Abstract
The objectives of this study were: (1) to determine carbon stock changes in the soil and forest floor of black pine (Pinus nigra subsp. pallasiana) plantations in Turkey; (2) to determine the effects of thinning on annual carbon accumulation in both; and, (3) to investigate relationships between annual carbon storage in soil and forest floor and stand characteristics of black pine. Samples were taken in 90 plots from stands at the pole (dbh = 11.0–19.9 cm) and sawlog (dbh = 20.0–35.9 cm) stages. Carbon analyses of soil and forest floor samples showed that in unthinned plantations significant organic carbon was sequestered an average of 1.47 Mg ha−1 a−1 in the soil and 0.20 Mg ha−1 a−1 in the forest floor. Organic carbon sequestered annually in the soil was significantly lower in thinned than in unthinned stands, while differences in the forest floor were insignificant. There were positive correlations between carbon sequestered in the soil and stand age, average DBH, mean stand height, basal area, and site index. Carbon sequestered the forest floor increased with basal area. As a result, carbon sequestered in the soil should not be ignored in greenhouse gas inventories because carbon stored belowground was approximately 70% of the aboveground pool.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Forest ecosystems are important carbon sinks that can mediate carbon dioxide emissions because of their biomass and the carbon stored in soils (Çömez 2012). In terrestrial ecosystems, more than 80% of the carbon bound above the soil and more than 70% of the entire soil organic carbon are tied up in the forest ecosystem (Jandl et al. 2007).
Under the Kyoto Protocol to address global climate change, countries submitted annual national inventory reports on greenhouse gas emissions from energy, transportation, wastes, agriculture, land use-land use change and forestry, and the amount of carbon bound from the atmosphere to the secretariat of the United Nations Climate Change Convention (Tolunay 2011; Güner and Makineci 2017).
There are two methodologies to determine the amount of atmospheric carbon sequestered in forest ecosystems. The first is stock change, while the second is the gain–loss method. The first relies on the carbon change between two periods; the second is based on the amount of carbon that accumulates in a year (IPCC 2003).
In greenhouse gas inventories, it is assumed that soil carbon remains unchanged in forested areas regardless of methods used (IPCC 2006). However, soil and forest floor carbon pools are known to change depending on stand development and silvicultural operations (Makineci 2005; Güner and Çömez 2017). It may be possible to include the carbon bound in forest soils to the national inventory as a gain with further scientific investigation at the national level, meaning that carbon storage increases. Nevertheless, no gain is envisaged in the guideline-based calculations.
Carbon is stored in below and aboveground parts of the forest ecosystem (trees and understory, forest floor and dead wood and in the soil). Several studies have determined carbon stocks in forest ecosystems (Laclau 2003; Peichl and Arain 2006; Yimer et al. 2006; Schulp et al. 2008; Makineci et al. 2015; Lee et al. 2016, 2018; Goussanou et al. 2018; Payne et al. 2019). However, there are limited studies on the amount of annual carbon stored in the soil and forest floor (Lettens et al. 2005; Prietzel et al. 2006; Miegroet et al. 2007; Berg et al. 2007; Black et al. 2009; Fonseca et al. 2011; Bárcena et al. 2014; Grüneberg et al. 2014; Wellbrock et al. 2017; Güner and Makineci 2017; Jiang et al. 2018). There is some research on the effects of thinning on soil carbon stocks by various tree species (Tolunay 1997; Tufekcioglu et al. 2005; Jurgensen et al. 2012; Ruiz-Peinado et al. 2013; Bravo-Oviedo et al. 2015; Ruiz-Peinado et al. 2016), however no data have been published related to black pine (Pinus nigra subsp. pallasiana (Lamb.) Holmboe).
There have been studies in black pine plantations in Turkey to determine the organic carbon stock stored in the soil (Karatepe 2004; Sevgi et al. 2011; Korkanç 2014; Polat et al. 2014; Güner et al. 2016; Güner and Çömez 2017) and in the forest floor (Karaöz 1993; Karatepe 2004; Sevgi et al. 2011; Güner and Çömez 2017). However, these studies focused on determination of the existing stock and not annual storage. This study looks at the annual organic carbon accumulated in the soil and forest floor.
This study was carried out: (1) to determine carbon stock changes; (2) to determine the effects of thinning on annual carbon accumulation in soil and forest floor; and, (3) to investigate the relationships between annual carbon storage and stand characteristics of black pine plantations. The findings will contribute to carbon-focused forest management and to the improvement of greenhouse gas inventories.
