Abstract
Genebank conservation of pollen is valuable because it makes genetic resources immediately available for use in breeding programs. In the case of Citrus species, conserved anthers or pollen can be easily transported and used to develop new varieties with pathogen resistance and desirable quality and yield traits. The aim of this study was to develop and improve air-desiccation cryopreservation protocols for Citrus cavaleriei and Citrus maxima anthers in genebanks. In the current study, warming, rehydration, and in vitro germination conditions were optimized to achieve high levels of in vitro germination in Citrus pollen for ten cultivars after liquid nitrogen (LN) exposure. The optimal warming, rehydration, and in vitro germination medium formulations affected the germination levels after pollen cryopreservation, with species- and cultivar-dependent effects. The Citrus anthers were dehydrated to the moisture content of 5–14% before LN exposure and warmed at 25 (cryopreserved Citrus anthers with a moisture content of lower than 10%) or 37°C (a moisture content of 10% or higher), then rehydrated, and cultured on medium with 150-g L−1 sucrose, 0.1-g L−1 boric acid, 1.0-g L−1 calcium nitrate, 0.1-g L−1 potassium nitrate, 0.3-g L−1 magnesium sulfate, and 10-g L−1 agar. After 2 yr of storage, in vitro germination levels of Citrus pollen after cryopreservation were significantly higher (> 22% for all ten cultivars) than those of samples that were stored at 4°C (0%). In vitro germination levels of pollen from six of ten cultivars after cryopreservation remained relatively high after 2 yr of storage (38–93%). The highest viability of 93% was obtained for C. cavaleriei ‘2–3’. The methods identified in the current study could be used to cryopreserve C. cavaleriei and C. maxima anthers.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Citrus spp., members of the family Rutaceae, are economically important fruit crops with global production levels of 131.3 t in 2012 (FAO 2015). High levels of genetic diversity among the species and cultivars of Citrus have been reported (Yang et al. 2015; Barbhuiya et al. 2016). Citrus germplasm collections are traditionally conserved in field repositories. For example, 1335 accessions belonging to 35 Citrus species are maintained in the field repository of the Citrus Research Institute, Chinese Academy of Agricultural Sciences (CAAS) (Zhang et al. 2014). Approximately 1372 accessions representing 96 Citrus species are maintained in field and screenhouse collections in the United States Department of Agriculture-Agricultural Research Service (USDA-ARS) National Plant Germplasm System (NPGS) and in the University of California Citrus Variety Collection (Volk et al. 2017). More than 750 accessions with wide genetic variability are conserved at the Brazilian Agricultural Research Corporation-Embrapa (dos Santos et al. 2015). These field and screenhouse conservation methods are expensive to maintain, require extensive acreage, and are vulnerable to extreme environmental conditions and to pests and diseases. Hence, alternative conservation techniques are under investigation, which includes the conservation of in vitro cultures and cryopreservation, i.e., the storage of plant organs and tissues under liquid nitrogen (LN, − 196°C) (Engelmann 2011).
Cryopreservation has been implemented in genebanks worldwide to conserve vegetatively propagated plant collections (Benson 2008; Chen et al. 2013). To date, cryopreservation methods for diverse propagules, including seeds, pollen, shoot tips, winter buds, embryos, and cell cultures, have been identified for over 200 plant species (Dulloo et al. 2010). Previous cryopreservation studies on Citrus germplasm focused on the applications of such techniques to seeds, embryos/embryonal axes, embryogenic/nucellar calluses, and shoot tips (Hao et al. 2002; Malik and Chaudhury 2006; Lambardi et al. 2007; Al Zoubi and Normah 2012; Malik et al. 2012; Yan et al. 2014; Rohini et al. 2016; Volk et al. 2017).
Genebank conservation of pollen/anthers is valuable because it provides access to genetic resources that can be readily used in breeding programs. Pollen cryopreservation methods have been reported for at least 170 species, including fruit and forest trees, staple crops, vegetables, forage grasses, and ornamental economic and medicinal plants (Ganeshan et al. 2008; Zhang et al. 2009; Chaudhury et al. 2010; Karun et al. 2014; Wang et al. 2015; Popova et al. 2016). Pollen cryopreservation has been implemented at genebanks for several species. For example, pollen from 60 peach (Prunus persica) cultivars was cryopreserved in Japan (Omura and Akihama 1980), and pollen from 71 ornamental species/cultivars was cryopreserved at Beijing Forestry University, Beijing, China (Xu et al. 2014). Pollen cryopreservation of Citrus spp. has been previously reported; pollen samples of Citrus limon (L.) Burm. f., Citrus aurantiifolia (Christm.) Swingle, Citrus sinensis (L.) Osbeck, and Poncirus trifoliata (L.) Raf. were collected and cryopreserved in LN (Ganeshan and Alexander 1991; Rajasekharan et al. 1995). In addition, gamma ray-irradiated fresh and cryopreserved pollen samples from C. limon, Citrus limetta Risso, and C. sinensis were reported to have germination levels of approximately 50% (Kundu et al. 2014).
