Abstract
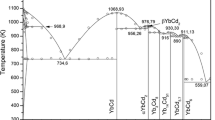
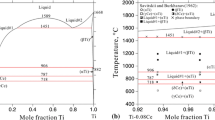
Using the CALculation of PHAse Diagrams (CALPHAD) method, the phase equilibria and thermodynamic basis for the Cd-Se and Pb-Se binary systems are critically studied in this work. The associated solution model is adopted for the liquid phases so as to reflect the V-shaped enthalpies of mixing, the abrupt change in activity plots, and the sharp maxima for the liquidus around intermediate phases. Due to the negligible solubilities in CdSe and PbSe, these two binary compounds are treated as stoichiometric phases. Solid Se, which has a hexagonal structure, is considered to be free of solubility with respect to Cd and Pb. The thermodynamic description provided in this work allows the experimental data to be well represented, thus providing a fundamental basis for semiconductor research.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
C. Ma, Y. Ding, D. Moore, X. Wang, and Z. Wang, J. Am. Chem. Soc. 126, 708 (2004).
M. Califano, A. Zunger, and A. Franceschetti, Appl. Phys. Lett. 84, 2409 (2004).
R.D. Schaller, M.A. Petruska, and V.I. Klimov, Appl. Phys. Lett. 87, 253102 (2005).
E. Hendry, M. Koeberg, F. Wang, H. Zhang, and C. De Melll Donga, Phys. Rev. Lett. 96, 057408 (2006).
W.D. Lawson, J. Appl. Phys. 22, 1444 (1951).
R.C. Sharma and Y.A. Chang, J. Phase Equilib. 17, 140 (1996).
R.C. Sharma and Y.A. Chang, J. Electrochem. Soc. 136, 1536 (1989).
J.C. Lin, R.C. Sharma, and Y.A. Chang, J. Phase Equilib. 17, 253 (1996).
N. Saunder and A.P. Miodownik, CALPHAD (Oxford: Pergamon Materials Series, Pergamon/Elsevier Science, 1998).
A. Reisman, M. Berkenblit, and M. Witzen, J. Phys. Chem. 66, 2210 (1962).
L.A. Sysoev, E.K. Raiskin, and V.R. Gurev, Izv. Akad. Nauk SSSR Neorg. Mater. 3, 390 (1967).
M. Chikashige and R. Hikosaka, Mem. Coll. Sci. Kyoto Impl. Univ. 2, 239 (1917).
D.R. Mason and D.F. O’Kane, International Conference on Semiconductor Physics, Prague, 1960 (New York: Academic Press Inc., 1961), pp. 1026–1032.
S. Deore, F. Xu, and A. Navrotsky, Am. Mineral. 93, 779 (2008).
W.J. Wosten, J. Phys. Chem. 65, 1949 (1961).
I.V. Korneeva, V.V. Sokolov, and A.V. Novoselova, Russ. J. Inorg. Chem. 5, 117 (1960).
J. Terpilowski and E. Ratajczak, Bull Acad. Polon. Sci. 12, 355 (1964).
L. Seacrist and Z.A. Munir, High Temp. Sci. 3, 340 (1971).
A.G. Sigai and H. Wiedemeir, J. Electrochem. Soc. 119, 910 (1972).
G. Vardi, K. Ieronimakis, and G. Trionfetti, Thermochim. Acta 129, 341 (1988).
P. Goldfinger and M. Jeunehomme, Trans. Faraday Soc. 59, 2851 (1963).
L.H. Shiozawa, J.M. Jost, Report No. ARL 62-365 (Mars. 1962) and (May, 1965), ARL-65-98, Aerospace Research Lab, U.S.A.F.
L.R. Shiozawa, J.M. Jost, Report No. ARL 62-366 (March 1962) and ARL 65-68 (May 1965), Aerospace Research Lab, U.S.A.F.
R.A. Burmeister and D.A. Stevenson, J. Electrochem. Soc. 114, 394 (1967).
K.C. Mills, Thermodynamic Data for Inorganic Sulphides (Butterworths: Selenides and Tellurides, 1974).
H. Jelinek and K.L. Komarek, Monatsh. Chem. 105, 689 (1974).
H. Pelabon and D. Gernez, Compt. Rend. 144, 1159 (1907).
K. Friedrich and A. Leroux, Metallurgie 5, 355 (1908).
R. Nozato and K. Isaki, Bull. Naniwa Univ. A (Jpn.) 3, 125 (1955).
B.E. Pelzel, Metallurgie 10, 717 (1956).
D. Seidman, I. Cadoff, K. Komarek, and E. Miller, Trans. Metall. AIME 221, 1269 (1961).
D.N. Seidman, Trans. Metall. AIME 236, 1361 (1966).
E. Miller and K.L. Komarek, Trans. Metall. AIME 236, 832 (1966).
M. Schneider and J.C. Guillaume, J. Phys. Chem. Solids 35, 471 (1974).
J. Greenwood and H.W. Worner, J. Inst. Met. 115, 433 (1939).
S.N. Hajiev, J. Chem. Thermodyn. 2, 765 (1970).
S. Boone and O.J. Kleppa, Thermochim. Acta 197, 109 (1992).
C.B. Finch and J.R. Wagner, J. Electrochem. Soc. 107, 932 (1960).
K.B. Sadykov and S.A. Semenkovich, Izv. Akad Nauk. Turk. SSR Seriya. Fiz. Khimi. Geo. Nauk. 3, 25 (1966).
M.S. Shamsuddin, Scrip. Metall. 7, 547 (1973).
K.P. Kotchi, R. Castanet, and J.C. Mathieu, Thermochim. Acta 76, 311 (1984).
A.T. Dinsdale, CALPHAD 15, 317 (1991).
F. Sommer, CALPHAD 2, 319 (1978).
B. Sundman, B. Jansson, and F. Andersson, CALPHAD 9, 153 (1985).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Liu, Y., Kang, Z., Sheng, G. et al. Phase Equilibria and Thermodynamic Basis for the Cd-Se and Pb-Se Binary Systems. J. Electron. Mater. 41, 1915–1923 (2012). https://doi.org/10.1007/s11664-012-2016-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11664-012-2016-7