Abstract
We aim to review the available literature on obese patients treated with laparoscopic Roux-en-Y gastric bypass (LRYGB) or laparoscopic sleeve gastrectomy (LSG) after failed laparoscopic adjustable gastric banding (LAGB), in order to compare the clinical outcomes of the two methods. A systematic literature search was performed in PubMed, CENTRAL, and Scopus databases, in accordance with the PRISMA guidelines. Eight studies met the inclusion criteria incorporating 635 patients. Both procedures are associated with comparable complications, conversions, mean hospital stay, and weight loss at 6 and 12 months. In the LRYGB group, % excess weight loss (%EWL) and BMI reduction after 24 months were increased. Well-designed, randomized controlled studies, comparing revisional LRYGB and LSG, are necessary to further assess their outcomes.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Obesity is a rising epidemic, and bariatric surgery continues to be the main therapeutic mode for a high rate of sustainable weigh loss [1]. Laparoscopic adjustable gastric banding (LAGB) is one of the most commonly performed procedures for morbid obesity, mainly due to its reversibility and minimal invasiveness. However, LAGB has been associated with high failure rates requiring revisional operation [2–5].
There are three available options to manage the complications of LAGB. Two of those, band removal without replacement and band revision, are associated with poor outcomes [6]. Conversion to an alternative bariatric procedure is another option, and laparoscopic Roux-en-Y gastric bypass (LRYGB) has been proposed as the main operation of choice [7, 8]. Recently laparoscopic sleeve gastrectomy (LSG) has gained increased popularity both as a primary operation for weight reduction [9] and as a revisional procedure after failed LAGB [10, 11]. On the other hand, the conversion to biliopancreatic diversion or duodenal switch is a demanding operation with long mean operative time and higher complication rates [12]. As the number of studies comparing the feasibility of LRYGB and LSG as salvage procedures after failed LAGB increases, it is necessary to examine whether the results between the two techniques are at least equivalent. The purpose of this study is to summarize the existing evidence comparing the surgical outcomes of LRYGB and LSG after failed LAGB.
Materials and Methods
Search Strategy and Articles Selection
The present study was conducted in accordance with the protocol agreed by all authors and the Preferred Reporting Items for Systematic Reviews and Meta-Analyses [13]. A thorough literature search was performed in Pubmed (Medline), Cochrane Central Register of Controlled Studies (CENTRAL), and Socpus (ELSEVIER) databases (last search: December 30, 2016) using the following terms in every possible combination: “failed adjustable gastric band,” “adjustable gastric banding,” “revisional,” “conversion,” “roux-en-y gastric bypass,” “gastric bypass,” “rygb,” “sleeve gastrectomy,” “sg.” Inclusion criteria were (1) original reports with ≥10 patients, (2) written in the English language, (3) published from 1970 to 2016, (4) conducted on human subjects, and (5) reporting outcomes of LRYGB or LSG performed after LAGB on obese patients. Two independent reviewers (DEM, VST) extracted the data from the included studies. Any discrepancies between the investigators about the inclusion or exclusion of studies were discussed with the guarantor author (DZ) in order to include articles that best matched the criteria, until consensus was reached. Moreover, the reference lists of all included articles were reviewed for additional potentially eligible studies. No search for unpublished literature was performed.
Data Extraction
For each eligible study, data were extracted relative to demographics (number of patients, mean age, sex, preoperative body mass index (BMI)), time from LAGB to conversion, along with the intraoperative parameters and outcomes (number of stages; mean operative time; length of hospital stay; conversion rate; intraoperative and postoperative complications; charges; BMI after 6, 12, and 24 months; and % excess weight loss (%EWL) after 6, 12, and 24 months). Moreover, categorical outcomes were 2 × 2 tabulated, referring patients presenting the outcome and patients free of the outcome, separately for LRYGB and LSG groups. Regarding continuous outcomes, we extracted the mean, the standard deviation, and the number of patients. In those cases where standard deviation was not available, it was calculated using the available data.
Statistical Analysis
Regarding the categorical outcomes, the odds ratio (ORs) and 95% confidence interval (CI) were calculated, based on the extracted data, by means of random-effects model (Mantel-Haenszel statistical method), where the number of studies providing data was sufficient. OR <1 denoted an outcome that was more frequent in the LSG group. Continuous outcomes were evaluated by means of weighted mean difference (WMD) with its 95% CI, using random-effects (inverse variance statistical method) models, appropriately to calculate pooled effect estimates. In cases where WMD < 0, values in the LSG group were higher. Between-study heterogeneity was assessed through Cochran Q statistic and by estimating I 2 [14]. A p value of less than 0.05 was set as the threshold indicating a statistically significant result.