In the current study, an air-desiccation cryopreservation protocol for conserving anthers of Citrus cavaleriei H. Lev. ex Cavalerie and Citrus maxima (Burm.) Merr. was developed and optimized. The effects of warming procedures, rehydration conditions, and germination medium formulations on the in vitro germination levels of cryopreserved pollen were investigated. A straightforward procedure for cryopreserving the anthers of Citrus spp. will aid in the establishment of large-scale pollen cryopreservation at the National Crop Genebank of China.
Materials and Methods
Plant materials
The anthers of Citrus cultivars were collected from healthy trees at the Citrus Germplasm Repository, Citrus Research Institute, CAAS, Chongqing, China, in 2014. The following experiments were conducted at the National Crop Genebank of China, Institute of Crop Science (ICS), CAAS, Beijing, China. Ten Citrus cultivars were used in the current study. These cultivars belonged to C. cavaleriei (cultivars ‘2–3’ and ‘2586’) and C. maxima (cultivars ‘14–7’, ‘Beibei’, ‘Guanxiang’, ‘Humi’, ‘Jintang’, ‘Kui’, ‘Sijipao’, and ‘Zuoshi’). The cultivars C. cavaleriei ‘2–3’ and C. maxima ‘14–7’ were used to first investigate the effect of in vitro germination medium formulations on the pollen germination after LN exposure. All ten Citrus cultivars were used to investigate the effects of warming conditions, rehydration treatment, and in vitro germination media on pollen germination after LN exposure and to monitor the pollen germination levels after 2 yr of cryopreservation (− 196°C) or storage at 4°C.
Storage procedures for Citrus anthers
Anthers were collected and removed from flowers by using tweezers and were put on a piece of paper for 16–24 h at 25°C to dry. The anthers were then packed in tubes and shipped on ice overnight to the National Crop Genebank of China (ICS, CAAS, Beijing). Upon arrival, the moisture content of the anthers for each cultivar was determined using the following formula: moisture content (%) = (M1 − M2) / M1 ∗ 100, where M1 corresponds to the weight of the anthers before drying and dry weight M2 refers to the weight of anthers after oven drying at 103°C for 3 h. The anthers were then placed under incandescent lamps (40 W, Philips, Beijing, China) with the light intensity of 36 ~ 54 μmol m−2 s−1 at 30°C until the moisture content decreased to 5–14% (Table 1). A subset of the desiccated anthers was then transferred to 1.8-mL cryovials (Nunc®, Thermo Fisher Scientific®, Roskilde, Denmark), which were directly plunged into and stored in a LN tank at − 196°C (CBS S3000-AB SERIES, Custom BioGenic Systems®, Romeo, MI) at the National Crop Genebank of China. A second subset of air-desiccated anthers was packed in tubes and stored at 4°C in refrigerators. When the stored anthers were warmed, the tubes or cryovials were rinsed in running tap water at 25°C for 15 min or placed in a water bath (DK-8D, Shanghai YIHENG Technical®, Shanghai, China) at 37°C for 5 min. The rehydration treatment was conducted by placing the open cryovials in a closed container which contained saturated copper sulfate solution with 100% humidity for 3 h at 25°C following the method of Connor and Towill (1993) and Hanna and Towill (1995).
Pollen viability assessment
Pollen viability was investigated by using an in vitro germination assay. Pollen was removed from anthers with a very fine brush. The brush was cleaned between samples by washing with distilled water, 70% (v/v) ethanol, and again with distilled water, and then subsequently air-dried. Pollen grains were germinated in liquid medium in the form of “hanging” drops or were dispersed on a solidified medium as described by Kundu et al. (2014). Four in vitro germination media formulations were M1 liquid medium (distilled water with 150-g L−1 sucrose, 0.1-g L−1 boric acid, 1.0-g L−1 calcium nitrate, 0.1-g L−1 potassium nitrate, and 0.3-g L−1 magnesium sulfate), M2 solid medium (M1 medium with 10-g L−1 agar), M3 liquid medium (distilled water with 150-g L−1 sucrose, 0.1-g L−1 boric acid, 0.3-g L−1 calcium nitrate, 0.1-g L−1 potassium nitrate, and 0.2-g L−1 magnesium sulfate), and M4 solid medium (M3 medium with 10-g L−1 agar) (Table 2). The agar was supplied by EKEAR®, Shanghai, China; the sugar was supplied by Beijing BioDee Biotechnology®, Beijing, China; the boric acid, calcium nitrate, potassium nitrate, and magnesium sulfate were supplied by XiLong Chemical®, Shantou, China. The germination media were autoclaved at 121°C for 15 min.
The effects of in vitro germination media on pollen germination after LN exposure were evaluated. After culture on the in vitro medium at 25°C for 8–16 h in the dark, the germinated pollen grains were counted using a microscope (SZ × 16, OLYMPUS®, Tokyo, Japan). The pollen grains were considered to have germinated when the pollen tube was, at least, as long as the diameter of the pollen grain (Dafni and Firmage 2000). Four microscope well slides were examined for each treatment. Four different fields of view, each with, at least, 50 grains were examined per microscope well slide.
Data analysis
The data were evaluated with SPSS 18.0 (http://www.spss.com/) and were presented as means with standard deviations. An analysis of variance, least significant difference test (P < 0.05), and t tests (P < 0.05 or P < 0.01) were performed to determine statistically significant differences among the treatments.
