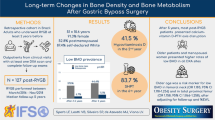
Abstract
Introduction
Bone mineral density (BMD) declines in the initial years after bariatric surgery, but long-term skeletal effects are unclear and comparisons between sleeve gastrectomy (SG) and Roux-en-Y gastric bypass (RYGB) are rare.
Design and Methods
An observational longitudinal study of obese patients undergoing SG or RYGB was performed. Whole-body (WB) BMD, along with BMD of the total hip (TH), femoral neck (FN), and lumbar spine (LS), was measured by dual-energy X-ray absorptiometry (DXA) before surgery and yearly thereafter for 4 years. Calciotropic hormones were also measured.
Results
Forty-seven patients undergoing RYGB surgery and 28 patients undergoing SG were included. Four years after RYGB, BMD declined by 2.8 ± 5.8% in LS, 8.6 ± 5% in FN, 10.9 ± 6.3% in TH, and 4.2 ± 6.2% in WB, relative to baseline. For SG, BMD declined by 8.1 ± 5.5% in FN, 7.7 ± 6% in TH, 2.0 ± 7.2% in LS, and 2.5 ± 6.4% in WB after 4 years, relative to baseline. Vitamin D levels increased with supplementation in both groups. Whereas parathyroid hormone levels increased slightly in the RYGB group, they decreased modestly in the SG group (P < 0.05 in both groups).
Conclusions
Bone loss after 4 years was comparable between the two procedures, although RYGB was associated with a slightly greater decrease at the TH than SG. Bone health should therefore be monitored after both RYGB and SG.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Bariatric surgery is the most effective treatment to achieve major, long-term weight loss [1,2,3,4]. Reflective of the obesity epidemic, the number of bariatric surgeries performed worldwide is also increasing. In 2014, the most commonly performed procedure was sleeve gastrectomy (SG), performed in 45.9% of total surgical procedures, followed by Roux-en-Y gastric bypass (RYGB) (39.6%), and adjustable gastric banding (AGB) (7.4%) [5]. Bariatric surgery is associated with beneficial metabolic effects, including the reduction of several obesity-related comorbidities and an overall decrease in mortality [6,7,8]. However, bariatric interventions are known to represent a challenge for bone physiology even though many aspects of this surgical complication remain unclear. The interaction of obesity with bone metabolism is complex, with many aspects of this interaction still poorly understood. It was initially thought that obesity had protective effects on skeletal health [9]; however, this notion has recently been challenged, with studies showing that obesity is associated with lower risk for certain fractures (wrist, hip, rib, pelvis) but higher risk for others (ankle, lower leg), along with a lack of protective effects after menopause [10]. Several longitudinal studies have shown that bone loss starts during the first year after RYGB and may continue for at least 24 months after RYGB. The overall decrease was consistently greatest during the first year [11,12,13,14,15,16,17,18,19], with the strongest effects observed in postmenopausal women [20]. Despite the strong associations between bariatric surgery and bone mineral density (BMD) loss, only one prospective study has assessed changes in BMD over a period of > 3 years after RYGB [21]. Furthermore, only a handful of studies have described the changes in BMD after other bariatric techniques such as AGB and SG [22,23,24,25,26].
The postoperative adjusted fracture risk has been shown to be higher in bariatric surgery patients relative to both obese (relative risk (RR) 1.38, 95% confidence interval (CI) 1.23 to 1.55) and non-obese control groups (RR 1.44, 95% CI 1.29 to 1.59) in several studies. Although these studies did show a decreased risk of distal lower limb fracture overall, the risk of upper limb, clinical spine, pelvic, hip, or femur fractures increased [27, 28].
Here, we compared the changes in BMD within the first 4 years after RYGB and SG in morbidly obese subjects.
