Abstract
As a major antioxidant in plants, ascorbic acid (AsA) plays a very important role in the response to aluminum (Al) stress. However, the effect of AsA on the mitigation of Al toxicity and the mechanism of nitrate nitrogen (NO3 −–N) uptake by plants under Al stress are unclear. In this study, a hydroponic experiment was conducted using peak 1 A rice (sterile line, Indica) with weaker resistance to Al and peak 1 superior 5 rice (F1 hybrid, Indica) with stronger resistance to Al to study the effects of exogenous AsA on the physiological and biochemical responses to NO3 −–N uptake by rice roots exposed to 50 μmol L−1 Al. Al stress induced increases in the concentrations of H2O2 and malondialdehyde (MDA) and in the activities of antioxidant enzymes [such as superoxide dismutase (SOD), peroxidase (POD), catalase (CAT), and ascorbate peroxidase (APX)]. Plasma membrane (PM) H+-ATPase and H+-pump activities, endogenous AsA content and NO3 −–N uptake in rice roots decreased under Al stress. After treatment with 2 mmol L−1 exogenous AsA combined with Al, concentrations of H2O2 and MDA in roots notably decreased, and endogenous AsA content and activities of SOD, POD, CAT, and APX in rice roots increased significantly; furthermore, the interaction of PM H+-ATPase and the 14-3-3 protein was also enhanced significantly compared with that in control plants without AsA treatment, which clearly increased NO3 −–N uptake. Based on all of these data, the application of AsA significantly reduced the accumulation of H2O2 and MDA and increased the activities of PM H+-ATPase and the H+-pump by increasing the endogenous AsA content, the antioxidant enzyme activities, and the interaction of PM H+-ATPase and the 14-3-3 protein in the roots of the two rice cultivars under Al stress, thereby improving the uptake of NO3 −–N in rice.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Approximately 50 % of the arable land in the world consists of acidic soils, and approximately 13 % of rice crops are distributed on acidic soils. In recent years, with the increase in acid rain and the use of chemical fertilizers, cultivated soils are being acidified, which is becoming a serious problem (Guo and others 2010). Soil acidification can lead to the release of active Al3+, which is toxic to plants (Liu and others 2014). Rice is not only a major global food crop but also China’s primary food crop. Aluminum (Al) has become one of the main factors restricting the growth and yield of rice in acidic upland and acid sulfate paddy fields (Vasconcelos and Rossiello 2002).
Al increases reactive oxygen species (ROS) production and enhances lipid peroxidation (Jones and others 2006; Sharma and Dubey 2007), especially in Al-sensitive genotypes (Giannakoula and others 2010; Ma and others 2012), which damages membrane lipids, cellular structures, proteins, and DNA. However, the in vivo antioxidant enzyme systems of plants respond to these changes to maintain metabolic balance. Excess ROS production can be prevented and ROS can be eliminated by antioxidant enzymes [such as superoxide dismutase (SOD), peroxidase (POD), catalase (CAT), and ascorbate peroxidase (APX)]. Al has been reported to decrease CAT activity in Oryza sativa (Sharma and Dubey 2007). In Allium cepa, Al was reported to inhibit CAT activity and significantly enhance the activities of SOD, POD, and APX (Achary and others 2008).
In plant cells, the plasma membrane (PM) is the first line of defense against outside stress. Therefore, the PM and proteins embedded in the PM can react first to stress factors. The PM H+-ATPase is known as the “dominant enzyme” in the activities of higher plants (Serrano 1989); it regulates many important physiological processes and plays important roles in maintaining the cytoplasmic pH and the steady-state levels of metabolites when plants must adapt to adverse environments. The PM H+-ATPase generates the electrochemical proton gradient across the membrane by hydrolyzing ATP and excluding H+ outside the cell membrane to provide a driving force for the uptake of nutrients and secondary transport in plants (Palmgren 2001; Zhang and others 2011). The 14-3-3 protein is a highly conserved regulatory protein in eukaryotic cells that can regulate PM H+-ATPase activity by interacting with phosphorylated PM H+-ATPase (Alterfjord and others 2004). Related studies have indicated that binding of the 14-3-3 protein to the C-terminal end of the PM H+-ATPase in Arabidopsis thaliana (AHA2) occurs via the last two phosphorylated threonine (Thr) residues, promoting the interaction of the 14-3-3 protein with PM H+-ATPase. This interaction enhances the activity of PM H+-ATPase and activates the proton pump (Fuglsang and others 1999). Therefore, the activity of PM H+-ATPase relates to its level of phosphorylation, which is regulated by many environmental factors (Heidi and others 2010).
Two major N sources (NH4 +–N and NO3 −–N) are taken up by plant roots (Chapman and Miller 2011). Nitrification is greatly restricted in flooded conditions. Consequently, the content of NH4 + greatly increases, and NH4 + becomes the main storage form of nitrogen in paddy fields. Rice absorbs much more NH4 +–N than NO3 −–N (Zhu and others 2011). Therefore, previous studies on the N nutrition of rice have primarily focused on NH4 +–N nutrition and neglected NO3 −–N nutrition. Notably, rice roots can secrete oxygen (O2), which can be used to convert NH4 +–N to NO3 −–N by nitrifying microorganisms. NO3 −–N on the surface of roots is instantly absorbed by rice. In fact, rice is in a mixed nutrient solution of NH4 +–N and NO3 −–N, even in completely flooded conditions (Luo and others 2013). An increasing number of studies have demonstrated that rice can take up and utilize two forms of nitrogen (Helali and others 2010). Under conditions of increased NO3 −–N content, some research has shown that rice roots, particularly lateral roots, have better growth and development than those exposed to NH4 +–N alone (Wang and others 2002). Therefore, the absorption and utilization of NO3 −–N in rice is significant in rice nitrogen nutrition.
