Abstract
This study was conducted to determine the changes in carbon stocks of oriental beech (Fagus orientalis) according to stand development stage in the Marmara Region of Türkiye. For this purpose, sample plots were taken from a total of 32 areas encompassing four stand development stages (young, middle age, mature and overmature stand). The diameter at breast height and height of all trees in the sample plots were measured, and only three dominant trees’s ages per plot were determined. Aboveground carbon stock was calculated using equations developed for beech forests, while the coefficients in the Agriculture, Forestry and Other Land Use guide were used to determine belowground carbon stocks. A soil pit was dug in each plot and soil samples were taken at different depths (0–10, 10–30, 30–60, 60–100 cm). In addition, litters were sampled from four different 25 × 25 cm sections in each plot, and then the physical and chemical properties of the soil and litters were analysed. The variations in carbon stocks in above- and below-ground tree mass, litter and soil, and in ecosystem carbon stocks according to development stage were examined by analysis of variance and Duncan test, and the relationships between the carbon stocks were investigated by correlation analysis. Aboveground (AG) and belowground (BG) tree, soil and ecosystem carbon stocks showed significant differences between the four stand development stages (P < 0.05), but not the litter carbon stocks (P > 0.05). AG and BG tree and ecosystem carbon stocks increased with progressive stand development stages, while the soil carbon stock was the highest at the young stage. These findings will contribute to the preparation of forest management plans and the national greenhouse gas inventory.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
International agreements to mitigate the effects of global climate change have obliged countries to report their carbon emissions and carbon pools. To ensure that inventory data is comparable between countries, an Agriculture, Forestry and Other-Land Use guide (AFOLU) for calculating these parameters has been developed (IPCC, 2006, 2013). This guide explains in detail how to calculate the annual change in carbon stocks in forest ecosystems. The guide divides forest ecosystem carbon pools into three classes: living belowground and aboveground biomass, dead organic matter (dead wood and litter), and organic carbon in soil. In addition, the guide includes some empirical coefficients in terms of climate zone, forest type and tree species to be used in the calculations of the pools in question, but suggests that these coefficients should be revealed through research on tree species at the local level for more accurate calculations (IPCC, 2003, 2006). Research has shown that the rate of carbon accumulation in carbon pools varies depending on environmental factors, tree species and tree organs (Bert & Danjon, 2006; Çömez, 2012; Laiho & Laine, 1997; Lamlom & Savidge, 2003; Thomas & Malczewski, 2007).
Türkiye's forest area is 22.9 million hectares; of this, the oriental beech (Fagus orientalis Lipsky) constitutes 8.2% of the country's forests, with a spread of 1.9 million hectares, and ranks second after oak genus (Quercus spp.) in terms of distributional area among broad-leaf species (GDF, 2021). Therefore, oriental beech is one of the priority species that needs to be researched (Güner, 2021). Native to Türkiye, the oriental beech spreads to Bulgaria, Romania, Greece, Crimea, the Caucasus and Northern Iran. It is widely distributed in Türkiye in the middle- and high elevations of the mountains running parallel to the Black Sea coast, and tends to establish along north-facing aspects, especially in pure and mixed forests. It is also seen occasionally in the Marmara, Central Anatolia and Eastern Mediterranean Regions. Oriental beech is distributed in regions with some temperate climate rich in moisture and with balanced precipitation (Anşin & Özkan, 1997; Atalay, 1992).
The aboveground biomass of oriental beech forests in the Eastern Black Sea Region of Türkiye was previously determined by Saraçoğlu (1998), while Güner et al. (2010) revealed both aboveground and belowground biomass in young oriental beech forests in the Eastern Black Sea Region (Artvin). Some researchers also studied the amount of belowground biomass in oriental beech forests in the Black Sea Region (Tüfekcioglu et al., 2005; Mısır & Mısır, 2013; Özbayram & Güvendi, 2016). Furthermore, the studies were carried out to investigate carbon stocks of Turkish forest soils (Tüfekcioglu et al., 2005), litters (Sargıncı et al., 2021) and ecosystems (Erkut, 2013). Additionally, aboveground carbon stocks in oriental beech forests in the Central Black Sea Region (Sinop) were determined by Kahyaoğlu (2017) and Kahyaoğlu et al. (2019). In general, these studies focused on the Black Sea Region and determined only one of the carbon pools in the ecosystem (such as aboveground tree mass, root mass, soil or litter). However, our study differs from previous studies because it covers all carbon pools in the oriental beech forest ecosystems, takes into account development stages and was conducted in the Marmara Region.
In this study, we aimed to investigate the carbon stocks and the relationships between these carbon pool stocks according to their development stages in oriental beech forests in the Marmara Region of Türkiye. The study hypothesised that there would be variations in the carbon pools among the stages of stand development and that there was a relationship between the carbon pools. The results of the study can be used in forest management plan development and greenhouse gas inventory reporting.