Materials and methods
Study area
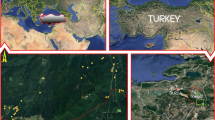
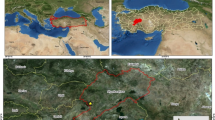
The research was conducted in an area between 38° 21ʹ–41° 31ʹ N and 27° 17ʹ–33° 48ʹ E, a transition zone from Western Inner Anatolia to the Black Sea, Marmara and Inner Aegean regions (Fig. 1). Dacite, rhyolite, basalt, andesite, granite, volcanic tuff, agglomerate, breccia, quartzite, mica schist, calcareous and serpentine are the common bedrock while the most common soil types are cambisols and luvisols, according to IUSS Working Group WRB (2015) (Güner et al. 2016; Güner and Çömez 2017).
Climatic data are from the nearest meteorological stations. Mean annual temperatures varies from 8.9 to 14.4 °C while annual precipitation ranges from 374 mm to 796 mm. Overall climate ranges from semi-arid to humid.
Table 1 provides stand and site characteristics of the sampling plots. Some 72% of the trees in the plots were at the pole stage (dbh = 11.0–19.9 cm), while 28% were sawlogs (dbh = 20.0–35.9 cm).
Experimental design
The first sampling was carried out on 67 plots in 2007 and 2017 to determine carbon sequestration. To extend the representation area, another sampling was performed in 23 additional plots from different geographical regions in 2011 and 2017. Therefore, data was obtained from two sampling periods, 2007–2017 and 2011–2017, 90 plots in total.
Plots were chosen for different aspects, elevation, slope position, inclination and stand development. Thirty-five of the 90 plots were moderately thinned (20–30% of the basal area) in different years between the sampling periods by the local forest authority. However, the timing and severity of these interventions could not be firmly determined due to the lack of data records.
Sampling procedure
The 100 to 200 m2 plots were established were square or rectangular in shape. During the initial sampling, diameter at breast height, tree heights and age of five representative trees in canopy were measured.
Slope position was calculated as a percentage in relation to the length of the whole slope. Aspect was recorded as azimuth (Q) measured from true north and converted to a radiation index using the following:
where RI is radiation index (dimensionless); Q is azimuth (degree)
This assigns a value of zero to an area that orients in a north-northeast direction, (typically the coolest and wettest orientation), and a value of 1 to warmer, drier, south-southwest facing slopes (Moisen and Frescino 2002; Aertsen et al. 2010).
During the two sampling periods in each area, mineral horizons were identified in 1-m deep soil pits at the same point from which soil samples were taken (90 sampling plots × 2 periods × 4–5 horizons = 840 soil samples). In the initial sampling in 2007 and 2011, soil samples were taken with steel cylinders in two sets, one set was used to calculate bulk density and coarse fragments while the second set was used for analysis. During the second sampling in 2017, disturbed soil samples were collected from the same points only for analysis using bulk densities calculated from the initial sampling. In years when soil sampling was carried out, forest floor samples were collected from a 25 × 25 cm area at different representative points (90 sampling plots × 2 periods × 4 repetitions = 720 forest floor samples) without considering the forest floor layers of leaf, fermentation and humus. All weighting and analyses of forest floor samples were performed separately on each replicate.
Analysis
Air-dried soil samples were crushed by mortar and pestle and sieved through 2-mm screens. The volume of the stones remaining on the screen was determined. Moisture was measured at 105 °C and samples classified according to particle size with a hydrometer; total carbonate was measured with a Scheibler calcimeter, and organic carbon determined by the Walkley–Black wet oxidation method (Carter and Gregorich 2008). Forest floor samples were dried at 65 °C for 24 h, weighed and ground for analysis. Carbon fractions were determined in the LECO CNH TruSpec elemental analyser (Leco Corporation, St. Joseph, MI, USA).
Data analysis
Equation 2 was used to determine soil organic carbon.
where SOC is soil organic carbon (Mg ha−1), i and j represent mineral soil horizons, BD is bulk density (< 2 mm) (g/L), HT is horizon thickness (mm), CF is carbon fraction of the soil horizon (%).
Oven-dried forest floor weights for a 25 × 25 cm area were extrapolated to one hectare and multiplied by their carbon concentration to calculate carbon stock.
Carbon stock changes in the soil and forest floor were evaluated with the one-sample t test statistics using Eqs. 3 and 4 (Hunt 1990). Annual carbon changes in the soil and forest floor were converted to relative values and analysed.
where RCA is the relative carbon accumulation, S the soil, FF the forest floor, ln the natural logarithm, SOC the soil organic carbon stock, FFC the forest floor carbon stock, T2−T1 the time between two measurements (2007–2017 = 10 years or 2011–2017 = 6 years)
The differences in carbon sequestered in the forest floor and the soil between thinned and unthinned stands were assessed by analysis of variance (ANOVA). Duncan’s test compared the means of carbon sequestered. The relationship between annual organic carbon accumulated in the forest floor and soil under unthinned plantations and stand characteristics were assessed by correlation analysis. Results with a value of α = 0.05 were considered statistically different. The SPSS package program was used for statistical analysis (SPSS v.22.0® 2015).