Experimental design
First, the effect of the medium on in vitro germination levels after pollen cryopreservation was preliminarily investigated. The cryopreserved anthers of cultivars C. cavaleriei ‘2–3’ and C. maxima ‘14–7’ were warmed at 25°C for 15 min or at 37°C for 5 min, without rehydration, and cultured on M1, M2, M3, or M4 medium. The in vitro germination levels of these treatments were analyzed to identify the optimal medium. Next, the effects of warming, rehydration, and in vitro germination medium on germination after pollen cryopreservation were investigated. After LN exposure for 1 wk, the cryopreserved anthers of all ten Citrus cultivars were cultured on M2 and M4 after the following treatments: warmed at 25°C for 15 min and rehydrated for 3 h, warmed at 25°C for 15 min without rehydration, warmed at 37°C for 5 min and rehydrated for 3 h, and warmed at 37°C for 5 min without rehydration, respectively. Finally, in vitro germination levels of pollen for all ten cultivars after 2 yr of storage at 4°C or at − 196°C were determined.
Results
Moisture content of air-desiccated Citrus anthers before storage
The anthers of Citrus cultivars were air-desiccated and the moisture content determined (Table 1). Anthers from two cultivars, C. cavaleriei ‘2–3’ and C. maxima ‘Jintang’, were air-desiccated to a moisture content of 5–7%. Anthers from seven cultivars, i.e., C. cavaleriei ‘2586’ and C. maxima ‘14–7’, ‘Guanxiang’, ‘Humi’, ‘Kui’, ‘Sijipao’, and ‘Zuoshi’, were air-desiccated to a moisture content of 10–12%, and anthers from C. maxima ‘Beibei’ were air-desiccated to a moisture content of 14%.
Effects of culture medium components on pollen germination after cryopreservation
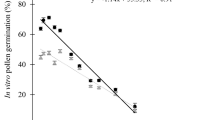
Two cultivars, C. cavaleriei ‘2–3’ and C. maxima ‘14–7’, were used to preliminarily assess the effects of in vitro germination medium. Relatively lower germination levels (lower than 20%) were obtained when the cryopreserved pollen was warmed and cultured on M1 (Fig. 1a–c), and the highest in vitro germination levels were 46% for C. cavaleriei ‘2–3’ when the cryopreserved pollen was warmed at 25°C, without rehydration, and cultured on M2 (Fig. 1b). For C. maxima ‘14–7’, higher germination levels were obtained when the cryopreserved pollen was warmed at 37°C, without rehydration, and germinated on M2 or M4, or warmed at 25°C without rehydration, and germinated on M2 (Fig. 1c).
Effects of in vitro germination medium and warming temperature on the pollen germination when the anthers were air-desiccated, LN exposed, warmed, without rehydrated, and cultured on M1, M2, M3, or M4 media. (A) Average pollen germination levels of all treatments for C. cavaleriei ‘2–3’ and C. maxima ‘14–7’. (B) Average pollen germination levels of all treatments for C. cavaleriei ‘2–3’. (C) Average pollen germination levels of all treatments for C. maxima ‘14–7’. Note: the four in vitro germination media formulations were M1 (distilled water with 150-g L−1 sucrose, 0.1-g L−1 boric acid, 1.0-g L−1 calcium nitrate, 0.1-g L−1 potassium nitrate, and 0.3-g L−1 magnesium sulfate), M2 (M1 medium with 10-g L−1 agar), M3 (distilled water with 150-g L−1 sucrose, 0.1-g L−1 boric acid, 0.3-g L−1 calcium nitrate, 0.1-g L−1 potassium nitrate, and 0.2-g L−1 magnesium sulfate), and M4 (M3 medium with 10-g L−1 agar). Values are means of four replicate experiments. Error bars represent standard deviations. Letters denote significant differences of the results for each treatment at 0.05 level (LSD test).
Effects of warming temperature and rehydration on germination levels after pollen cryopreservation
There were no significant differences between the two warming temperatures when all the germination data from the two species were combined (Fig. 2a). However, there was a significant difference between 25 and 37°C warming treatments when the average germination levels of C. maxima were analyzed separately (Fig. 2c). The effects of rehydration after warming on the germination levels are presented in Fig. 2d–e. The rehydration treatment after the cryopreserved anthers were warmed promoted pollen germination significantly (Fig. 2d). There were no significant differences between germination media M2 and M4 when all the germination data of cryopreserved anthers from two species was combined (Fig. 2g–i).