Materials and methods
Study area
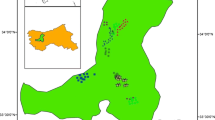
The research was carried out in pure oriental beech forests in the Marmara Region of Türkiye (Fig. 1). The research areas were located on granite, granodiorite and quartz diorite bedrock (GDMRE, 2021). The soil type commonly found in the research areas was Luvisol (IUSS Working Group WRB, 2015). In addition, the beech forests in the research area were spread on different soil texture classes, without carbonate and salt.
The annual mean temperature in the sample plots ranged from 6.8 to 10.2 °C, with high temperatures from 26.5 to 29.9 °C, and low temperatures from -9.2 to -5.8 °C. Mean temperature of the coldest month was -3.4 °C, while mean temperature of the hottest month was 20.1 °C. Furthermore, mean annual rainfall varied between 903 and 1276 mm, with rainfall range of 24–34 mm in the driest month, 241–340 mm in the spring months (March + April + May), and 161–227 mm in the summer months (June + July + August) (GDM, 2020). According to the Erinç method, the climate types of the research areas were semi-humid and humid (Özyuvacı, 1999).
The dominant tree species in the research area was Fagus orientalis, and the Trachystemo orientalis–Fagetum orientalis plantassociation was widespread at 600–1100 m. Its distinctive species included Trachystemon orientalis (G.Don), Cardamine bulbifera (L.) Crantz and Campanula rotundifolia var. olympica (Boiss). While 54.5% of the plant taxa included in its floristic composition belong to the Euro-Siberian region, 1.8% belong to the Mediterranean flora. Plant taxa with multiple or unknown regions constitute 43.7% of the total flora in the area (Akman et al., 1979; Türe et al., 2005).
Experimental design and sampling procedure
The research was conducted in a total of 32 sample plots, within 8 areas for each of 4 development stages: young (diameter at breast height, dbh = 0–7.9 cm), middle age (dbh = 8–19.9 cm), mature (dbh = 20–35.9 cm) and overmature stands (dbh ≥ 36 cm). Sampling plots were generally selected among 3 dense-cover (canopy cover = 70–100%; one mature and two overmature sampling plots) and among 2 moderate-cover (canopy cover = 40–70%) plots to ensure that different growing environments were represented. The sample plots were square or rectangular in shape and large enough to include at least 15 trees (Çepel et al., 1977). The slope, elevation, aspect and slope position characteristics of the sample plots were recorded, the diameter at breast height and total height of all trees were measured, and the age of three dominant trees was measured with increment borer in per sample area (Table 1). Then, a soil pit was dug in each of the sample plots, and soil samples were taken using a steel cylinder with a volume of 1000 cm3 at the following depth levels: 0–10, 10–30, 30–60 and 60–100 cm. Litter were sampled from 4 different points representing the sample plots and in areas of 0.0625 m2 (25 × 25 cm) in size (32 sample plots × 4 replications = 128 litter samples). All litter was collected in these areas, placed in bags and transported to the laboratory.
Laboratory analysis
The litter samples were dried to constant weight at 70 °C, weighed, and then the amount per unit area was calculated. The dried litter samples were ground and analysed for carbon content using a CNH elemental analyser (Leco Corporation, St. Joseph, Michigan, USA). The soil samples were air dried, the soil clods (aggregates) were crushed in a porcelain mortar and sieved through sieves of 2 mm mesh, and the weights of the fine earth and coarse fragments were determined. The fine earth was dried to a constant weight at 105 °C, weighed, and its moisture content determined (Carter & Gregorich, 2008). For the soil samples, pH-values were determined in pure water at a v/5v ratio (TS ISO 10390, 2013), soil texture was analysed by the Bouyoucous hydrometer method (Kroetsch & Wang, 2008) and organic carbon content was determined using the Walkley–Black wet combustion method (TS8336, 1990).
Calculations and statistical analysis
Equation 1, developed by Erkut (2013), was used to calculate the aboveground carbon stocks of trees in the sample plots. Then, the aboveground carbon stocks in a one-hectare area were calculated using the values obtained for the sample plots and the conversion coefficients to hectares. The coefficients in the AFOLU guide (IPCC, 2006) were used to determine belowground carbon stocks. For this purpose, coefficients of 0.46 were used in areas with aboveground tree mass < 75 t ha−1, 0.23 in areas with 75–150 t ha−1 and 0.24 in areas with > 150 t ha−1. By adding the aboveground and belowground carbon stocks of the sample plots, the carbon stocks of all trees in a hectare area were found.
where AGCS is aboveground carbon stock (kg), and d is diameter at breast height (cm).