Results and discussion
Carbon sequestration in forest floor and soil
Forest floor mass was 13.1–19.0 Mg ha−1 in the sample plots in 2007–2017 while it was 23.4–26.0 Mg ha−1 in 2011–2017. The average forest floor sequestration rate was 0.5 Mg ha−1 a−1 (Table 2). Tolunay and Çömez (2008) reported that forest floor rates ranged from 6.2 to 82.4 Mg ha−1 (27.9 Mg ha−1) in black pine plantations in Turkey. Our results are consistent with their findings.
There was an overall increase in soil organic carbon in the plots in 2017 compared to 2007 and 2011 (Table 3). Güner and Makineci (2017) also reported average soil organic carbon contents in Scots pine stands were 0.2–4.4% in 2003, and 0.6–4.9% in 2013.
Carbon concentration in the forest floor samples were found to 49.9–48.6% in 2007–2017 while they were found to 48.1–46.6% in 2011–2017. There was a slight decrease in carbon concentrations over the periods (Table 3). Güner and Makineci (2017) likewise found a decline in forest floor carbon concentrations from 2003 (42.8–52.8%) to 2013 (38.7–51.6%) in natural Scots pine forests in Turkey. The decline in carbon levels over time was due to the decomposition of the forest floor. Studies on coniferous species in Turkey showed that forest floor carbon was 36.8–50.4% for Scots pine (Çömez 2012), 36.8–45.9% for cedar (Karataş et al. 2017), 36.1–44.1% for maritime pine (Tolunay et al. 2017) and 35.6–45.8% fostone pine (Tolunay et al. 2017). The results in this study are consistent with the results on Scots pine, whereas the cedar, maritime pine and stone pine results were higher. Forest floor carbon content varies, depending on not only the species, but also site productivity and degree of decomposition of the forest floor. Furthermore, forest floor dynamics of stands at a rapid development stage, i.e., from young to mature, may be different from those of over mature stands (Yildiz et al. 2011). Therefore, forest floor characteristics of stands at a relatively young stage may be expected to change over 10 years. On the other hand, considering the turnover of forest floor, the measurement of the carbon pool corresponds to a comparison between pools at a given time due to the fact that some of the forest floor carbon is transferred to the soil. However, the carbon dynamics of these ecosystems cannot be fully understood only through the comparison between carbon pools. For this reason, an accurate assessment can be made if the input and output dynamics of these pools are considered.
Soil carbon stock in the unthinned plots was 85.84–85.96 Mg ha−1 for 2007–2017 while it was 61.08–84.11 Mg ha−1 for 2011–2017. Annual carbon sequestration was 1.47 Mg ha−1 (Table 4). The amount of annual organic carbon sequestrated was statistically significant (P < 0.001). In a review of black pine plantations in Turkey, soil organic carbon ranged from 9.3 to 174.6 Mg ha−1, with a mean of 65.1 Mg ha−1 (Tolunay and Çömez 2008). The soil organic carbon in this study was in the similar range. There are several studies that report on soil organic carbon (Berger et al. 2002; Korkanç 2014; Makineci et al. 2015). However, their results widely vary, possibly due to differences in the depth and bulk density of the soils. In a study to determine annual rates of carbon sequestration in natural Scots pine forests in Turkey, Güner and Makineci (2017) reported a value of 2.88 Mg ha−1 a−1. The value was twice that of our findings. This difference is may be due to possible differences between the two species and in the way the forests were established. In a study of second generation humid tropical forests in Costa Rica, the rate of soil carbon sequestration was 1.1 Mg ha−1 a−1 (Fonseca et al. 2011). In two different Scots pine ecosystems in Germany, soil carbon sequestration rates were 0.2 and 0.4 Mg ha−1 a−1 (Prietzel et al. 2006). Moreover, the studies conducted in Germany demonstrated that soil carbon sequestration was 0.4 Mg ha−1 a−1 in the first 30-cm layer (Grüneberg et al. 2014; Wellbrock et al. 2017). Lettens et al. (2005) reported that the annual rate of soil carbon sequestration was 1.0 Mg ha−1 in Belgium coniferous forests. The annual carbon sequestration rates were 2.2–2.5 Mg ha−1 in upper 30-cm soil layer in a Sitka spruce forest in Ireland (Black et al. 2009). In a study conducted in spruce-fir forests in southeastern United States, annual carbon sequestration rates were 1.0–1.4 Mg ha−1 in the top 50-cm soil layer (Miegroet et al. 2007). In this study, soil organic carbon sequestration rates are, overall, consistent with those of the abovementioned studies. However, soil organic carbon sequestration is highly variable. For example, it increases initially with the wastes after silvicultural thinnings, whereas it may decrease depending on the decline in litterfall and amount of fine roots. In addition, soil and litter carbon stocks may increase after stand closure until a balance is reached, while the annual rate of sequestration may decrease as the stand ages. Eker et al. (2013) reported that the rate of forest floor carbon sequestration decreased from the seedling stage to the thicket stage in natural brutian pine forests, and then increased, reaching a maximum level during the polewood stage; litterfall and rates of decomposition were balanced during the over mature stage.
