Abstract
Adiponectin is a polypeptide known to inhibit cardiac fibrosis via the activation of adenosine monophosphate-activated protein kinase (AMPK). Statins can also activate AMPK, resulting in the secretion of adiponectin. We determined whether atorvastatin inhibits angiotensin II-induced cardiac fibrosis (AICF) in the presence or absence of adiponectin. Adiponectin knockout (APN-KO, n = 44) and wild type (WT, n = 44) mice were received subcutaneous angiotensin II (1.5 mg/kg/day), and atorvastatin (10 mg/kg/day) was administered orally for 15 days. The mRNA expression levels of collagen type I and III, as well as AMPK phosphorylation levels in cardiac tissue were then measured. In the APN-KO mice, collagen type I (p < 0.001) and type III (p = 0.001) expression was significantly greater when treated with angiotensin II, while their expression was significantly reduced in the presence of angiotensin II and atorvastatin. Relative AMPK phosphorylation levels in APN-KO mice were also significantly higher in the angiotensin II + atorvastatin group when compared with angiotensin II group alone. We conclude that atorvastatin attenuates AICF independently from adiponectin by activating AMPK. These data suggest potential cardioprotection beyond lipid modulation potentially supporting statin pleiotropic hypothesis.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Adenosine monophosphate-activated protein kinase (AMPK) is a key player in maintaining cellular homeostasis. Adenosine triphosphate (ATP) levels are often depleted during cardiac diseases, stimulating AMPK to impact key metabolic enzymes and transcription factors in order to replenish cellular ATP content [1, 2]. While the primary function of AMPK is regulating of cellular energy, this enzyme is also crucial for cell protection preventing cardiomyocyte hypertrophy and fibrosis of the extracellular matrix [3,4,5]. Importantly, AMPK can be activated by various external stimuli, including adiponectin and metabolic demands [6]. Adiponectin is a potent cardioprotective adipokine with antidiabetic and anti-inflammatory properties that are mainly mediated by the activation of AMPK [7,8,9].
Statins are 3-hydroxy-3-methylgutaryl-coenzyme A reductase inhibitors commonly prescribed for cholesterol control. However, they have also been reported to exhibit potent cardioprotective effects by inhibiting cardiac hypertrophy and fibrosis supporting the pleiotropic hypothesis [10, 11]. Despite some sporadic observations [12,13,14,15,16], the precise mechanisms of extra antifibrotic effects have not been ascertained, although some studies have demonstrated that the inhibition of cardiac hypertrophy and fibrosis can occur via AMPK activation [15, 16]. Additionally, it is well established that statins increase serum concentrations of adiponectin [17,18,19,20]. However, it remains unclear whether AMPK activation by statins occurs via statin-dependent adiponectin secretion, and the roles of statin-AMPK and statin-adiponectin-AMPK interactions are unknown.
We here determined whether atorvastatin activates AMPK and decreases angiotensin II-induced cardiac fibrosis(AICF) with an associated increase in adiponectin secretion in an animal model.
Materials and Methods
Animals
The study was conducted in accordance with criteria outlined in the Guide for the Care and Use of Laboratory Animals and approved by the Ethics Committee for Animal Experimentation of Dong-A University (protocol approval number: DIACUC-12-21). The in vivo experiments were performed using 44 wild-type (WT) and 44 adiponectin knockout (APN-KO) (Ad −/−) male mice with pure C57BL/6 J backgrounds (The Jackson Laboratory, Bar Harbor, ME, USA). The mice were between 12 and 14 weeks, weighed between 25 and 30 g, and were maintained at a fixed temperature of 22 °C and fed regular chow. None of the experimental animals died of any causes prior to euthanasia, and all were humanely euthanized with pentobarbital sodium for tissue collection. The matched 44 WT and 44 APN-KO mice were evenly assigned to eight groups, each consisting of 11 WT or 11 APN-KO animals, predefined as (1) saline, (2) saline + atorvastatin, (3) angiotensin II, and (4) angiotensin II + atorvastatin groups. The study flow chart algorithm is outlined in Fig. 1.