In cases where multiple studies analyzed the same population (ie, series from the same hospital), only the larger study or the one with the longest follow-up (if the sample was similar) was included in the meta-analysis.
Quality and Publication Bias Assessment
The Newcastle-Ottawa Quality Assessment Scale (NOS) [15] was used as an assessment tool to evaluate non-RCTs. The scale’s range varies from zero to nine stars, and studies with a score equal to or higher than five were considered to have adequate methodological quality to be included. There were no RCTs in the literature to be included. Two reviewers (DEM, VST) rated the studies independently and final decision was reached by consensus.
The existence of publication bias could not be evaluated using the Egger’s formal statistical test, by the funnel plot method [16], because the number of the included in the analyses studies was not adequate (less than 10). As a result, the power of the test was substantially compromised and was not used [16].
Results
Article Selection and Patient Demographics
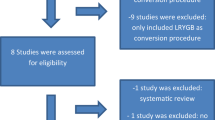
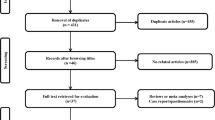
The flow diagram of the search of the literature is shown in Fig. 1 and the Prisma Checklist is shown in Table S1. The characteristics of the included studies are summarized in Table 1. Eight studies were included in the qualitative synthesis [17–24]. The study design was retrospective in all studies and no randomized controlled studies were identified. The included studies were conducted in France [17, 21], the UK [18], Kuwait [20], and USA [19, 22–24] and were published between 2013 and 2015. The LRYGB sample size ranged from 12 to 74 patients, while the LSG ranged from 13 to 72. The total sample size was 635 patients; 355 patients treated with LRYGB, and 280 patients treated with LSG. Pre-conversion mean BMI was >30 kg/m2 in all included patients. Time from LAGB to conversion ranged from 30.6 to 82.5 months.
Indications for Conversion
The indications for conversion are presented in Table 2. The most common indication in both groups was the insufficient weight loss (67.88 and 63.93% for LRYGB and LSG, respectively). The second most common indication was problems related to band (i.e. slippage or erosion) (8.39 and 17.81% for LRYGB and LSG, respectively). In most cases of band erosion, conversion was performed in two stages [17–20, 22]. The band was removed at the first stage and the revision was planned at a later step. Other common indications were persistent dysphagia (LRYGB 4.74%; LSG 3.2%), gastric pouch dilation (LRYGB 5.47%; LSG 3.65%), and intractable gastroesophageal reflux (LRYGB 2.19%; LSG 1.83%).
Number of Stages
The number of stages of the bariatric procedures performed is presented in Table 3 . In five studies [18–22], both one-stage and two-stage procedures were performed. In one study [24], all patients were treated with one-stage operations, while in one study [17], two-stage surgery was performed in all patients. In total, 63.3% of the patients reported in the included studies were treated with one-stage procedure and 36.7% patients with two-stage.
Mean Operative Time and Mean Hospital Stay
Mean operative time ranged from 142 to 224 min for the LRYGB and from 108.4 to 156 min for the LSG (Table 3). According to our two-arm analysis, mean operative time was significantly increased in the RYGB group (WMD 37.67 [95% CI: 6.54, 68.80]; p = 0.02). The mean hospital stay was 3 days for LRYGB and ranged from 2 to 3 days for the LSG group (Table 3). No significant difference was reported (WMD −0.35 [95% CI −1.02, 0.32]; p = 0.31).
Conversions and Mortality
Conversion rate in the LRYGB group was 0–1.9% and in LSG group, it was 0%, as shown in Table 3. No deaths were reported.
%EWL
The results of meta-analysis of %EWL for 6, 12, and 24 months are shown in Table 4 . Three studies [17, 19, 20] assessed the %EWL after 6 months, and no significant difference was found (WMD −3.00 [95% CI −14.63, 8.64]; p = 0.61). Six studies [17–20, 22, 23] reported on %EWL after 12 months, and it was significantly greater in the LRYGB group (WMD 5.95 [95% CI −1.84, 13.75]; p = 0.13) (Fig. S1). Four studies assessed the %EWL after 24 months, and it was significantly greater in the LRYGB group (WMD 11.19 [95% CI 0.97, 21.42]; p = 0.03) (Fig. 2).