Research on growth models for black pine plantations in Turkey, the average increment was 5.6 m3 ha−1 in a stand with a mean age of 35 years. This was also the result in this research with a moderate density (792 trees ha−1) and average site class II (Yavuz et al. 2004). In another study to determine carbon stocks in black pine plantations, the biomass conversion and expansion factor (BCEF) was 0.613 Mg m−3, root to shoot ratio 0.179, and weighted carbon of total tree mass was 53.8% (Güner and Çömez 2017). According to these results, the annual rate of carbon sequestration in aboveground and belowground biomass is 2.17 Mg ha−1 in black pine plantations. Therefore, soil carbon sequestered in unthinned plantations was approximately 70% of the annual carbon sequestered. This suggests that carbon accumulation in the soil should not be ignored in greenhouse gas inventories.
Forest floor carbon stock was 6.4–9.1 Mg ha−1 for 2007–2017 and 11.3–12.0 Mg ha−1 for 2011–2017 (Table 4). In the black pine plantations, the amount of carbon that accumulated in the forest floor was 0.20 Mg ha−1 a−1, which is statistically significant (P < 0.001). Berg et al. (2007) also reported a similar carbon sequestration value for forest floor to our result, with a mean of 0.18 Mg ha−1 a−1 in Swedish forests. Bárcena et al. (2014) found that annual carbon sequestration rate by forest floors in northern European forests was 0.35 Mg ha−1 a−1 for coniferous forests, and 0.10 Mg ha−1 a−1 for broadleaved and mixed forest floors after afforestation. Our results on the annual rate of forest floor carbon sequestration are consistent with those of the above studies. Annual rate of forest floor carbon sequestration was 0.02 Mg ha−1 a−1 in natural Scots pine forests in Turkey, which was statistically insignificant (Güner and Makineci 2017). This was possible because forest floor decomposition in the ecosystem might be at an optimum level. The difference between these two studies in Turkey may be the result of differences between species, habitats and establishment methods.
Effects of thinning on carbon sequestration
Thinning of black pine plantations did not have a significant effect on forest floor mass and carbon stock but significantly affected soil carbon. The rates of soil carbon sequestration in thinned plots were lower than in unthinned ones (Fig. 2). In other words, silvicultural thinnings decreased the annual rate of soil carbon sequestration. This may be the result of living biomass decline after thinning. In fact, carbon inputs into the soil are primarily through litterfall (Pausas 1997) and fine root decomposition (Berg and McClaugherty 2003). However, research on this topic is variable. For example, there was no difference in soil and forest floor characteristics 5 years after thinning at varying degrees in Scots pine stands at the young stage (Tolunay 1997). It has also been reported that thinning operations at varying degrees in Scots pine plantations did not affect organic and mineral soil carbon stock (Ruiz-Peinado et al. 2016). Similarly, in a study in Scots pine populations in southern Europe, light and moderate thinning did not affect soil and forest floor carbon stock (Bravo-Oviedo et al. 2015). In oriental beech stands at thicket stage, thinning did not affect soil organic carbon concentrations (Tufekcioglu et al. 2005). In red pine (Pinus resinosa Ait.) and broadleaved stands, thinning operations at varying degrees did not affect the upper 30 cm soil layer and forest floor carbon stocks (Jurgensen et al. 2012). Light and heavy thinning of maritime pine stands did not affect organic and mineral soil carbon sequestration rates compared tocontrol plots (Ruiz-Peinado et al. 2013). However, in a study by Makineci (2005) in sessile oak (Quercus petraea (Matt.) Liebl.) coppice forests, soil organic carbon levels increased with the intensity of thinning operations 8 years later, while total forest floor carbon sequestration decreased. Besides, he concluded that soil organic carbon increased because the organic matter was carried deep into the soil from the forest floor. In addition, the author observed that the upper portions of the better developed herbaceous cover turned into litter after thinning, and attributed the decrease in the litter mass to the increased litter decomposition after thinning.