Angiotensin II and Atorvastatin Administration
The mice were anesthetized with pentobarbital sodium (50 mg/kg, intraperitoneally) (Hanlim Pharmaceuticals Inc., Seoul, Republic of Korea). A2900 angiotensin II acetate salt (Sigma-Aldrich Inc., St. Louis, MO, USA) was dissolved in normal saline and infused by Alzet® model 2002 mini-osmotic pumps (Alzet Osmotic Pumps, Cupertino, CA, USA). The pumps containing angiotensin II or normal saline control were implanted subcutaneously in the posterior cervical region, and infused continuously (at 1.5 mg/kg/day) for 15 days [21, 22]. The groups treated with atorvastatin (Chong Kun Dang Pharm Corporation, Seoul, Republic of Korea) received a dose of 10 mg/kg/day mixed with regular chow during the treatment phase [15, 23, 24]. Treatment with atorvastatin began 2 days before pump implantation and continued for 15 days. After infusion, the hearts were excised and frozen at –80 °C in liquid nitrogen until subsequent extraction of RNA or protein.
Blood Collection
The blood samples were collected from the abdominal vein under anesthesia and placed at room temperature for 20 min and then on ice for 20 min. The samples were allowed to clot, before centrifugation at 3000 rpm for 10 min at 4 °C. The serum samples were stored at −80 °C until further analysis.
Myocardial Tissue Collection
Under anesthesia induced by pentobarbital sodium, the hearts were removed and briefly rinsed with normal saline to remove blood and foreign substances, blotted dry, and weighed. All heart specimens were immediately stored in liquid nitrogen at –80 °C until protein and RNA extraction.
Serum Adiponectin
The high molecular weight adiponectin levels in plasma were measured using enzyme-linked immunosorbent assay (ELISA) (96-well plate, mouse adiponectin ELISA kit; ALPCO, Salem, NH, USA), according to the manufacturer’s protocol. The protein samples and standard solution were quantified at an absorbance wavelength of 492 nm.
Protein Extraction and Western Blot Analysis
The snap-frozen specimens were homogenized in 10 mL of Invitrogen™ FNN0071 tissue extraction reagent (Invitrogen Corporation, Carlsbad, CA, USA) per 1 g of heart tissue for the extraction of protein. The reagent was supplemented with an A2714 protease inhibitor cocktail (Sigma-Aldrich) immediately prior to its use. The homogenates were centrifuged at 10,000 rpm for 5 min and total protein concentrations were measured using bicinchoninic acid protein assay kits (Sigma-Aldrich).Samples were separated by electrophoresis on 15% sodium dodecyl sulfate–polyacrylamide gels, and then transferred to a polyvinylidene fluoride membrane (General Electric Healthcare, Piscataway, NJ, USA). The membranes were treated with 5% bovine serum albumin while being gently agitated for 1 h at room temperature and overnight at 4 °C. After blocking, the membranes were incubated with a Thr 172 phospho-AMPKα primary antibody (Cell Signaling Technology, Danvers, MA, USA) at a 1:1,000 dilution while being gently agitated overnight at 4 °C. The membranes were washed three times before incubation with a secondary horseradish peroxidase-linked antibody (1:1000, Cell Signaling Technology). After incubation with the secondary antibody, the membranes were washed three times. Bands were detected with ECL™ Western Blotting Detection Reagent (Pierce Chemical Corporation, Rockford, IL, USA).
Polymerase Chain Reaction
Total RNA was isolated from the snap-frozen, homogenized heart samples using Trizol reagent (Invitrogen).The expression levels of collagen type I and type III mRNA as markers of cardiac fibrosis were measured by reverse transcription polymerase chain reaction with a SYBR Green PCR kit and Rotor-Gene Q sequence detection system (QIAgen, Hilden, Germany). The primer sequences for mouse collagen type I, collagen type III, and glyceraldehyde 3-phosphate dehydrogenase were designed by Bioneer Corporation (Daejeon, Republic of Korea), with the following primers used: mouse collagen type I, 5′-GTCCCAACCCCCAAAGAC-3′ (forward) and 5′-CATCTTCTGAGTTTGGTGATACGT-3′ (reverse); mouse collagen type III, 5′-TGGTTTCTTCTCACCCTTCTTC-3′ (forward) and 5′-TGCATCCCAATTCATCTACGT-3′ (reverse); glyceraldehyde 3-phosphate dehydrogenase (GAPDH), 5′-CCTGCACCACCAACTGCTTA-3′ (forward) and 5′-TCATGAGCCCTTCCACAATG-3′ (reverse). Collagen type I and III mRNA levels were reported relative to the internal control (GAPDH).