Equation 2, developed by Atıcı (1998), was used to calculate the bark trunk volumes in the sample plots.
where ln is logarithm, V is the volume of trunk with bark (dm3), d is diameter at breast height (cm), and h is tree height (m).
The carbon stocks in the forest litter (forest floor) at the sample plot were estimated by multiplying the amount of litter in the sample plot and the carbon rates found as a result of the analysis, and then these values were converted into hectares.
The carbon stock per volume of 1000 cm3 was determined by multiplying the percentage of carbon values in the soil by the amount of fine earth in a volume of 1000 cm3. Then, these values were multiplied by the thickness of the depth layers to calculate the carbon amount of 1 m depth and 1 m2 area (i.e. 1 m3), and this value was converted to hectares by multiplying by 10,000.
The total carbon stock in one hectare of each sample area was measured by adding up the amount of carbon stocked in the trees, litter and soil. The relationships between the soil, litter, tree and ecosystem carbon stock and stand characteristics (age, mean diameter at breast height of the stand, mean height of the stand, age, basal area and bark trunk volume) were examined by Pearson correlation analysis. Normal distribution of the data set was controlled by Shapiro–Wilk tests, while homogeneity of the variances was evaluated by Levene's test. All data were normally distributed (P > 0.05) and homogeneous variance (P > 0.05). Then the differences between stand characteristics and tree, litter, soil and ecosystem carbon stock at different development stages were examined by analysis of variance. (ANOVA). Duncan’s multiple-range test was used for comparison of the means. The results were considered statistically different at the α = 0.05 level. The SPSS package program was used for statistical analysis (SPSS, 2015).
Results and discussion
Stand parameters
In this study, significant differences were found between the stand development stages in terms of stand age, mean diameter at breast height of the stand, mean height of the stand, number of trees, basal area and bark trunk volume (P < 0.05). Four similar groups were formed in terms of the stand age, stand mean diameter at breast height and stand mean height; three similar groups were formed in terms of the number of trees and bark trunk volume; and two similar groups were formed in terms of basal area. While the stand age, stand mean diameter at breast height, stand mean height and bark trunk volume increased with increasing development stage, the number of trees decreased. The stand basal area was found to be highest (48.6 m2 ha−1) in stands at the mature stage and lowest (30.6 m2 ha−1) in stands at the young stage (Table 2). Similarly, Makineci et al. (2015) showed for oak that stand age, stand mean diameter at breast height, stand mean height and basal area increased with successive development stages, while the number of trees decreased. The stand volume was found 122, 356, 793, and 1108 m3 ha−1 in the development stages young, middle age, mature, and overmature respectively, depending on the progression of the stand development stage. Similar findings have been found in studies on the subject (Çömez, 2012; Güner & Çömez, 2017; Karataş et al., 2017; Tolunay et al., 2017).
Tree carbon stock
Significant differences (P < 0.05) were determined between the stand development stages in terms of the aboveground, belowground and whole-tree carbon stocks. Three similar groups were established in all three carbon stocks, and the lowest carbon stock was found in stands in the young stage. The aboveground, belowground and whole-tree carbon stocks were found to be highest in the stands at the mature and overmature stages, and the differences between these stands were found to be statistically insignificant (P > 0.05) (Table 3). The high level of the aboveground, belowground and whole-tree carbon stocks in the stands in the mature and overmature development stages can be explained by the fact that they have more stand volume and tree mass compared to the other development stages. As seen in the Table 2, the average stand volumes in stands at the young, middle age, mature and overmature development stages were 122, 356, 793 and 1108 m3 ha−1, respectively. Likewise, a study for oak, the aboveground tree carbon stocks of the young, middle age and mature development stages (Makineci et al., 2015) were reported 13, 54 and 89 t ha−1, respectively. Furthermore, for black pine (Pinus nigra Arnold. subsp. pallasiana (Lamb.) Holmboe) afforestation sites, the carbon stock of all trees varied significantly between the stand development stages, with the lowest carbon stock in the young stands and the highest in the mature stands (Güner & Çömez, 2017). Another study for Pinus strobus L. plantations aged at 2, 15, 30 and 65-year-old, mean aboveground tree carbon stocks were 0.5, 66, 92 and 176 t ha−1 respectively (Peichl & Arain, 2006). Aboveground carbon stock in young, middle age, mature and overmature stages are found at 42.8; 122.6; 253.6 and 344.4 t ha−1 respectively. All sample plots have been evaluated together, and an average of 190.9 t C ha−1 was found. A similar study in the eastern beech forests in the Eastern Black Sea Region (middle age, mature, and overmature development stage) was found 175.9 t ha−1 (Erkut, 2013), which lower than our research findings. Belowground carbon stock in young, middle age, mature and overmature development stages were 12,6; 29,3; 60,9 and 82,7 t ha−1 respectively, and the average of all sample areas was 46.4 t C ha−1. There are studies investigating belowground mass and carbon stock in eastern beech forests in Türkiye (Tüfekcioglu et al., 2005; Mısır & Mısır, 2013; Özbayram & Güvendi, 2016). In these studies, the cylinder method was used, and the mass of belowground in a certain diameter class (such as Ø ≤ 10 mm or Ø > 50 mm) was determined. Although no study has been found revealing the entire belowground mass and carbon stock in eastern beech, belowground mass and carbon stock of the broad-leaf species ash (Fraxinus angustifolia Vahl.) and alder (Alnus glutinosa L.) were studied (Sariyildiz et al., 2023). In this study, belowground carbon stock was found 80.3 t ha−1 for ash and 25.2 t ha−1 for alder (Sariyildiz et al., 2023). These values determined for ash and alder are similar to values we determined for eastern beech.