Relationship between carbon sequestration and stand properties
Basal area was positively correlated with forest floor mass and the amount of carbon sequestration in unthinned plantations (Table 5). This is one of the most important indicators of biomass in a unit area. As basal area increased, litterfall and annual forest floor carbon sequestered also increased. Clark et al. (2001) reported a similar relationship between aboveground biomass and annual litterfall in tropical forests. A significant relationship between stand basal area and litterfall was reported for brutian pine forests in Turkey (Erkan et al. 2018). In a study of natural Scot pine forests, basal area and site index explained 67% of the variation in litterfall (Çömez et al. 2019).
Soil organic carbon sequestration had a positive relationship with stand age, mean stand diameter and height, site index and basal area, while it was negatively related with the number of trees (Table 5). With regards to these relationships, as the stand ages, competition between the trees and the number of trees after thinning decreases. Starting from the establishment of a plantation, basal area, average stand diameter, and height increase as the stand ages, and also results in an increase in stand biomass. Aboveground and belowground biomass increases also lead to higher litterfall and greater decomposition of fine roots, which will increase soil organic carbon. Likewise, in eastern cottonwood (Populus deltoides Bartr. ex Marsh) plantations, soil organic carbon stock increased with tree age upper 30-cm layer was 0.75 Mg ha−1 a−1 in 7-year old stands, while it was 1.81 Mg ha−1 a−1 in 11-year old stands (Arora et al. 2014). In a study in second generation humid tropical forests in Costa Rica, a positive relationship was found between soil carbon accumulation and stand age (Fonseca et al. 2011). The positive relationship between site index and annual soil organic carbon accumulation was due to faster growth rates on fertile habitats, and thus there was more organic matter input into the soil and organic matter decomposition was close to the optimum level.
Conclusions
Significant amounts of carbon are stored in the soil and forest floor in the unthinned forests. These sequestrations should be included in greenhouse gas inventories; otherwise, carbon stocks will be underestimated.
Thinning had a considerable effect on soil organic carbon sequestered. Therefore, heavy thinnings should be avoided in forest management plans in which carbon sequestration is an objective, and the intervals between the silvicultural thinnings should be extended.
As the stand biomass increased, soil and forest floor organic carbon sequestration also increased. Thus, areas with the best habitat conditions should a priority for plantation establishment for the purpose of carbon sequestration.
Under this scope, there is a need for further research regarding the effects on the carbon budget and changes in forest ecosystems and forestry practices on carbon management in the ecosystem.
References
Aertsen W, Kınt V, Orshoven J, Özkan K, Muys B (2010) Comparison and ranking of different modelling techniques for prediction of site index in Mediterranean mountain forests. Ecol Model 221:1119–1130. https://doi.org/10.1016/j.ecolmodel.2010.01.007
Arora G, Chaturvedi S, Kaushal R, Nain A, Tewari S, Alam NM, Chaturvedi OP (2014) Growth, biomass, carbon stocks, and sequestration in an age series of Populus deltoides plantations in Taria region of central Himalaya. Turk J Agric For 38:550–560. https://doi.org/10.3906/tar-1307-94
Bárcena TG, Kiaer LP, Vesterdal L, Stefánsdóttır HM, Gundersen P, Sigurdsson BD (2014) Soil carbon stock change following afforestation in Northern Europe: a meta-analysis. Glob Change Biol 20(8):2393–2405. https://doi.org/10.1111/gcb.12576
Berg B, McClaugherty C (2003) Plant litter decomposition, humus formation, carbon sequestration. Springer, Berlin, p 286
Berg B, Gundersen P, Akselsson C, Johansson MB, Nilsson A, Vesterdal L (2007) Carbon sequestration rates in Swedish forest soils—a comparison of three approaches. Silva Fennica 41(3):541–558
