Abstract
The seedlings of two soybean genotypes, Al-tolerant PI 416937 (PI) and Al-sensitive Young, were cultured in the solution containing 0, 25 or 50 µM Al (AlCl3·6H2O) for 24, 36 or 48 h in the hydroponics, and the calluses induced from two genotypes were cultured in medium containing 0, 10, 50 or 100 µM Al for 5, 10 or 15 days, respectively. The effects of Al on growth of seedling roots and calluses, antioxidant enzyme activities of superoxide dismutase (SOD) and peroxidase (POD) and lipid peroxidation were investigated. Under Al stress, PI was more tolerant to Al toxicity than Young at both intact plant and tissue levels and lower concentrations of Al significantly stimulated the root and callus growth of PI. Al application enhanced the activities of SOD and POD and lipid peroxidation in both roots and calluses of two genotypes. Although the differences of SOD activities between two genotypes in response to Al toxicity depended on Al concentration and durations of treatment, SOD activities in the roots of PI were higher than those in the roots of corresponding Young in the presence of Al for 36 or 48 h. Meanwhile, the POD activities in PI roots increased as the Al levels and durations of treatment increased, significantly higher than those in the corresponding Young roots. Moreover, Al-treated PI had significantly lower lipid peroxidation than Young at both root and callus levels. These results suggest that the enhanced antioxidant-related enzyme activities and reduced lipid peroxidation in PI might be one of Al-tolerant mechanisms.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Al toxicity is a major factor limiting crop productivity on acid soils. Micromolar concentration of Al can inhibit root growth within minutes or hours in many agricultural plant species (Kochian 1995). The Al toxicity mainly inhibits root growth, thus causing increased crop sensitivity to drought and decreased nutrition uptake (Foy et al. 1992; Delhaize and Ryan 1995). Significant genotypic differences in Al tolerance were found in plant species. Al resistance can be divided into mechanisms facilitating Al exclusion from root apex (Al exclusion) and conferring the ability of plants to tolerate Al in the plant symplasm (Al tolerance) (Kochian 1995). The former includes the immobilization of Al at cell wall, selective permeability of the plasma membrane and the detoxification of organic acids and other exudates in the rhizosphere. The latter includes complexation by Al-binding proteins, compartmentalization in the vacuoles, chelation by ligands in the cytosol, induction of Al-tolerant enzymes (Keltjens and Ulden 1987; Kasai et al. 1992; Taylor et al. 1997).
When subjected to environmental stress, Al-tolerant plants may develop a defense system. At the cellular level, the primary target of Al is the plasma membrane. Abiotic environmental stresses, such as Al toxicity, drought, excess salt and heavy metals, can induce the formation of reactive-oxygen species in plant cells (Hippeli et al. 1999; Breusegem et al. 2001). Al toxicity led to an increase in enzyme activity related to ROS (reactive-oxygen species) and lipid peroxidation in vitro or vivo conditions, suggesting that Al could alter plasma membrane properties and permeability and induce oxidative stress (Cakmak and Horst 1991; Ikegawa et al. 2000; Yamamoto et al. 2003). Al-induced lipid peroxidation is the most prominent symptom of oxidative stress in plants and animals (Yamamoto et al. 2001).
It was found that superoxide dismutase (SOD) is to be involved in detoxification mechanisms when maize, soybean and barley were subjected to Al stress (Cakmak and Horst 1991; Boscolo et al. 2003; Guo et al. 2004). A significant increase in MDA concentration and a stimulation of SOD and peroxidase (POD) activities were observed in tolerant barley subjected to low pH or Al stress, while higher malonaldehyde (MDA) concentration and lower SOD activity were noted in the sensitive genotypes (Guo et al. 2004). The activities of SOD and POD and MDA concentrations in soybean increased under Al stress, and a close correlation existed between the MDA and root elongation rate induced by Al (Cakmak and Horst 1991). In contrast, Al exposure was not found to induce the increase of MDA in both Al-sensitive and tolerant maize inbred lines (Boscolo et al. 2003). Furthermore, Al stress induced the dose- and time-dependent formation of SOD and POD in an Al-sensitive maize inbred line, but not in an Al-tolerant line (Boscolo et al. 2003). In addition, POD and APX, the H2O2 consuming enzymes were activated following similar patterns of expression and exhibited significant correlation between their elevated activities and root growth inhibition (Šimonovicová et al. 2004). Cultured plant cells or tissues have been used as a model system for investigation of cellular mechanisms of Al toxicity in plant. At the tissue level, Al enhanced activities of SOD both in roots of intact plants and suspension-cultured cells of tea plants adapted to acid soils (Ghanati et al. 2005).
