Abstract
Castor oil can be used in industry. The molecular species of triacylglycerols containing hydroxy fatty acids (FA) in castor oil have been identified. We report here the identification of twelve diacylglycerols (DAG) containing hydroxy FA in castor oil using positive ion electrospray ionization mass spectrometry of the lithium adducts. They were RR (diricinolein, R is ricinoleate), RL, RS, R-diOH18:0, R-diOH18:1, R-diOH18:2, R-triOH18:0, R-triOH18:1, R-triOH18:2, diOH18:0-diOH18:1, diOH18:1-diOH18:1 and diOH18:1-diOH18:2. The MS2 fragment ions, [M + Li − FA]+ and [FA + Li]+, from the lithium adducts of DAG containing hydroxy FA (one or two hydroxy FA), were used for the identification. The additional fragment ions from the neutral losses of FA lithium salts [M + Li − FALi]+ were used for the identification of eleven DAG containing two normal FA in a soybean oil bioconversion product. The MS2 fragment ions from the neutral losses of FA lithium salts [M + Li − FALi]+ were not detected from the DAG containing hydroxy FA. The DAG containing FA with more hydroxyl groups than the other FA on the same DAG molecule tended to have a prominent fragment ion [FA + Li]+ and an undetectable fragment ion [M + Li − FA]+ while the FA was the more hydroxylated FA. Also the less hydroxylated FA of a DAG tended to have a prominent fragment ion [M + Li − FA]+ and an undetectable fragment ion [FA + Li]+ while the FA was the less hydroxylated FA.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Triacylglycerols (TAG) containing hydroxy fatty acids (FA), e.g., castor oil, have many industrial uses such as the manufacture of biodegradable aviation lubricant, plastic, paint, nylons and cosmetics, because of the hydroxyl group on the FA constituents. Castor oil is the only commercial source of TAG containing hydroxy FA. Diacylglycerols (DAG) containing hydroxy FA can also be used in industry. DAG are the intermediate molecules in the biosynthesis of TAG and phospholipids. We have earlier detected a small amount (0.14 %) of diricinolein (RR), DAG containing two ricinoleic acids (OH18:1), in castor oil by HPLC [1]. RR was later identified by mass spectrometry (MS) [2]. We report here the identification of twelve DAG containing hydroxy FA (one or two hydroxy FA) in castor oil by the MS.
Positive ion electrospray ionization mass spectrometry (ESI-MS) of lithium adducts of TAG containing three normal (non-hydroxy) FA has been reported [3−5]. The same method has also been used for the identification of TAG containing hydroxy FA in castor oil [2, 6−11]. The MS2 spectra of the lithium adducts of TAG containing three normal FA showed the prominent fragment ions from the neutral losses of the three constituent FA [M + Li − FA]+ or [M + Li − RCO2H]+, and the three constituent FA lithium salts [M + Li − FALi]+ or [M + Li − RCO2Li]+ [4, 5]. The fragmentation pathways of these ions have been proposed [4, 5]. The MS2 spectra of the lithium adducts of TAG containing hydroxy FA in castor oil showed the fragment ions from the neutral losses of three constituent FA [M + Li − FA]+, an aldehyde [M + Li − C6H13CHO]+ and a ketene [M + Li − C5H11CH=C=O]+, and FA lithium adducts [FA + Li]+ [2, 7−11]. We have reported the MS2 spectra of five DAG containing hydroxy FA in castor oil [8, 10, 11] and the characteristic fragment ions were similar to those of the TAG containing hydroxy FA in castor oil. The MS2 spectra of the lithium adduct of DAG containing two normal FA have been rarely reported. The MS2 spectrum of the lithium adduct of distearin (SS) showed the fragment ions [M + Li − FALi]+ and [M + Li − FA]+ [12]. We report here the identification of twelve DAG containing hydroxy FA in castor oil by MS2 of the lithium adducts of DAG. The MS2 spectra were compared with those of DAG containing two normal FA in the soybean oil bioconversion product [13].
Material and Methods
Materials
Castor oil, lithium acetate and the standards of DAG of PP, LL and LnLn were obtained from Sigma (St. Louis, MO, USA). High purity methanol and 2-propanol (Honeywell Burdick & Jackson) for LC and MS were purchased from VWR International (West Chester, PA, USA). High purity nitrogen for MS was acquired from Praxair (Oakland, CA, USA). Research grade helium (Praxair) was used as a collision gas of MS. The soybean oil bioconversion product was obtained as described in Ref. 13.