Forest plot describing the differences in a % excess weight loss (%EWL) after 24 months, b BMI after 24 months. a %EWL after 24 months was significantly greater in laparoscopic Roux-en-Y gastric bypass. b BMI after 24 months was significantly greater in laparoscopic sleeve gastrectomy indicating that BMI reduction was greater in the laparoscopic Roux-en-Y gastric bypass group
Post-Operative BMI
The results of meta-analysis of BMI after 6, 12, and 24 months are shown in Table 4. Two studies assessed the BMI after 6 months and no significant difference was found (WMD −0.46 [95% CI −5.65, 4.73]; p = 0.86). Three studies reported on BMI after 12 months and there was no significant difference between the two groups (WMD −1.97 [95% CI −6.24, 2.30]; p = 0.37) (Fig. S1). Three studies assessed the BMI after 24 months and it was significantly lower in the LRYGB group (WMD −3.61 [95% CI −5.16, −2.05]; p < 0.00001) (Fig. 2).
Complications
The results of meta-analysis on complications are shown in Table 4 and figures. Six studies [17, 18, 20–22, 24] reported on leaks and there was no significant difference between the LRYGB and LSG groups (OR 1.36 [95% CI 0.43, 4.31]; p = 0.60) (Fig. S2). Moreover, no significant difference was found regarding post-operative bleeding (OR 2.62 [95% CI 0.51, 13.54]; p = 0.25) (Fig. S2), abscess (OR 2.43 [95% CI 0.28, 20.92]; p = 0.42), incisional hernias (OR 1.10 [95% CI 0.09, 14.09]; p = 0.94), and internal hernias (OR 2.12 [95% CI 0.23, 19.78]; p = 0.51). Furthermore, no significant difference between the two groups was reported regarding intestinal obstruction (OR 3.48 [95% CI 0.59, 20.36]; p = 0.17), marginal ulcer (OR 1.97 [95% CI 0.20, 19.43]; p = 0.56), stenosis (OR 0.23 [95% CI 0.04, 1.46]; p = 0.12), and chronic abdominal pain (OR 3.27 [95% CI 0.37, 28.80]; p = 0.29).
Quality and Publication Bias
Quality assessment of each study according to Newcastle-Ottawa Assessment Scale is shown in Table 1 . Publication bias was not calculated for the outcomes because of the small number of the studies that were included.
Discussion
This meta-analysis identified eight articles comparing LRYGB and LSG as two alternative revisional bariatric procedures performed after failed LAGB, measuring patients’ outcomes, and published between 2013 and 2015. No similar meta-analysis was identified through literature search. Only one similar systematic review [25] published in 2013 was identified which did not include quantitative analysis. Since 2013, the number of comparative studies, along with the need for a meta-analysis has increased. The articles included in this study bring us closer to the implementation of either method with improved standards of safety and efficiency.
In the past, the LAGB was one of the most commonly performed bariatric operations. However, it has been associated with high rates of complications requiring revision [2–5]. According to our findings, insufficient weight loss was the most common indication for revisional surgery (67.88 and 63.93% for LRYGB and LSG, respectively). This trend is in accordance with previous findings [25] and the reported high pre-conversion mean BMI (>40 in all studies). Problems related to band, such as slippage and erosion, gastric pouch dilation, and persistent dysphagia, were the main complications requiring salvage operation. An interesting point is that one study [17] reported on four cases with intractable gastroesophageal reflux that underwent LSG as revisional procedure. A possible explanation may be the multiform, instead of just restrictive, mechanism of action that the authors of that study [17] attribute to LSG. There is no comment made by the authors [17] regarding the effect of LSG on those patients. In the same study [17], four patients underwent revisional surgery due to catheter disconnection. In those cases, the catheter was not reconnected possibly due to the coexistence of additional indications for conversion.
The present study demonstrates that both LRYGB and LSG are well-tolerated and safe surgical approaches. According to our analysis, complication rates were similar between the LRYGB and LSG approaches. Leaks and hemorrhage are the main risks of bariatric procedures, due to the long stapled lines and gastrointestinal anastomosis. According to our findings, the incidence of leaks and hemorrhage was comparable between the two groups. Mean operative time was significantly greater in the LRYGB group. Mean hospital stay was similar between the LRYGB and LSG groups. The rate of conversions to open was low and no deaths were reported in either group.
