Abstract
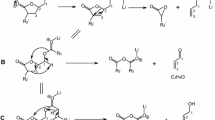
We have examined the biosynthetic pathway of triacylglycerols containing ricinoleate to determine the steps in the pathway that lead to the high levels of ricinoleate incorporation in castor oil. The biosynthetic pathway was studied by analysis of products resulting from castor microsomal incubation of 1-palmitoyl-2-[14C]oleoyl-sn-glycero-3-phosphocholine, the substrate of oleoyl-12-hydroxylase, using high-performance liquid chromatography, gas chromatography, mass spectrometry, and/or thin-layer chromatography. In addition to formation of the immediate and major metabolite, 1-palmitoyl-2-[14C]rici-noleoyl-sn-glycero-3-phosphocholine, 14C-labeled 2-linoleoyl-phosphatidylcholine (PC), and 14C-labeled phosphatidylethanolamine were also identified as the metabolites. In addition, the four triacylglycerols that constitute castor oil, triricinolein, 1,2-diricinoleoyl-3-oleoyl-sn-glycerol, 1,2-diricinoleoyl-3-linoleoyl-sn-glycerol, 1,2-diricinoleoyl-3-linolenoyl-sn-glycerol, were also identified as labeled metabolites in the incubation along with labeled fatty acids: ricinoleate, oleate, and linoleate. The conversion of PC to free fatty acids by phospholipase A2 strongly favored ricinoleate among the fatty acids on the sn-2 position of PC. A major metabolite, 1-palmitoyl-2-oleoyl-sn-glycerol, was identified as the phospholipase C hydrolyte of the substrate; however, its conversion to triacylglycerols was blocked. In the separate incubations of 2-[14C]ricinoleoyl-PC and [14C]ricinoleate plus CoA, the metabolites were free ricinoleate and the same triacylglycerols that result from incubation with 2-oleoyl-PC. Our results demonstrate the proposed pathway: 2-oleoyl-PC. Out results demonstrate the proposed pathway: 2-oleoyl-PC→2-ricinoleoyl-PC→ricinoleate →triacylglycerols. The first two steps as well as the step of diacylglycerol acyltransferase show preference for producing ricinoleate and incorporating it in triacylglycerols over oleate and linoleate. Thus, the productions of these triacylglycerols in this relatively short incubation (30 min), as well as the availability of 2-oleoyl-PC in vivo, reflect the in vivo drive to produce triricinolein in castor bean.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- DGDG:
-
digalactosyl diacylglycerol
- FAB:
-
fast atom bombardment
- FAME:
-
fatty acid methyl esters
- FFA:
-
free fatty acids
- GC:
-
gas chromatography
- HPLC:
-
high-performance liquid chromatography
- 2-linolenoyl-PC:
-
1-acyl-2-linolenoyl-sn-glycero-3-phosphocholine
- 2-linoleoyl-PC:
-
1-acyl-2-linoleoyl-sn-glycero-3-phosphocholine
- MGDG:
-
monogalactosyl diacylglycerol
- MS:
-
mass spectrometry or mass spectrum
- NAPE:
-
N-acyl-phosphatidylethanolamine
- 2-oleoyl-PC:
-
1-acyl-2-oleoyl-sn-glycero-3-phosphocholine
- PC:
-
phosphatidylcholine
- PE:
-
phosphatidylethanolamine
- PI:
-
phosphatidylinositol
- 2-ricinoleoyl-PC:
-
1-acyl-2-ricinoleoyl-sn-glycero-3-phosphocholine
- TLC:
-
thin-layer chromatography
References
van de Loo, F.J., Broun, P., Turner, S., and Somerville, C. (1995) An Oleate 12-Hydroxylase from Ricinus communis L. Is a Fatty Acyl Desaturase Homolog, Proc. Natl. Acad. Sci. USA 92, 6743–6747.
Broun, P., Hawker, N., and Somerville, C.R. (1997) Expression of Castor and L. fendleri Oleate 12-Hydroxylases in Transgenic Plants, Physiology, Biochemistry and Molecular Biology of Plant Lipids (Williams, J.P., Khan, M.U., and Lem, N.W., eds.) pp. 342–344, Kluwer, Dordrecht.
Bafor, M., Smith, M.A., Jonsson, L., Stobart, K., and Stymne, S. (1991) Ricinoleic Acid Biosynthesis and Triacylglycerol Assembly in Microsomal Preparations from Developing Castor Bean (Ricinus communis) Endosperm, Biochem. J. 280, 507–514.
Moreau, R.A., and Stumpf, P.K. (1981) Recent Studies of the Enzymic Synthesis of Ricinoleic Acid by Developing Castor Beans, Plant Physiol. 67, 672–676.
Lin, J.T., McKeon, T.A., Goodrich-Tanrikulu, M., and Stafford, A.E. (1996) Characterization of Oleoyl-12-hydroxylase in Castor Microsomes Using the Putative Substrate, 1-Acyl-2-oleoyl-sn-glycero-3-phosphocholine, Lipids 31, 571–577.
