Abstract
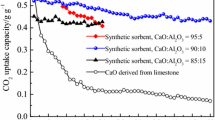
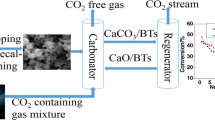
A novel calcium-based pellet was prepared by extrusion of sol-gel CaO powder and cement with high aluminum-based content. Limestone was used for comparison. The cyclic CO2 capture performance and carbonation kinetics of the sorbents were investigated in a thermogravimetric analyzer (TGA). The changes in phase and microstructure were characterized by X-ray diffraction (XRD), scanning electron microscopy (SEM) and Brunauer Emmet Teller (BET) surface area, respectively. The results indicate that the pellet consisted of CaO and Ca12Al14O33 after initial calcination. Limestone reactivity decreased dramatically with the increase in the cycle number, whereas the pellet showed a relatively stable cyclic CO2 capture performance with high reactivity. The CO2 capture capacity of the pellet achieved 0.43 g CO2/g sorbent after 50 cycles at 650 °C and 850 °C for carbonation and calcination, respectively. Moreover, the pellet obtained fast carbonation rates with slight decay after multiple cycles. The porous microstructure of the pellet contributed to the high reactivity of the sorbent during high temperature reactions, and the support material of Ca12Al14O33, enhanced the cyclic durability of the calcium-based sorbents.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
N. MacDowell, N. Florin, A. Buchard, J. Hallett, A. Galindo, G. Jackson, C. S. Adjiman, C.K. Williams, N. Shah and P. Fennell, Energy Environ. Sci., 3, 1645 (2010).
F.-C. Yu, N. Phalak, Z. Sun and L.-S. Fan, Ind. Eng. Chem. Res., 51, 2133 (2012).
H. An, T. Song, L. Shen, L. Zhang and B. Feng, Ind. Eng. Chem. Res., 51, 13046 (2012).
M. Zaman and J. H. Lee, Korean J. Chem. Eng., 30, 1497 (2013).
U. Zahid, Y. Lim, J. Jung and C. Han, Korean J. Chem. Eng., 28, 674 (2011).
J.H. Choi, C.K. Yi, S.H. Jo, H.J. Ryu and Y.C. Park, Korean J. Chem. Eng., 31, 194 (2014).
C. C. Dean, J. Blamey, N. H. Florin, M. J. Al-Jeboori and P. S. Fennell, Chem. Eng. Res. Des., 89, 836 (2011).
J. Blamey, E.J. Anthony, J. Wang and P.S. Fennell, Prog. Energy Combust, 36, 260 (2010).
S. Wang, S. Fan, Y. Zhao, L. Fan, S. Liu and X. Ma, Ind. Eng. Chem. Res., 53, 10457 (2014).
C.Q. Cao, K. Zhang, C.C. He, Y.A. Zhao and Q. J. Guo, Chem. Eng. Sci., 66, 375 (2011).
J. C. Abanades, Chem. Eng. J., 90, 303 (2002).
B. Feng, W.Q. Liu, X. Li and H. An, Energy Fuels, 20, 2417 (2006).
F. Fang, Z. S. Li and N. S. Cai, Korean J. Chem. Eng., 26, 1414 (2009).
T. Witoon, Ceram. Int., 37, 3291 (2011).
G. S. Grasa and J. C. Abanades, Ind. Eng. Chem. Res., 45, 8846 (2006).
W.Q. Liu, H. An, C.L. Qin, J. J. Yin, G.X. Wang, B. Feng and M.H. Xu, Energy Fuels, 26, 2751 (2012).
R.Y. Sun, Y. J. Li, H.L. Liu, S.M. Wu and C.M. Lu, Appl. Energy, 89, 368 (2012).
V. Manovic and E. J. Anthony, Environ. Sci. Technol., 43, 7117 (2009).
C.-C. Li, U.-T. Wu and H.-P. Lin, J. Mater. Chem. A, 2, 8252 (2014).