Histological Examination of Cardiac Tissue
The extent of cardiac fibrosis was examined by light microscopy. Immediately following harvest, the specimens were fixed in 10% phosphate-buffered formalin, embedded in paraffin, sliced into 5-mm-thick sections, and stained with hematoxylin-eosin and Masson’s trichrome (Sigma-Aldrich). The slides were inspected at 400× magnification with an Olympus 4 light microscope, and images were obtained with Aperio Image Socpe software (Leica Biosystems, Newcastle Ltd., UK). Areas of connective tissue deposition were measured using ImageJ software (National Institutes of Health, Bethesda, MD, USA). The measurements are expressed as relative area of connective tissue stained with Masson’s trichrome per total myocardial tissue area in each field.
Statistical Analysis
The data are expressed as mean ± standard error (SEM). Significant differences between two groups were ascertained by independent t test, with one-way analysis of variance and Scheffe’s test for post hoc analyses used for comparisons among multiple groups. All statistical comparisons were two-sided and p values of <0.05 were considered statistically significant. All analyses were performed using SPSS version 12.0 (SPSS Inc., Chicago, IL, USA).
Results
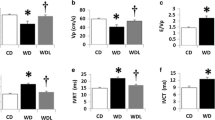
Effects of Angiotensin II and Atorvastatin on Body and Heart Weights
In both WT and APN-KO mice, the heart weight (HW)/body weight (BW) ratios were consistently higher in the angiotensin II and angiotensin II + atorvastatin group than in the saline or saline + atorvastatin group (Table 1). However, the HW/BW ratio was not significantly different between two groups.
Adiponectin Levels
To assess the effect of atorvastatin on adiponectin secretion, we analyzed mean serum high molecular weight adiponectin levels in WT mice. There were no significant differences observed (p = 0.258), suggesting that the doses of angiotensin II administered did not act as a stress stimuli for adiponectin in WT mice (see Fig. 2 for details).
Collagen Type I and III Expression
The mRNA expression levels of collagen type I and type III were assessed as quantitative markers of angiotensin II-induced cardiac fibrosis. In WT mice, no significant between-group differences in the mRNA levels of collagen type I (p = 0.261) or III (p = 0.135) were observed. In the APN-KO mice, relative collagen type I (p < 0.001) and type III (p = 0.001) expression was significantly higher in the angiotensin II group (Fig. 3), while levels of collagen type I (2.09 ± 0.11 vs. 4.76 ± 0.47; p < 0.001) and type III (1.43 ± 0.06 vs. 3.00 ± 0.55; p = 0.027) were significantly reduced in the angiotensin II + atorvastatin group when compared to the angiotensin II only group.
Effect of adiponectin and atorvastatin treatment on collagen type I and III mRNA expression as a marker of cardiac fibrosis. In WT mice, there were no significant between-group differences in collagen type I and III mRNA levels. In APN-KO mice, expression of both transcripts was significantly higher in the angiotensin II group and significantly lower in the angiotensin II + atorvastatin when compared to angiotensin II treatment alone. (n = 4 mice per group. *p < 0.05, **p < 0.01, ***p < 0.001. Ang II angiotensin II, Ator atorvastatin)
Cardiac Fibrosis
Masson’s trichrome staining techniques revealed denser and wider areas of connective tissue deposition in APN-KO mice in the angiotensin II group than in the other groups (Fig. 4a). Connective tissue deposition was also significantly higher in the angiotensin II-treated APN-KO mice (p = 0.002), while the extent of connective tissue deposition was borderline significantly reduced in the angiotensin II + atorvastatin group compared with the angiotensin II group (14.23 ± 2.99 vs. 32.33 ± 6.48%, p = 0.049). However in the WT mice, there were no significant between-group differences in terms of connective tissue deposition (p = 0.064) (Fig. 4b).