HPLC Fractionation of the Molecular Species of Acylglycerols in Castor Oil and the Soybean Oil Bioconversion Product
The fractionation of the molecular species of TAG and DAG in castor oil and the soybean oil bioconversion product were as previously reported [1]. Chromatographic fractionation was performed using a Waters HPLC (Waters Associate, Milford, MA, USA) and a C18 analytical column (Gemini, 250′ 4.6 mm, 5 µ, C18, Phenomenex, Torrance, CA, USA). First, 1 mg of the sample in ethanol (50 μL) was chromatographed at 22 °C (room temperature) with a linear gradient from 100 % methanol to 100 % 2-propanol over 40 min, at a 1 mL/min flow rate, and detected at 205 nm. Fractions were collected every 30 s and corresponding fractions were pooled from seven HPLC runs. HPLC fractions were used for MS studies. The final methanol solutions of samples were prepared for direct infusion into the mass spectrometer by combining half of each HPLC fraction collected with 50 µL of a methanol solution of 100 mM lithium acetate and diluting to a total volume of 250 µL.
Electrospray Ionization Mass Spectrometry
An LCQ Advantage ion-trap mass spectrometer (MS 2.0) with Xcalibur 2.0 SR2 software (ThermoFisher Scientific, San Jose, CA, USA) was used for MS analysis of the various molecular species of DAG. The infusion at a 2.5 μL/min flow rate from a syringe (250 μL) pump produced stable singly-charged lithiated parent ions which were subsequently fragmented for MS2 analysis. ESI source conditions were as follows: sheath gas flow rate, 10 arbitrary units (au); aux/sweep gas flow rate, 0 au; spray voltage, 4 kV; capillary temperature, 200°C; capillary voltage, 5 V; tube lens offset, 15 V. Scan conditions were as follows: isolation width, 1.5 ; normalized collision energy, 27–42 %; scan ranges, 100–1500 m/z. Acquire time was 3 min.
Results and Discussion
Castor oil and the soybean oil bioconversion product containing TAG and DAG of both hydroxy FA and normal FA [13] were fractionated by HPLC. The HPLC fractions were used for the analysis of DAG by positive ion electrospray ionization mass spectrometry (ESI-MS) of the lithium adducts. The hydroxy FA of acylglycerols in castor oil have been identified including the numbers and locations of hydroxyl groups and double bonds on the acyl chains [8, 10, 11]. They were biosynthesized from ricinoleate presumably with the addition of OH groups at C-11 and/or C-13 positions and with the addition of double bonds at the C-13 or C-14 positions [2] or the saturation of double bond at the C-9 position.
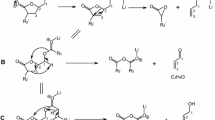
Figure 1 shows the MS2 spectrum of diricinolein (RR) in castor oil from [M + Li]+ at m/z 659.4. This is a simple spectrum. The prominent fragment ions were ricinoleate lithium adduct [R + Li]+ at m/z 305.1, and from the neutral losses of ricinoleate [M + Li − R]+ at m/z 361.1 and an aldehyde [M + Li − C6H15CHO]+ at m/z 545.3. The fragment ion from the neutral loss of the aldehyde was due to the cleavage between C-11 and C-12 on the C18 acyl chain containing a double bond at the C-9 position and a hydroxyl group at the C-12 position, e.g., ricinoleate [8]. This ion was also the common fragment ion on the MS2 spectra of the lithium adducts of TAG containing hydroxy FA in castor oil [7−12]. None of the MS2 spectra of the TAG [8−12] and DAG (Figs. 1, 2; Table 1) [8, 10, 11] containing hydroxy FA (one, two or three hydroxy FA) in castor oil showed the fragment ions from the neutral losses of FA lithium salts [M + Li − FALi]+. While the fragment ions from the neutral losses of FA lithium salts [M + Li − FALi]+ were the prominent ions from the TAG and DAG containing all normal FA [5, 6, 12]. We have recently obtained the MS2 spectra of TAG containing three normal FA in castor oil [2] and they showed the prominent fragment ions of both [M + Li − FA]+ and [M + Li − FALi]+.
Figure 2 is the MS2 spectrum of ricinoleoyl-stearoyl-glycerol (RS) in castor oil from [M + Li]+ at m/z 645.3. The prominent fragment ions were ricinoleate lithium adduct [R + Li]+ at m/z 305.0, and from the neutral losses of stearate [M + Li − S]+ at m/z 361.0 and an aldehyde [M + Li − C6H15CHO]+ at m/z 531.2. No fragment ions of stearate lithium adduct [S + Li]+ at m/z 291.0 and that from the neutral loss of ricinoleate [M + Li − R]+ at m/z 347.0 were detected. The MS2 spectra of DAG containing both hydroxy FA and normal FA (e.g., RS, RL) seems that hydroxy FA tend to show as lithium adducts only and normal FA tend to show as those from the neutral losses of the normal FA only. Both Figs. 1 and 2 are simple spectra. The MS2 spectra of the TAG including three hydroxy FA and three normal FA and the mix of both showed the fragment ions from the neutral losses of all of the three FA constituents [3, 5, 6, 8−12].