Ascorbic acid (AsA) is the most crucial and important non-enzyme antioxidant in plants, playing an important role in mediating certain biotic and abiotic stress-induced oxidative stresses. AsA can improve a plant’s growth and enhance its ability to resist stress. Exogenous AsA significantly relieved the effects of Al toxicity in tomatoes (Ma and others 2015), ginger (Huang and others 2008), and Hydrilla verticillata (Xu and others 2006), and it reduced Al accumulation in root tips and the absorption capacity of Al in cell walls, indicating that AsA could be involved in the regulation of plant resistance to Al toxicity. AsA is the first line of plant defense against oxidative stress through the removal of a variety of free radicals, such as O2 −·, HO·, and H2O2. The ascorbic acid-glutathione (AsA-GSH) cycle indirectly clears APX substrates of H2O2 in an AsA-GSH loop (Munne-Bosch and Alegte 2002). The synthesis of AsA in vivo (endogenous AsA) is directly involved in the removal of the ROS, and the addition of exogenous AsA can inhibit lipid peroxidation and decrease the content of malondialdehyde (MDA) in plant tissues, thereby improving the antioxidant capacity of plants (Zheng and others 2006). However, few studies have reported the physiological and biochemical response mechanism of AsA to promote the uptake of NO3 −–N in rice under Al stress. Our previous study (Zhou and others 2015) showed that 2 mmol L−1 AsA could significantly alleviate the inhibitory effect of Al stress on two types of rice seedlings; therefore, 2 mmol L−1 AsA was considered a suitable concentration to study the physiological changes in rice in this study. In the present study, both peak 1 A (PA1) rice (with weaker resistance to Al) and peak 1 superior 5 (PS5) rice (with stronger resistance to Al) cultivars were used as experimental materials. The concentrations of H2O2 and MDA, the activities of certain antioxidant enzymes, the activity of PM H+-ATPase and its interaction with the 14-3-3 protein, and the uptake of NO3 −–N were determined in the presence of Al or Al in combination with exogenous AsA to analyze the effect of exogenous AsA on physiological and biochemical parameters and on the uptake of NO3 −–N by both types of rice. The results demonstrate that the application of exogenous AsA can alleviate Al toxicity and enhance NO3 −–N uptake in two rice cultivars.
Materials and Methods
Plant Materials and Growth Conditions
The rice [Oryza sativa L.; PA1 (sterile line, Indica) and PS5 (F1 hybrid rice, Indica)] seeds were sterilized in 10 % H2O2 for 5 min and then washed in deionized water for 5 min. The seeds were fully imbibed with deionized water for 24 h. Then, the seeds were germinated at 25 °C in the dark. When seedlings with roots reached a height of approximately 1 cm, they were sown in floating mesh in a polypropylene pot with 5 L of a 0.5 mM CaCl2 solution at pH 4.5, which was renewed daily. Seedlings at the 1-leaf stage and rice seedlings of a similar size were selected and transferred into ¼-strength normal rice nutrient solution (IRRI, pH 4.5) for 1 week. Then, the seedlings were transferred into normal rice nutrient solution containing the following macronutrients in mmol L−1: NH4NO3, 2.86; KH2PO4, 0.3; K2SO4, 0.35; CaCl2, 1.0; MgSO4·7H2O, 1.0; MnCl2·4H2O, 9.0 × 10−3; H3BO3, 2.0 × 10−2; (NH4)6Mo7O24·4H2O, 0.39 × 10−4; CuSO4·5H2O, 3.2 × 10−4; Na2SiO3·9H2O, 5.0 × 10−4; ZnSO4·7H2O, 7.7 × 10−4; and Na2EDTA-Fe(II), 2.0 × 10−2. The solution was adjusted to pH 4.5 with 1 mol L−1 HCl and renewed every 2 days. All experiments were conducted under greenhouse conditions at day/night temperatures of 28 °C/20 °C with 12 h of light (1000 μmol m−2 s−1). Seedlings at the 4-leaf stage that were similar in size were selected and exposed to 50 μmol L−1 AlCl3 alone or 50 μmol L−1 AlCl3 in combination with 2 mmol L−1 AsA in the rice nutrient solution (pH 4.5) for different periods of time. Each treatment was repeated three times. After treatment, the root tips (0–20 mm) of each treatment were harvested, immediately frozen in liquid nitrogen and stored at −80 °C for measurement of their physiological and biochemical indices and antioxidant enzyme activities.
Determination of Root Activity and NO3 − Absorption
Root activity was analyzed by the TTC method (Zhang and Wei 2003). Roots (0.2 g) were dipped in a mixture of 5 mL of 0.4 % TTC and 5 mL of phosphate buffer and incubated in the dark for 1 h at 37 °C; then, 2 mL of 1 mol L−1 H2SO4 was added to terminate the reaction (at the same time, 2 mL of 1 mol L−1 H2SO4 was added to the root samples as a blank). The roots were sampled, ground with 3–4 mL of ethyl acetate, and diluted to 10 mL. With the blank serving as the control, the absorbance of the supernatant was determined at 485 nm using a spectrophotometer. The NO3 −–N concentration in the solution was determined by ultraviolet spectrophotometry, referring to the standard of the People’s Republic of China environmental protection industry (HJ/T346-2007) with slight modifications. NO3 −–N and dissolved organic matter both absorb at a wavelength of 220 nm; however, dissolved organic matter also absorbs at 275 nm, whereas NO3 −–N does not. Therefore, with deionized water as the control, the absorbance was determined at 220 nm (OD220) and 275 nm (OD275) and subsequently corrected according to the following formula: OD=OD220 − 2OD275. The NO3 −–N content was calculated according to a standard curve. The same volume of the rice nutrient solution (pH 4.5) containing different amounts of Al3+ and AsA was renewed daily. The reduction in the NO3 −–N content represented the quantity of NO3 −–N in the liquid that was taken up by the rice every day.