Berger TW, Neubauer C, Glatzel G (2002) Factors controlling soil carbon and nitrogen stores in pure stands of Norway spruce (Picea abies) and mixed species stands in Austria. For Ecol Manag 159:3–14. https://doi.org/10.1016/S0378-1127(01)00705-8
Black K, Byrne KA, Mencuccini M, Tobin B, Nieuwenhuis M, Reidy B, Bolger T, Saiz G, Green C, Farrell ET, Osborne B (2009) Carbon stock and stock changes across a Sitka spruce chronosequence on surface-water gley soils. Forestry 82(3):255–272. https://doi.org/10.1093/forestry/cpp005
Bravo-Oviedo A, Ruiz-Peinado R, Modrego P, Alonso R, Montero G (2015) Forest thinning impact on carbon stock and soil condition in Southern European populations of P. sylvestris L. For Ecol Manag 357:259–267. https://doi.org/10.1016/j.foreco.2015.08.005
Carter MR, Gregorich EG (2008) Soil sampling and methods of analysis. CRC Press, Boca Raton, p 1224
Clark DA, Brown S, Kicklighter DW, Chambers JQ, Thomlinson JR, Ni J, Holland EA (2001) Net primary production in tropical forests: an evaluation and synthesis of existing field data. Ecol Appl 11:371–384
Çömez A (2012) Determination of carbon sequestration in Scots pine (Pinus sylvestris L.) stands on Sündiken Mountain-Eskişehir. Research Institute for Forest Soil and Ecology Press, Eskişehir, p 123 (in Turkish)
Çömez A, Tolunay D, Güner ŞT (2019) Litterfall and the effects of thinning and seed cutting on carbon input into the soil in Scots pine stands in Turkey. Eur J Forest Res 138(1):1–14. https://doi.org/10.1007/s10342-018-1148-6
Eker M, Acar HH, Özçelik R, Alkan H, Gürlevik N, Çoban HO, Korkmaz M, Yılmaztürk A (2013) Investigation of the availability of harvest residues in forestry. The Scientific and Technological Research Council of Turkey (TUBİTAK), Project Number: 110O435, Isparta, p 435 (in Turkish)
Erkan N, Comez A, Aydin AC, Denli O, Erkan S (2018) Litterfall in relation to stand parameters and climatic factors in Pinus brutia forests in Turkey. Scand J For Res 33(4):338–346. https://doi.org/10.1080/02827581.2017.1406135
Fonseca W, Benayas JMR, Alice FE (2011) Carbon accumulation in the biomass and soil of different aged secondary forests in the humid tropics of Costa Rica. For Ecol Manag 262:1400–1408. https://doi.org/10.1016/j.foreco.2011.06.036
Goussanou CA, Guendehou S, Assogbadjo AE, Sinsin B (2018) Application of site-specific biomass models to quantify spatial distribution of stocks and historical emissions from deforestation in a tropical forest ecosystem. J For Res 29(1):205–213. https://doi.org/10.1007/s11676-017-0411-x
Grüneberg E, Zıche D, Wellbrock N (2014) Organic carbon stocks and sequestration rates of forest soils in Germany. Glob Change Biol 20:2644–2662. https://doi.org/10.1111/gcb.12558
Güner ŞT, Çömez A (2017) Biomass equations and changes in carbon stock in afforested black pine (Pinus nigra Arnold. subsp. pallasiana (Lamb.) Holmboe) stands in Turkey. Fresenius Environ Bull 26(3):2368–2379
Güner ŞT, Makineci E (2017) Determination of annual organic carbon sequestration in soil and forest floor of Scots pine forests on The Türkmen Mountain (Eskişehir, Kütahya). J Fac For Istanb Univ 67(2):109–115. https://doi.org/10.17099/jffiu.199494
Güner ŞT, Çömez A, Özkan K, Karataş R, Çelik N (2016) Modelling the productivity of Anatolian black pine plantations in Turkey. J Fac For Istanb Univ 66(1):159–172. https://doi.org/10.17099/jffiu.18731(ın Turkish)
Hunt R (1990). Relative growth rates. In: Basic growth analysis. Springer, Dordrecht, pp 25–34
IPCC (2003) Good practice guidance for land use, land-use change and forestry. In: IGES, Penman J, Gytarsky M, Hiraishi T, Krug T, Kruger D, Pipatti R, Buendia L, Miwa K, Ngara T, Tanabe K, Wagner F (eds) IPCC/OECD/IEA/IGES, Hayama, Japan. http://www.ipcc-nggip.iges.or.jp/public/gpglulucf/gpglulucf_contents.html. Accessed 14 Dec 2015
IPCC (2006) IPCC guidelines for national greenhouse gas inventories, prepared by the National Greenhouse Gas Inventories Programme. In: IGES, Japan (Eggleston HS, Buendia L, Miwa K, Ngara T, Tanabe K) (eds) http://www.ipcc-nggip.iges.or.jp/public/2006gl/index.html. Accessed 21 June 2019
IUSS Working Group WRB (2015) World reference base for soil resources 2014, update 2015. International soil classification system for naming soils and creating legends for soil maps. World Soil Resources Reports No. 106. FAO, Rome, p 192