Litter carbon stock
Differences between the stand development stages in terms of litter mass, carbon content and carbon stock were insignificant (P > 0.05). When all sample areas were evaluated together, the average litter mass was 12.5 t ha−1, the carbon content was 43.9% and carbon stock was 5.5 t ha−1 (Table 4). It is reported that many factors such as location, climate characteristics, landform, tree type, forest age, canopy closure, soil properties and soil organisms influence litter mass (Kantarcı, 2000). In general, as stand development progresses, the tree mass per unit area and, as a result, the litter mass increases. A study in Iran for oriental beech forests showed that litter thickness (cm) and carbon content (%) increased with advancing development stages (Kooch et al., 2021). Likewise, for oak, litter mass showed significant differences among the young, middle age and mature development stages with the values of 4.5, 6.4 and 10.6 t ha−1 respectively (Makineci et al., 2015). Similar results were reported for coniferous tree species; Pinus nigra Arn. (Güner & Çömez, 2017), Cedrus libani A. Rich. (Karataş et al., 2017), Pinus pinea L. and Pinus pinaster Ait. (Makineci, 2021). For brutia pine (Pinus brutia Ten.), a significant relationship was found between a number of parameters such as basal area per hectare, total volume and canopy cover, and the amount of litterfall (Erkan et al., 2018). However, in our study, differences between the stand development stages in terms of litter mass, carbon content and carbon stock were insignificant. This can be explained by the addition of high amounts of cutting residue to the litter as a result of natural regeneration studies (seeding, overhead release and final cutting). When all data were evaluated together, the average litter mass was 12.5 t ha−1, carbon content was 43.9% and carbon stock was 5.5 t ha−1. Similar to our findings, for oriental beech forests in the middle age, mature and overmature development stages in the Eastern Black Sea Region, the litter mass was 4.8–27 t ha−1, litter carbon content was 34.9% and litter carbon stock was 2–7 t C ha−1 (Erkut, 2013). The results of another study from oriental beech forests in Türkiye also showed similar findings to our study, with litter mass of 8.7 (2.6–36.6) t ha−1 and litter carbon stock of 3.2 (1.0–9.4) t C ha−1 reported (Tolunay & Çömez, 2008). Similarly, for oriental beech forests in Iran, the carbon content of litter was 36.2% for the young stands and 52.5% for the old stands (Kooch et al., 2021).
Soil carbon stock
The changes in soil properties in the research area at different depth levels are given in Table 5. Differences between depth levels in terms of bulk density, amount of fine earth (Ø < 2 mm), percent sand, silt, clay, pH-values and organic carbon were significant (P < 0.05). In general, bulk density, fine earth, percent sand and pH-values increased with soil depth, while silt, clay and organic carbon values decreased (Table 5).
