Abstract
In this retrospective study, we sought to investigate the prevalence and the clinical significance of thyroid uptake discovered by FDG PET/CT within the thyroid gland during imaging investigation of non-thyroid disorders. This study included 5,877 subjects with no previous history of thyroid malignancy referred to our PET/CT center (1,596 for cancer screening and 4,281 for primary staging or restaging purposes) from June 2006 to April 2009. All of the patients had at least 12 months of follow-up or were censored on the date of the last follow-up. The disease status was analyzed for correlation with the maximum standardized uptake value (SUVmax). Receiver operating characteristics (ROC) curves were constructed to determine the optimal cutoff values for SUVmax. In the screening group (n = 1,596), 55 patients (4%) had thyroid FDG uptake. Twenty-two subjects were referred for cytological or histological assessment and 4 (18%) were diagnosed with thyroid carcinoma. In the primary staging/restaging group (n = 4,281), 165 patients (4%) had thyroid FDG-avid sites. Thirty-three patients were referred for cytological or histological assessment and 4 (12%) were found to have thyroid carcinoma. Of the 8 malignant lesions, 7 were papillary thyroid carcinoma, and 1 medullary thyroid carcinoma. Surgical resection was performed in seven out of eight patients. Three patients had lymph node metastasis (one case detected by preoperative PET/CT) and three bilateral malignant diseases (all false negative in PET/CT scans). Of the 47 benign lesions, 35 were nodular goiter or nodular hyperplasia, 6 multinodular goiter, and 6 autoimmune thyroid disease. The average value of SUVmax of malignant thyroid lesions was significantly higher than that of benign thyroid lesions (8.2 ± 4.5 vs. 5.6 ± 3.2, P = 0.048). However, we were unable to identify an optimal SUVmax cutoff because the total area under the curve was small. The prevalence of thyroid uptake on FDG PET/CT in this study is 3.7%, which is consistent with previous observations. Of patients who received verification by cytology or histology, 14% (8/55) were found to have thyroid malignancies. However, we were unable to establish an optimal SUVmax cutoff value to differentiate benign from malignant lesions. FDG PET/CT scans were false negative in a significant proportion of patients with thyroid carcinoma and regional lymph node metastases or separate tumor nodule(s) in a contralateral lobe. Further diagnostic tests (thyroid ultrasound/fine needle aspiration) are recommended in presence of focal thyroid uptake detected by FDG PET/CT scans.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Background
Under physiological conditions, the thyroid parenchyma is characterized by low-intensity uptake of 18-fluoro-deoxyglucose (FDG). Therefore, positron emission tomography (PET) using FDG does not visualize the normal thyroid gland [1]. Of note, the thyroid gland is a rare target for metastasis from other primary cancers [2]. With the increasing use of FDG PET/CT, unexpected diffuse or focal areas of FDG uptake within the thyroid gland have been repeatedly described during imaging investigation of non-thyroid disorders [3]. To assess whether hypermetabolic thyroid uptake are malignant, cytological or histological verification should be pursued. Unfortunately, complete pathological data are not available for all reported series. Therefore, the prevalence and the clinical significance of thyroid FDG uptake remain uncertain.
In Taiwan, FDG PET is one of the most widely used imaging modalities for cancer screening, primary staging, restaging, and evaluation of cancer treatment. Therefore, we planned a retrospective study to investigate the prevalence and the clinical significance of thyroid uptake discovered by PET/CT during imaging investigation of non-thyroid disorders among Taiwanese individuals.
Materials and methods
Patient population
This retrospective study included 5,877 subjects with no previous history of thyroid malignancy who were referred to our PET/CT center for cancer screening (1,596 patients) or primary staging or restaging purposes (4,281 patients) from June 2006 to April 2009. Patients with incidental FDG accumulation within the thyroid gland were included in the study. The nature of focally or diffusely increased thyroid uptake was determined by image-guided fine needle aspiration cytology or by histological analysis of surgical resection specimens. When the diagnosis was not based on cytology or histology, close follow-up with ultrasound or laboratory examinations was pursued for at least 12 months. All patients gave written informed consent for the FDG PET/CT study and for evaluation of their cytological or histological and clinical records. The study protocol was approved by our local Ethics Committee.