Effect of adiponectin and atorvastatin on connective tissue deposition as a marker of cardiac fibrosis. a Representative light microscope images of cardiac sections at 400× magnification with Masson’s trichrome stain. The areas of connective tissue deposition are denser and wider in response to angiotensin II treatment in the APN-KO mice. b Quantification of connective tissue deposition following the indicated treatments in APN-KO mice. (n = 3 mice per group. *p < 0.05, **p < 0.01, WT wild type, APN-KO adiponectin knockout, Ang II angiotensin II, Ator atorvastatin)
AMPK Phosphorylation
There were no significant between-group differences (p = 0.248) in terms of relative AMPK phosphorylation levels among WT mice (Fig. 5a). However, in the APN-KO mice, relative AMPK phosphorylation levels were significantly increased (p = 0.011) in the angiotensin II + atorvastatin group (Fig. 5b). However, the phosphorylation levels among the saline, saline + atorvastatin, and angiotensin II groups were similar.
Effects of atorvastatin and adiponectin treatment on AMPK phosphorylation in mouse cardiac tissue. a Angiotensin II and atorvastatin treatment has no significant effect on AMPK phosphorylation in WT mice. b In APN-KO mice, AMPK phosphorylation levels increase only in the presence of both angiotensin II + atorvastatin. (n = 4 mice per group. *p < 0.05. Ang II angiotensin II, Ator atorvastatin)
Discussion
The main finding of the index study is the fact that atorvastatin attenuates cardiac fibrosis independently from adiponectin by modulating adenosine monophosphate-activated protein kinase. These data have clinical implications suggesting cardioprotection beyond lipid modulation supporting statin pleiotropic hypothesis. As we initially hypothesized, atorvastatin treatment activated AMPK in cardiac tissue and attenuated angiotensin II-induced cardiac fibrosis in APN-KO mice. The index data are in agreement with a recent study reported that statins can inhibit cardiac fibrosis via AMPK activation [16], since it is well known that the potent cardioprotective effects of adiponectin are directly mediated by AMPK [7,8,9]. In light of these facts, we applied APN-KO mice to exclude the effect of adiponectin secretion in order to understand better the underlying molecular events responsible for such association. In fact, use of the genetic knockout model confirmed that atorvastatin treatment both activates AMPK and inhibits cardiac fibrosis in the absence of adiponectin. In our murine model, cardiac fibrosis was induced by a continuous subcutaneous infusion of angiotensin II with an osmotic mini-pump. Angiotensin II is a prominent stressor that induces tissue fibrosis by activating mitogen-activated protein kinases such as extracellular signal-regulated kinase (ERK) 1/2 [25], which in turn can be suppressed by activated AMPK [4]. In a similarly designed experimental study, extensive cardiac fibrosis was induced in APN-KO mice treated with angiotensin II [8]. It appears that the relatively low dose of angiotensin II selected did not induce significant amounts of cardiac fibrosis in WT mice with normal serum concentrations of adiponectin, but had considerable effects on cardiac fibrosis in APN-KO mice. The observation of extensive cardiac fibrosis only in the APN-KO mice suggests that adiponectin is a strong antifibrotic promoter.
While a prior experimental study has reported that atorvastatin activates AMPK in cardiomyocytes and suppresses cardiac fibrosis in WT mice [15], the relatively low doses (10 mg/kg/day) of atorvastatin used in the present study significantly attenuated cardiac fibrosis in our APN-KO mice. These observations suggest that the preventive role played by statins against cardiac fibrosis may be amplified in conditions associated with low serum concentrations of adiponectin, and may be clinically relevant.