So far, some Al-tolerant or sensitive soybean genotypes have been identified. Horst and Klotz (1990) identified PI as one of the two most tolerant lines after screening over 1,000 genotypes of soybean. Al-tolerant soybean genotype PI and sensitive genotype Young have been widely studied for the understanding of the mechanisms of Al tolerance (Goldman et al. 1989; Foy et al. 1992; Bianchi-Hall et al. 1998; Bushamuka and Zobel 1998; Bianchi-Hall et al. 2000; Nian et al. 2004). Although differences in Al tolerance of these soybean genotypes have been studied genetically and physiologically, the mechanisms responsible for Al tolerance are not clearly understood. Therefore, the objective of this study was to investigate the effect of increasing Al concentrations on the seedling and callus growth, lipid peroxidation (MDA), SOD and POD activities in two contrasting soybean genotypes, as well as their relationship to Al tolerance.
Materials and methods
Plant materials and treatments
Two soybean genotypes differing in Al tolerance were used in the experiments. For in vivo test, the seeds of Al-tolerant PI and Al-sensitive Young were germinated in fine sand. Uniform 3-day-old seedlings were transferred to 1.5 l plastic pots containing 0.5 mM CaCl2 solution with 0, 25 or 50 μM Al (AlCl3·6H2O) at pH 4.5 and grown for 24, 36 or 48 h under controlled conditions at 25°C and 12 h/12 h (light/dark) cycles. The root growth was measured and relative root elongation, [(elongation in the presence of Al) (elongation in the absence of Al)−1 × 100], was calculated.
For in vitro tests, the calluses from two soybean genotypes were induced and sub-cultured in Murashige–Skoog (MS) medium amended with 2,4 d (2 mg/l), 6-BA (0.5 mg/l), sucrose (30 g/l) and Karaoke powder (8 g/l) at pH 5.8. One gram of callus from each genotype was suspended to liquid medium containing 0, 10, 50 or 100 µM Al (AlCl3·6H2O) at pH 4.5 and cultured for 5, 10 or 15 days. The medium was a modified MS with 2,4 d (2 mg/l), 6-BA (0.5 mg/l), sucrose (30 g/l) and Karaoke powder (8 g/l), while EDTA and KH2PO4 were excluded to avoid reduction of Al toxicity. Potassium (K) was added as K2SO4 to supply the same amount of K in KH2PO4. The sterilized medium (30 ml) was poured into each culture vessel and sterilized cotton (0.2 g) and a piece of filter paper were placed in liquid medium to establish a flat surface to plant the callus. The calluses were harvested at 5, 10 or 15 days after Al treatment and fresh weights were measured. All experiments were replicated three times.
Extraction and assay of the enzymes
To determine enzyme activities, 0.5 g of root tip (<2 cm) or callus was sampled following Al treatments and homogenized with chilled pestle and mortar in 5-ml extract buffer containing 50 mM phosphate buffer (pH 7.0). The homogenate was centrifuged at 12,000g for 15 min and the resulting supernatant was used to determine the enzyme activity. The whole extraction procedure was performed at 4°C. SOD was measured using the photochemical NBT method (Beauchamp and Fridovich 1971) as modified by Dhindsa and Matowe (1981). The reaction mixture (3 ml) contained: 1.75 ml of 50 mM phosphate buffer (pH 7.8), 0.3 ml of 0.1 mM EDTA, 0.3 ml of 130 mM methionine, 0.3 ml of 750 µM nitroblue tetrazolium (NBT), 0.3 ml of 20 µM riboflavin and 100 µl of the supernatant. Riboflavin was added immediately before the reaction and the reaction was initiated by placing the tubes under two 15 W fluorescent lamps. The reaction was terminated after 10 min by removing the reaction tubes from the light source. Non-illuminated and illuminated reactions without supernatant served as calibration standards. Reaction product was measured at 560 nm. The volume of the supernatant corresponding to 50% inhibition of the reaction was assigned a value of one enzyme unit.