X. Zhang, Z. Li, Y. Peng, W. Su, X. Sun and J. Li, Chem. Eng. J., 243, 297 (2014).
J. Yin, C. Qin, B. Feng, L. Ge, C. Luo, W. Liu and H. An, Energy Fuels, 28, 307 (2014).
C. Qin, J. Yin, C. Luo, H. An, W. Liu and B. Feng, Chem. Eng. J., 228, 75 (2013).
Y. J. Li, R.Y. Sun, H. L. Liu and C. M. Lu, Ind. Eng. Chem. Res., 50, 10222 (2011).
H. C. Chen, C. S. Zhao, L. B. Duan, C. Liang, D. J. Liu and X. P. Chen, Fuel Process. Technol., 92, 493 (2011).
C. Luo, Y. Zheng, N. Ding, Q.L. Wu, G.A. Bian and C.G. Zheng, Ind. Eng. Chem. Res., 49, 11778 (2010).
M. Broda, A. M. Kierzkowska and C.R. Müller, ChemSusChem, 5, 411 (2012).
A. Akgsornpeak, T. Witoon, T. Mungcharoen and J. Limtrakul, Chem. Eng. J., 237, 189 (2014).
E.T. Santos, C. Alfonsín, A. J. S. Chambel, A. Fernandes, A.P. Soares Dias, C. I. C. Pinheiro and M. F. Ribeiro, Fuel, 94, 624 (2012).
P. Xu, M. Xie, Z. Cheng and Z. Zhou, Ind. Eng. Chem. Res., 52, 12161 (2013).
S.D. Angeli, C. S. Martavaltzi and A. A. Lemonidou, Fuel, 127, 62 (2014).
A.M. Kierzkowska and C.R. Muller, Energy Environ. Sci., 5, 6061 (2012).
C. Luo, Y. Zheng, C.G. Zheng, J. J. Yin, C.L. Qin and B. Feng, Int. J. Greenh. Gas Con., 12, 193 (2013).
Z. S. Li, Y. Liu and N. S. Cai, Chem. Eng. Sci., 89, 235 (2013).
C. Luo, Y. Zheng, N. Ding, Q.L. Wu and C.G. Zheng, Chin. Chem. Lett., 22, 615 (2011).
Z. Zhou, Y. Qi, M. Xie, Z. Cheng and W. Yuan, Chem. Eng. Sci., 74, 172 (2012).
K. Wang, X. Guo, P.F. Zhao and C.G. Zheng, Appl. Clay. Sci., 50, 41 (2010).
V. Manovic and E. J. Anthony, Ind. Eng. Chem. Res., 49, 6916 (2010).
Z. S. Li, N. S. Cai, Y.Y. Huang and H. J. Han, Energy Fuels, 19, 1447 (2005).
C. S. Martavaltzi and A. A. Lemionidou, Ind. Eng. Chem. Res., 47, 9537 (2008).
V. Manovic and E. J. Anthony, Ind. Eng. Chem. Res., 48, 8906 (2009).
Z. S. Li, N. S. Cai and Y. Y. Huang, Ind. Eng. Chem. Res., 45, 1911 (2006).
S. K. Bhatia and D. D. Perlmutter, AIChE J., 29, 79 (1983).
C. Luo, Q.W. Shen, N. Ding, Z.X. Feng, Y. Zheng and C.G. Zheng, Chem. Eng. Technol., 35, 547 (2012).
T. Witoon, T. Mungcharoen and J. Limtrakul, Appl. Energy, 118, 32 (2014).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Luo, C., Zheng, Y., Xu, Y. et al. Cyclic CO2 capture characteristics of a pellet derived from sol-gel CaO powder with Ca12Al14O33 support. Korean J. Chem. Eng. 32, 934–938 (2015). https://doi.org/10.1007/s11814-014-0291-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-014-0291-0