Patients and Methods
Data Source
We performed an observational longitudinal study of morbidly obese patients undergoing SG or RYGB between November 2007 and October 2014 at University Hospital. The institutional review board approved the study protocol, and all subjects provided written informed consent.
Patients
The criteria used to assess eligibility for bariatric surgery were in accordance with the French guidelines for bariatric surgery [29]. To be included in the study, patients had to be followed at least 4 years after the procedure and to have a yearly measurement of BMD. Preoperative assessments included abdominal ultrasound, bone densitometry, functional respiratory tests, upper gastrointestinal endoscopy, and blood analysis including nutritional parameters. The surgical procedure was determined by a multidisciplinary staff, taking into consideration the patient’s overall state of health.
Follow-up visits with both the surgeon and endocrinologists were performed 1, 3, 6, 12, 18, and 24 months after surgery, with yearly visits thereafter. Blood analyses were performed at each visit, and vitamin supplementation was adapted according to blood tests. After bariatric surgery, patients were maintained on polyvitamins all life. In addition, specific supplementation for vitamin D was given according to the blood concentration of vitamin D. If blood concentration of vitamin D was between 20 and 30 ng/ml (50–75 nmol/l), patients were maintained on colecalciferol 100,000 UI every 15 days for 1 months and a half and then every 3 months. If it was between 10 and 20 ng/ml (25–50 nmol/l), 100,000 UI of colecalciferol was administered every 15 days for 2 moths and then every 3 months. Observance was not specifically monitored.
Anthropometric data (body weight, BMI [kg/m2], weight loss, percentage of weight loss, percentage of excess weight loss, and physical activity score), and blood test variables, including calcium, phosphorus, albumin, creatinine, glomerular filtration rate (GFR), 25-hydroxyvitamin D (25OHD), intact parathyroid hormone (PTH), alkaline phosphatase (ALP), C-reactive protein (CRP), fasting insulin, fasting glucose, and homeostasis model assessment of insulin (HOMA-IR), were collected at baseline and at each follow-up. Areal BMD was measured at baseline and every year thereafter.
Surgical Technique
All procedures were performed in the same center. RYGB intervention consisted of a non-banded pouch and a transmesocolic and retrogastric gastrojejunostomy. A 120-cm alimentary limb and a 40-cm biliopancreatic limb were built.
SG intervention consisted of a resection of the greater curvature including the complete fundus, which was resected from the distal antrum (6 cm proximal to the pylorus) to the angle of His over a 32-French bougie.
During hospitalization and after hospital discharge, patients were asked to follow a dietary protocol and were given the same dietary advice.
Physical Activity Score
A 20-point non-validated local score assessing sedentary behavior (5 points), physical activity (5 points), and physical condition (10 points) was used. Patients also performed a validated walk (6-min walk test) as well as validated strength, balance, and flexibility tests as an objective assessment of their physical condition. Assessment of physical activity and sedentary behavior was subjective. Patients were asked about their travel patterns and the frequency of their physical activity lasting at least 30 min.
Bone Mineral Densitometry
Areal BMD (g/cm2) was measured by dual-energy X-ray absorptiometry (DXA) using a Lunar Expert XL (GE Healthcare, USA). Lumbar spine (LS) (L2–L4), femoral neck (FN), total hip (TH), and whole-body (WB) BMD were measured independently. Z-score is defined as the number of standard deviations above or below the mean for the patient’s age, sex, weight, and ethnicity [30]. We did not select the T-score to have consistent data for all patients due to the inclusion of both young women and men in the cohort.
Biochemical Analyses
25OHD was measured by Automate Liaison (DiaSorin). PTH and insulin were measured by chemiluminescence using a Unicel DXi 800 (Beckman Coulter). GFR was estimated using the CKD-EPI creatinine equation (2009).