Peroxidase (POD) activities were determined with guaiacol at 470 nm. One hundred microliter of the enzyme extract was added to the reaction mixture containing 0.05 ml guaiacol solution and 0.03 ml hydrogen peroxide solution in 3 ml phosphate buffer solution (pH 7.0). The solution was then mixed well and kept until the absorbance at 470 nm reached 0.05. The time for the absorbance to increase by 0.1 was recorded. The enzyme activity was calculated using the extinction coefficient of guaiacol dehydrogenation product under the specified conditions.
The level of lipid peroxidation was expressed as MDA concentration and determined as 2-thiobarbituric acid (TBA) reactive metabolites. Soybean fresh root or tissues (0.5 g) were homogenized and extracted in 10 ml of 0.25% (w/v) TBA made in 10% (v/v) trichloroacetic acid (TCA). Extract was heated at 95°C for 30 min and then immediately cooled on ice. After centrifugation at 10,000g for 10 min, the absorbance of the supernatant was measured at 532 nm. Correction of non-specific turbidity was made by subtracting the absorbance value measured at 600 and 450 nm. The level of lipid peroxidation was expressed as nmol g−1 fresh weight.
Results
Effects of Al exposure on soybean root and callus growth
The relative root elongation of two genotypes in the presence of Al was significantly different, with Al-tolerant PI being higher than Young at both Al concentrations. When Al-treated PI was exposed to 25 µM Al for 24, 36 or 48 h, root growth was markedly stimulated, increasing 30.8, 24.9 and 12.9% when compared with Al-free control roots, whereas root elongation of Young was inhibited by Al treatments (Table 1).
No difference in callus growth in both genotypes was observed after 5-day Al exposure (Fig. 1). However, the callus growth of PI at all Al concentrations was significantly stimulated after 10- or 15-day Al treatment, while that of Al-sensitive Young was slightly promoted only by 10 or 50 µM Al. The callus growth for Young was inhibited after exposure to 100 μM Al for 15 days, while that for PI was still significantly enhanced (Fig. 1), suggesting that PI was more tolerant to Al than Young at tissue level. These results also revealed consistent responses of two genotypes to Al toxicity at both in vitro and in vivo levels.
Effects of Al exposure on SOD activities in the plant roots and calluses
Al presence induced the dose- and time-dependent increase of SOD activity in both Al-tolerant PI and Al-sensitive Young in soybean roots (Fig. 2). After 24-h of Al treatment, the SOD activities in both PI and Young increased with the increase of Al contents, and no significant difference in the root SOD activities was observed in two soybeans. In the presence of 50 µM Al for 36 h, however, SOD activity in PI roots was significantly higher than that in Young roots. In addition, SOD activity in PI was significantly higher than that in Young roots after exposure to 25 µM Al for 48 h, whereas SOD activities in both genotypes were significantly reduced when exposed to 50 µM Al for 48 h.
Effects of Al application on SOD activities in the roots of Al-tolerant PI and Al-sensitive Young. Three-day-old seedlings of PI and Young were cultured in 1.5-l plastic pots containing 0.5 mM CaCl2 solution with 0, 25 or 50 µM AlCl3. The SOD activities in the roots were measured after Al exposure of 24, 36 or 48 h. The error bars represent mean ± SD (n = 3)
In vitro results showed that 5-day Al treatment caused the enhancement of SOD activities in the calluses of both genotypes at all the Al treatments (Fig. 3). SOD activities in the callus of PI tended to increase with increasing Al concentrations or durations of treatment, while those in Young increased as durations of treatment increased only in the presence of 10 or 50 µM Al. Moreover, SOD activity in PI was significantly higher than that in Young after 15-day exposure of 100 µM Al stress, whereas SOD activity in Young was significantly higher than that in PI in the presence of 10 or 25 µM Al.