To shed further light on the molecular mechanisms responsible for the antifibrotic effects of atorvastatin, we measured myocardial AMPK phosphorylation levels. Atorvastatin alone did not increase AMPK phosphorylation in the anigotensin II-treated WT mice, showing that the low dose of angiotensin II used was not sufficient to act as a stressor of AMPK phosphorylation in the presence of adiponectin. However, in the angiotensin II-treated APN-KO mice, significant AMPK phosphorylation was induced by atorvastatin and resulted in the inhibition of cardiac fibrosis. This observation suggests that atorvastatin activates AMPK independently from adiponectin secretion. Importantly, we applied low-dose angiotensin II, compared with the similar study which assessed much higher amounts (3.2 mg/kg/day) causing severe cardiac fibrosis in adiponectin deficient mice (21, 22). Also, we wanted to certify the protective function of adiponectin. With regard to the statin dose pick, previous studies used simvastatin or rosuvastatin (20 mg/kg/day orally), we also applied lower doses. In fact, we used half-dose 10 mg/kg/day atorvastatin (23, 24) resulted in the similar impact.
We were unable to assess transforming growth factor (TGF)-β1 or ERK phosphorylation levels as markers of tissue fibrosis. Angiotensin II causes tissue fibrosis by inducing the synthesis of TGF-β1 [26] and by activating ERK 1/2 [25]. Conversely, the synthesis of TGF-β1 and activation of ERK 1/2 can be inhibited by activated AMPK [4]. We cannot confirm whether atorvastatin, by activating AMPK, actually downregulates TGF-β1 synthesis or ERK phosphorylation. The administration of atorvastatin may also have had effects on matrix metalloproteinase, Smad, Rho kinase, and mitogen-activated protein kinases pathways [12,13,14]. There is some growing evidence that statin pleiotropy may be the real finding, rather than random laboratory artifact. For example, statins universally inhibit platelet protease-activated receptor-1 thrombin receptor reducing thrombosis burden in patients with metabolic syndrome [26]. These results indicate that the statin-AMPK regulatory axes are likely to play an important role in the prevention of cardiac fibrosis in the absence of adiponectin. Our findings also suggest that the role of statins in the activation of AMPK and inhibition of cardiac fibrosis may be greater in clinical disorders associated with low serum concentrations of adiponectin, such as obesity, diabetes mellitus, coronary artery disease, and heart failure [27].
Our study has some obvious limitations. The major shortcoming is how to extrapolate even elegant basic science animal data into a clinically meaningful message. Also, the mechanistic explanation of the observed findings is lacking; therefore, these data should be considered preliminary.
In conclusion, atorvastatin activates myocardial AMPK and inhibits angiotensin II-induced cardiac fibrosis in the complete absence of adiponectin. Low doses of atorvastatin also activated AMPK and caused prominent cardiac antifibrotic effects in the absence of adiponectin.
Change history
13 October 2017
In the original published article, Victor Serebruany was designated as the corresponding author in error. The corresponding author is Jong Sung Park. Dr. Park can be reached by email at thinkmed@dau.ac.kr.