Statistical Analysis
Quantitative variables were presented as either the mean with standard deviation, or as the median with interquartile range. Qualitative variables were presented as percentages. A Shapiro-Wilk test was used to test the assumption of a normal distribution for each quantitative variable. Due to the low sample size, non-parametric tests were performed to compare quantitative variables. A Mann-Whitney U test was used to compare independent samples (comparisons between RYGB vs. SG), and a paired Wilcoxon rank test was used for matched data (comparisons between time points within the same group). Qualitative variables were compared using Fisher’s exact test. Correlation analyses were performed using non-parametric Spearman’s correlation coefficient. All statistical analyses were performed using STATA SE 13.1 software, with P values < 0.05 considered statistically significant.
Results
Baseline
Forty-seven patients undergoing RYGB surgery and 28 patients undergoing SG were included in this study. Eleven patients were excluded because they had no BMD measurement before bariatric surgery and 17 were excluded because they had no yearly BMD measurement during the 4 years after bariatric surgery. In addition, 3 other patients were excluded from the study because of a pancreatic cancer for one patient, a terminal kidney insufficiency that needed dialysis for a second one and because sleeve gastrectomy was converted to gastric bypass during follow-up for the third one. Their clinical characteristics are shown in Table 1. Age, sex/menopause status, smoking status, body weight, BMI, comorbidity, physical status score, and physical activity levels were not significantly different between groups. At baseline, LS and TH BMD were significantly higher in the RYGB group than in the SG group. WB BMD tended to be higher in the RYGB group than in the SG group. There was no difference in FN BMD between the two groups.
The biological characteristics of the two groups are shown in Table 2. At baseline, no significant differences were observed in albumin, calcium, calcium corrected for albumin, creatinine, GFR, PTH, 25OHD, ALP, CRP, glycemia, plasma insulin concentrations, or HOMA-IR scores between groups. The average vitamin D concentration was below the optimal threshold, but only 5% of patients had PTH levels above the laboratory standard.
Weight Loss
Weight loss evolution in each group is shown in Fig. 1. Four years after bariatric surgery, the average weight decrease was 27.4 ± 10.5% in the RYGB group versus 19.5 ± 13.6% in the SG group (P < 0.01).
Changes in Bone Mineral Density
LS BMD scores were available at baseline and at 1-year follow-up for all patients (75), at 2-year follow-up for 70 patients (93%), at 3-year follow-up for 66 patients (88%), and for 69 patients (92%) at the last follow-up, 4 years after surgery. TH and FN BMD scores were available at baseline and at 1-year follow-up for 74 patients (99%), at 2-year follow-up for 69 patients (92%), at 3-year follow-up for 65 (87%) patients, and for 68 patients (91%) at 4-year follow-up. WB BMD scores were available at baseline and 1-year follow-up for 68 patients (91%), at 2-year and 3-year follow-up for 62 patients (83%), and for 63 patients (84%) at the end of the 4 years of follow-up.
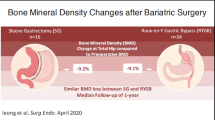
At year 4 in the RYGB group, the rates of bone loss were 2.8 ± 5.8% in LS, 8.6 ± 5% in FN, 10.9 ± 6.3% in TH, and 4.2 ± 6.2% in WB, respectively (Fig. 2). At year 4 in the SG group, the rates of bone loss were 2.0 ± 7.2% in LS, 8.1 ± 5.5% in FN, 7.7 ± 6.4% in TH, and 2.5 ± 6.4% in WB.
At the WB, the 2-year percentage of bone loss was greater in the RYGB than in the SG group (P < 0.05). A higher rate of bone loss was also found in the RYGB group compared to the SG group for the TH at year 3 and 4 (P < 0.05). The other percentages of bone loss were comparable (Fig. 2).
In the RYGB group, mean BMD values were significantly lower at year 4 than at baseline and year 1 for all bone sites (P < 0.05) (Supplementary Table 1). In the SG group, both FN and TH BMD values were significantly lower at year 4 than at baseline (P < 0.05). LS BMD at year 4 was significantly lower than at year 1 (P < 0.05), but comparable to baseline values. WB BMD values did not vary significantly throughout the follow-up (Supplementary Table 2).