Effects of Al application on SOD activities in the calluses of Al-tolerant PI and Al-sensitive Young. Callus from PI or Young was exposed to liquid medium containing 0, 10, 50 or 100 µM AlCl3 at pH 4.5. The POD activities were measured after 5, 10 or 15 days of treatment. The values are mean ± SD of three replicates
Effects of Al exposure on POD activities in the soybean roots and calluses
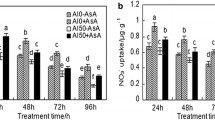
The POD activities in PI were enhanced by the Al treatments and tended to increase with the increase in Al concentration and durations of treatment (Fig. 4). In general, PI roots had higher POD activities than Young, showing a trend of positive association between POD activity and the root growth (Table 1; Fig. 4). POD activity in Young root decreased with the prolongation of treatment of 50 µM Al with POD activities reduced to the control level after 48-h Al treatment.
Effects of Al application on POD activities in the roots of Al-tolerant PI and Al-sensitive Young. Three-day-old seedlings were treated in 1.5-l plastic pots containing 0.5 mM CaCl2 solution with 0, 25 or 50 μM AlCl3. The POD activities in the roots were measured after Al exposure of 24, 36 or 48 h. The error bars represent mean ± SD (n = 3) and U means \( \Updelta \text{OD}_{470\,{\text{min}^{-1}}} \)
Al increased POD activities in callus at all the concentrations (Fig. 5). POD activities of PI at all the Al levels and those in Young treated with 10 or 50 μM Al increased with the increase in exposure times. POD activity in Young callus was 58.9% higher than that in PI sampled after 5-day treatment with 100 µM Al, whereas POD activities in PI were 22.6 and 122.4% higher than those in Young in the presence of 100 µM Al stress for 10 and 15 days, respectively.
Effects of Al application on POD activities in the calluses of Al-tolerant PI and Al-sensitive Young. Callus from PI or Young was exposed to liquid medium containing 0, 10, 50 or 100 µM AlCl3 at pH 4.5. The POD activities were measured after 5, 10 or 15 days of treatment. The error bars represent mean ± SD (n = 3) and U means \( \Updelta \text{OD}_{470\,{\text{min}^{-1}}} \)
Effects of Al exposure on MDA in the soybean roots and calluses
Al exposure caused a significant increase in the MDA in the roots of two genotypes, which increased with increasing the Al concentrations and durations of treatment (Fig. 6). The Al-sensitive Young showed higher MDA content than Al-tolerant PI at all the treatments, and a significant negative association between MDA and root relative elongation were observed (Table 1; Fig. 6).
Effects of Al application on lipid peroxidation in the roots of Al-tolerant PI and Al-sensitive Young. Three-day-old seedlings were treated in 1.5-l plastic pots containing 0.5 mM CaCl2 solution with 0, 25 or 50 µM AlCl3. The MDA concentrations in the roots were measured after Al exposure of 24, 36 or 48 h. The error bars represent mean ± SD (n = 3)
Al treatment caused MDA increase in callus, too (Fig. 7). The contents of MDA in PI calluses increased as Al concentrations and durations of treatment increased, whereas those in Young depended on Al levels and durations of treatment. The Young callus in the presence of 100 µM Al showed higher MDA than that in PI after 5-day exposure, and a similar response in both genotypes with 50 µM Al was observed after 10 or 15-day Al exposure.
Effects of Al application on lipid peroxidation in the calluses of Al-tolerant PI and Al-sensitive Young. Callus from PI or Young was exposed to liquid medium containing 0, 10, 50 or 100 µM AlCl3 at pH 4.5. The MDA concentration was measured after 5, 10 or 15 days of treatment. The error bars represent mean ± SD (n = 3)
Discussion
Although it is generally recognized that Al is a major factor limiting plant root growth, some reports did show beneficial effect of Al on plant growth (Mullette 1975; Konishi et al. 1985; Haridasan 1988; Huang and Bachelard 1993; Malkanthi et al. 1995; Osaki et al. 1997; Watanabe et al. 2005). This beneficial effect often occurs at low or moderate Al concentrations. In the present study, we also found that Al exposure could stimulate PI root growth at 25 µM Al level (Table 1), and callus growth for PI was stimulated at all the Al treatments (Fig. 1). After 10- or 15-day Al exposure, however, 10 or 50 µM Al only slightly stimulated the callus growth of Young and 100 µM Al inhibited it. These results may also explain the high tolerance of PI to Al stress, indicating consistency of Al tolerance at both in vitro and in vivo levels. Osaki et al. (1997) proposed that Al-stimulating effect was ascribed not only to the alleviation of H+ toxicity, but also to the increase of root activity, such as P uptake. Furthermore, there were reports that the beneficial effect of Al was induced by the combination of Al and nutrients (Watanabe et al. 1997, 2005). However, our results could not support this reasoning as the soybean roots were exposed to Al in the 0.5 mM Ca solution (pH 4.5) without other nutrients in our experiment. A similar result was obtained from the experiment using suspension-cultured tea cells pretreated with or without Al in simple salt solution (Ghanati et al. 2005).These two results suggest that other mechanisms contribute to this stimulation. Therefore, further study is warranted to fully understand this physiological response.