Abbreviations
- AMPK:
-
Adenosine monophosphate-activated protein kinase
- ATP:
-
Adenosine triphosphate
- APN-KO:
-
Adiponectin knockout
- AICF:
-
Angiotensin II-induced cardiac fibrosis
- BW:
-
Body weight
- ERK:
-
Extracellular signal-regulated kinase
- HW:
-
Heart weight
- TGF:
-
Transforming growth factor
References
Beauloye C, Bertrand L, Horman S, Hue L (2011) AMPK activation, a preventive therapeutic target in the transition from cardiac injury to heart failure. Cardiovasc Res 90(2):224–233
Hardie DG (2007) AMP-activated/SNF1 protein kinases: conserved guardians of cellular energy. Nat Rev Mol Cell Biol 8(10):774–785
Zhang P, Hu X, Xu X, Fassett J, Zhu G, Viollet B, Xu W, Wiczer B, Bernlohr DA, Bache RJ, Chen Y (2008) AMP activated protein kinase-alpha2 deficiency exacerbates pressure-overload-induced left ventricular hypertrophy and dysfunction in mice. Hypertension 52(5):918–924
Du J, Guan T, Zhang H, Xia Y, Liu F, Zhang Y (2008) Inhibitory crosstalk between ERK and AMPK in the growth and proliferation of cardiac fibroblasts. Biochem Biophys Res Commun 368(2):402–407
Stuck BJ, Lenski M, Böhm M, Laufs U (2008) Metabolic switch and hypertrophy of cardiomyocytes following treatment with angiotensin II are prevented by AMP-activated protein kinase. J Biol Chem 283(47):32562–32569
Yamauchi T, Kamon J, Minokoshi Y, Ito Y, Waki H, Uchida S, Yamashita S, Noda M, Kita S, Ueki K, Eto K, Akanuma Y, Froguel P, Foufelle F, Ferre P, Carling D, Kimura S, Nagai R, Kahn BB, Kadowaki T (2002) Adiponectin stimulates glucose utilization and fatty-acid oxidation by activating AMP-activated protein kinase. Nat Med 8(11):1288–1295
Xu A, Vanhoutte PM (2012) Adiponectin and adipocyte fatty acid binding protein in the pathogenesis of cardiovascular disease. Am J Physiol Heart Circ Physiol 302(6):H1231–H1240
Fujita K, Maeda N, Sonoda M, Ohashi K, Hibuse T, Nishizawa H, Nishida M, Hiuge A, Kurata A, Kihara S, Shimomura I, Funahashi T (2008) Adiponectin protects against angiotensin II-induced cardiac fibrosis through activation of PPAR-alpha. Arterioscler Thromb Vasc Biol 28(5):863–870
Konishi M, Haraguchi G, Ohigashi H, Ishihara T, Saito K, Nakano Y, Isobe M (2011) Adiponectin protects against doxorubicin-induced cardiomyopathyby anti-apoptotic effects through AMPK up-regulation. Cardiovasc Res 89(2):309–319
Zhou Q, Liao JK (2010) Pleiotropic effects of statins-Basic research and clinical perspectives. Circ J 74(5):818–826
Yamamoto C, Fukuda N, Jumabay M, Saito K, Matsumoto T, Ueno T, Soma M, Matsumoto K, Shimosawa T (2011) Protective effects of statin on cardiac fibrosis and apoptosis in adrenomedullin-knockout mice treated with Ang II and high salt loading. Hypertens Res 34(3):348–353
An Z, Yang G, He YQ, Dong N, Ge LL, Li SM, Zhang WQ (2013) Atorvastatin reduces myocardial fibrosis in a rat model with post-myocardial infarction heart failure by increasing the matrix metalloproteinase-2/tissue matrix metalloproteinase inhibitor-2 ratio. Chin Med J (Engl) 126(11):2149–2156
Rodrigues Díez R, Rodrigues-Díez RR, Lavoz C, Rayego-Mateos S, Civantos E, Rodríguez-Vita J, Mezzano S, Ortiz A, Egido J, Ruiz-Ortega M (2010) Statins inhibit angiotensin II/Smad pathway and related vascular fibrosis, by a TGF-β-independent process. PLoS One 5(11):e14145
Rupérez M, Rodrigues-Díez R, Blanco-Colio LM, Sánchez-López E, Rodríguez-Vita J, Esteban V, Carvajal G, Plaza JJ, Egido J, Ruiz-Ortega M (2007) HMG-CoA reductase inhibitors decrease angiotensin II-induced vascular fibrosis: role of RhoA/ROCK and MAPK pathways. Hypertension 50(2):377–383
Sun W, Lee TS, Zhu M, Gu C, Wang Y, Zhu Y, Shyy JY (2006) Statins activate AMP-activated protein kinase in vitro and in vivo. Circulation 114(24):2655–2662