Z-scores fell consistently within the high normal range throughout the 4 years of the study (Table 3). At baseline, none of the patients had Z-scores < − 2 at any site. Four years after bariatric surgery, only a single 41-year-old woman who underwent RYGB had a Z-score < − 2 at the LS.
At baseline, Z-scores in LS and TH were significantly lower in the SG group, relative to the RYGB group, but not in either FN or WB. Four years after SG, there were no significant differences between Z-scores. FN and TH Z-scores 4 years after SG or RYGB were significantly lower than those seen at baseline. Average LS and WB Z-scores remained unchanged 4 years after SG or RYGB.
Changes in Biological Parameters
Changes in biological parameters over time are shown in Tables 4 and 5. Four years after bariatric surgery, albumin, calcium, calcium corrected for albumin, creatinine, GFR, ALP, vitamin D, glycaemia, insulin, and HOMA-IR values were not significantly different between the RYGB and SG groups. Mean CRP was lower in the RYGB group than in the SG group (2.4 ± 3.1 mg/L vs. 4.6 ± 4.2 mg/L, respectively; P < 0.05). Mean PTH levels were higher in the RYGB group relative to the SG group (55.5 ± 17.4 ng/mL vs. 46.6 ± 11.4 ng/mL, respectively; P < 0.05).
Predictors of Skeletal Changes
No correlations were found between any of the biological parameters, including PTH and CRP, and bone loss at year 4 for either bariatric surgery, when considered together. There was a positive correlation between the percentage of weight loss and the percentage of BMD loss in FN, TH, and WB at year 4 (r = 0.394, P < 0.001; r = 0.472, P < 0.001; and r = 0.289, P = 0.021, respectively) but not in LS (r = 0.153, P = 0.21).
Discussion
When rates of bone loss were compared between the two procedures, only bone loss in TH was significantly higher in the RYGB group compared with the SG group. Bone loss continues at a slower rate after the first year following bariatric surgery, and it might be interesting to have a control group to compare with normal physiological loss.
Overall, bone loss in our population was similar than that reported in reviews of long-term retrospective studies [32, 33]; however, few studies assessed changes in BMD over more than 2 years following bariatric surgery. In a 5-year prospective study of 21 patients after RYGB, vertebral BMD decreased by 7.8% after 5 years, whereas the average BMD of FN and TH declined by 15.3% and 14.1%, respectively. Bone loss was greatest within the first 2 years after surgery, with slower, but continued, bone loss seen thereafter [21]. A second long-term prospective study reported that the initial decrease in BMD within the first postoperative period was 10% in FN and 3% in the spine. Thereafter, the rate of bone loss decreased to rates of 2.7% and 3.1%, respectively, between the first and the third year after RYGB [15].
To determine whether the observed reductions in BMD were clinically significant with regard to the subsequent risk of fracture, Z-scores were calculated in each patient. Even though the mean Z-scores were lower in FN and TH at year 4 compared to baseline, they remained above the range of age-adjusted normal values. A decline in LS and WB Z-scores was not observed, although this discrepancy may be explained by the fact that Z-scores are adjusted to body weight with the GE-Lunar devices which is not the case for the Hologic devices [30, 34].