Determination of MDA, H2O2, and AsA Contents
The MDA content was determined by the 2-thiobarbituric acid (TBA) method described by Zhang and Wei (2003). The H2O2 content was determined using the methyl orange method described by Gay and Gebicki (2003), and AsA was determined according to the method of Law and others (1983).
Measurement of the Proline Content and Activities of SOD, POD, CAT, and APX
The proline content was determined by the acidic ninhydrin method (Demiral and Türkan 2005). The SOD activity was determined by the nitroblue tetrazolium (NBT) light reduction method described by Giannopolitis and Ries (1977), with slight modifications. The reaction mixture consisted of 50 mmol L−1 Tris–HCl (pH 7.0), 3.0 μmol L−1 EDTA, 14.5 mmol L−1 methionine, 2.25 mmol L−1 NBT, and 60 μmol L−1 riboflavin. An appropriate quantity of enzyme extract was added to the reaction mixture. The reaction was initiated by placing the tubes below two 15-W fluorescent lamps for 15 min. The reaction was terminated by keeping the tubes in the dark for 10 min, and the absorbance was read at 560 nm. One unit of SOD activity was defined as the quantity of SOD enzyme required to cause 50 % inhibition of NBT reduction under the experimental conditions. The POD activity was measured according to the method described by Chance and Mehley (1955), with slight modifications. The mixture contained 50 mmol L−1 Tris–HCl (pH 7.0), 10 mmol L−1 guaiacol and 5 mmol L−1 H2O2. Then, 50 μL of the enzyme extract was added into the reaction mixture. The increase in the absorbance at 470 nm was measured at 0.5 min intervals up to 2 min. The POD activity was defined as the change in the OD470 per minute per milligram of protein. The CAT activity was assayed by monitoring the consumption of H2O2 at 240 nm for 2 min according to the method described by Aebi (1984). The APX activity was determined as described by Nakano and Asada (1981).
Assay of PM H+-ATPase Activity and PM H+-Pump Activity
PM H+-ATPase was extracted and determined as described by Shen and others (2005). The protein concentration was quantified according to the method of Bradford. The PM protein purity, PM H+-ATPase activity, and PM H+-pump activity were determined as described by Guo and others (2013).
Co-immunoprecipitation and Western Blot Analysis
To detect the interaction between the 14-3-3 protein and PM H+-ATPase via co-immunoprecipitation (Co-IP), a total of 500 μg of PM protein plus 2 μg of a specific phosphorylation antibody targeted against VHA2 were incubated at 4 °C for 6 h with occasional shaking (40 rpm). Then, 20 μL of protein A/G plus-agarose (Santa Cruz Biotech, Santa Cruz, CA) was added to the protein solution, which was then incubated at 4 °C for 12 h. The protein samples were centrifuged to precipitate the proteins. The protein pellets were then washed several times, resuspended, and separated by 10 % SDS-PAGE for Western blot analysis. The separated proteins were transferred to PVDF membranes using a semi-dry transfer system. The membranes were first treated with the specific phosphorylation antibody targeted against VHA2 or the Malus domestica 14-3-3 protein antibody and subsequently treated with a goat anti-rabbit IgG conjugated with peroxidase. The Western signal bands on the membrane were quantified using a densitometer (Bio-Rad, Hercules, CA) and expressed as a percentage of the samples without Al treatment (referred to as Al treatment at 0 h and set as 1.0).
Statistical Analysis
We utilized a randomized complete design. All experiments were replicated 3–5 times. All data were analyzed with Excel and SAS software (SAS Institute 1998). ANOVA was performed for each variable with a comparison of means by Duncan’s multiple range test with a significance level of 0.05.
Results
Assay of Rice Root Activity
As shown in Table 1, in the early period of single Al stress, the root activity of the two rice types was similar. The reduction capacity of the root systems of the two types of rice gradually declined with prolonged stress. When the stress was applied for 96 h, the root reduction activity of PA1 and PS5 was only 38 and 54 % that of the control, respectively, and the results indicate that the plant roots were damaged at that point. However, after treatment with exogenous AsA, the reduction activity of the roots recovered. The reduction activity of PA1 and PS5 was elevated to 53 and 72 % of the control, respectively, and the reduction activity in the plant roots was higher than that of non-AsA at the same time point.
The Effect of AsA on the Proline Content in the Root Tips of Rice Plants Under Al Stress
Crops contain little free proline under normal conditions. An increase in the free proline content is a physiological and biochemical response of plants to adverse stress. Simultaneously, the accumulation of proline is related to the production of ROS and plays an important role in removing endogenous ROS in plants. As shown in Table 2, the free proline content gradually increased with the extension of the time of exposure to Al stress to a maximum at 48 h for PA1 and 72 h for PS5 and then decreased slowly. However, after treatment with exogenous AsA, the free proline content of the root tip increased at each time point, except for the decrease in free proline in PA1 at 96 h compared with the single Al stress plants. PA1 increased by 0.3- to 0.5-fold during the period of 12–48 h, and PS5 increased by 0.2- to 0.4-fold during the period of 6–72 h.
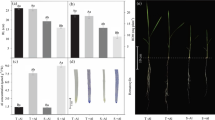
Effect of AsA on NO3 − Uptake Under Al Stress
As shown in Fig. 1, compared with the control, the uptake capacity of NO3 −–N in the two rice cultivars notably decreased with Al stress or the extension of the Al stress period. After treatment with exogenous AsA, the uptake capacity of NO3 −–N increased in both cultivars; however, the amount of the increase decreased with Al stress and the extension of the stress period, and the uptake capacity of NO3 −–N in the absence of Al stress was higher than that under Al stress. At different time points, the NO3 −–N absorption ability of the two cultivars presented obvious differences from that of the control. Comparing AsA-treated with non-AsA-treated plants at the same time point, the NO3 −–N absorption capacity of PA1 (Fig. 1a) was significantly different from that of PS5 during the entire period. However, PS5 (Fig. 1b) was significantly different at 24 h and was not obviously different during the period of 48–96 h.