Effects of warming temperature and rehydration on germination levels after pollen cryopreservation. Average pollen germination levels for all Citrus cultivars, C. cavaleriei, and C. maxima cultivars were presented, when the anthers were air-desiccated, LN exposed, warmed on 25 or 37°C, with or without rehydrated, and cultured on M1, M2, M3, or M4 media. Average pollen germination levels for warming temperature for all Citrus cultivars (A), C. cavaleriei cultivars (B), and C. maxima cultivars (C). Average pollen germination levels for rehydration treatments for all Citrus cultivars (D), C. cavaleriei cultivars (E), and C. maxima cultivars (F). Average pollen germination levels for media formulations for all Citrus cultivars (G), C. cavaleriei cultivars (H), and C. maxima cultivars (I). Note: the four in vitro germination media formulations were M1 (distilled water with 150-g L−1 sucrose, 0.1-g L−1 boric acid, 1.0-g L−1 calcium nitrate, 0.1-g L−1 potassium nitrate, and 0.3-g L−1 magnesium sulfate), M2 (M1 medium with 10-g L−1 agar), M3 (distilled water with 150-g L−1 sucrose, 0.1-g L−1 boric acid, 0.3-g L−1 calcium nitrate, 0.1-g L−1 potassium nitrate, and 0.2-g L−1 magnesium sulfate), and M4 (M3 medium with 10-g L−1 agar). NR represents non-rehydration, R represents rehydration. The rehydration treatment was conducted by placing the open cryovials in a closed container which contains the saturated copper sulfate solution with 100% humidity for 3 h at 25°C. Values are means of four replicate experiments. Error bars represent standard deviations. Letters denote significant differences of the results for each treatment at 0.05 level (LSD test). * and ** denote significant differences between the non-rehydration treatments and rehydration treatments, for each treatment at 0.05 level and 0.01 level, respectively (t test).
The germination levels of cryopreserved pollen after LN exposure for 1 wk, under all treatments for each Citrus cultivar, are presented in Table 1. The highest average in vitro germination levels of 84 and 79% for C. cavaleriei ‘2–3’ and ‘2586’, respectively, were obtained when the cryopreserved anthers were warmed at 25 or 37°C and rehydrated. For C. maxima, the relatively high average in vitro germination levels of 71 and 60% for ‘Kui’ and ‘Zuoshi’, respectively, were obtained when the cryopreserved anthers were warmed at 37°C and rehydrated. Moderate average in vitro germination levels were obtained for cryopreserved anthers from C. maxima ‘Sijipao’ (52%), ‘Guanxiang’ (49%), ‘Humi’ (48%), ‘14–7’ (44%), ‘Beibei’ (42%), and ‘Jintang’ (40%).
Pollen viability after 2 yr of storage at 4°C or cryopreservation
After 2 yr of storage at 4°C, the in vitro germination level of stored pollen of all ten cultivars was 0% (Table 3). The in vitro germination levels of cryopreserved pollen of ten cultivars were also screened (Table 3). For C. cavaleriei ‘2–3’ and C. maxima ‘14–7’, ‘Jintang’, ‘Kui’, ‘Sijipao’, and ‘Zuoshi’, the rewarming and rehydration parameters and germination medium were the same as in the previous procedures, which resulted in the highest in vitro germination levels of cryopreserved pollen after LN exposure for 1 wk. For C. cavaleriei ‘2586’ and C. maxima ‘Beibei’, ‘Guanxiang’, ‘Sijipao’, and ‘Humi’, the rehydration parameters and germination medium were modified to obtain higher germination levels. The cryopreserved pollen was warmed at 37°C, rehydrated, and cultured on M2. After 2 yr of storage in a LN tank, in vitro germination levels of C. cavaleriei ‘2–3’ and ‘2586’ were higher (93 and 80%) than after 1 wk of storage. For C. maxima, the in vitro germination levels of ‘Beibei’, ‘Guanxiang’, and ‘Sijipao’ were 55, 53, and 65%, respectively, and the in vitro germination levels of the other five cultivars, i.e., C. maxima ‘14–7’, ‘Jintang’, ‘Humi’, ‘Kui’, and ‘Zuoshi’, decreased.
Discussion
Cryopreservation of Citrus cultivar pollen/anthers
It is crucial to dry pollen to an optimal level prior to LN exposure. Moisture content plays an important role during pollen/ anther cryopreservation, because excess free water in pollen forms ice crystals and reduces the viability (Towill 1985). The moisture content of the pollen or anthers decreased during pollen shed and might be around 20% (Nepi et al. 2010). It was deemed important to keep materials with a moisture content lower than 20% for successful cryopreservation (Vendrame et al. 2008). The measurement of moisture content of pollen or anthers was essential. However, previous studies preferred the pollen desiccation duration. For example, 24 h for Citrus pollen and 8 h for peach pollen were reported as the optimal desiccation durations (Kundu et al. 2014; Song and Li 2015). Ganeshan and Sulladmath (1983) and Ganeshan and Alexander (1991) studied C. limon pollen storage in LN with desiccation of pollen at 35°C for 15 min; however, the moisture content of the pollen was not reported. It is difficult to reproduce the results of cryopreservation procedures from different laboratories because detailed information on the initial moisture content and the moisture content after desiccation was not reported (Dafni and Firmage 2000). Thus, in the current study, the detailed moisture content (5–14%) of Citrus anthers for successful cryopreservation is reported.
Optimal moisture content of cryopreserved pollen for some species has been investigated, and it is found that there is variation of moisture content among different species, such as lower than 7.5% for English walnut (Juglans regia, Luza and Polito 1988) and 13–21% for apricot (Prunus spp., Wang and Li 1996). Although the moisture content of anthers and pollen may not be the same, in the current study, results demonstrated that cryopreserved anthers with low moisture content had better germination levels when they were warmed at 25°C, rehydrated, and cultured on M2. For example, C. cavaleriei ‘2–3’ and C. maxima ‘Jintang’ (moisture content of 7 and 5%) had average in vitro germination levels of 84 and 40%, respectively, which were higher than the pollen germination levels obtained by warming at 37°C with or without rehydration.