The amount of fine earth (t ha−1), soil organic carbon content (%) and soil carbon stock (t ha−1) showed statistically significant differences between the stand development stages (P < 0.05). The amount of fine earth was the lowest in the young stands (8,96 t ha−1), and the highest in the middle (11,22 t ha−1) and mature (10,98 t ha−1) age stands (Table 6). The fact that the amount of fine earth was lowest in young stands is likely because these stands are generally located on upper elevation land (Table 1). The amount of fine earth and carbon content affect the soil carbon stock. In addition, it is reported that organic matter input, the decomposition rate of organic matter (Tolunay & Çömez, 2007) and fine root decomposition in the soil (Berg & McClaugherty, 2003) influence the carbon content of the soil. In this study, soil organic carbon content and soil carbon stock were the highest at the young stage, followed by a decrease at the middle age and mature stand development stages. Soil organic carbon content and soil carbon stock increased after the mature development stage and reached 1.09% and 105 t C ha−1, respectively, in the overmature development stage (Table 6). The fact that the soil organic carbon stock and organic carbon content were highest in young stands, then decreased, reaching their lowest level in mature stands, before increasing again in overmature stands, can be explained by the addition of high amounts of cutting residue to the litter as a result of natural regeneration studies. Due to the reduction in canopy closure via the overhead release and final cutting, the amount of precipitation and temperature within the stand increased. Therefore, abundant cutting residues decomposed faster, increasing the organic carbon content of the soil. As a result of regeneration activities, the cutting residues added to the litter decreased with decomposition and reached a balance in mature stands, and then, due to the increase in tree mass, both litterfall and fine root decomposition provided organic matter to the soil, causing an increase in the soil organic carbon stock. Likewise, in a study conducted on the sessile oak (Quercus petraea (Matt.) Liebl.), the carbon stock in 0–30 cm deep soil was found to be 31.5, 10.6, 24.7 and 55.0 t ha−1 in stands at young, middle age, mature and overmature development stages, respectively. The carbon stock decreased from the young stage to the middle age stage, reached a balance in the middle age stage, and increased again, reaching the highest level in the overmature stage (Savacı & Aktaş-Tümer, 2022). In our study, when all sampling areas were evaluated together, the average soil organic carbon stock was 101.4 t ha−1. Likewise, the value of 77.9 (27.8–227.3) t ha−1 given by Tolunay and Çömez (2008) as the average carbon stock in Türkiye's beech forests is consistent with the findings of this study. Again, in a study conducted in eastern beech forests (middle age, mature and overmature development stage) in the Eastern Black Sea Region, soil organic carbon stock was found 81.1 t ha−1, which is similar to our research findings (Erkut, 2013).
Ecosystem carbon stock
Differences between stand development stages in terms of ecosystem carbon stock were found to be significant (P < 0.05). The lowest ecosystem carbon stock was in the young (187.5 t C ha−1) and middle age (255.4 t C ha−1) stands development stages, followed by mature (396.4 t C ha−1) and overmature (537.4 t C ha−1) stages (Table 7). This difference between stand development stages in terms of ecosystem carbon stock was largely due to tree carbon stock (Fig. 2); similar findings have been reported in many studies on the subject (Çömez, 2012; Güner & Çömez, 2017; Karataş et al., 2017; Makineci et al., 2015; Tolunay et al., 2017). The carbon stocks in the components of the ecosystem (trees, litter, soil) varied according to development stage. While tree mass carbon stock constituted 30% of the ecosystem carbon stock in stands at the young stage, this rate increased up to 80% in the overmature development stage stands. While the ratio of soil carbon stock to ecosystem carbon stock was 67% in stands at the young stage, this ratio decreased to 19% in stands at the overmature stage. The ratio of litter carbon stock to ecosystem carbon stock was 3%, 2%, 1% and 1% in stands of the young, middle age, mature and overmature development stages, respectively (Fig. 3). According to the average values, the ecosystem carbon stock in the oriental beech forests in the Marmara Region was found to be 344 t ha−1. The ecosystem carbon stock (average of 257 t C ha−1) reported in a study conducted on oriental beech forests (middle age, mature and overmature development stages) in the Eastern Black Sea Region (Giresun) was found to be lower than that in our study since belowground biomass was not included in the ecosystem carbon stock (Erkut, 2013).
Comparison of carbon stocks by development stage for different carbon pools (each column representing the mean value of the carbon stock for the related development stages). Error bars indicate ± SE. Carbon stocks sharing the same letter in columns are not significantly different from each other at the α = 0.05 level, according to the ANOVA results for the biomass, litter, soil and ecosystem
Relationships between carbon stocks
The relationships between tree, litter and soil carbon stocks are given in Table 8. A positive relationship was seen between the litter carbon stock and the soil carbon stock at the 0.05 significance level, while the relationships between other carbon stocks were statistically insignificant (P > 0.05). As one of the sources of soil carbon was litter, the positive relationship between litter carbon stock and soil carbon stock was an expected outcome. Previous studies found positive relationships between tree carbon stock and litter carbon stock (Bradford et al., 2009; Çömez, 2012; Güner & Çömez, 2017). The fact that no significant relationship was detected between the tree carbon stock and litter carbon stock in our study can be attributed to the introduction of high amounts of cutting residue into the stands in the development stage through regeneration studies. In a study conducted on Taurus cedar (Cedrus libani A. Rich.) afforestation area, a positive relationship was found between tree carbon stock and soil carbon stock. This situation has been explained as more carbon input into the soil due to litterfall and fine root decomposition resulting from the increase in tree mass (Karataş et al., 2017).