The negative skeletal effects of bariatric surgery are presumably multifactorial and are not yet fully understood [19, 33, 35, 36]. A positive correlation was found between the percentages of weight loss and the associated bone loss in FN, TH, and WB, with no association seen in LS. We do not have the statistical power to do more accurate analysis. These results are consistent with previous reports, in which a strong association between weight loss and bone loss after bariatric surgery were observed in most, but not all, studies, especially at bone sites other than TH and FN [32, 37]. The effects of skeletal muscle on static and dynamic loading have been shown to have direct beneficial effects on bone. A recent study showed that a supervised exercise program halved bone loss in both LS and TH one year after RYGB [38]. Vitamin D, calcium, and protein supplementation with exercise have also been shown to have positive effects on bone protection 2 years following bariatric surgery (RYGB or SG) [39]. In contrast, no correlation was found between PTH, calcium, phosphorus, and vitamin D levels throughout the follow-up, with no association to bone loss 4 years after surgery, although < 10% of RYGB patients and no SG patients had a PTH level above the normal range 4 years after surgery. This rate was higher in some other studies, but not all [37], with some studies reporting striking declines in BMD even in the absence of significant changes in circulating vitamin D or PTH levels [32]. A recent study evaluating the rate of secondary hyperparathyroidism (SHPT) (serum PTH above the normal limit) in obese subjects after different bariatric surgeries reported SHPT rates of 21.0% before surgery, 35.4% 1 year after surgery, and 63.3% after 5 years. Single anastomosis (mini-) gastric bypass (SAGB) had the highest prevalence of SHPT (50.6%), followed by RYGB (33.2%), LAGB (25.8%), and SG (17.8%) 1 year after surgery. Five years after surgery, SAGB still had the highest prevalence of SHPT (73.6%), followed by RYGB (56.6%), LAGB (38.5%), and SG (41.7%) [40]. A careful monitoring of vitamin D supplementation may explain the low rate of SHPT in our study. No correlation was found between bone loss and CRP, glycaemia, insulin, or HOMA-IR 4 years after bariatric surgery, although the present study was not designed to highlight a potential role of these parameters.
Mean baseline TH and LS BMD values differed significantly between the two groups, with higher values in the RGYB group. Also, baseline WB BMD tended to be higher in the RYGB group than in the SG group. Although these may be explained by cumulative non-significant clinical baseline differences between groups; The mean age of the RYGB patients was 44.4 ± 10.6 years compared to 47.3 ± 11.5 years in the SG group (P = 0.22), and the percentages of men and postmenopausal women were 32% and 21%, respectively, in the RYGB group compared to 21% and 39%, respectively, in SG group (P = 0.23). The higher bone loss in the RYGB group is concordant with changes in calciotropic hormone levels, along with a higher percentage of weight loss in the RYGB group compared to the SG group (27.4 ± 10.5% vs. 19.5 ± 13.6%; P < 0.01), and the regression towards the mean. On the other hand, the modest differences in the clinical characteristics of the 2 groups at baseline above mentioned (sex/menopause status and age) may have contributed to the greater bone loss seen in the SG group relative to the RYGB group and may explain why differences were not found in LS, FN, or WB. A recent study comparing bone changes in 21 patients 1 year after RYGB and SG revealed a higher degree of bone loss in TH and FN after RYGB compared to SG, with no significant differences in LS between groups [22]. Among a cohort of 43 non-menopausal women, a tendency towards greater bone loss at the FN, LS, and WB was observed in the RYGB group compared to the SG 1 year after surgery [41]. Another study reported similar changes in LS and FN BMD 1 year after RYGB or SG, although baseline data were not available to assess these changes over time [23]. Finally, a study involving only 15 patients who underwent bariatric surgery did not show significant differences in either LS or TH bone loss 1 year after RYGB or SG [24].
One of the strengths of this study is the long-term follow-up. Furthermore, all BMD measurements were performed by the same team with the same DXA device throughout the study, which reduces possible measurement biases and allows for external validity. Moreover, the rigorous selection of patients before bariatric surgery and the careful collection of all biological parameters are likely to strengthen the validity of the present results. Nevertheless, patients were not randomized, and the two groups are not fully comparable, which might have influenced some of the comparisons in BMD during the follow-up period. In addition, size measurements and fracture events are missing. Of note, it must be emphasized that BMD is not the only factor involved in risk of bone fractures. Indeed, we did not investigate some important factors like family history of osteoporosis, prior bone fractures, ethnicity, alcohol intake, and medication use. Moreover, we do not have a control group without surgery to compare bone loss.