FDG PET protocol
All FDG PET/CT scans were done on a Discovery ST 16 PET/CT scanner (GE Healthcare, Milwaukee, MI, USA). Images were acquired 50 min after i.v. injection of FDG (333–407 MBq). Sixteen-detector multi-slice CT images were acquired using speed of rotation and couch movement of 0.5 s/rotation and 35 mm/s, respectively. The images were reconstructed in 3.3 mm slice width during normal respiration. CT images were recombined from a 512 × 512 matrix to a 128 × 128 matrix and matched to the pixel size of the PET data in order to match the in-slice resolution of the PET emission images. The CT images were subsequently converted to maps of PET attenuation coefficients. Without changing the patient position, a whole-body PET emission scan was performed over the same area as was covered by CT with six bed positions. All acquisitions were carried out in 2D mode, the protocol comprising an emission scan with 3 min/bed position. PET images were reconstructed using CT attenuation maps. Transaxial emission images of 3.3 × 3.3 × 3.27 mm3 (in plane matrix size 128 × 128, 47 slices per bed position) were reconstructed using ordered subsets expectation maximization with four iterations and ten subsets. The axial field of view was 157 mm, with acquisition of 47 slices per bed position.
Statistical analysis
We used logistic regression analysis to examine the independent contribution of the general characteristics of the study participants (age, gender), the maximum standardized uptake value (SUVmax), and the pattern of FDG uptake (unilateral or bilateral) to having benign or malignant thyroid lesions. Receiver operating characteristics (ROC) curves were constructed to establish an optimal SUVmax cutoff value to differentiate benign from malignant lesions. All statistical analyses were performed using SPSS software, version 16.0 (SPSS Inc., Chicago, IL, USA).
Results
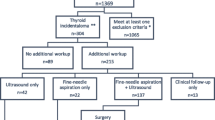
The flow of participants through the study is shown in Fig. 1. The general characteristics of the study patients are given in Table 1. We observed no relationship of age, gender and the pattern of FDG uptake with an increased occurrence of thyroid carcinoma. A total of 8 malignant thyroid lesions (7 papillary thyroid carcinoma and 1 medullary thyroid carcinoma) were correctly identified by FDG PET/CT (Table 2). No metastasis from other primary tumors was detected. Among patients who underwent surgical resection, we identified 5 false negative lesions based on final histology (carcinoma in the contralateral thyroid lobe and/or neck lymph node metastases in three patients). Only a case of ipsilateral regional lymph node metastasis was correctly diagnosed preoperatively (Table 3).
Flow of the participants (n = 5,877) through the study. A total of 1,596 patients underwent FDG PET for cancer screening and 4,281 for primary staging or restaging purposes. Among the 1,596 patients undergoing cancer screening, subsequent examinations revealed that 4 patients were harboring thyroid cancer. In 4 patients examined for primary staging or restaging purposes, ensuing examinations confirmed the existence of malignant thyroid tumors
Cancer screening group
In the screening group (n = 1,596), 55 patients (4%) had focally or diffusely increased FDG thyroid uptake. Twenty-two subjects were referred for cytological or histological assessment and 4 (19%) of these lesions were malignant. No metastasis from other primary tumors was detected. Of the 4 malignant lesions, 3 were papillary carcinoma, and 1 medullary thyroid carcinoma. Of the 3 patients with papillary carcinoma, 2 had carcinoma in the contralateral thyroid lobe and/or neck lymph node metastases according to the histopathological results obtained after thyroidectomy and/or neck lymph node dissection. However, only one metastatic neck lymph node from medullary thyroid carcinoma was correctly diagnosed preoperatively by FDG PET/CT.