It is still debatable whether the option of one-stage or two-stage revisional procedure is better. According to a recent meta-analysis [26], both options have similar rates of complications, morbidity, and mortality. The majority of the patients reported in the included studies were treated with one-stage procedure.
Since the main indication for revision is insufficient weight loss, it has been debatable whether performing LSG, which is a restrictive procedure, after a failed restrictive procedure would be effective in reducing weight. According to our findings, both procedures demonstrate similar %EWL and BMI reduction in 6 and 12 months. This is reflective of the additional hormonal effects, except for the restrictive principles, of sleeve gastrectomy [27]. Moreover, those findings are in accordance with previous reported outcomes of both procedures as primary operations [28]. However, %EWL and BMI reduction were significantly greater in the RYGB group after 24 months.
This meta-analysis demonstrates the need for additional studies comparing RYGB and LSG as revisional procedures after failed LAGB. Ideally, these would be randomized controlled studies, with prospective design and longer follow-up. For rare events, such as conversions and mortality, a large sample is needed. The studies included offer specific linkage to patient outcomes, complications, and weight loss.
The limitations of this systematic review and meta-analysis reflect the limitations of the studies included. All studies included were retrospective. There were no randomized controlled studies between the comparative studies included, thus posing a certain limitation in this study.
On the other hand, the strengths of this study are (1) the clear data-extraction protocol, (2) the well-specified inclusion-exclusion criteria, (3) the search in three different databases, (4) the quality assessment of the included studies, and (5) the detailed presentation of the results of data-extraction and analysis.
Conclusion
This meta-analysis identified eight unique peer-reviewed studies of revisional LRYGB and LSG procedures after failed LAGB with patient outcomes data. These studies suggest that LRYGB and LSG are associated with comparable clinical outcomes, mean hospital stay, and complications. Mean operative time was increased in the LRYGB group. There was no significant difference between the two groups regarding the %EWL and BMI reduction after 6 and 12 months. However, %EWL and BMI reduction were greater after 24 months in the LRYGB group. These results should be interpreted with caution due to the lack of randomized controlled studies. Future studies with greater clarity in significant outcomes and complications, such as the %EWL and complications, are necessary to demonstrate the differences in efficacy between LRYGB and LSG after LAGB.
References
Colquit JL, Picot J, Loveman E, et al. Surgery for obesity. Cochrane Database Syst Rev. 2009;2:CD003641.
Chapman AE, Kiroff G, Game P, et al. Laparoscopic adjustable gastric banding in the treatment of obesity: a systematic literature review. Surgery. 2004;3:326–51.
Suter M, Calmes JM, Paroz A, et al. A 10-year experience with laparoscopic gastric banding for morbid obesity: high long-term complication and failure rates. Obes Surg. 2006;16:829–35.
Himpens J, Cadière GB, Bazi M, et al. Long-term outcomes of laparoscopic adjustable gastric banding. Arch Surg. 2011;7:802–7.
Jennings NA, Boyle M, Mahawar K, et al. Revisional laparoscopic Roux-en-Y gastric bypass following failed laparoscopic adjustable gastric banding. Obes Surg. 2013;23:947–52.
Müller MK, Attigah N, Wildi S, et al. High secondary failure rate of rebanding after failed gastric banding. Surg Endosc. 2008;22:448–53.
Mognol P, Chosidow D, Marmuse JP. Laparoscopic conversion of laparoscopic gastric banding to Roux-en-Y gastric bypass: a review of 70 patients. Obes Surg. 2004;14:1349–53.
Ardestani A, Lautz DB, Tavakkolizadeh A. Band revision versus Roux-en-Y gastric bypass conversion as salvage operation after laparoscopic adjustable gastric banding. Surg Obes Relat Dis. 2011;7:33–7.
Magouliotis DE, Tasiopoulou VS, Sioka E, et al. Robotic versus laparoscopic sleeve gastrectomy for morbid obesity: a systematic review and meta-analysis. Obes Surg. 2017;27:245. doi:10.1007/s11695-016-2444-1.
Patel S, Szomstein S, Rosenthal RJ. Reasons and outcomes of reoperative bariatric surgery for failed and complicated procedures (excluding adjustable gastric banding). Obes Surg. 2011;21:1209–19.
Patel S, Eckstein J, Acholonu E, et al. Reasons and outcomes of laparoscopic revisional surgery after laparoscopic adjustable gastric banding for morbid obesity. Surg Obes Relat Dis. 2010;6:391–8.