Effects of exogenous AsA on NO3 − absorption in PA1 (a) and PS5 (b) roots under Al stress for different periods of time. The values represent the mean ± SD (n = 3). Different letters in the same column or cultivar indicate that the values are significantly different at the 0.05 level, according to Duncan’s multiple range test
Effect of AsA on the Contents of MDA and H2O2 Under Al Stress
The data in Table 3 show that the patterns of change in the MDA contents of the roots of the two rice cultivars were similar to those of the H2O2 contents. The contents of H2O2 and MDA in the two cultivars tended to increase with the extension of the period of Al stress. When exposed to Al stress for 96 h, the MDA and H2O2 contents of PA1 were approximately 3.8-fold and 4-fold greater than those of the control, respectively, whereas the MDA and H2O2 contents in PS5 were approximately 4-fold and 1.9-fold higher than those of the control, respectively. After treatment with exogenous AsA, the MDA and H2O2 in the roots both declined compared with those of the single Al stress plants. The MDA and H2O2 in the roots of the two cultivars were significantly reduced with a treatment time of 12 h or more. When the AsA treatment time was 96 h, the MDA and H2O2 contents were approximately 2.4-fold and 3.0-fold higher in PA1 than in the control, respectively, and approximately 2.8-fold and 1.6-fold higher in PS5 than in the control, respectively. The obvious decrease in the MDA content indicates that the degree of membrane lipid peroxidation induced by Al significantly decreased, and the damage due to Al stress in the plants was effectively alleviated.
Effect of AsA on the Activities of SOD, POD, CAT, and APX Under Al Stress
SOD is a key enzyme in the antioxidant system, scavenging free radicals. Its activity is an indicator of plant resistance to stress. As shown in Fig. 2a, b, with an increase in the Al treatment time, the SOD activity in PA1 increased initially and then essentially stabilized, and the SOD activity in PS5 gradually increased over the period of Al stress. The SOD activity in PA1 (Fig. 2a) was 2.8-fold higher than that of the control at 48 h, and the SOD activity in PS5 (Fig. 2b) was 2.6-fold higher than that of the control. Furthermore, the SOD activities in the roots of the two cultivars increased with the extension of the Al stress period. However, after treatment with an exogenous AsA, compared to a lack of AsA treatment, the SOD activity in PA1 (Fig. 2a) was 3.6-fold higher than that of the control at 48 h, whereas that of PS5 (Fig. 2b) was higher than that of the control by 3.3-fold.
Effects of exogenous AsA on the activities of antioxidant enzymes in PA1 and PS5 roots under Al stress for different periods of time. The activities of SOD in PA1 (a) and PS5 (b) roots, of POD in PA1 (c) and PS5 (d) roots, of CAT in PA1 (e) and PS5 (f) roots, and of APX in PA1 (g) and PS5 (h) roots are shown. The values represent the mean ± SD (n = 3). Different letters in the same column or cultivar indicate that the values are significantly different at the 0.05 level, according to Duncan’s multiple range test
As an adaptive enzyme in the antioxidant system, POD plays an important role in protecting the membrane lipids from peroxidation and reducing the cell damage caused by oxidative stress in plants. As shown in Fig. 2c, d, the POD activities in the roots of the two cultivars increased initially and then decreased during the period of Al stress. However, the POD activity did not change significantly from 0 to 3 h after Al treatment. Subsequently, the POD activity increased to its maximal level at 24 h. The POD activity in the PA1 roots was twice that in the control roots (Fig. 2c), and this activity in the PS5 roots (Fig. 2d) was 2.4-fold greater than that in the control roots at 24 h after Al stress. After treatment with exogenous AsA, the POD activities in the roots of the two cultivars increased with the extension of the Al stress period. The POD activity in PA1 (Fig. 2c) was 2.5-fold higher than that of the control, whereas that in PS5 (Fig. 2d) was 2.9-fold higher than that of the control at 24 h. The POD activities in the two cultivars increased with AsA treatment; the activity of both PA1 and PS5 was significantly higher than that without AsA treatment after 6 h and 12 h, respectively. The POD activity increased under Al stress. However, the addition of AsA could further enhance the POD activity. Thus, POD might play an important role in the response of AsA to Al stress.
CAT is an important protective enzyme that plays an essential role in plants’ resistance to adverse environments. The CAT activities in the roots of both cultivars increased initially and subsequently declined with extension of the Al stress period (Fig. 2e, f). The CAT activity in PA1 increased to its highest level at 24 h after Al treatment, reaching an activity level that was 2.3-fold greater than that of the control (Fig. 2e), whereas PS5 reached its maximum at 48 h (approximately 2.6-fold higher than that of the control) (Fig. 2f). However, the CAT activity in plants with AsA treatment was higher than that in plants without AsA treatment. After 12 h of treatment, the CAT activity of PA1 (Fig. 2e) was significantly higher than that of plants that had not been treated with AsA, whereas the CAT activity of PS5 (Fig. 2f) treated with AsA from 6 to 48 h was significantly higher than that of untreated plants. After 24 h of AsA treatment, the CAT activities of PA1 and PS5 were 1.3-fold and 1.2-fold higher than the CAT activity of untreated plants, respectively. These results suggest that the antioxidant enzyme activity in both cultivars was stronger than that in the control roots under Al stress and the exogenous AsA treatment further enhanced the CAT activity in the roots of both cultivars.
APX is a type of peroxidase that uses AsA as an electron donor and is the main enzyme scavenged by hydrogen peroxide. As shown in Fig. 2g, h, under Al stress conditions, the APX activity in PA1 roots increased initially and subsequently decreased slowly with the increase in the stress period. The APX activity in PA1 increased to its highest level at 48 h after Al treatment, which was 2.8-fold higher than that of the control (Fig. 2g), whereas the APX activity in PS5 increased over the entire Al treatment period and was 2.4-fold higher than that of the control at 72 h (Fig. 2h). The APX activity in the roots of both cultivars increased with AsA treatment compared to without AsA treatment. The APX activity in PA1 (Fig. 2g) was notably higher than that in plants without AsA after 12 h of treatment, whereas the APX activity in PS5 (Fig. 2h) was notably higher than that in plants without AsA after 6 h of treatment. The APX activity of PA1 and PS5 was elevated to 3.4-fold and 2.9-fold that of the control at 48 h after AsA treatment, respectively. The increased APX activity in both cultivars suggests that the crops could further remove superoxide.