Cancer primary staging/restaging group
In this group (n = 4,281), 165 patients (4%) had thyroid FDG-avid sites. Of these 165 patients, 67 (41%) were referred for staging of head and neck cancers, 34 (21%) for lung cancer, 27 (16%) for gynecological cancers, 11 (7%) for breast cancer, 9 (5%) for colorectal cancer, 3 (2%) for unknown primary cancers, and 1 for urological cancer. Thirty-three patients were referred for cytological or histological assessment and 4 (12%) were found to have thyroid carcinoma. Of these 4 patients, three were referred for primary staging of head and neck cancers (1 oral cancer, 1 laryngeal cancer, 1 buccal cancer) and one for restaging of ovarian cancer. All of these thyroid malignancies were papillary thyroid carcinoma. No metastasis from other primary tumors was detected. No false negative lesion was identified by histopathology after thyroidectomy.
Benign thyroid lesions
Forty-seven patients with thyroid uptake had benign thyroid lesions (35 nodular goiter or nodular hyperplasia, 6 multinodular goiter, and 6 autoimmune thyroid disease) according to ultrasound and following cytological or histological findings. All of the 6 patients with autoimmune thyroid disease had diffusely increased thyroid uptake and 5 of them were presented with elevated serum thyroid-stimulating hormone (TSH) or thyroid peroxidase (TPO) antibody which corresponds to previous reported experiences [4, 5].
Role of SUVmax in predicting malignant lesions
The average value of SUVmax of malignant thyroid lesions was significantly higher than that of benign thyroid lesions (8.2 ± 4.5 vs. 5.6 ± 3.2, P = 0.048, Fig. 2). However, there was high intersubject variability in the SUVmax values of benign and malignant thyroid lesions (Fig. 3). Unfortunately, we were unable to identify an optimal SUVmax cutoff because the total area under the curve was small (0.677).
Discussion
The prevalence of thyroid uptake on FDG PET/CT in this study (3.7%) is consistent with previous observations (1.2–4.3%) [6, 7]. The overall prevalence of thyroid malignancies identified by an increased thyroid uptake and subsequently verified by cytological or histological analysis was 14% (8/55).
Although FDG PET plays a key role in the diagnosis of distant metastases, in this study we failed to identify metastatic lesions from other primary tumors. In the present report, most of the FDG-avid lesions of the thyroid gland were benign in nature (86%, 47/55) with a wide range of SUVmax values. These data are in good agreement with previous reports showing that uptake of FDG may be prominent in benign thyroid lesions (e.g., thyroiditis or Hürthle cell adenoma) [8]. Although a high uptake of 18F-FDG has been reported for some high-grade thyroid carcinomas, certain well-differentiated papillary carcinoma of the thyroid may be negative on FDG PET [9]. This phenomenon may explain the overlap in SUVmax values observed between benign and malignant thyroid lesions (Fig. 2). In this study, we failed to identify a reliable cutoff value of SUVmax for differentiating malignant and benign thyroid lesions. In this context, a threshold value for SUVmax alone cannot be employed to differentiate between malignancy and benignity.
Only one female subject out of 15 patients with bilateral thyroid uptake of FDG in this study had a diagnosis of thyroid carcinoma. However, she had focal uptake of FDG in both lobes of the thyroid gland rather than diffuse thyroid uptake. For those patients diagnosed of autoimmune thyroid disease, there was correlation between diffuse thyroid uptake, laboratory, and cytological findings. False negative FDG PET findings were not uncommon in our patients receiving thyroidectomy. Most of them were tumors or lymph nodes less than 0.5 cm in size of well-differentiated papillary thyroid carcinoma. These lesions may be underestimated either because of the partial volume effect of PET or cell differentiation. Indeed, only a single case of metastatic node involvement from medullary thyroid carcinoma was correctly identified by FDG PET in the preoperative period.
For the 14% of malignant rate found in this study and absence of optimal cutoff value of SUVmax for diagnostic help in FDG PET, follow-up with ultrasound or fine needle aspiration when detecting focal thyroid FDG uptake is highly recommended. As for diffuse thyroid FDG uptake, laboratory study for TSH and TPO antibody should be adequate.
