Abstract
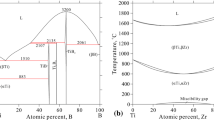
The Sb-Te system is re-modeled using the calculation of phase diagram (CALPHAD) technique. The liquid phase is modeled as (Sb, Sb2Te3, Te) using the associate model and as (Sb3+) p (Te2−,Te,Va) q using the ionic model. The solution phases rhom(Sb) and hex(Te) are described as substitutional solutions. Two compounds, delta and gamma, are treated as (Sb)0.4(Sb,Te)0.6 according to their homogeneity ranges, while the compound Sb2Te3 follows a strict stoichiometry. A set of self-consistent thermodynamic parameters is obtained. Using these thermodynamic parameters, the experimental Sb-Te phase diagram, mixing enthalpies of liquid at 911 K and 935 K, activities of Sb and Te in liquid at 911 K and 1023 K, and Gibbs energy of liquid at 911 K, is well reproduced by the calculations. And the calculated enthalpy of formation, enthalpy of fusion, and heat capacity of Sb2Te3 are also in fairly good agreement with all the available experimental data.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
K. Rajasekar, L. Kungumadevi, A. Subbarayan, and R. Sathyamoorthy, Ionics 14, 69 (2008).
F.-H. Li, W. Wang, Y.-L. Gong, and J.-Y. Li, J. Electron. Mater. 41, 3039 (2012).
Y.T. Kim and S.-I. Kim, App. Phy. Lett. 103, 121906 (2013).
E.T. Kim, J.Y. Lee, and Y.T. Kim, Phys. Status Solidi (RRL) 3, 103 (2009).
D. Hsieh, Y. Xia, D. Qian, L. Wray, F. Meier, J.H. Dil, J. Osterwalder, L. Patthey, A.V. Fedorov, H. Lin, A. Bansil, D. Grauer, Y.S. Hor, R.J. Cava, and M.Z. Hasan, Phys. Rev. Lett. 103, 146401 (2009).
H.J. Zhang, C.X. Liu, X.L. Qi, X. Dai, Z. Fang, and S.C. Zhang, Nat. Phys. 5, 438 (2009).
G. Wang, X. Zhu, J. Wen, X. Chen, K. He, L. Wang, X. Ma, Y. Liu, X. Dai, Z. Fang, J. Jia, and Q. Xue, Nano Res. 3, 874 (2010).
G. Ghosh, H.L. Lukas, and L. Delaey, Z. Metallkd. 80, 731 (1989).
The SGTE (Scientific Group Thermodata Europe) Substance Data File, Version Summer 1986.
Y. Feutelais, B. Legendre, S. Misra, and T.J. Anderson, J. Phase Equili. 15, 171 (1994).
http://www.thermocalc.com/resources. SGTE pure elements (unary) database, Version 5.1 (2010).
B.W. Howlett, S. Misra, and M.B. Bever, Trans. AIME 230, 1367 (1964).
V.M. Zhdanov, Zh. Fiz. Khim. 45, 2388 (1971).
E.V. Gul’tyaev and A.V. Petrov, Fiz. Tverd. Tela. 1, 368 (1965).
R.A. Medzhidov and S.M. Rasulov, Russ. J. Phys. Chem. 53, 100 (1979).
B. Legendre, Y. Feutelais, and J.R. Didry, J. Therm. Anal. 34, 345 (1988).
A.S. Pashinkin, A.S. Malkova, and M.S. Mikhailova, Russ. J. Phys. Chem. A 82, 878 (2008).
G. Ghosh, J. Phase Equili. 15, 349 (1994).
Y. Feutelais, B. Legendre, and G. Morgant, J. Therm. Anal. 34, 1093 (1988).
B. Onderka and K. Fitzner, Z. Metallkd. 86, 313 (1995).
H. Fay and H.E. Ashley, Am. Chem. J. 27, 95 (1902).
H. Pélabon, Compt. Rend. 142, 207 (1906).
W. Haken, Ann. Phys. 32, 291 (1910).
K. Honda and T. Soné, Sci. Rep. Tohoku Univ. 2, 9 (1913).
V.I. Veraksa, V.N. Lange, and T.I. Lange, Zh. Fiz. Khim. 37, 2308 (1963).
K. Honda and E. Endo, J. Inst. Met. 37, 29 (1927).
G. Offergeld and J. Van Cakenberghe, J. Phys. Chem. Solids 11, 310 (1959).
G. Offergeld and J. Van Cakenberghe, Nature 184, 185 (1959).
A.C. Glatz, Proceedings of the 2nd International Conference on Thermal Analysis, ed., R.E Schwenker and P.D. Gain (New York: Academic Press, 1969), pp. 1411–1422.
A.N. Krestonikov, V.M. Glazov, V.A. Evseev, and O.V. Situlina, Tsvetn Met. 2, 79 (1967).