Optimal warming temperature will be useful to avoid ice formation (Zhang et al. 2006). An ambient temperature or 22°C for warming cryopreserved pollen was suggested (Ganeshan 1985; Ganeshan 1986; Sacks and St Clair 1996). Chen et al. (1990) also found that warming cryopreserved pollen with moisture content of 30% at 40°C resulted in a viability of 71% for Sinoj ackia. In the current study, a warming temperature of 37°C significantly increased germination levels of cryopreserved Citrus pollen over those obtained when the cryopreserved pollen was warmed at 25°C. However, when the moisture content of air-desiccated Citrus anthers was low (for instance, 5–7%, in the current study), a warming temperature of 25°C effectively promoted germination of cryopreserved pollen.
Assessing the pollen viability by in vitro germination on medium is practical and effective. The concentrations and the ratios of sucrose, boric acid, calcium nitrate, and agar affected the pollen germination and therefore must be optimized for different species (Parton et al. 2002; Bou Daher et al. 2009; Mortazavi et al. 2010; Borghezan et al. 2011). Sucrose and boric acid in the culture medium were essential for pollen germination (Borghezan et al. 2011). Sucrose provided the osmotic pressure and the energy to promote pollen tube growth, and a sucrose level of 10–15% in the medium was sufficient for pollen germination. Boric acid improved germination, perhaps formed an ionizable sugar-boron complex (Soares et al. 2015). The calcium concentration affected pollen tube rigidity (Bhojwani and Bhatnagar 1974) and it had significant effects on pollen germination levels (Mortazavi et al. 2010). In addition, the consistency of the medium was a critical factor for successful in vitro germination. Ganeshan and Sulladmath (1983) found that hanging drop methods were preferable for Citrus pollen germination. Kundu et al. (2014) indicated that in vitro germination levels of cryopreserved C. sinensis pollen obtained on solid and liquid germination media showed no significant differences. In the current study, a high concentration of calcium nitrate (1.0 g L−1) in M1 germination medium resulted in a relatively low germination level, which may have led to pollen rupture. Pollen germination assays conducted on solid medium (M2 and M4) were found to promote higher germination.
Studies on the effects of rehydration treatment after cryopreservation and warming on germination levels are relatively scarce. Rehydration after warming was deemed necessary for some species (Parton et al. 2002). For example, incubation of the pollinia from Apocynaceae on slides in a humidity chamber (relative humidity = 90%) for 4–5 h was practical and effective for germination (Shashikumar 2006). In previous studies of pollen cryopreservation in Citrus, the effects of rehydration on in vitro germination levels were not investigated. In the current study, the in vitro germination levels of the cryopreserved anthers were significantly affected by the rehydration treatment. The highest average in vitro germination levels for C. cavaleriei ‘2–3’ (84%) and ‘2586’ (79%) were obtained when the cryopreserved anthers were warmed and rehydrated. For C. maxima, the relatively high average in vitro germination levels for ‘Kui’ (71%) and ‘Zuoshi’ (60%) were obtained when the cryopreserved anthers were warmed and rehydrated.
Comparison studies of cryopreservation and low temperature storage
Pollen can be stored at 4, − 20°C, and LN temperature. However, cryopreservation significantly extended the lifespan of stored pollen, for instance in studies for litchi (Litchi chinensis), kiwi (Apteryx spp.), date palm (Phoenix dactylifera), and Dendrobium hybrids (Vendrame et al. 2008; Mortazavi et al. 2010; Borghezan et al. 2011; Wang et al. 2015). In the current study, the in vitro germination levels of Citrus pollen of all ten cultivars were 0% after 2 yr of storage at 4°C, whereas the cryopreserved Citrus pollen was viable, with germination levels ranging from 22 to 93%. Kundu et al. (2014) reported that the highest in vitro germination levels (53 and 55%) were from cryopreserved C. sinensis pollen. In the current study, the highest germination levels (93%) were obtained for pollen from C. cavaleriei ‘2–3’ after cryostorage for 2 yr. The cryopreserved pollen from five cultivars (two of C. cavaleriei and three of C. maxima) after 2 yr of cryostorage showed in vitro germination levels of 50–93%. The in vitro germination levels of the cryopreserved pollen from five other cultivars (of C. maxima) ranged from 22 to 38%. Furthermore, the in vitro germination levels of three cultivars, i.e., C. cavaleriei ‘2–3’ and C. maxima ‘Beibei’ and ‘Sijipao’, were significantly increased to 93, 55, and 65%, respectively. The increased germination levels after cryostorage that were obtained for these cultivars may result from ultra-cold stimulation of pollen, which has been observed in other studies (Barnabas and Raiki 1976; Van Der Walt and Littlejohn 1996; Karun et al. 2006; Zhang et al. 2009).