Jandl R, Lindner M, Vesterdal L, Bauwens B, Baritz R, Hagedorn F, Johnson DW, Minkinen K, Byrne KA (2007) How strongly can forest management influence soil carbon sequestration? Geoderma 137:253–268. https://doi.org/10.1016/j.geoderma.2006.09.003
Jiang L, Zhao W, Lewis BJ, Wei Y, Dai L (2018) Effects of management regimes on carbon sequestration under the Natural Forest Protection Program in northeast China. J For Res 29(5):1187–1194. https://doi.org/10.1007/s11676-017-0542-0
Jurgensen M, Tarpey R, Pickens J, Kolka R, Palik B (2012) Long-term effect of silvicultural thinnings on soil carbon and nitrogen pools. Soil Sci Soc Am J 76:1418–1425. https://doi.org/10.2136/sssaj2011.0257
Karaöz MÖ (1993) The amounts and nutrient contents of forest floor of some coniferous stands at the Atatürk arboretum near İstanbul. J Fac For Istanb Univ A 43(1):93–115 (ın Turkish)
Karataş R, Çömez A, Güner ŞT (2017) Determination of carbon stocks in cedar (Cedrus libani A. Rich.) afforestation areas. Journal of Forestry Research 4(2):107–120. https://doi.org/10.17568/ogmoad.338029(ın Turkish)
Karatepe Y (2004) Amount of nitrogen and organic carbon in soil and nitrogen and organic matter in forest floor of black pine (Pinus nigra Arn. supsp. pallasiana (Lamb.) Holmboe) stands developed in Gölcük (Isparta). Fac For J Süleyman Demirel Univ A(2):1–16 (ın Turkish)
Korkanç SY (2014) Effects of afforestation on soil organic carbon and other soil properties. CATENA 123:62–69. https://doi.org/10.1016/j.catena.2014.07.009
Laclau P (2003) Biomass and carbon sequestration of ponderosa pine plantations and native cypress forests in northwest Patagonia. For Ecol Manag 180:317–333. https://doi.org/10.1016/S0378-1127(02)00580-7
Lee J, Tolunay D, Makineci E, Çömez A, Son YM, Kim R, Son Y (2016) Estimating the age-dependent changes in carbon stocks of Scots pine (Pinus sylvestris L.) stands in Turkey. Ann For Sci 73(2):523–531. https://doi.org/10.1007/s13595-016-0546-5
Lee J, Makineci E, Tolunay D, Son Y (2018) Estimating the effect of abandoning coppice management on carbon sequestration by oak forests in Turkey with a modeling approach. Sci Total Environ 640–641:400–405. https://doi.org/10.1016/j.scitotenv.2018.05.341
Lettens S, Orshoven JV, Wesemael BV, Muys B, Perrin D (2005) Soil organic carbon changes in landscape units of Belgium between 1960 and 2000 with reference to 1990. Glob Change Biol 11:2128–2140. https://doi.org/10.1111/j.1365-2486.2005.001074.x
Makineci E (2005) Thinning effects on diameter increment and some soil properties in sessile oak (Quercus petraea (Matlusch) Lieb.) coppice forest. Fac For J Süleyman Demirel Univ A(2):1–10 (ın Turkish)
Makineci E, Ozdemir E, Caliskan S, Yilmaz E, Kumbasli M, Keten A, Beskardes V, Zengin H, Yilmaz H (2015) Ecosystem carbon pools of coppice-originated oak forests at different development stages. Eur J For Res 134(2):319–333. https://doi.org/10.1007/s10342-014-0854-y
Miegroet HV, Moore PT, Tewksbury CE, Nicholas NS (2007) Carbon sources and sinks in high-elevation spruce-fir forests of the Southeastern US. For Ecol Manag 238:249–260. https://doi.org/10.1016/j.foreco.2006.10.020
Moısen GG, Frescıno TS (2002) Comparing five modelling techniques for predicting forest characretistics. Ecol Model 157:209–225. https://doi.org/10.1016/S0304-3800(02)00197-7
Pausas JG (1997) Litter fall and litter decomposition in Pinus sylvestris forests of the eastern Pyrenees. J Veg Sci 8:643–650. https://doi.org/10.2307/3237368
Payne NJ, Cameron DA, Leblanc JD, Morrison IK (2019) Carbon storage and net primary productivity in Canadian boreal mixedwood stands. J For Res 30(5):1667–1678. https://doi.org/10.1007/s11676-019-00886-0
Peichl M, Arain MA (2006) Above- and belowground ecosystem biomass and carbon pools in an age-sequence of temperate pine plantation forests. Agric For Meteorol 140:51–63. https://doi.org/10.1016/j.agrformet.2006.08.004
Polat S, Polat O, Kantarcı MD, Tüfekçi S, Aksay Y (2014) Relationships between some environmental characteristics and site indices (H38) of Taurus cedar (Cedrus libani A. Rich.) and black pine (Pinus nigra Arnold.) afforestation areas in the Kadıncık Basin of Mersin. Journal of Forestry Research 1(1):22–37. https://doi.org/10.17568/oad.86449(ın Turkish)