Topart P, Becouarn G, Ritz P. Biliopancreatic diversion with duodenal switch or gastric bypass for failed gastric banding: retrospective study from two institutions with preliminary results. Surg Obes Relat Dis. 2007;3(5):521–5.
Liberati A, Altman DG, Tetzlaff J, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. PLoS Med. 2009;6:e1000100.
Higgins JPT, Green S. Cochrane handbook for systematic reviews of interventions version 5.1.0 [updated March 2011]. The Cochrane Collaboration. 2011 Available from www.cochrane-handbook.org
Stang A. Critical evaluation of the Newcastle-Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur J Epidemiol. 2010;25:603–5.
Egger M, Smith DG, Schneider M, et al. Bias in meta-analysis detected by a simple, graphical test. BMJ. 1997;315(7109):629–34.
Carandina S, Maldonado PS, Tabbara M, et al. Two-step conversion surgery after failed laparoscopic adjustable gastric banding. Comparison between laparoscopic Roux-en-Y gastric bypass and laparoscopic gastric sleeve. Surg for Obes Relat Dis. (2014) 00–00. doi:10.1016/j.soard.2014.03.017.
Carr WRJ, Jennings NA, Boyle M, et al. A retrospective comparison of early results of conversion of failed gastric banding to sleeve gastrectomy or gastric bypass. Surg Obes Relat Dis. (2014) 00–00. doi:10.1016/j.soard.2014.07.021
Gonzalez-Heredia R, Masrur M, Patton K, et al. Revisions after failed gastric band: sleeve gastrectomy and Roux-en-Y gastric bypass. Surg Endosc. 2015;29:2533. doi:10.1007/s00464-014-3995-7.
Khoursheed M, Al-Bader I, Mouzannar A, et al. Sleeve gastrectomy or gastric bypass as revisional bariatric procedures: retrospective evaluation of outcomes. Surg Endosc. 2013; doi:10.1007/s00464-013-3038-9.
Liu KH, Diana M, Vix M, et al. Revisional surgery after failed adjustable gastric banding: institutional experience with 90 consecutive cases. Surg Endosc. 2013; doi:10.1007/s00464-013-3056-7.
Marin-Perez P, Betancourt A, Lamota M, et al. Outcomes after laparoscopic conversion of failed adjustable gastric banding to sleeve gastrectomy or Roux-en-Y gastric bypass. BJS. 2014;101:254–60. doi:10.1002/bjs.9344.
Moon RC, Teixeira AF, Jawad MA. Conversion of failed laparoscopic adjustable gastric banding: sleeve gastrectomy or Roux-en-Y gastric bypass? Surg Obes Relat Dis. 2013;9:901–7. doi:10.1016/j.soard.2013.04.003.
Yeung L, Durkan B, Barrett A, et al. Single-stage revision from gastric band to gastric bypass or sleeve gastrectomy: 6- and 12-month outcomes. Surg Endosc. 2015; doi:10.1007/s00464-015-4498-x.
Coblijn UK, Verveld CJ, van Wagensveld BA, et al. Laparoscopic Roux-en-Y gastric bypass or laparoscopic sleeve gastrectomy as revisional procedure after adjustable gastric band—a systematic review. Obes Surg. 2013;23:1899–914. doi:10.1007/s11695-013-1058-0.
Dang JT, Switzer NJ, Wu J, et al. Gastric band removal in revisional bariatric surgery, one-step versus two-step: a systematic review and meta-analysis. Obes Surg. 2016; doi:10.1007/s11695-016-2082-7.
Cohen R, Uzzan B, Bihan H, et al. Ghrelin levels and sleeve gastrectomy in super-superobesity. Obes Surg. 2005;15(10):1501–2.
Karamanakos SN, Vagenas K, Kalfarentzos F, et al. Weight loss, appetite suppression, and changes in fasting and postprandial ghrelin and peptide-YY levels after Roux-en-Y gastric bypass and sleeve gastrectomy: a prospective, double blind study. Ann Surg. 2008;247(3):401–7.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The authors declare that they have no conflicts of interest.
Ethical Approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed Consent
Does not apply.
Rights and permissions
About this article
Cite this article
Magouliotis, D.E., Tasiopoulou, V.S., Svokos, A.A. et al. Roux-En-Y Gastric Bypass versus Sleeve Gastrectomy as Revisional Procedure after Adjustable Gastric Band: a Systematic Review and Meta-Analysis. OBES SURG 27, 1365–1373 (2017). https://doi.org/10.1007/s11695-017-2644-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-017-2644-3