Species- and cultivar-specific responses in cryopreservation of Citrus anthers
The differential responses to cryo-exposure among pollen could be attributed to their inherent genetic traits, physiological status, and the level of maturity among the samples collected (Normah et al. 2013). The present study suggested that the differences in the in vitro germination levels of cryopreserved pollen between the two Citrus species were mainly due to genotypic variation. In the field, C. cavaleriei cultivars can tolerate low temperatures, whereas C. maxima cultivars are generally more tropical (unpublished data). In the current study, the in vitro germination levels showed significant differences between tropical and temperate species. The in vitro germination levels for C. cavaleriei ‘2–3’ and ‘2586’ anthers after cryopreservation (> 79%) were significant higher than those obtained for the eight C. maxima cultivars (< 65%). C. maxima ‘14–7’, ‘Beibei’, ‘Zuoshi’, ‘Humi’, ‘Kui’, ‘Jintang’, and ‘Guanxiang’ cultivars were from Sichuan Province and Chongqing City, and they may have relatively high levels of cold tolerance. Moreover, it was found in a previous study that pollen of C. maxima cultivars originated from the tropical regions in Southeast Asia, namely ‘Qiangdele’, ‘Menglunzao’, ‘Wanbai’, ‘Wendan’, and ‘Shaokemokete’, showed extremely low viability after cryopreservation (< 20%, unpublished data).
The most effective warming temperature and rehydration treatments for the cryopreserved pollen of each cultivar varied. For example, for C. maxima ‘Kui’ and ‘Zuoshi’, rehydration treatment after warming at 37°C significantly promoted pollen germination; for C. maxima ‘Humi’, rehydration treatment after warming at 37°C significantly decreased the in vitro germination level; and for C. maxima ‘Jintang’, rehydration treatment after warming at 25°C significantly increased the in vitro germination level. The cultivar-specific responses to the treatment conditions were in agreement with the findings of Kundu et al. (2014) from other Citrus species.
Applications of cryopreservation protocols for Citrus anthers/pollen in genebank
The Pollen Cryobank can provide a constant supply of viable and fertile pollen to allow supplementary pollinations for breeding research and can become an important component of national genebanks. In India, pollen from more than 650 accessions belonging to 40 species was conserved in NBPGR and the Indian Institute of Horticultural Research, and pollen from 64 accessions belonging to C. limon, Citrus aurantifolia, and Pyrus trifoliata were cryopreserved (Mandal 2000; Ganeshan and Rajasekharan 2000); In the USA, the NCGRP conserves pollen of 13 pear (Pyrus spp.) cultivars and 24 Pyrus species (Reed et al. 2000). Based on results in the current study, improved air-desiccation cryopreservation protocols for C. cavaleriei and C. maxima anthers is now used in the National Crop Genebank of China (Beijing, CAAS). The study reinforces that Citrus anthers with the moisture content of 5–14% were critical for successful cryopreservation. Considering the effects of the warming, rehydration, and in vitro germination medium formulations on the in vitro germination, it is practical and feasible to warm the cryopreserved Citrus anthers with a moisture content of lower than 10% at 25°C and a moisture content of 10% or higher at 37°C, respectively, and then rehydrate and culture the pollen on M2 medium. In addition, the genetic traits of species or cultivars of C. cavaleriei and C. maxima will benefit by the large-scale application of cryopreservation protocols in genebanks.
Conclusions
This study identified optimal air-desiccation cryopreservation protocols for C. cavaleriei and C. maxima anthers. The Citrus anthers were dehydrated to the moisture content of 5–14% before LN exposure and warmed at 25°C (cryopreserved Citrus anthers with a moisture content of lower than 10%) or 37°C (a moisture content of 10% or higher), then rehydrated, and cultured on solid germination medium containing 150-g L−1 sucrose, 0.1-g L−1 boric acid, 1.0-g L−1 calcium nitrate, 0.1-g L−1 potassium nitrate, 0.3-g L−1 magnesium sulfate, and 10-g L−1 agar. After 2 yr of storage, in vitro germination levels of Citrus pollen after cryopreservation were significantly higher (in vitro germination levels were higher than 22% for all ten cultivars) than those for anthers that were stored at 4°C (no viable pollen). In conclusion, C. cavaleriei and C. maxima can be successfully conserved as cryopreserved anthers in genebanks. These cryopreservation procedures will aid in the establishment of large-scale pollen cryopreservation protocols at the National Crop Genebank of China.