The susceptibility of plants to some environmental stresses was found to correlate with their antioxidant response (Bowler et al. 1992; Foyer et al. 1994). Modulation of these antioxidants is, therefore, an important adaptive response to withstand adverse conditions (Kochian 1995; Okamoto et al. 2001). In the previous studies, PI was found to exhibit strong Al tolerance in both short-term hydroponics and long-term sand or soil pot cultures (Goldman et al. 1989; Pantalone et al. 1999) and Al-induced organic acid exudation in roots was not involved in the different Al tolerance in PI and Young (Nian et al. 2004). In this study, we showed that Al induced an increase in SOD activity in both seedling roots and calluses of Al-tolerant PI and sensitive Young, which was dependent on the time of exposure and Al concentration, and partly related to root growth or Al tolerance (Table 1; Figs. 1, 2). The consistent response of SOD activities at both plant root and callus levels suggests the possible existence of a common tolerance mechanism related to antioxidant enzyme activities under both in vivo and in vitro conditions. In the previous reports, PI was found to be tolerant to both drought and salt stresses (Goldman et al. 1989; Abd-Alla et al. 1998). Therefore, these results suggest that the enhanced antioxidant-related enzyme activity in PI might be one of common tolerance mechanisms under these stresses.
Cakmak and Horst (1991) found that Al increased the activities of SOD and POD in the soybean root tips, but no information on their relationship with Al tolerance was available because only one soybean genotype was used in their experiment. Our data demonstrated that SOD activities in the roots of Al-tolerant PI in the presence of high Al concentrations were higher than those in Al-sensitive Young, which tended to increase with the increase in Al concentrations, revealing an association between the Al tolerance and SOD activity (Table 1; Fig. 2). However, Al sensitivity or tolerance was not clearly associated with the SOD activity in plants receiving a mild stress—24 h exposure or 25 μM Al (Fig. 2). This may be ascribed to the less damage of Young root (8% reduction in relative root elongation) in comparison with the corresponding treatment without Al. Moreover, both genotypes showed low SOD activities with severe inhibition of soybean root growth in the presence of 50 μM Al for 48 h, suggesting that Al-damaged soybeans could not continuously elevate SOD activity (Table 1; Fig. 2).
Similarly, SOD activities in calluses of PI increased under Al application, and related increase of callus growth was observed as well (Figs. 1, 3); when callus was subjected to 100 µM Al for 15 days, however, callus growth of Young was markedly reduced with no increase in SOD activity (Figs. 1, 3). These results suggest that SOD should be correlated with Al tolerance at cellular level. Al could not affect callus growth of both genotypes in 5-day treatment, but SOD activities were significantly enhanced (Fig. 3). These results suggest that the increase in SOD activities could have taken place before the growth inhibition or stimulation of the callus growth.
In this experiment, PI root had significantly higher POD activities than Young root at higher Al concentration (50 µM Al), and root relative elongation of PI was much higher than Young (Table 1; Fig. 4), revealed that different Al tolerance between two soybeans, to some extent, was positively correlated with POD activity. At the callus level, higher Al concentration (100 µM Al) increased POD activities in PI with the increasing durations of treatment, but reverse was true for Young, indicating an association between POD activity and Al tolerance.