In summary, we found that bone loss after both RYGB and SG continued after the first year, with slower rates of bone loss observed after the first 2 years. The amount of bone loss was comparable between the two procedures after 4 years, with the exception of the TH with a greater bone loss in the RYGB than in the SG group. Further studies are needed to determine the mechanisms of bone loss, as well as to investigate therapeutic strategies to preserve skeletal health in patients receiving bariatric surgery.
References
Adams TD, Halverson RC, LaMonte MJ, et al. Long-term mortality after gastric bypass surgery. N Engl J Med. 2007;9.
Maciejewski ML, Arterburn DE, Van Scoyoc L, et al. Bariatric surgery and long-term durability of weight loss. JAMA Surg. 2016;151(11):1046–55. https://doi.org/10.1001/jamasurg.2016.2317.
O’Brien PE, Hindle A, Brennan L, et al. Long-term outcomes after bariatric surgery: a systematic review and meta-analysis of weight loss at 10 or more years for all bariatric procedures and a single-centre review of 20-year outcomes after adjustable gastric banding. Obes Surg. 2019;29(1):3–14. https://doi.org/10.1007/s11695-018-3525-0.
Golzarand M, Toolabi K, Farid R. The bariatric surgery and weight losing: a meta-analysis in the long- and very long-term effects of laparoscopic adjustable gastric banding, laparoscopic Roux-en-Y gastric bypass and laparoscopic sleeve gastrectomy on weight loss in adults. Surg Endosc. 2017;31(11):4331–45. https://doi.org/10.1007/s00464-017-5505-1.
Angrisani L, Santonicola A, Iovino P, et al. Bariatric surgery and endoluminal procedures: IFSO worldwide survey 2014. Obes Surg. 2017;27(9):2279–89. https://doi.org/10.1007/s11695-017-2666-x.
Sjöström L. Review of the key results from the Swedish Obese Subjects (SOS) trial - a prospective controlled intervention study of bariatric surgery. J Intern Med. 2013;273(3):219–34. https://doi.org/10.1111/joim.12012.
Arterburn DE, Olsen MK, Smith VA, et al. Association between bariatric surgery and long-term survival. JAMA. 2015;313(1):62–70. https://doi.org/10.1001/jama.2014.16968.
Cardoso L, Rodrigues D, Gomes L, et al. Short- and long-term mortality after bariatric surgery: a systematic review and meta-analysis: CARDOSO et al. Diabetes Obes Metab. 2017;19(9):1223–32. https://doi.org/10.1111/dom.12922.
De Laet C, Kanis JA, Odén A, et al. Body mass index as a predictor of fracture risk: a meta-analysis. Osteoporos Int. 2005;16(11):1330–8. https://doi.org/10.1007/s00198-005-1863-y.
Compston JE, Watts NB, Chapurlat R, et al. Obesity is not protective against fracture in postmenopausal women: GLOW. Am J Med. 2011;124(11):1043–50. https://doi.org/10.1016/j.amjmed.2011.06.013.
Fleischer J, Stein EM, Bessler M, et al. The decline in hip bone density after gastric bypass surgery is associated with extent of weight loss. J Clin Endocrinol Metabol. 2008;93(10):3735–40. https://doi.org/10.1210/jc.2008-0481.
Casagrande DS, Repetto G, Mottin CC, et al. Changes in bone mineral density in women following 1-year gastric bypass surgery. Obes Surg. 2012;22(8):1287–92. https://doi.org/10.1007/s11695-012-0687-z.
Stein EM, Carrelli A, Young P, et al. Bariatric surgery results in cortical bone loss. J Clin Endocrinol Metabol. 2013;98(2):541–9. https://doi.org/10.1210/jc.2012-2394.