We have identified twelve DAG containing hydroxy FA in castor oil by MS as shown in Table 1. The characteristic ions (m/z) and their relative abundance of the DAG are also given. Among the twelve DAG in Table 1, five are newly reported here and they are RL, RS, R-diOH18:2, diOH18:0-diOH18:1 and R-triOH18:0. We did not detect RO in castor oil and oleate is the precursor of ricinoleate [14]. Previously reported were the MS2 spectra of DAG lithium adducts of R-diOH18:1 [8], diOH18:1-diOH18:1 [10], R-triOH18:1 [10], diOH18:1-diOH18:2 [11], and R-triOH18:2 [11]. The most abundant fragment ions (base peaks) were usually from the neutral losses of aldehyde, [M + Li − C6H13CHO]+ as shown in Table 1. The fragment ions of the two FA lithium adducts combined, [FA + Li]+, were usually more abundant than those from the neutral losses of the two FA combined, [M + Li − FA]+. Among the DAG containing two different FA, the fragment ions from the losses of the less hydroxylated FA, [M + Li − FA]+, were more abundant than those from the losses of the more hydroxylated FA. The fragment ions of the lithium adducts, [FA + Li]+, of the less hydroxylated FA were less abundant than those of the more hydroxylated FA. The extreme case is shown as Fig. 2 of RS, a DAG containing both hydroxylated FA and normal FA and both [M + Li − R]+ at m/z 347.0 and [S + Li]+ at m/z 291.0 were not detectable (Table 1).
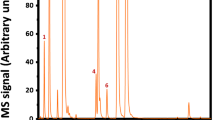
We have recently used MS2 of DAG lithium adducts to identify the DAG containing hydroxy FA in the soybean oil bioconversion product and the presence or lack of [M + Li − R]+ and [FA + Li]+ were similar to those from the DAG containing hydroxy FA in castor oil [13]. Figure 3 is an example of the identification by MS of DAG containing hydroxy FA in the soybean oil bioconversion product. This is the MS2 spectrum of three DAG, OH18:1-OH:18:1, diOH18:2-S and diOH18:1-O, combined. For the DAG, diOH18:2-S and diOH18:1-O containing both hydroxy FA and normal FA, the fragment ions of hydroxy FA of lithium adducts as [diOH18:2 + Li]+ at m/z 319.1 and [diOH18:1 + Li]+ at m/z 321.1 were prominent, while the fragment ions from the neutral losses of hydroxy FA [M + Li − diOH18:2]+ at m/z 347.1 and [M + Li − diOH18:1]+ at m/z 345.1 were either not detected or trace only. For the same two DAG, the fragment ions from the neutral losses of normal FA, [M + Li − S]+ at m/z 375.1 and [M + Li − O]+ at m/z 377.1, were prominent and the fragment ions of the lithium adducts of these two normal FA, [S + Li]+ at m/z 291.1, [O + Li]+ at m/z 289.1, were not detected. For DAG OH18:1-OH18:1 containing two monohydroxy FA, both [OH18:1 + Li]+ at m/z 305.1 and [M + Li − OH18:1]+ at m/z 361.1 were prominent. Again this example showed that the more hydroxylated FA of a DAG tend to have prominent fragment ion [FA + Li]+ and undetectable fragment ion [M + Li − FA]+. Also the less hydroxylated FA of a DAG tend to have prominent fragment ion [M + Li − FA]+ and undetectable fragment ion [FA + Li]+. We have quantified DAG containing two normal FA in the soybean oil bioconversion product by HPLC using an evaporative light scattering detector [13].
The MS spectra of DAG containing hydroxy FA (one or two hydroxy FA) were simple as shown in Figs. 1 and 3. The MS spectra of DAG containing two normal FA were more complicated as shown in Fig. 4. Figure 4 is the MS2 spectrum of DAG, OL, in the soybean oil bioconversion product from [M + Li]+ at m/z 625.4 [13]. The most abundant fragment ion was [M + Li − H2O]+ at m/z 607.4. The other prominent fragment ions were [M + Li − L]+ at m/z 345.1, [M + Li − O]+ at m/z 343.1, [M + Li − LLi]+ at m/z 339.1, [M + Li − OLi]+ at m/z 337.1, [O + Li]+ at m/z 289.1 and [L + Li]+ at m/z 287.1. Their relative abundances are shown in Table 2. Table 2 shows the MS2 characteristic ions of thirteen DAG containing two normal FA in the soybean bioconversion product (LL and LnLn were the standards). Some of the DAG in Table 2 might not be commercially available (e.g., LP, OL, OP, OS, OA, SA and PB). We have shown that the MS2 spectrum of PP standard was identical to that of PP in the soybean oil bioconversion product. The relative abundances of the fragment ions are also given in Table 2. Consistently the most abundant fragment ions (base peak) were [M + Li − H2O]+ and these fragment ions were also shown for the MS2 spectra of DAG and TAG containing hydroxy FA at much lower abundances (Figs. 1, 2) [7−11]. The relative abundance of [M + Li − FA]+ and [M + Li − FALi]+ were generally more abundant than those of [FA + Li]+.