Hermida N, Markl A, Hamelet J, Van Assche T, Vanderper A, Herijgers P, van Bilsen M, Hilfiker-Kleiner D, Noppe G, Beauloye C, Horman S, Balligand JL (2013) HMG CoA reductase inhibition reverses myocardial fibrosis and diastolic dysfunction through AMP-activated protein kinase activation in a mouse model of metabolic syndrome. Cardiovasc Res 99(1):44–54
Qu HY, Xiao YW, Jiang GH, Wang ZY, Zhang Y, Zhang M (2009) Effect of atorvastatin versus rosuvastatin on levels of serum lipids, inflammatory markers and adiponectin in patients with hypercholesterolemia. Pharm Res 26(4):958–964
Blanco-Colio L, Martín-Ventura JL, Gómez-Guerrero C, Masramon X, de Teresa E, Farsang C, Gaw A, Gensini G, Leiter LA, Langer A, Egido J (2008) Adiponectin plasma levels are increased by atorvastatin treatment in subjects at high cardiovascular risk. Eur J Pharmacol 586(1–3):259–265
Sugiyama S, Fukushima H, Kugiyama K, Maruyoshi H, Kojima S, Funahashi T, Sakamoto T, Horibata Y, Watanabe K, Koga H, Sugamura K, Otsuka F, Shimomura I, Ogawa H (2007) Pravastatin improved glucose metabolism associated with increasing plasma adiponectin in patients with impaired glucose tolerance and coronary artery disease. Atherosclerosis 194(2):e43–e51
Inami N, Nomura S, Shouzu A, Omoto S, Kimura Y, Takahashi N, Tanaka A, Nanba M, Shouda Y, Iwasaka T (2007) Effects of pitavastatin on adiponectin in patients with hyperlipidemia. Pathophysiol Haemost Thromb 36(1):1–8
Essick EE, Ouchi N, Wilson RM, Ohashi K, Ghobrial J, Shibata R, Pimentel DR, Sam F (2011) Adiponectin mediates cardioprotection in oxidative stress-induced cardiac myocyte remodeling. Am J Physiol Heart Circ Physiol 301(3):H984–H993
Zhong J, Basu R, Guo D, Chow FL, Byrns S, Schuster M, Loibner H, Wang XH, Penninger JM, Kassiri Z, Oudit GY (2010) Angiotensin-converting enzyme 2 suppresses pathological hypertrophy, myocardial fibrosis, and cardiac dysfunction. Circulation 122(7):717–728
Zadelaar S, Kleemann R, Verschuren L, de der Vries-Van Weij J, van der Hoom J, Princen HM, Kooistra T (2007) Mouse models for atherosclerosis and pharmaceutical modifiers. Arterioscler Thromb Vasc Biol 27(8):1706–1721
Nie P, Li D, Hu L, Jin S, Yu Y, Cai Z, Shao Q, Shen J, Yi J, Xiao H, Shen L, He B (2014) Atorvastatin improves plaque stability in ApoE-knockout mice by regulating chemokines and chemokine receptors. PLoS One 9(5):e97009
Hunyady L, Catt KJ (2006) Pleiotropic AT1 receptor signaling pathways mediating physiological and pathogenic actions of angiotensin II. Mol Endocrinol 20(5):953–970
Serebruany VL, Miller M, Pokov AN, Malinin AI, Lowry DR, Tanguay JF, Hennekens CH (2006) Effect of statins on platelet PAR-1 thrombin receptor in patients with the metabolic syndrome (from the PAR-1 inhibition by statins [PARIS] study). Am J Cardiol 97(9):1332–1336
Han SH, Quon MJ, Kim JA, Koh KK (2007) Adiponectin and cardiovascular disease: response to therapeutic interventions. J Am Coll Cardiol 49(5):531–538
Acknowledgements
The authors declare no conflicts of interest. Part of this work was supported by the “Brain Pool” program funded by the Korea Ministry of Science and Technology to Dr. Serebruany.
Author information
Authors and Affiliations
Corresponding author
Additional information
An erratum to this article is available at https://doi.org/10.1007/s11745-017-4296-4.
About this article
Cite this article
Choi, S.Y., Park, J.S., Roh, M.S. et al. Inhibition of Angiotensin II-Induced Cardiac Fibrosis by Atorvastatin in Adiponectin Knockout Mice. Lipids 52, 415–422 (2017). https://doi.org/10.1007/s11745-017-4246-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11745-017-4246-1