Relationships between carbon stocks and stand parameters
A positive relationship was found between stand age, stand mean diameter at breast height, stand mean height, basal area and bark trunk volume, and tree carbon stock and ecosystem carbon stock at a significance level of 0.01. Furthermore, negative relationships were found between the number of trees and tree carbon stock and ecosystem carbon stock at the 0.05 significance level, as well as between the medium stand height and soil carbon stock at the 0.05 significance level (Table 9). The basis of these relationships is the mass of trees per unit area; with the increase in tree mass, tree carbon stock and ecosystem carbon stock increased. Similar results were obtained in studies conducted on Taurus cedar (Karataş et al., 2017) and black pine afforestation area (Güner & Çömez, 2017).
Conclusions
This study investigated to the variations in carbon stocks in different ecosystem components of the oriental beech forests of the Marmara Region, Türkiye, with stand development stages. Tree carbon stocks showed significant differences between the stand development stages, reaching 55, 152, 315 and 427 t ha−1 in the young, middle age, mature and overmature stages, respectively. No differences were found between the development stages in terms of litter mass, litter carbon content and litter carbon stock. For oriental beech forests in the Marmara Region, mean litter mass was 12.5 t ha−1, the litter carbon content and stock were 43.9% and 5.5 t ha−1 respectively. The obtained values can be used in future calculations for the same region and similar habitats. Mean soil carbon stock showed significant differences between the four development stages, as 127, 98, 77 and 105 t ha−1 for the young, middle age, mature and overmature stages, respectively. Since many factors such as stand characteristics, soil properties, litter decomposition rate and climate characteristics influence soil carbon stock, more comprehensive modelling studies are needed. Despite this, the soil carbon data presented in this study can be used until more detailed studies are conducted. Ecosystem carbon stock showed differences between development stages and was found to be 188, 255, 396 and 537 t ha−1 at the young, middle age, mature and overmature stages, respectively. The data revealed by this research can be used in forest management plan development and greenhouse gas inventory reporting.
Data availability
The datasets analyzed during the current study are available from the corresponding author upon reasonable request.
References
Akman, Y., Barbero, M., & Quezel, P. (1979). Contribution to the study of the forest vegetation of Mediterranean Anatolia. Phytocoenologia, 5(3), 277–346.
Anşin, R., & Özkan, Z. C. (1997). Spermatophyta. Karadeniz Technical University Faculty of Forestry Press.
Atalay, İ. (1992). The ecology of beech (Fagus orientalis Lipsky) forests and their regioning in terms of seed transfer. Forest Tree Seeds and Tree Breeding Research Institute Press.
Atıcı, E. (1998). Increment and growth in uneven aged beech (Fagus orientalis Lipsky). Istanbul University PhD thesis, Graduate School of Science.
Berg, B., & Mcclaugherty, C. (2003). Plant litter decomposition, humus formation, carbon sequestration. Springer-Verlag.
Bert, D., & Danjon, F. (2006). Carbon concentration variation in the roots, stem and crown of mature Pinus pinaster (Ait.). Forest Ecology and Management, 222, 279–295. https://doi.org/10.1016/j.foreco.2005.10.030
Bradford, J., Weishampel, P., Smith, M. L., Kolka, R., Birdsey, R. A., Ollinger, S. V., & Ryan, M. G. (2009). Detrital carbon pools in temperate forests: Magnitude and potentialfor landscape-scale assessment. Canadian Journal of Forest Research, 39, 802–813. https://doi.org/10.1139/X09-010
Carter, M. R., & Gregorich, E. G. (2008). Soil Sampling and Methods of Analysis. CRC Press.
Çepel, N., Dündar, M., & Günel, A. (1977). Relationship between edaphic and physiological attributes and growth of Scotch pine in important forest areas. The Scientific and Technological Research Council of Turkey Press.
Çömez, A. (2012). Determination of carbon sequestration in Scots pine (Pinus sylvestris L.) stands on Sündiken Mountain-Eskişehir. Eskişehir: Research Institute for Forest Soil and Ecology Press.
Erkan, N., Çömez, A., Aydın, A. C., Denli, O., & Erkan, S. (2018). Litterfall in relation to stand parameters and climatic factors in Pinus brutia forests in Turkey. Scandinavian Journal of Forest Research, 33(4), 338–346. https://doi.org/10.1080/02827581.2017.1406135
Erkut, S. (2013). Capacity of carbon stock in pure beech stands of Giresun Forest District Directorate Akkuş Forest Enterprise. Karadeniz Technical University Master of science thesis, Graduate School of Science.
GDF. (2021). Forestry Statistics 2020. Turkish General Directorate of Forestry. https://www.ogm.gov.tr/tr/e-kutuphane/resmi-istatistikler Accessed 22 October 2021
GDM. (2020). Meteorological data. https://mgm.gov.tr/ Accessed 14 February 2020
GDMRE. (2021). 1:500,000 Scale Geological Inventory Map Series of Turkey. http://www.mta.gov.tr/v3.0/hizmetler/500bas Accessed 04 January 2021
Güner, ŞT. (2021). Relationships between site index and ecological variables of beech forests in Marmara region of Turkey. Fresenius Environmental Bulletin, 30(6A), 6920–6927.