The lipid peroxidation is the most prominent symptom of oxidative stress. MDA, an oxidized product of membrane lipids, can indicate the extent of oxidative stress. In a previous report, Al exposure could not induce the increase of MDA in both Al-sensitive and tolerant maize inbred lines (Boscolo et al. 2003). However, in our experiment, MDA contents tended to increase with the increase in Al concentrations in the soybean roots and a negative association between MDA and Al tolerance was observed under Al stress (Table 1; Fig. 6). This is in agreement with a previous report that MDA concentration of soybean was increased under Al stress, and a close relationship existed between lipid peroxidation and inhibition of root-elongation rate induced by Al (Cakmak and Horst 1991). These results suggest that MDA could be used as a useful index for screening Al-tolerant soybeans.
References
Abd-Alla MH, Vuong TD, Harper JE (1998) Genotypic differences in dinitrogen fixation response to NaCl stress in intact and grafted soybean. Crop Sci 38:72–77
Beauchamp C, Fridovich I (1971) Superoxide dismutase: improved assays and an assay applicable to acrylamide gels. Anal Biochem 44:276–287
Bianchi-Hall CM, Carter Jr TE, Rufty TW, Arellano C, Boerma HR, Ashley DA, Burton JW (1998) Heritability and resource allocation of aluminum tolerance derived from soybean PI 416937. Crop Sci 38:513–522
Bianchi-Hall CM, Carter Jr TE, Bailey MA, Mian MAR, Rufty TW, Ashley DA, Boerma HR, Arellano C, Hussey RS, Parrott WA (2000) Aluminum tolerance associated with quantitative trait loci derived from soybean PI 416937 in hydroponics. Crop Sci 40:538–544
Boscolo PRS, Marcelo M, Jorge RA (2003) Aluminum-induced oxidative stress in maize. Phytochemistry 62:181–189
Bowler C, Van Montagu M, Inze D (1992) Superoxide dismutases and stress tolerance. Annu Rev Plant Physiol Plant Mol Biol 43:83–116
Breusegem FV, Vranová E, Dat JF, andInzé D (2001) The role of active oxygen species in plant signal transduction. Plant Sci 161:405–414
Bushamuka VN, Zobel RW (1998) Differential genotypic and root type penetration of compacted soil layers. Crop Sci 38:776–781
Cakmak I, Horst WJ (1991) Effects of aluminum on lipid peroxidation, superoxide dismutase, catalase and peroxidase activities in root tips of soybean (Glycine max). Physiol Plant 83:463–468
Delhaize E, Ryan PR (1995) Aluminum toxicity and tolerance in plants. Plant Physiol 107:315–321
Dhindsa RS, Matowe W (1981) Drought tolerance in two mosses: correlated with enzymatic defense against lipid peroxidation. J Exp Bot 32:79–91
Foy CD, Duke JA, Devine TE (1992) Tolerance of soybean germplasm to an acid tatum subsoil. J Plant Nutr 15:527–547
Foyer C, Lelandais M, Kunert JJ (1994) Photooxidative stress in plant. Physiol Plant 92:696–717
Ghanati F, Morita A, Yokota H (2005) Effects of aluminum on the growth of tea plant and activation of antioxidant system. Plant Soil 276:1–2
Goldman IL, Carter TE, Patterson RP (1989) Differential genotypic response to drought stress and subsoil aluminum in soybean. Agron J 81:330–334
Guo T, Zhang G, Zhou M, Wu F, Chen J (2004) Effects of aluminum and cadmium toxicity on growth and antioxidant enzyme activities of two barley genotypes with different Al resistance. Plant Soil 258:241–248
Haridasan M (1988) Performance of Miconia albicans (SW.) Triana, an aluminum accumulating species, in acidic and calcareous soils. Commun Soil Sci Plant Anal 19:1091–1103
Hippeli S, Heiser I, Elstner EF (1999) Activated oxygen and free oxygen radicals in pathology: new insights and analogies between animals and plants. Plant Physiol Biochem 37:167–178
Horst WJ, Klotz F (1990) Screening soybean for aluminum tolerance adaptation to acid soils. In: Bassam NEl (ed) In genetic aspects of plant mineral nutrition. Kluwer, Dordrecht, pp 355–360