The Effect of AsA on the Activity of PM H+-ATPase and the H+-Pump Under Al Stress
As shown in Fig. 3a, b, the PM H+-ATPase activity in the roots of both rice cultivars was significantly lower than that of the control at 24 h under Al stress. However, after treatment with AsA, the PM H+-ATPase activity in the roots was significantly higher than that in plants without AsA treatment. To further investigate whether AsA improved the PM H+-pump activity, this study analyzed the change in the PM H+-pump activity in rice roots. The results in Fig. 3c, d show that the changes in the PM H+-pump activity in PA1 and PS5 root tips were consistent with the changes in the H+-ATPase activity in the PM, accounting for the fact that AsA treatment also significantly increased the H+-ATPase activity and H+-pump activity in the root PM of rice.
Effects of exogenous AsA on the activities of PM H+-ATPase and H+-pump in PA1 and PS5 roots under Al stress for 24 h. The activities of PM H+-ATPase in PA1 (a) and PS5 (b) roots, and the activities of H+-pump in PA1 (c) and PS5 (d) roots. Data are the mean ± SD of three independent experiments. Different letters in the same column or cultivar indicate that the values are significantly different at the 0.05 level, according to Duncan’s multiple range test
AsA-Enhanced Expression and Interaction of PM H+-ATPase and the 14-3-3 Protein in Rice Roots Under Al Stress
The interaction of PM H+-ATPase and the 14-3-3 protein can activate PM H+-ATPase activity. To further investigate whether the AsA-enhanced NO3 −–N absorption is associated with an enhanced interaction between PM H+-ATPase and 14-3-3 proteins under Al stress, we extracted PM proteins from PA1 and PS5 root tips that underwent different treatments for 24 h and analyzed the co-immunoprecipitation (Co-IP; Fig. 4). The change in the phosphorylation level of PM H+-ATPase agreed with the change in the interaction with the 14-3-3 protein in PA1 (Fig. 4a, b) and PS5 (Fig. 4c, d) roots. Moreover, the relative expression of the PM H+-ATPase protein and the 14-3-3 protein was analyzed in silico. The data in Fig. 4 demonstrate that the relative expression of the PM H+-ATPase-bound 14-3-3 protein in the roots of PA1 and PS5 (Fig. 4e) was 0.88-fold and 0.91-fold higher than that of the control at 24 h under Al stress, respectively. When PA1 and PS5 roots were treated with Al combined with AsA, the relative expression of the PM H+-ATPase-bound 14-3-3 protein was elevated to 1.21-fold and 1.3-fold that of the control, respectively. At the same time, as shown in Fig. 4, the relative expression of the 14-3-3 protein-bound PM H+-ATPase in the PA1 and PS5 (Fig. 4f) roots was 0.52-fold and 0.61-fold higher than that of the control at 24 h under Al stress, respectively. The phosphorylation level of PM H+-ATPase and the interaction with the 14-3-3 proteins were significantly increased by treatment with both Al and AsA. The relative expression of the 14-3-3 protein-bound PM H+-ATPase in the PA1 and PS5 roots was elevated to 1.2-fold and 1.5-fold that of the control, respectively. These results demonstrate that the AsA-enhanced interaction of PM H+-ATPase with the 14-3-3 protein was higher in the PS5 roots than that in the PA1 roots.
Effects of exogenous AsA on the co-immunoprecipitation (Co-IP) of PM H+-ATPase and 14-3-3 proteins in PA1 and PS5 roots under Al stress for 24 h. The experiments were conducted more than three times, and one set of representative results is presented. Co-IP and Western blot analyses were performed to examine the interaction between phosphorylated PM H+-ATPase (a, c) and 14-3-3 protein (b, d) in PA1 and PS5 roots, respectively. The relative quantification of PM H+-ATPase that bound to the 14-3-3 protein (e) is shown in a and c, and the relative quantification of 14-3-3 protein that bound to PM H+-ATPase (f) is shown in b and d. The data are the mean ± SD of the three independent experiments. Different letters in the same column or cultivar indicate that the values are significantly different at the 0.05 level, according to Duncan’s multiple range test
The Effect of Exogenous AsA on the Endogenous AsA Content
As shown in Fig. 5, under Al stress, the endogenous AsA content in the roots of both cultivars decreased with the increase in the stress period. After 96 h of Al stress, the AsA content in PA1 was 35 % of the control content (Fig. 5a), and the AsA content in PS5 was 46 % of the control content (Fig. 5b); the AsA content remained constant in the control roots. The endogenous AsA content in the roots of both cultivars increased with AsA treatment compared to without AsA treatment. The AsA content in PA1 was 161 % of that in untreated plants (Fig. 5a), whereas it was 147 % of that of untreated plants in PS5 at 96 h after combined AsA and Al treatment (Fig. 5b). In rice roots, the increased endogenous AsA was positively related to the exogenous AsA treatment.