Güner, ŞT., & Çömez, A. (2017). Biomass equations and changes in carbon stock in afforested black pine (Pinus nigra Arnold. subsp. pallasiana (Lamb.) Holmboe) stands in Turkey. Fresenius Environmental Bulletin, 26(3), 2368–2379.
Güner, S., Yagci, V., Tilki, F., & Celik, N. (2010). The effects of initial planting density on above- and below-ground biomass in a 25-year-old Fagus orientalis Lipsky plantation in Hopa Turkey. Scientific Research and Essays, 5(14), 1856–1860.
IPCC. (2003). Good Practise Guidance For Land Use, Land-use Change and Forestry, In: IGES, Eds. Penman, J., Gytarsky, M., Hiraishy, T., Krug, T., Kruger, D., Pipatti, R., Buendia, L., Miwa, K., Ngara, T., Tanabe, K., Wagner, F., IPCC/OECD/IEA/IGES, Hayama, Japan. http://www.ipcc-nggip.iges.or.jp/public/gpglulucf/gpglulucf_contents.html Accessed 17 February 2017
IPCC. (2006). IPCC Guidelines for national greenhouse gas inventories, prepared by the National Greenhouse Gas Inventories Programme, In: IGES, Japan (Eds.: H.S. Eggleston, L. Buendia, K. Miwa, T. Ngara and K. Tanabe). http://www.ipcc-nggip.iges.or.jp/public/2006gl/index.html Accessed 04 January 2016
IPCC. (2013). Revised supplementary methods and good practice guidance arising from the Kyoto protocol, Intergovernmental Panel on Climate Change. http://www.ipcc-nggip.iges.or.jp/public/kpsg/. Accessed 15 Feb 2024
IUSS Working Group WRB. (2015). World reference base for soil resources 2014, update 2015. International soil classification system for naming soils and creating legends for soil maps. World Soil Resources Reports No. 106. FAO, Rome.
Kahyaoğlu, N., Güvendi, E., & Kara, Ö. (2019). Determination of aboveground biomass amount in pure oriental beech stands (a case of Sinop-Türkeli). Anatolian Journal of Forest Research, 5(2), 79–85.
Kahyaoğlu, N. (2017). Determination of the biomass and carbon storage of the oriental beech (Fagus orientalis Lipsky.) forest in Sinop region. Karadeniz Technical University PhD thesis, Graduate School of Science.
Kantarcı, M. D. (2000). Soil Science. Istanbul University Press.
Karataş, R., Çömez, A., & Güner, ŞT. (2017). Determination of carbon stocks in cedar (Cedrus libani A. Rich.) afforestation areas. Turkish Journal of Forest Research, 4(2), 107–120. https://doi.org/10.17568/ogmoad.338029
Kooch, Y., Parsapour, M. K., Egli, M., & Moghimian, N. (2021). Forest floor and soil properties in different development stages of Oriental beech forests. Applied Soil Ecology, 161, 103823. https://doi.org/10.1016/j.apsoil.2020.103823
Kroetsch, D., & Wang, C. (2008). Particle size distribution, in section VI. Soil physical analysis, Section Ed. By D.A. Angers, F.J. Larney. In M. R. Carter & E. G. Gregorich (Eds.), Soil sampling and methods of analysis (2nd ed., pp. 713–725). Boca Raton: CRC Press.
Laiho, R., & Laine, J. (1997). Tree stand biomass and carbon content in an age sequence of drained pine mires in southern Finland. Forest Ecology and Management, 93, 161–169. https://doi.org/10.1016/S0378-1127(96)03916-3
Lamlom, S. H., & Savidge, R. A. (2003). A reassessment of carbon content in wood: Variation within and between 41 North American species. Biomass and Bioenergy, 25, 381–388. https://doi.org/10.1016/S0961-9534(03)00033-3
Makineci, E. (2021). Nitrogen accumulation in forest floors with introduced Pinus pinea and Pinus pinaster in dune site. Environmental Monitoring and Assessment, 193, 327. https://doi.org/10.1007/s10661-021-09100-3
Makineci, E., Ozdemir, E., Caliskan, S., Yilmaz, E., Kumbasli, M., Keten, A., Beskardes, V., Zengin, H., & Yilmaz, H. (2015). Ecosystem carbon pools of coppice-originated oak forests at different development stages. European Journal of Forest Research, 134(2), 319–333. https://doi.org/10.1007/s10342-014-0854-y
Mısır, M., & Mısır, N. (2013). Root biomass and carbon storage for Fagus orientalis Lipsky. (Northeastern Anatolia). International Journal of Education and Research, 1(4), 1–8.