Conclusions
The prevalence of thyroid FDG uptake observed in this study is in substantial agreement with previous reports. Some cases proved to be false negative mainly due to the absence of FDG uptake and the presence of well-differentiated papillary thyroid carcinomas. Of note, some small-sized tumors or lymph nodes have revealed false negative findings on PET scans. Another notable finding was that SUVmax values alone may not enable adequate differentiation between benign and malignant lesions which is in agreement with a systemic review [10]. Thus, the identification of focal FDG uptake in the thyroid gland on PET/CT scans should prompt a cytological or histological verification, as it may lead to significant management changes in selected patients.
References
Y. Nakamoto, M. Tatsumi, D. Hammoud, C. Cohade, M.M. Osman, R.L. Wahl, Normal FDG distribution patterns in the head and neck: PET/CT evaluation. Radiology 234, 879–885 (2005)
K.Y. Lam, C.Y. Lo, Metastatic tumors of the thyroid gland: a study of 79 cases in Chinese patients. Arch. Pathol. Lab. Med. 122, 37–41 (1998)
A. Van den Bruel, A. Maes, T. De Potter, L. Mortelmans, M. Drijkoningen, B. Van Damme, P. Delaere, R. Bouillon, Clinical relevance of thyroid fluorodeoxyglucose-whole body positron emission tomography incidentaloma. J. Clin. Endocrinol. Metab. 87, 1517–1520 (2002)
T.Y. Kim, W.B. Kim, J.S. Ryu, G. Gong, S.J. Hong, Y.K. Shong, 18F-fluorodeoxyglucose uptake in thyroid from positron emission tomogram (PET) for evaluation in cancer patients: high prevalence of malignancy in thyroid PET incidentaloma. Laryngoscope 115, 1074–1078 (2005)
D. Karantanis, T.V. Bogsrud, G.A. Wiseman, B.P. Mullan, R.M. Subramaniam, M.A. Nathan, P.J. Peller, R.S. Bahn, V.J. Lowe, Clinical significance of diffusely increased 18F-FDG uptake in the thyroid gland. J. Nucl. Med. 48, 896–901 (2007)
Y.K. Chen, H.J. Ding, K.T. Chen, Y.L. Chen, A.C. Liao, Y.Y. Shen, C.T. Su, C.H. Kao, Prevalence and risk of cancer of focal thyroid incidentaloma identified by 18F-fluorodeoxyglucose positron emission tomography for cancer screening in healthy subjects. Anticancer Res. 25, 1421–1426 (2005)
K.W. Kang, S.K. Kim, H.S. Kang, E.S. Lee, J.S. Sim, I.G. Lee, S.Y. Jeong, S.W. Kim, Prevalence and risk of cancer of focal thyroid incidentaloma identified by 18F-fluorodeoxyglucose positron emission tomography for metastasis evaluation and cancer screening in healthy subjects. J. Clin. Endocrinol. Metab. 88, 4100–4104 (2003)
L.F. de Geus-Oei, G.F. Pieters, J.J. Bonenkamp, A.H. Mudde, C.P. Bleeker-Rovers, F.H. Corstens, W.J. Oyen, 18F-FDG PET reduces unnecessary hemithyroidectomies for thyroid nodules with inconclusive cytologic results. J. Nucl. Med. 47, 770–775 (2006)
U. Feine, R. Lietzenmayer, J.P. Hanke, J. Held, H. Wohrle, W. Muller-Schauenburg, Fluorine-18-FDG and iodine-131-iodide uptake in thyroid cancer. J. Nucl. Med. 37, 1468–1472 (1996)
P. Shie, R. Cardarelli, K. Sprawls, K.G. Fulda, A. Taur, Systematic review: prevalence of malignant incidental thyroid nodules identified on fluorine-18 fluorodeoxyglucose positron emission tomography. Nucl. Med. Commun. 30, 742–748 (2009)
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Additional information
An erratum to this article can be found at http://dx.doi.org/10.1007/s12020-011-9480-3
Rights and permissions
About this article
Cite this article
Ho, TY., Liou, MJ., Lin, KJ. et al. Prevalence and significance of thyroid uptake detected by 18F-FDG PET. Endocrine 40, 297–302 (2011). https://doi.org/10.1007/s12020-011-9470-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-011-9470-5