Shanbhogue VV, Støving RK, Frederiksen KH, et al. Bone structural changes after gastric bypass surgery evaluated by HR-pQCT: a two-year longitudinal study. Eur J Endocrinol. 2017;176(6):685–93. https://doi.org/10.1530/EJE-17-0014.
Vilarrasa N, San José P, García I, et al. Evaluation of bone mineral density loss in morbidly obese women after gastric bypass: 3-year follow-up. Obes Surg. 2011;21(4):465–72. https://doi.org/10.1007/s11695-010-0338-1.
Coates PS, Fernstrom JD, Fernstrom MH, et al. Gastric bypass surgery for morbid obesity leads to an increase in bone turnover and a decrease in bone mass. J Clin Endocrinol Metab. 2004;89(3):1061–5. https://doi.org/10.1210/jc.2003-031756.
Mahdy T, Atia S, Farid M, et al. Effect of Roux-en Y gastric bypass on bone metabolism in patients with morbid obesity: Mansoura experiences. Obes Surg. 2008;18(12):1526–31. https://doi.org/10.1007/s11695-008-9653-1.
Yu EW, Bouxsein ML, Putman MS, et al. Two-year changes in bone density after roux-en-Y gastric bypass surgery. J Clin Endocrinol Metab. 2015;100(4):1452–9. https://doi.org/10.1210/jc.2014-4341.
Gagnon C, Schafer AL. Bone health after bariatric surgery. JBMR Plus. 2018;2(3):121–33. https://doi.org/10.1002/jbm4.10048.
Schafer AL, Kazakia GJ, Vittinghoff E, et al. Effects of gastric bypass surgery on bone mass and microarchitecture occur early and particularly impact postmenopausal women. J Bone Miner Res. 2018;33(6):975–86. https://doi.org/10.1002/jbmr.3371.
Lindeman KG, Greenblatt LB, Rourke C, et al. Longitudinal 5-year evaluation of bone density and microarchitecture after Roux-en-Y gastric bypass surgery. J Clin Endocrinol Metab. 2018;103(11):4104–12. https://doi.org/10.1210/jc.2018-01496.
Bredella MA, Greenblatt LB, Eajazi A, et al. Effects of Roux-en-Y gastric bypass and sleeve gastrectomy on bone mineral density and marrow adipose tissue. Bone. 2017;95:85–90. https://doi.org/10.1016/j.bone.2016.11.014.
Vilarrasa N, de Gordejuela AGR, Gómez-Vaquero C, et al. Effect of bariatric surgery on bone mineral density: comparison of gastric bypass and sleeve gastrectomy. Obes Surg. 2013;23(12):2086–91. https://doi.org/10.1007/s11695-013-1016-x.
Nogués X, Goday A, Peña MJ, et al. Pérdida de masa ósea tras gastrectomía tubular: estudio prospectivo comparativo con el bypass gástrico. Cir Esp. 2010;88(2):103–9. https://doi.org/10.1016/j.ciresp.2010.04.008.
Ruiz-Tovar J, Oller I, Priego P, et al. Short- and mid-term changes in bone mineral density after laparoscopic sleeve gastrectomy. Obes Surg. 2013;23(7):861–6. https://doi.org/10.1007/s11695-013-0866-6.
Carrasco F, Ruz M, Rojas P, et al. Changes in bone mineral density, body composition and adiponectin levels in morbidly obese patients after bariatric surgery. Obes Surg. 2009;19(1):41–6. https://doi.org/10.1007/s11695-008-9638-0.
Rousseau C, Jean S, Gamache P, et al. Change in fracture risk and fracture pattern after bariatric surgery: nested case-control study. BMJ. 2016:i3794. https://doi.org/10.1136/bmj.i3794.
Zhang Q, Chen Y, Li J, et al. A meta-analysis of the effects of bariatric surgery on fracture risk: bariatric surgery and fracture. Obes Rev. 2018;19(5):728–36. https://doi.org/10.1111/obr.12665.
obesity_surgery_-_guidelines.pdf.
