Benvenga, F. Trimarchi, Changed presentation of Hashimoto’s thyroiditis in north-eastern Sicily and Calabria (southern Italy) based on a 31-year experience. Thyroid 18(4), 429–441 (2008)
G. Mazziotti, L.D. Premawardhana, A.B. Parkes et al., Evolution of thyroid autoimmunity during iodine prophylaxis—the Sri Lankan experience. Eur. J. Endocrinol. 149(2), 103–110 (2003)
P. Vitti, E. Martino, F. Aghini-Lombardi et al., Thyroid volume measurement by ultrasound in children as a tool for the assessment of mild iodine deficiency. J. Clin. Endocrinol. Metab. 79(2), 600–603 (1994)
F. Pacini, T. Vorontsova, E.P. Demidchik et al., Post Chernobyl thyroid carcinoma in Belarus children and adolescents: comparison with naturally occurring thyroid carcinoma in Italy and French. J. Clin. Endocrinol. Metab. 82(11), 3563–3569 (1997)
M.J. Favus, A.B. Scneider, M.E. Stachura et al., Thyroid cancer occurring as late as consequence of head-and-neck irradiation. Evaluation of 1056 patients. N. Engl. J. Med. 294, 1019–1025 (1976)
L. García-Pascual, M.J. Barahona, M. Balsells et al., Complex thyroid nodules with nondiagnostic fine needle aspiration cytology: histopathologic outcomes and comparison of the cytologic variants (cystic vs. acellular). Endocrine. 39(1), 33–40 (2011)
L. García-Pascual, M. Balsells, M. Fabbi et al., Prognostic factors and follow-up of patients with differentiated thyroid carcinoma with false negative or nondiagnostic FNAC before surgery Comparison with a control group. Endocrine 40(3), 423–431 (2011)
D.T. Hughes, M.R. Haymart, B.S. Miller et al., The most commonly occurring papillary thyroid cancer in the United States is now a microcarcinoma in a patient older than 45 years. Thyroid 21(3), 231–236 (2011)
J.M. Stulak, C.S. Grant, D.R. Farley et al., Value of preoperative ultrasonography in surgical management of initial and reoperative papillary thyroid cancer. Arch. Surg. 141, 489–494 (2006)
T.Y. Ho, M.J. Liou, K.J. Lin et al., Prevalence and significance of thyroid uptake detected by 18F-FDG PET. Endocrine 40(2), 297–302 (2011)
Y. Ito, T. Higashiyama, Y. Takamura et al., Risks factors for recurrence to the limph node in papillary thyroid carcinoma patients without preoperatively detectable lateral node metastasis: validity of prophylactic modified radical neck dissection. World J. Surg. 31, 2085–2091 (2007)
E.L. Mazzaferri, S.M. Jhiang, Long-term impact of initial surgical and medical therapy on papillary and follicular thyroid cancer. Am. J. Med. 97, 418–428 (1994)
E. Sampson, J.D. Brierley, L.W. Le et al., Clinical management and outcome of papillary and follicular (differentiated thyroid cancer) presenting with distant metastasis at diagnosis. Cancer 110, 1451–1456 (2007)
M.G. Castagna, L. Brilli, T. Pilli et al., Limited value of repeat recombinant human thyrotropin (rhTSH)-stimulated thyroglobulin testing in differentiated thyroid carcinoma patients with previous negative rhTSH-stimulated thyroglobulin and undetectable basal serum thyroglobulin levels. J. Clin. Endocrinol. Metab. 93(1), 76–81 (2008)
R.M. Tuttle, T. Hernan, J. Shas et al., Estimating risk of recurrence in differentiated thyroid cancer after total thyroidectomy and radioactive iodine remnant ablation: using response to therapy variables to modify the initial risk estimates predicted by The New Thyroid Association Staging System. Thyroid 20(12), 1341–1349 (2010)
Acknowledgments
This research did not receive any specific grant from any funding agency in the public, commercial, or not-to-profit sector.
Conflict of interest
Nothing to declare.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pagano, L., Caputo, M., Samà, M.T. et al. Clinical–pathological changes in differentiated thyroid cancer (DTC) over time (1997–2010): data from the University Hospital “Maggiore della Carità” in Novara. Endocrine 42, 382–390 (2012). https://doi.org/10.1007/s12020-012-9612-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-012-9612-4