References
Al Zoubi OM, Normah MN (2012) Desiccation sensitivity and cryopreservation of excised embryonic axes of Citrus suhuiensis cv. limau madu, Citrumelo [Citrus paradisi macf. × Poncirus trifoliata (L.) raf.] and Fortunella polyandra. CryoLetters 33(3):241–251
Barbhuiya AR, Khan ML, Dayanandan S (2016) Genetic structure and diversity of natural and domesticated populations of Citrus medica L. in the Eastern Himalayan region of Northeast India. Ecol Evol 6(12):3898–3911
Barnabas B, Raiki E (1976) Storage of maize (Zea mays L.) pollen at − 196°C in liquid nitrogen. Euphytica 25(1):747–752
Benson EE (2008) Cryopreservation of phytodiversity: a critical appraisal of theory practice. Crit Rev Plant Sci 27:141–219
Bhojwani SS, Bhatnagar SP (1974) Fertilization. In: The embryology of angiosperms. New Delhi: Skylark Printers, pp 102–120
Borghezan M, Clauman AD, Steinmacher DA, Guerra MP, Orth AI (2011) In vitro viability and preservation of pollen grain of kiwi (Actinidia chinensis var. deliciosa (A. Chev.) A. Chev). Crop Breed Appl Biotechnol 11(4):338–344
Bou Daher F, Chebli Y, Geitmann A (2009) Optimization of conditions for germination of cold-stored Arabidopsis thaliana pollen. Plant Cell Rep 28(3):347–357
Chaudhury R, Malik SK, Rajan S (2010) An improved pollen collection and cryopreservation method for highly recalcitrant tropical fruit species of mango (Mangifera Indica L.) and litchi (Litchi Chinensis Sonn.) CryoLetters 31(3):268–278
Chen PL, He SA, Jin W (1990) Cryopreservation of pollen from Eucommia ulmoides Oliu. and Sinoj ackia xylocarpa Hu. Acta Bot Sin 32(4):288–291 (in Chinese)
Chen XL, Zhang JM, Xia X, Huang B, Lu XX (2013) Progress on cryopreservation state and research of plant germplasm resources. J Plant Genet Resour 14(3):414–427 (in Chinese)
Connor KF, Towill LE (1993) Pollen-handling protocol and hydration/dehydration characteristics of pollen for application to long-term storage. Euphytica 68:77–84
Dafni A, Firmage D (2000) Pollen viability and longevity: practical, ecological and evolutionary implications. Plant Syst Evol 222(1):113–132
dos Santos ARA, de Souza EH, Souza FVD, Fadini M, Girardi EA, Soares WD (2015) Genetic variation of Citrus and related genera with ornamental potential. Euphytica 205(2):503–520
Dulloo ME, Hunter D, Borelli T (2010) Ex situ and in situ conservation of agricultural biodiversity: major advances and research needs. Not Bot Horti Agrobot Cluj-Napoca 38:123–355
Engelmann F (2011) Use of biotechnologies for the conservation of plant biodiversity. In Vitro Cell Dev-Plant 47:5–16
FAO (2015) FAOSTAT. Agricultural statistics database. World Agricultural Information Center, 2009 Rome. http://faostat.fao.org/site/567/default.aspx#ancor
Ganeshan DS, Rajasekharan PE, Shashikumar SS, Decruze SW (2008) Cryopreservation of pollen. In: Reed (ed) Plant cryopreservation: a practical guide. Springer, Berlin, pp 443–464
Ganeshan S (1985) Cryogenic preservation of grape pollen. Vitis 24:169–173
Ganeshan S (1986) Cryogenic preservation of papaya pollen. Sci Hortic 28:65–70
Ganeshan S, Alexander MP (1991) Cryogenic preservation of lemon (Citrus limon Burm.) pollen. Hortic Sci 56:228–230
Ganeshan S, Rajasekharan PE (2000) Current status of pollen cryopreservation research: relevance to tropical horticulture. Cryopreservation of tropical plant germplasm programs and application JIRCAS/IPGRI publication (Eds: Engelmann and H.Takagi), pp 360–365
Ganeshan S, Sulladmath VV (1983) Pollen storage studies on Citrus limon Burm. varietal differences and influence of flower types. Hortic Sci 48(2):51–54
Hanna WW, Towill LE (1995) Long-term pollen storage. Plant Breed Rev 13:179–207
Hao YJ, You CX, Deng XX (2002) Effects of cryopreservation on developmental competency, cytological and molecular stability of Citrus callus. CryoLetters 23:27–35
Karun A, Sajini KK, Nair M, Kumaran PM, Samsudheen K (2006) Cryopreservation of coconut (Cocos nucifera L.) pollen. J Plant Crops 34(3):568–571
Karun A, Sajini KK, Niral V, Amarnth CH, Remya P, Rajesh MK, Samsudeen K, Jerard BA, Engelmann F (2014) Coconut (Cocos Nucifera L.) pollen cryopreservation. CryoLetters 35(5):407–417
Kundu M, Dubey A, Srivastav M, Malik S, Singh B (2014) Effect of gamma ray irradiation and cryopreservation on pollen stainability, in vitro germination, and fruit set in Citrus. Turk J Biol 38(1):1–9
Lambardi M, Halmagyi A, Benelli C, De CA, Vettori C (2007) Seed cryopreservation for conservation of ancient Citrus germplasm. Adv Hortic Sci 21:198–202
Luza JG, Polito VS (1988) Cryopreservat ion of English walnut ( Juglans regia L. ) pollen. Euphytica 37:141–148
Malik SK, Chaudhury R (2006) The cryopreservation of embryonic axes of two wild and endangered Citrus species. Plant Genet Resour 4(3):204–209