Carey JJ, Delaney MF, Love TE, et al. DXA-generated Z-scores and T-scores may differ substantially and significantly in young adults. J Clin Densitom. 2007;10(4):351–8. https://doi.org/10.1016/j.jocd.2007.06.001.
Wallace TM, Lety JC, Matthews DR. Use and abuse of HOMA modeling. Diabetes Care. 2004.
Yu EW. Bone metabolism after bariatric surgery. J Bone Miner Res. 2014;29(7):1507–18. https://doi.org/10.1002/jbmr.2226.
Hage MP, El-Hajj Fuleihan G. Bone and mineral metabolism in patients undergoing Roux-en-Y gastric bypass. Osteoporos Int. 2014;25(2):423–39. https://doi.org/10.1007/s00198-013-2480-9.
Tothill P, Laskey MA, Orphanidou CI, et al. Anomalies in dual energy X-ray absorptiometry measurements of total-body bone mineral during weight change using Lunar, Hologic and Norland instruments. Br J Radiol. 1999;72(859):661–9. https://doi.org/10.1259/bjr.72.859.10624323.
Folli F, Sabowitz BN, Schwesinger W, et al. Bariatric surgery and bone disease: from clinical perspective to molecular insights. Int J Obes. 2012;36(11):1373–9. https://doi.org/10.1038/ijo.2012.115.
Brzozowska MM, Sainsbury A, Eisman JA, et al. Bariatric surgery, bone loss, obesity and possible mechanisms: bariatric surgery and bone. Obes Rev. 2013;14(1):52–67. https://doi.org/10.1111/j.1467-789X.2012.01050.x.
Stein EM, Silverberg SJ. Bone loss after bariatric surgery: causes, consequences, and management. Lancet Diab Endocrinol. 2014;2(2):165–74. https://doi.org/10.1016/S2213-8587(13)70183-9.
Campanha-Versiani L, Pereira DAG, Ribeiro-Samora GA, et al. The effect of a muscle weight-bearing and aerobic exercise program on the body composition, muscular strength, biochemical markers, and bone mass of obese patients who have undergone gastric bypass surgery. Obes Surg. 2017;27(8):2129–37. https://doi.org/10.1007/s11695-017-2618-5.
Muschitz C, Kocijan R, Haschka J, et al. The impact of vitamin D, calcium, protein supplementation, and physical exercise on bone metabolism after bariatric surgery: the BABS study. J Bone Miner Res. 2016;31(3):672–82. https://doi.org/10.1002/jbmr.2707.
Wei J-H, Lee W-J, Chong K, et al. High incidence of secondary hyperparathyroidism in bariatric patients: comparing different procedures. Obes Surg. 2018;28(3):798–804. https://doi.org/10.1007/s11695-017-2932-y.
Carrasco F, Basfi-fer K, Rojas P, et al. Changes in bone mineral density after sleeve Gastrectomy or gastric bypass: relationships with variations in vitamin D, ghrelin, and adiponectin levels. Obes Surg. 2014;24(6):877–84. https://doi.org/10.1007/s11695-014-1179-0.
Acknowledgements
We would like to thank the Bordeaux University Hospital for allowing this work. We would also like to thank all of the subjects who participated in this study.
Funding
This research did not receive any specific grant from any funding agency in the public, commercial, or not-for-profit sector.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
ESM1
(PDF 260 kb)
Rights and permissions
About this article
Cite this article
Cadart, O., Degrandi, O., Barnetche, T. et al. Long-Term Effects of Roux-en-Y Gastric Bypass and Sleeve Gastrectomy on Bone Mineral Density: a 4-Year Longitudinal Study. OBES SURG 30, 3317–3325 (2020). https://doi.org/10.1007/s11695-020-04568-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-020-04568-3