Effects of exogenous AsA on the endogenous AsA content in PA1 and PS5 under Al stress for different periods of time. The endogenous AsA content in PA1 (a) and PS5 (b) roots. The values represent the mean ± SD (n = 3). Different letters in the same column or cultivar indicate that the values are significantly different at the 0.05 level, according to Duncan’s multiple range test
Discussion
Root reduction activity is an important index of physiological characteristics and is correlated with respiration in plant roots. The root activity of the two rice cultivars decreased with prolonged Al stress. As shown in Table 1, the root activity in PA1 was 38 % of the control activity at 96 h after Al stress, whereas the activity in PS5 was 54 % of the control activity. The root activity in PS5 was less affected by Al stress than that in PA1; the root reduction activity recovered to the level of the control 24 h after treatment with exogenous AsA combined with Al. However, after 24 h of treatment with exogenous AsA combined with Al, the root reduction activity of both cultivars was below that of the control but higher than that of non-AsA-treated plants, indicating that exogenous AsA can improve the root reduction activity under Al stress. Greater root reduction activity indicates greater respiration intensity, which provides more energy for root activity and enhances the ability to provide nutrients and moisture to aboveground plant parts to ensure normal plant growth in a stressful environment.
AsA is a highly abundant, small antioxidant molecule in plant tissues that can remove ROS both directly (by reacting with and simultaneously reducing ROS) and indirectly (by functioning as an enzymatic substrate in ROS scavenging) (Du and others 2001). AsA is an important component of the antioxidant defense system in plants and serves as a reductant for the peroxidative removal of H2O2 (Noctor and Foyer 1998). In the present study, the endogenous AsA content in the roots of both cultivars initially decreased with the increase in Al stress duration, and the exogenous AsA treatment increased the endogenous AsA content (Fig. 5) and significantly decreased the active oxygen content compared with the treatment of rice roots with Al alone, which is consistent with the findings of Wang and Kao (2007). In addition, we observed a higher AsA content in PS5 (Al-tolerant) than in PA1 (Al-sensitive) under Al stress. This increased AsA could react with additional ROS to significantly reduce the ROS content and prevent oxidative damage. We suggest that the effect of exogenous AsA on Al tolerance could be related to the increased level of endogenous AsA and direct removal of ROS.
Under normal growth conditions, ROS are present at modest levels, and plant cells experience only mild oxidative stress; however, many stressors enhance ROS production. ROS have a positive effect on the resistance to cell invasion by pathogens and the expression of genes related to disease resistance. However, plants can experience oxidative damage if they produce an amount of ROS that exceeds the removal ability of the antioxidant system. The Al stress-related damage to rice plants was proposed to be related to oxidative injury. Lipid peroxidation is the most typical symptom of biological oxidative stress. MDA is one of the products of lipid peroxidation, and its content can reflect the degree of oxidative stress in plants. Our investigation revealed that MDA and H2O2 increased significantly in rice roots after Al stress. The contents of MDA and H2O2 were higher in PA1 than in PS5 under Al stress (Table 3). At 96 h after Al treatment, the H2O2 content in PA1 and PS5 roots was 4-fold and 1.9-fold higher than that in the control, respectively. However, after adding AsA, the H2O2 content was only 3-fold and 1.6-fold higher than that of the control, and the MDA content was 3.8-fold and 4-fold higher than that of the control, respectively. Nevertheless, after adding AsA, the MDA content was 2.4-fold and 2.8-fold higher that of the control in PA1 and PS5 roots, respectively. This conclusion is consistent with the increased MDA content in rice plants under Al stress observed by Kuo and Kao (2003) and Tewari and others (2004). Therefore, exogenous AsA can markedly reduce the accumulation of H2O2 and MDA caused by Al stress. Reducing the damage caused by oxidative stress improved the Al tolerance of rice. This result is also consistent with that of Wang and Kao (2007) and could imply that AsA plays a crucial role in scavenging ROS by increasing the endogenous AsA level.
When exposed to environmental stress, to alleviate oxidative stress, many plants have evolved mechanisms to scavenge ROS via the upregulation of antioxidant enzymes, such as SOD, POD, CAT, and APX, and the enhancement of their activities. These antioxidant enzymes function synergistically to scavenge active oxygen free radicals to protect plants from oxidative stress and to alleviate the membrane lipid peroxidation that leads to cell membrane damage. Many studies have indicated that Al not only induces oxidative stress but also induces the expression of antioxidant enzymes, leading to an increase in their activity. As the enzyme activity increases, the ability to remove ROS increases, as does the plant’s resistance. As shown in Fig. 2, the activities of SOD, POD, CAT, and APX initially increased and subsequently declined in rice seedling root tips under Al stress. The levels of SOD, POD, CAT, and APX in PA1 were 2.8-fold, 2-fold, 2.3-fold, and 2.8-fold higher than those in the control, respectively, and those in PS5 were 2.6-fold, 2.4-fold, 2.6-fold, and 2.4-fold higher than those in the control, respectively. After AsA treatment, the activity of SOD, POD, CAT, and APX increased in the roots of both rice cultivars. The levels of SOD, POD, CAT, and APX in PA1 were elevated to 3.6-fold, 2.5-fold, 2.8-fold, and 3.4-fold those of the control, respectively, and those in PS5 were 3.3-fold, 2.9-fold, 3.2-fold, and 2.9-fold higher than those in the control, respectively. The increase in antioxidant enzymes effectively decreased the accumulation of H2O2 induced by Al stress in rice roots, which reduced the oxidative damage caused by ROS stress and the membrane lipid peroxidation. These results demonstrate that AsA might be involved in an important response mechanism to Al stress by regulating the activity of antioxidant enzymes in rice plants.
The enhanced production of ROS under Al stress can pose a threat to cells, but it is also thought that ROS serve as signaling molecules to activate the stress response and defense pathways (Wang and others 2004). Al enhanced the activities of several antioxidant enzymes, including SOD, POD, CAT, and APX, associated with increased ROS generation. Meanwhile, the level of endogenous AsA and antioxidant enzyme activity increased after treatment with exogenous AsA and Al. The increased levels of endogenous AsA and antioxidant enzyme activity indicate that Al-induced stress tolerance in rice may be caused by increased endogenous AsA and antioxidant activity, which, in turn, synergistically reduces the level of H2O2 and oxidative injury to membranes. This alleviation of oxidative injury was attributed to the protection of rice against growth inhibition due to Al stress and to increased NO3 −–N uptake.
