Abstract
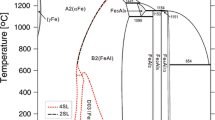
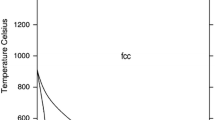
The Pt-Te and the Pt-Sb-Te systems are modeled using the calculation of phase diagram (CALPHAD) technique. In the Pt-Te system, the liquid phase is modeled as (Pt, PtTe2, Te) using the associate model, and four intermediates, PtTe2, Pt2Te3, Pt3Te4 and PtTe, are treated as stoichiometric compounds and their enthalpies of formation are obtained by means of first-principles calculations. The solution phases, fcc(Pt) and hex(Te), are described as substitutional solutions. Combined with the thermodynamic models of the liquid phase in the Pt-Sb and Sb-Te systems in the literature, the liquid phase of the Pt-Sb-Te ternary system is modeled as (Pt, Sb, Te, Sb2Te3, PtTe2) also using the associate model. The compounds, PtTe2, Pt2Te3, Pt3Te4 and PtTe in the Pt-Te system and PtSb2, PtSb, Pt3Sb2 and Pt7Sb in the Pt-Sb system are treated as line compounds Pt m (Sb,Te) n in the Pt-Sb-Te system, and the compound Pt5Sb is treated as (Pt,Sb)5(Pt,Sb,Te). A set of self-consistent thermodynamic parameters is obtained. Using these thermodynamic parameters, the experimental Pt-Te phase diagram, the experimental heat capacities of PtTe and PtTe2, the enthalpies of formation from first-principles calculations for PtTe2, Pt2Te3, Pt3Te4, and PtTe, and the ternary isothermal sections at 873 K, 923 K, 1073 K and 1273 K are well reproduced.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
J. Yang, W. Zhu, X. Gao, S. Bao, X. Fan, X. Duan, and J. Hou, J. Phys. Chem. B 110, 4599 (2006).
L.J. Cabri, Platinum-Group Elements: Mineralogy, Geology, Recovery (Washington, DC: Geology Division of CIM, 1981).
W.-S. Kim, J. Alloys Compd. 252, 166 (1997).
M.L. Gimpl, C.E. Nelson, and N. Fuschillo, Trans. Am. Soc. Metals 56, 209 (1963).
Y.C. Bhan, T. Godecke, and K. Schubert, J. Less-Common Metals 19, 121 (1969).
W.-S. Kim, Met. Mater. 2, 9 (1996).
V.K. Karzhavin, Geochem. Int. 45, 931 (2007).
F. Grønvold, T. ThurmannMoe, E.F. Westrum Jr, and E. Chang, J. Chem. Phys. 35, 1665 (1961).
E.F. Westrum Jr, H.J. Carlson, F. Grønvold, and A. Kjekshus, J. Chem. Phys. 35, 1670 (1961).
G. Ghosh, H.L. Lukas, and L. Delaey, Z. Metallkd. 80, 731 (1989).
C. Guo, C. Li, and Z. Du, J. Electron. Mater. 43, 4082 (2014).
J. Liu, Y. Zhang, and C. Guo, Int. J. Nonferrous Metall. 2, 95 (2013).
C. Guo, C. Li, and Z. Du, Int. J. Mater. Res. 105, 525 (2014).
M. El-Boragy and K. Schubert, Z. Metallkd. 62, 667 (1971).
W.-S. Kim and G.Y. Chao, Can. Mineral. 28, 675 (1990).
W.-S. Kim, J. Alloys Compd. 284, 218 (1999).
W.-S. Kim, J. Alloys Compd. 407, 235 (2006).
http://www.thermocalc.com/resources, SGTE pure elements (unary) database, Version 5.1 (2010).
M. Hillert and L.I. Staffansson, Acta Chem. Scand. 24, 3618 (1970).
B. Sundman and J. Ǻgren, J. Phys. Chem. Solids 42, 297 (1981).
J.P. Perdew, K. Burke, and M. Ernzerhof, Phys. Rev. Lett. 77, 3865 (1996).
Y. Ohmasa, I. Yamamoto, M. Yao, and H. Endo, J. Phys. Soc. Jpn. 64, 4766 (1995).
C. Soulard, P.E. Petit, P. Deniard, M. Evain, S. Jobic, M.H. Whangbo, and A.C. Dhaussy, J. Solid State Chem. 178, 2008 (2005).
J.O. Andersson, T. Helander, L.H. Hoglund, P.F. Shi, and B. Sundman, Calphad 9, 273 (2002).
Y.M. Muggianu, M. Gambino, and J.-P. Bros, J. Chim. Phys. 72, 83 (1975).
C. Guo, C. Li, P.J. Masset, and Z. Du, Calphad 36, 100 (2012).
Acknowledgements
This work was supported by National Natural Science Foundation of China (NSFC) (Grant Nos. 51371029 and 51171017) and the National High Technology Research and Development Program of China (Grant No. 2013AA031601).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Guo, C., Huang, L., Li, C. et al. Thermodynamic Modeling of the Pt-Te and Pt-Sb-Te Systems. J. Electron. Mater. 44, 2638–2650 (2015). https://doi.org/10.1007/s11664-015-3676-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11664-015-3676-x