Huang J, Bachelard EP (1993) Effects of aluminium on growth and cation uptake in seedlings of Eucalyptus mannifera and Pinus radiate. Plant Soil 149:121–127
Ikegawa H, Yamamoto Y, Matsumoto H (2000) Response to aluminum of suspension-cultured tobacco cells in a simple calcium solution. Soil Sci Plant Nutr 46:503–514
Kasai M, Sasaki M, Yamamoto Y, Matsumoto H (1992) Aluminum stress increases K+ efflux and activities of ATP and PPi-dependent H+ pumps of tonoplast-enriched membrane vesicles from barley roots. Plant Cell Physiol 33:1035–1039
Keltjens WG, Ulden PSR (1987) Effect of Al on nitrogen (NH4 and NO3) uptake, nitrate reductase activity and proton releaser in two sorghum cultivars different in Al tolerance. Plant Soil 104:227–234
Kochian LV (1995) Cellular mechanisms of aluminum toxicity and resistance in plants. Annu Rev Plant Physiol Plant Mol Biol 46:237–260
Konishi S, Miyamoto S, Taki T (1985) Stimulatory effects of aluminum on tea plants grown under low and high phosphorus supply. Soil Sci Plant Nutr 31:361–368
Malkanthi DRR, Moritsugu M, Yokoyama K (1995) Effect of low pH and Al on absorption and translocation of some essential nutrients in excised Barley roots. Soil Sci Plant Nutr 41:253–262
Mullette KJ (1975) Stimulation of growth in eucalyptus due to aluminium. Plant Soil 42:495–499
Nian H, Yang ZM, Huang H, Yan X, Matsumoto H (2004) Citrate secretion induced by aluminum stress may not be the key mechanism responsible for the differential aluminum tolerance of some soybean genotypes. J Plant Nutr 27:2047–2066
Okamoto O, Pinto E, Latorre LR, Bechara EJH, Colepicolo P (2001) Antioxidant modulation in response to metal-induced oxidative stress in algal chloroplasts. Arch Environ Comtam Toxicol 40:18–24
Osaki M, Watanabe T, Tadano T (1997) Beneficial effect of aluminum on growth of plants adapted to low pH soils. Soil Sci Plant Nutr 43:551–563
Pantalone VR, Rebetzke GJ, Burton JW, Carter Jr TE, Israel DW (1999) Soybean PI 416937 root system contributes to biomass accumulation in reciprocal grafts. Agron J 91:840–844
Šimonovicová M, Tamás L, Huttová J, Mistrík I (2004) Effect of aluminum on oxidative stress related enzymes activities in barley roots. Biol Plant 48:261–266
Taylor GJ, Basu A, Basu U, Slaski JJ (1997) Al-induced 51-kilodalton membrane-bound proteins are associated with resistance to Al in a segregating population of wheat. Plant Physiol 114:363–372
Watanabe T, Osaki M, Tadano T (1997) Aluminium-induced growth stimulation in relation to calcium, magnesium and silicate nutrition in Melastoma malabathricum L.: an Al accumulator plant. Plant Soil 43:827–837
Watanabe T, Janshen S, Osaki M (2005) The beneficial effect of aluminium and the role of citrate in Al accumulation in Melastoma malabathricum. New Phytol 165:773–780
Yamamoto Y, Kobayashi Y, Matsumoto H (2001) Lipid peroxidation is an early symptom triggered by aluminum, but is not the primary cause of elongation inhibition in pea roots. Plant Physiol 125:199–208
Yamamoto Y, Kobayashi Y, Devi SR, Rikiishi S, Matsumoto H (2003) Oxidative stress triggered by aluminum in plant roots. Plant Soil 255:239–243
Acknowledgments
This study was supported by National Public Benefit Research Foundation (nyhyzx07-004-11), 948 and 863 projects and Key Projects in the National Science and Technology Pillar Program (2007BAD89B14). We are grateful to Dr. Zhanyuan Zhang with Division of Plant Sciences of University of Missouri for revising this paper.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by A. Aniol.
Rights and permissions
About this article
Cite this article
Du, B., Nian, H., Zhang, Z. et al. Effects of aluminum on superoxide dismutase and peroxidase activities, and lipid peroxidation in the roots and calluses of soybeans differing in aluminum tolerance. Acta Physiol Plant 32, 883–890 (2010). https://doi.org/10.1007/s11738-010-0476-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11738-010-0476-z