The uptake of NO3 −–N in root cells is an important step in nitrogen metabolism in plants. At the absorption stage, the co-transport of NO3 −/2H+ energized by the H+-ATPase of the cell membrane is currently recognized as the NO3 − transmembrane transport mode (Mcclure and others 1990). The membrane transport of substances in the reverse concentration gradient is an energy-consuming process, but plant cells cannot directly make use of the energy from ATP hydrolysis to promote the transmembrane transport of the material. The PM H+-ATPase activated H+-pump activity and generated an electrochemical gradient across the membrane by hydrolyzing ATP and excluding H+ outside the cell membrane to provide a driving force for the uptake of nutrients and secondary transport in plants. The results of this study showed that exogenous AsA can enhance PM ATPase activity, H+-pump activity, and the interaction of 14-3-3 proteins and PM H+-ATPase in the roots of both rice cultivars (Fig. 4), which can significantly increase the uptake of nitrate nitrogen.
Conclusions
Al treatment caused oxidative stress in the roots of two rice varieties with different tolerances to Al, and PS5 had stronger resistance to Al than PA1. During the induction of oxidative stress due to Al stress, the expression of antioxidant enzymes was also induced, which increased the antioxidant enzyme activity and the ability to scavenge ROS, enhancing the plants’ resistance to Al. Exogenous AsA plays an important role in scavenging ROS by enhancing the endogenous AsA content and further improving the activities of SOD, POD, CAT, and APX in the two types of rice roots, which more effectively removes accumulated H2O2 and reduces the level of peroxidation of membrane lipids and the plant damage due to Al stress. Meanwhile, AsA increases the activity of PM H+-ATPase and the ability to pump H+ by enhancing the interaction of PM H+-ATPase and the 14-3-3 protein in rice roots, which provides a large amount of H+ and energy for the absorption of NO3 −–N, improving the NO3 −–N uptake ability in rice.
References
Achary VMM, Jena S, Panda KK, Panda BB (2008) Aluminium induced oxidative stress and DNA damage in root cells of Allium cepa L. Ecotoxicol Environ Saf 70(2):300–310
Aebi H (1984) Catalase in vitro. Methods Enzymol 105(C):121–126
Alterfjord M, Sehnke PC, Arkell A (2004) Plasma membrane H+-ATPase and 14-3-3 isoforms of Arabidopsis leaves: evidence for isoform specificity in the 14-3-3/H+-ATPase interaction. Plant Cell Physoi 45(9):1202–1210
Baohui Ma, Gao Lu, Hongxiao Zhang, Jin Cui, Zhenguo Shen (2012) Aluminum-induced oxidative stress and changes in antioxidant defenses in the roots of rice varieties differing in Al tolerance. Plant Cell Rep 31(4):687–696
Chance B, Mehley AC (1955) Assay of catalase and peroxidase. In: Colowick SP, Kaplan NO (eds) Methods in enzymology, 2nd edn. Academic Press, New York, pp 764–775
Chapman N, Miller T (2011) Nitrate transporters and root architecture. In: Geisler M, Venema K (eds) Transporters and pumps in plant signaling, signaling and communication in plants. Springer, Berlin, pp 165–190
Demiral T, Türkan I (2005) Comparative lipid peroxidation, antioxidant defense systems and proline content in roots of two rice cultivars differing in salt tolerance. Environ Exp Bot 53(3):247–257
Du XM, Yin WX, Zhao YX, Zhang H (2001) The production and scavenging of reactive oxygen species in plants. Chin J Biotechnol 17(2):121–125
Fuglsang AT, Visconti S, Drumm K, Palmgren MG (1999) Binding of 14-3-3 protein to the plasma membrane H+-ATPase AHA2 involves the three C-terminal residues Tyr(946)-Thr-Val and requires phosphorylation of Thr(947). Biochem 274(51):74–80
Gay CA, Gebicki JM (2003) Measurement of protein and lipid hydro peroxides in biological systems by the ferric-xylenol orange method. Anal Biochem 315(1):29–35
Giannakoula A, Moustakas M, Syros T, Yupsanis T (2010) Aluminum stress induces up-regulation of an efficient antioxidant system in the Al-tolerant maize line but not in the Al-sensitive line. Environ Exp Bot 67(3):487–494
Giannopolitis CN, Ries SK (1977) Superoxide dismutase I. Occurrence in high plants. Plant Physiol 59(2):309–314
Guo JH, Liu XJ, Zhang Y, Zhang JL, Shen WX, Han WF, Zhang P, Christie KW, Goulding PM, Zhang FS (2010) Significant acidification in major Chinese croplands. Science 327(5968):1008–1010
Guo CL, Chen Q, Zhao XL, Chen XQ, Zhao Y, Wang L, Li KZ, Yu YX, Chen LM (2013) Al-enhanced expression and interaction of 14-3-3 protein and plasma membrane H+ -ATPase is related to Al-induced citrate secretion in an Al-resistant black soybean. Plant Mol Bio Rep 31(4):1012–1024
Helali SMR, Nebli H, Kaddour R, Mahmoudi H (2010) Influence of nitrate–ammonium ratio on growth and nutrition of Arabidopsis thaliana. Plant Soil 336(2):65–74
Huang JL, Wang SHH, Zhang ZHX (2008) Effect of external AsA on the photoinhibition of ginger leaves in vitro. Acta Bot Boreal Occident Sin 28(10):2041–2046
Jones DL, Blancaflor EB, Kochian LV, Gilroy S (2006) Spatial coordination of aluminium uptake, production of reactive oxygen species, callose production and wall rigidification in maize roots. Plant Cell Environ 29(7):1309–1318