L.V. Poretskaya, NKh Abrikosov, and V.M. Glazov, Zh. Neorg. Khim. 8, 1204 (1963).
Y. Kimata, Mem. Coll. Sci. Kyoto Univ. 16, 115 (1915).
N.S. Konstantinov and V.I. Smirnov, Izv. St. Petersburg Politekh. Inst. Imp. Petra Velik 23, 713 (1915).
N. Kh. Abrikosov, V.E Bankina, L.V. Poretskaya, L.E. Shelimova, and E.V. Skudnova, Semiconducting II-VI, IV-VI and V-VI Compounds, (New York: Plenum Press, 1969), pp. 159-242.
NKh Abrikosov, L.V. Poretskaya, and I.E. Ivanova, Zh. Neorg. Khim. 4, 2525 (1959).
S. Bordas, M.T. Clavaguera-Mora, B. Legendre, and C. Hancheng, Thermochim. Acta 107, 239 (1986).
A. Brown and B. Lewis, J. Phys. Chem. Solids 23, 1597 (1962).
V.E. Eckerlin and A. Stegherr, Acta Crystallogr. 21, A78 (1966).
N.K. Abrikosov and M.M. Stasova, Inorg. Mater. 21, 1758 (1985).
L.E. Shelimova, O.G. Karpinskii, M.A. Kretova, V.I. Kosyakov, V.A. Shestakov, V.S. Zemskov, and F.A. Kuznetsov, Inorg. Mater. 36, 768 (2000).
P.M. Imamov and S.A. Semiletov, Kristallografiya 15, 972 (1970).
K. Kifune, Y. Kubota, T. Matsunaga, and N. Yamada, Acta Crystallogr. Sect. B 61, 492 (2005).
C.W. Sun, J.Y. Lee, M.S. Youm, and Y.T. Kim, Phys. Stat. Sol. 1, R25 (2007).
C.W. Sun, J.Y. Lee, M.S. Youm, and Y.T. Kim, Jpn. J. Appl. Phys. 45, 9157 (2006).
N. Frangis, S. Kuypers, C. Manolikas, G. Van Tendeloo, J. Van Landuyt, and S. Amelinckx, J. Solid State Chem. 84, 314 (1990).
K. Kifune, T. Fujita, T. Tachizawa, Y. Kubota, N. Yamada, and T. Matsunaga, Cryst. Res. Technol. 48, 1011 (2013).
T. Maekawa, T. Yokokawa, and K. Niwa, J. Chem. Thermodyn. 4, 153 (1972).
Ya.I. Gerasimov and A.V. Nikoskaya, Problems of the Metallurgy and Physics of Semiconductors (Moscow : Izv. Akad. Nauk SSSR, 1961).
A.A. Vecher, L.A. Mechkovskii, and A.S. Skoropanov, lzv. Akad. Nauk SSSR, Neorg. Mater. 10, 2140 (1974).
V.M. Glazov, R.A. Kuliev, and A.N. Krestovnikov, Zh. Fiz. Khim. 45, 2671 (1971).
C. Mallika and O.M. Sreedharan, J. Chem. Thermodyn. 18, 727 (1986).
E. Herwig and M. Wobst, Z. Metallkd. 83, 35 (1992).
G.E. Guhskaya and I.V. Evfimovskii, Zh. Neorg. Khim. 7, 1615 (1962).
V.M. Glazov, A.N. Krestovnikov, and N.N. Glagoleva, Izv. Akad. Nauk SSSR Neorg. Mater. 2, 453 (1966).
V.M. Glazov, A.N. Krestovnikov, N.N. Glagoleva, and S.B. Evgenev, Izv. Akad. Nauk SSSR Neorg. Mater. 2, 1477 (1966).
V.M. Glazov, A.N. Krestovnikov, and V.A. Evseev, Dokl. Akad. Nauk SSSR 169, 868 (1966).
M. Wobst, Wiss. Z. Tech. Hochsch. Karl-Marx-Stadt 12, 393 (1970).
R. Blakeway, Philos. Mag. 20, 965 (1969).
A.S. Jordan, Metall. Trans. 1, 239 (1970).
M.T. Clavaguera-Mora and N. Clavaguera, J. Phys. Chem. Solids 43, 963 (1982).
E. Summer, Z. Metallkd. 73, 72 (1982).
M. Hillert, B. Jansson, B. Sundman, and J. Agren, Metall. Trans. A 16, 261 (1985).
J.O. Andersson, T. Helander, L.H. Hoglund, P.F. Shi, and B. Sundman, Calphad 9, 273 (2002).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Guo, C., Li, C. & Du, Z. Thermodynamic Re-modeling of the Sb-Te System Using Associate and Ionic Models. J. Electron. Mater. 43, 4082–4089 (2014). https://doi.org/10.1007/s11664-014-3299-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11664-014-3299-7