Prietzel J, Stetter U, Klemmt H-J, Rehfuess KE (2006) Recent carbon and nitrogen accumulation and acidification in soils of two Scots pine ecosystems in Southern Germany. Plant Soil 289:153–170. https://doi.org/10.1007/s11104-006-9120-5
Ruiz-Peinado R, Bravo-Oviedo A, Lopez-Senespleda E, Montero G, del Río M (2013) Do thinnings influence biomass and soil carbon stocks in Mediterranean maritime pinewoods. Eur J Forest Res 132:253–262. https://doi.org/10.1007/s10342-012-0672-z
Ruiz-Peinado R, Bravo-Oviedo A, Montero G, del Río M (2016) Carbon stocks in a Scots pine afforestation under different thinning intensities management. Mitig Adapt Strat Glob Change 21:1059–1072. https://doi.org/10.1007/s11027-014-9585-0
Schulp CJE, Nabuurs G-J, Verburg PH, Waal RW (2008) Effect of tree species on carbon stocks in forest floor and mineral soil and implications for soil carbon inventories. For Ecol Manag 256:482–490. https://doi.org/10.1016/j.foreco.2008.05.007
Sevgi O, Makineci E, Karaöz Ö (2011) The forest floor and mineral soil carbon pools of six different forest tree species. Ekoloji 20(81):8–14
SPSS v.22.0® (2015) SPSS 22.0 guide to data analysis. Prentice Hall, Upper Saddle River
Tolunay D (1997) Effects of stand treatments on nutrient cycling in young Scots pine (Pinus sylvestris L.) stands on Aladağ Mountain in Bolu. Ph.D. Dissertation, İstanbul University, Graduate School of Sciences, İstanbul, p 213 (in Turkish)
Tolunay D (2011) Total carbon stocks and carbon accumulation in living tree biomass in forest ecosystems of Turkey. Turk J Agric For 35:265–279. https://doi.org/10.3906/tar-0909-369
Tolunay D, Çömez A (2008) Organic carbon stocks in soils and forest floor in Turkish forests. In: Sympossium on air pollution and it’s control, 22–25 October 2008, Hatay, Turkey, pp. 750–765 (in Turkish)
Tolunay D, Makineci E, Şahin A, Özturna AG, Pehlivan S, Abdelkaım MMA. 2017. Carbon sequestration in maritime pine (Pinus pinaster Ait.) and stone pine (Pinus pinea L.) plantations on İstanbul-Durusu sand dunes. The Scientific and Technological Research Council of Turkey (TUBİTAK), Project Number: 114O797, İstanbul, p 148 (in Turkish)
Tufekcioglu A, Guner S, Tilki F (2005) Thinning effects on production, root biomass and some soil properties in a young oriental beech stand in Artvin, Turkey. J Environ Biol 26(1):91–95
Wellbrock N, Grüneberg E, Riedel T, Polley H (2017) Carbon stocks in tree biomass and soils of German forests. Cent Eur For J 63:105–112. https://doi.org/10.1515/forj-2017-0013
Yavuz H, Mısır N, Mısır M (2004) Growth models for black pine plantations. The Scientific and Technological Research Council of Turkey (TUBİTAK), Project Number: 2747, Trabzon, p 223 (in Turkish)
Yildiz O, Cromack K Jr, Radosevich SR, Martinez-Ghersa MA, Baham JE (2011) Comparison of 5th- and 14th-year Douglas-fir and understory vegetation responses to selective vegetation removal. For Ecol Manage 262:586–597. https://doi.org/10.1016/j.foreco.2011.04.015
Yimer F, Ledin S, Abdelkadir A (2006) Soil organic carbon and total nitrogen stocks as affected by topographic aspect and vegetation in the Bale Mountains, Ethiopia. Geoderma 135:335–344. https://doi.org/10.1016/j.geoderma.2006.01.005
Acknowledgements
This study was presented orally as “Changes in Carbon Stocks of Soil and Forest Floor in Black Pine Plantations”, and its abstract was published in the Proceedings of the Conference “10th International Soil Congress 2019” in Ankara, Turkey, 17–19 June 2019, p 305.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Project funding: This study was conducted as part of the project, “Changes in carbon stocks on soil and forest floor in black pine plantations (ESK-30(6321)/2017-2018)” funded by the Turkish General Directorate of Forestry.
The online version is available at http://www.springerlink.com.
Corresponding editor: Yu Lei.
Rights and permissions
About this article
Cite this article
Güner, Ş.T., Güner, D. Changes in carbon stocks of soil and forest floor in black pine plantations in Turkey. J. For. Res. 32, 339–347 (2021). https://doi.org/10.1007/s11676-019-01073-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11676-019-01073-x