Kuo MC, Kao CH (2003) Aluminum effects on lipid peroxidation and antioxidative enzyme activated in rice leaves. Biol Plant 46(1):149–152
Law MY, Charles SA, Halliwell B (1983) Glutathione and ascorbic acid in spinach (Spinacia oleracea) chloroplasts. Biochem 210:899–903
Liu J, Piñeros MA, Kochian LV (2014) The role of aluminum sensing and signaling in plant aluminum resistance. J Integr Plant Biol 56(3):221–230
Luo J, Qin J, He F, Li H, Liu T, Polle A, Peng C, Luo ZB (2013) Net fluxes of ammonium and nitrate in association with H+ fluxes in fine roots of Populus popularis. Planta 237(4):919–931
Ma YX, Zhang YX, Lu J, Yu JH, Wang XW (2015) Mitigation of autotoxicity stress of tomato by ascorbic acid. J Agro Environ Sci 34(7):1247–1253
Mcclure PR, Kochian LV, Spanswick RM, Shaff JE (1990) Evidence for cotransport of nitrate and protons in maize roots II. Measurement of NO3- and H+ fluxes with ion-selective microelectrodes. Plant Physiol 93(1):290–294
Munne-Bosch S, Alegte L (2002) Interplay between ascorbic acid and lipophilic antioxidant defenses in chloroplasts of water-stressed Arabidopsis plants. FEBS Lett 524(1–3):145–148
Nakano K, Asada K (1981) Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplasts. Plant and Cell Physiol 22(5):867–880
Noctor G, Foyer CH (1998) Ascorbate and glutathione: keeping active oxygen under control. Annu Rev Plant Physiol Plant Mol Biol 49(1):249–279
Palmgren MG (2001) Plant plasma membrane H+-ATPases: powerhouses for nutrient uptake. Annu Rev Plant Physiol 52(1):817–845
Pertl Heidi, Pöckl Magdalena, Blaschke Christian, Obermeyer Gerhard (2010) Osmoregulation in Lilium pollen grains occurs via modulation of the plasma membrane H+-ATPase activity by 14-3-3 proteins. Plant Physiol 154(4):1921–1928
Serrano R (1989) Structure and function of plasma membrane ATPase. Annu Rev Plant Physiol 40:61–94
Sharma P, Dubey R (2007) Involvement of oxidative stress and role of antioxidative defense system in growing rice seedlings exposed to toxic concentrations of aluminum. Plant Cell Rep 26(11):2027–2038
Shen H, He LF, Sasaki T, Yamamoto Y, Zheng SJ, Ligaba A, Yan XL, Ahn SJ, Yamaguchi M, Sasakawa H, Matsumoto H (2005) Citrate secretion coupled with the modulation of soybean root tip under aluminum stress. Upregulation of transcription, translation, and threonine-oriented phosphorylation of plasma membrane H+-ATPase. Plant Physiol 138(1):287–296
Tewari RK, Kumar P, Tewari N, Srivastava S, Sharma PN (2004) Macronutrient deficiencies and differential antioxidant responses influence on the activity and expression of superoxide dismutase in maize. Plant Sci 66(3):687–694
Vasconcelos SS, Rossiello ROP (2002) Differential root responses to aluminum stress among Brazilian rice genotypes. Plant Nutr 25(3):655–669
Wang JM, Kao CH (2007) Protective effect of ascorbic acid and glutathione on AlCl3 induced growth of rice roots. Biol Plant 51(3):493–500
Wang XB, Wu P, Hu B, Chen QS (2002) Effects of nitrate on the growth of lateral root and nitrogen absorption in rice. Acta Bot Sin 44(6):678–683
Wang YS, Wan GJ, Yang ZM, Wang QY, Lo B, Li SQ, Lu YP, Wang SH, Sun X (2004) Salicylic Acid modulates aluminum-induced oxidative stress in roots of cassia tora. Acta Bot Sin 46(7):819–828
Xu BG, Shi GX, Xu QS, Wang X, Zhao J, Hu JZH (2006) Protective effects of exogenous ascorbic acid on antioxidant system in Hydrilla verticillata under Cd2+ stress. Chin JAppl Ecol 17(9):1768–1770
Zhang ZL, Wei JQ (2003) Plant physiology experiment guidance, 3rd edn. Higher Education Press, Beijing
Zhang RP, Liu G, Wu N, Gu M, Zeng HQ, Zhu YY, Xu GH (2011) Adaptation of plasma membrane H+-ATPase and H+-pump to P deficiency in rice roots. Plant Soil 349(1):3–11
Zheng QW, Wang XK, Xie JQ, Feng ZZ, Feng ZW, Ni XW, Ou Yang ZY (2006) Effects of exogenous ascorbate acid on membrane protective system of in situ rice leaves under O3 stress. Acta Ecol Sin 26(4):1131–1137
Zhou XH, Gu ZH, Xu HN, Chen LM, Li KZ (2015) The effects of exogenous ascorbic acid on photosynthetic characteristics in Oryza sativa L. under aluminum stress. J Yangzhou Univ (Agricultural and Life Science Edition) 36(3):73–78
Zhu YY, Zeng HQ, Di TJ, Xu GH, Shen QR (2011) Involvement of Plasma Membrane H+-ATPase in Adaption of Rice to Ammonium Nutrient. Chin J Rice Sci 18(4):335–342
Acknowledgments
The work in the authors' lab was funded by the Ministry of Science and Technology of the People Republic of China (Grant No. 2014CB138701) and National Natural Science Fund of China (Grant Nos. 31260297, 31560351, 31260063).
Author information
Authors and Affiliations
Corresponding author
Additional information
An erratum to this article is available at http://dx.doi.org/10.1007/s00344-017-9679-5.
Rights and permissions
About this article
Cite this article
Zhou, X., Gu, Z., Xu, H. et al. The Effects of Exogenous Ascorbic Acid on the Mechanism of Physiological and Biochemical Responses to Nitrate Uptake in Two Rice Cultivars (Oryza sativa L.) Under Aluminum Stress. J Plant Growth Regul 35, 1013–1024 (2016). https://doi.org/10.1007/s00344-016-9599-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00344-016-9599-9