We have found only one MS2 spectrum of DAG containing two normal FA (SS) recently reported as far as we aware by Bowden et al. [12]. The fragment ions of SS were the same as those shown in Table 2. In this recent report, fragment ions were also selected for the detection of seven DAG containing normal FA using selected reaction monitoring (SRN) [12].
Conclusions
We have used the MS2 fragment ions of [M + Li − FA]+ and [FA + Li]+ for the identification of DAG containing hydroxy FA (one or two hydroxy FA) in castor oil. The additional fragment ions, [M + Li − FALi]+, were used for the identification of DAG containing two normal FA. The MS2 spectra of DAG containing hydroxy FA (Figs. 1, 2) were simple compared to those of DAG containing two normal FA (Fig. 4), mainly because the fragment ions from the neutral losses of FA lithium salts, [M + Li − FALi]+, were not detectable for the DAG containing hydroxy FA. The MS2 spectra of TAG of lithium adducts showed a similar pattern.
References
Lin JT, Turner C, Liao LP, McKeon TA (2003) Identification and quantification of the molecular species of acylglycerols in castor oil by HPLC using ELSD. J Liq Chromatogr Relat Technol 26:773–780
Lin JT, Chen GQ (2012) Identification of minor acylglycerols less polar than triricinolein in castor oil by mass spectrometry. J Am Oil Chem Soc 89:1773–1784
Hsu FF, Turk J (2005) Electrospray ionization with low-energy collisionally activated dissociation tandem mass spectrometry of complex lipids: structural characterization and mechanisms of fragmentation. In: Byrdwell WC (ed) Modern methods for lipid analysis. AOCS Press, Champaign, pp 61–178
Hsu FF, Turk J (1999) Structural characterization of triacylglycerols as lithiated adducts by electrospray ionization mass spectrometry using low-energy collisionally activated dissociation on a triple stage quadrupole instrument. J Am Soc Mass Spectrom 10:587–599
Hsu FF, Turk J (2010) Electrospray ionization multiple-stage linear ion-trap mass spectrometry for structural elucidation of triacylglycerols: assignment of fatty acyl groups on the glycerol backbone and location of double bond. J Am Soc Mass Spectrom 21:657–669
Lin JT (2010) The use of lithiated adducts for structural analysis of acylglycerols by mass spectrometry with electrospray ionization. In: AOCS Lipid Library webpage. http://lipidlibrary.aocs.org/topics/Lin-Li-adducts/index.htm
Lin JT, Arcinas A (2007) Regiospecific analysis of diricinoleoylacylglycerols in castor (Ricinus communis L.) oil by electrospray ionization-mass spectrometry. J Agric Food Chem 55:2209–2216
Lin JT, Arcinas A, Harden AH (2009) Identification of acylglycerols containing dihydroxy fatty acids in castor oil by mass spectrometry. Lipids 44:359–365
Lin JT (2009) Ratios of regioisomers of triacylglycerols containing dihydroxy fatty acids in castor oil by mass spectrometry. J Am Oil Chem Soc 86:1031–1035
Lin JT, Chen GQ (2010) Acylglycerols containing trihydroxy fatty acids in castor oil and the regiospecific quantification of triacylglycerols. J Am Oil Chem Soc 87:1371–1379
Lin JT, Chen GQ (2011) Identification of diacylglycerol and triacylglycerol containing 11,12,13-trihydroxy-9,14-octadecadienoic acid in castor oil. New Biotechnol 28:203–208
Bowden JA, Albert CJ, Barnaby OS, Ford DA (2011) Analysis of cholesteryl esters and diacylglycerols using lithiated adducts and electrospray ionization-tandem mass spectrometry. Anal Biochem 417:202–210
Hou CT, Lin JT (2012) Production of polyol oils from soybean oil through bioprocess. Biocat Agric Biotech (submitted)
Lin JT et al (1998) Biosynthesis of triacylglycerols containing ricinoleate in castor microsomes using 1-acyl-2-oleoyl-sn-glycerol-3-phosphocholine as the substrate of oleoyl-12-hydroxylase. Lipids 33:59–69
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Lin, JT., Chen, G.Q. & Hou, C.T. Mass Spectrometry of the Lithium Adducts of Diacylglycerols Containing Hydroxy FA in Castor Oil and Two Normal FA. J Am Oil Chem Soc 90, 33–38 (2013). https://doi.org/10.1007/s11746-012-2136-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11746-012-2136-5