Özbayram, A. K., & Güvendi, E. (2016). Relationships between coarse root biomass and habitat-stand properties in oriental beech (Fagus orientalis Lipsky) stands in Sinop. Düzce University Journal of Forestry, 12(2), 27–33.
Özyuvacı, N. (1999). Meteorology and Climatology. Istanbul University Faculty of Forestry Press.
Peichl, M., & Arain, M. A. (2006). Above and belowground ecosystem biomass and carbon pools in an age-sequence of temperate pine plantation forests. Agricultural and Forest Meteorology, 140, 51–63. https://doi.org/10.1016/j.agrformet.2006.08.004
Saraçoğlu, N. (1998). Biomass tables of beech (Fagus orientalis Lipsky). Turkish Journal of Agriculture and Forestry, 22, 93–100.
Sargıncı, M., Yıldız, O., Tolunay, D., Toprak, B., & Temür, Ş. (2021). Leaf litter dynamics in Western Black Sea mountainous forest ecosystems. Canadian Journal of Forest Research, 51(12), 1821–1832. https://doi.org/10.1139/cjfr-2020-0489
Sariyildiz, T., Tani, M., & Parlak, S. (2023). First assessment of root biomass and root carbon and nitrogen stocks in Turkish floodplain forests. Environmental Monitoring and Assessment, 195(1), 148. https://doi.org/10.1007/s10661-022-10758-6
Savacı, G., & Aktaş-Tümer, G. (2022). Effect of development stages on soil organic carbon and nitrogen stocks of sessile oak (Quercus petrea (Matt.) Liebl): A case study of Taşköprü Kastamonu. ArtGRID, 4(2), 240–253. https://doi.org/10.57165/artgrid.1203364
SPSS. (2015). SPSS 22.0 guide to data analysis, published by Prentice Hall, Upper Saddle River. New Jersey
Thomas, S. C., & Malczewski, G. (2007). Wood carbon content of tree species in Eastern China: Interspecific variability and the importance of the volatile fraction. Journal of Environmental Management, 85, 659–662. https://doi.org/10.1016/j.jenvman.2006.04.022
Tolunay, D., & Çömez, A. (2007). Carbon storage in forest soils and the situation in Turkey. Forests Symposium in Solving Global Climate Change and Water Problems (pp. 97–107). İstanbul
Tolunay, D., & Çömez, A. (2008). Organic carbon stocks in soils and forest floor in Turkish forests. Symposium on Air Pollution and It’s Control (pp. 750–765). Hatay
Tolunay, D., Makineci, E., Şahin, A., Özturna, A. G., Pehlivan, S., & Abdelkaım, M. M. A. (2017). Carbon sequestration in maritime pine (Pinus pinaster Ait.) and stone pine (Pinus pinea L.) plantations on İstanbul-Durusu sand dunes. Project Number: 114O797. İstanbul: The Scientific and Technological Research Council of Turkey.
TS 8336. (1990). Soils-Determination of organic matter. Turkish Standards Institution Press.
TS ISO 10390. (2013). Soil quality-Determination of pH. Turkish Standards Institution Press.
Tüfekcioglu, A., Güner, S., & Tilki, F. (2005). Thinning effects on production, root biomass and some soil properties in a young oriental beech stand in Artvin Turkey. Journal of Environmental Biology, 26(1), 91–95.
Türe, C., Tokur, S., & Ketenoğlu, O. (2005). Contribution to the syntaxonomy and ecology of the forest and shrub vegetation in Bithynia, Northwestern Antolia, Turkey. Phyton (austria), 45, 81–115.
Acknowledgements
This study was conducted as part of the projects, “Change of carbon stocks according to development stage in oriental beech forests in the Marmara Region [ESK-46(6330)/2021-2022]” funded by the Turkish General Directorate of Forestry.
Funding
This work was supported by the Turkish General Directorate of Forestry ESK-46(6330).
Author information
Authors and Affiliations
Contributions
Şükrü Teoman Güner: Conceptualization, Methodology, Data collection, Formal analysis, Data curation, Writing—original draft, Writing—review & editing, Visualization. Özgür Kiracıoğlu: Writing—review & editing. Abdullah Sarımehmetoğlu: Writing—review & editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
All authors have read, understood, and have complied as applicable with the statement on “ethical responsibilities of authors.”
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Güner, Ş.T., Kiracioğlu, Ö. & Sarimehmetoğlu, A. Changes in carbon stocks according to stand development stages in oriental beech forests in the Marmara Region of Türkiye. Environ Monit Assess 196, 571 (2024). https://doi.org/10.1007/s10661-024-12727-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10661-024-12727-7