McKeon, T.A., Lin, J.T., Tanrikulu, M.G., and Stafford, A.E. (1997) Ricinoleate Biosynthesis in Castor Microsomes, Industrial Crops Products 6, 383–389.
Lin, J.T., McKeon, T.A., and Stafford, A.E. (1995) Gradient Reversed-Phase High-Performance Liquid Chromatography of Saturated, Unsaturated and Oxygenated Free Fatty Acids and Their Methyl Esters, J. Chromatogr. A 699, 85–91.
Singleton, J.A., and Stikeleather, L.F. (1995) High-Performance Liquid Chromatography Analysis of Peanut Phospholipids. II Effect of Postharvest Stress on Phospholipid Composition, J. Am. Oil Chem. Soc. 72, 485–488.
Lin, J.T., Woodruff, C.L., and McKeon, T.A. (1997) Non-Aqueous Reversed-Phase High-Performance Liquid Chromatography of Synthetic Triacylglycerols and Diacylglycerols, J. Chromatogr. A 782, 41–48.
Tanrikulu, M.G., Stafford, A.E., Lin, J.T., Makapugay, M.I., Fuller, G., and McKeon, T.A. (1994) Fatty Acids Biosynthesis in Novel ufa Mutants of Neurospora crassa, Microbiology 140, 2683–2690.
Kates, M. (1986) Techniques of Lipidology. Isolation, Analysis and Identification of Lipids, 2nd edn., Vol. 3, part 2 in Laboratory Techniques in Biochemistry and Molecular Biology, (Burdon, R.H., and van Knippenberg, P.H., eds.) Elsevier, Amsterdam.
Joyard, J., and Douce, R. (1993) Galactolipid Synthesis, in Lipid Metabolism in Plants (Moore, T.S., ed.) pp. 215–274.
Chapman, K.D., and Moore, T.S. (1993) Catalytic Properties of a Newly Discovered Acyltransferase That Synthesizes N-Acylphosphatidylethanolamine in Cottonseed (Gossypium hirsutum L.) Microsomes, Plant Physiol. 102, 761–769.
Sparace, S.A., Wagner, L.K., and Moore, T.S. (1981) Phosphatidylethanolamine Biosynthesis in Castor Bean Endosperm, Plant Physiol. 67, 922–925.
Shin, S., and Moore, T.S. (1990) Phosphatidylethanolamine Synthesis by Castor Bean Endosperm, a Base Exchange Reaction, Plant Physiol. 93, 148–153.
Kinney, A.J. (1993) Phospholipid Head Groups, in Lipid Metabolism in Plants (Moore, T.S., ed.) pp. 259–284, CRC Press, Boca Raton.
Stahl, U., Banas, A., and Stymne, S. (1995) Plant Microsomal Phospholipid Acyl Hydrolases Have Selectivities for Uncommon Fatty Acids, Plant Physiol. 107, 953–962.
Wang, X., Dyer, J.H., and Zheng, L. (1993) Purification and Immunological Analysis of Phospholipase D from Castor Bean Endosperm, Arch. Biochem. Biophys. 306, 486–494.
Stobart, K., Mancha, M., Lenman, M., Dahlqvist, A., and Stymne, S. (1997) Triacylglycerols Are Synthesized and Utilized by Transacylation Reaction in Microsomal Preparations of Developing Safflower (Carthamus tinctorius L.) Seeds, Planta 203, 58–66.
Mancha, M., and Stymne, S. (1997) Remodelling of Triacylglycerols in Microsomal Preparations from Developing Castor Bean (Ricinus communis L.) Endosperm, Planta 203, 51–57.
Smith, M.A., Jonsson, L., Stymne, S., and Stobart, K. (1992) Evidence for Cytochrome b 5 as an Electron Donor in Ricinoleic Acid Biosynthesis in Microsomal Preparations from Developing Castor Bean (Ricinus communis L.), Biochem. J. 287, 141–144.
Bafor, M., Jonsson, L., Stobart, A.K., and Stymne, S. (1990) Regulation of Triacylglycerol Biosynthesis in Embryos and Microsomal Preparations from the Developing Seeds of Cuphea lanceolata, Biochem. J. 272, 31–38.
Vogel, G., and Browse, J. (1996) Cholinephosphotransferase and Diacylglycerol Acyltransferase, Plant Physiol. 110, 923–931.
Wiberg, E., Tillberg, E., and Stymne, S. (1994) Substrates of Diacylglycerol Acyltransferase in Microsomes from Developing Oil Seeds, Phytochemistry 36, 573–577.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Lin, JT., Woodruff, C.L., Lagouche, O.J. et al. Biosynthesis of triacylglycerols containing ricinoleate in castor microsomes using 1-acyl-2-oleoyl-sn-glycero-3-phosphocholine as the substrate of oleoyl-12-hydroxylase. Lipids 33, 59–69 (1998). https://doi.org/10.1007/s11745-998-0180-3
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11745-998-0180-3