Malik SK, Chaudhury R, Pritchard HW (2012) Long-term, large scale banking of citrus species embryos: comparisons between cryopreservation and other seed banking temperatures. CryoLetters 33(6):453–464
Mandal BB (2000) Cryopreservation research in India: current status and future perspectives. In: Engelmann F, Takagi H (eds) Cryopreservation of tropical plant germplasm—current research progress and applications. IPGRI/JIRCAS, Rome/Tsukuba, pp 282–286
Mortazavi SMH, Arzani K, Moieni A (2010) Optimizing storage and in vitro germination of date palm (Phoenix dactylifera) pollen. J Agric Sci Technol 12(2):181–189
Nepi M, Cresti L, Guarnieri M, Pacini E (2010) Effect of relative humidity on water content, viability and carbohydrate profile of Petunia hybrida and Cucurbita pepo pollen. Plant Syst Evol 284(1–2):57–64
Normah MN, Chin HF, Reed BM (2013) Conservation of tropical plant species. Springer, New York, pp 65–76
Omura N, Akihama TA (1980) Pollen preservation of fruit trees for genebanks in Japan. Plant Genet Resour Newsl 1988:28–31
Parton E, Vervaeke I, Delen R, Vandenbussche B, Deroose R, Proft MD (2002) Viability and storage of bromeliad pollen. Euphytica 125(2):155–161
Popova E, Kim HH, Saxena PK, Engelmann F, Pritchard HW (2016) Frozen beauty: the cryobiotechnology of orchid diversity. Biotechnol Adv 34(4):380–403
Rajasekharan PE, Ganeshan S, Thamizharasu V (1995) Expression of trifoliate leaf character in Citrus limonia x Poncirus trifoliata hybrids through cryostored pollen. J Hortic Sci 70(3):485–490
Reed BM, DeNoma J, Chang Y (2000) Application of cryopreservation protocols at a clonal genebank. In: Engelmann F, Takagi H (eds) Cryopreservation of tropical plant germplasm—current research progress and applications. IPGRI/JIRCAS, Rome/Tsukuba, pp 246–249
Rohini MR, Malik SK, Choudhary R, Kaur S, Uchoi A, Chaudhury R (2016) Storage behavior and cryopreservation studies in Indian rough lemon (Citrus jambhiri): a promising rootstock for long-term conservation. Turk J Agric For 40:865–873
Sacks EJ, St Clair DA (1996) Cryogenic storage of tomato pollen: effect on fecundity. Hortscience 31(3):447–448
Shashikumar S (2006) Pollen biology of few horticulturally important plants. PhD thesis. Bangalore University, Bangalore
Soares TL, de Souza EH, de Carvalho Costa MAP, de Oliveira S, dos Santos-Serejo JA (2015) Viability of pollen grains of tetraploid banana. Bragantia 75:145–151
Song CM, Li QH (2015) Study on germination rate of in vitro culture and cryopreservation for Prunus pseudocerasu cv. ‘Yanhong’. Seed 34(11):92–99
Towill LE (1985) Low temperature and freeze/vacuum-drying preservation of pollen. In: Kartha KK (ed) Cryopreservation of plant cells and organs. CRC Press, Boca Raton, pp 171–198
Van Der Walt D, Littlejohn GM (1996) Storage and viability testing of protea pollen. J Am Soc Hortic Sci 121(5):804–809
Vendrame WA, Carvalho VS, Dias JMM, Maguire I (2008) Pollination of Dendrobium hybrids using cryopreserved pollen. Hortscience 43(1):264–267
Volk GM, Bonnart R, Shepherd A, Yin ZF, Lee R, Polek M, Krueger R (2017) Citrus cryopreservation: viability of diverse taxa and histological observations. Plant Cell Tissue Organ Cult 128(2):327–334
Wang CH, Li JR (1996) Study of low and super low temperature storage in pollens of apricot. J Laiyang Agric Coll 13(2):169–173 (in Chinese)
Wang LM, Wu JF, Chen JZ, Fu DW, Zhang CY, Cai CH, Ou LX (2015) A simple pollen collection, dehydration, and long-term storage method for litchi (Litchi chinensis Sonn.) Sci Hortic 188:78–83
Xu J, Li L, Liu Q, Shi Y, Peng JG, Jia MX, Liu Y (2014) Wide-scale pollen banking of ornamental plants through cryopreservation. CryoLetters 35(4):312–319
Yan Q, Wen B, Zhang N, Yin SH, Ji MY (2014) Cryopreservation strategies for pomelo seeds from Xishuangbanna, South China. Seed Sci Technol 42(2):202–213
Yang XM, Li H, Liang M, Xu Q, Chai LJ, Deng XX (2015) Genetic diversity and phylogenetic relationships of citron (Citrus medica L.) and its relatives in southwest China. Tree Genet Genomes 11(6):129
Zhang JM, Xin X, Yin GK, Lu XX, Chen XL (2014) In vitro conservation and cryopreservation in National Crop Genebank of China. Acta Hortic 1039:309–317
Zhang YL, Chen RD, Huang CJ, Liu Y (2009) Cryo-banking of Prunus mume pollen and its application in cross-breeding. CryoLetters 30(3):165–170
Zhang YL, Shang XQ, Liu Y (2006) Advances in research of pollen cryopreservation. J Beijing Forest Univ 28(4):139–147 (in Chinese)
Acknowledgments
This study was supported by the Utilization Special Grant from the Ministry of Agriculture (Grant number: 2016NWB036-09), the Agricultural Science and Technology Innovation Program/Crop Germplasm Resources Preservation and Sharing Innovation Team (CAAS, ASTIP), the National Key Technology R&D Program (Grant number: 2013BAD01B01), and the Crop Germplasm Resources Protection, Cooperation Research on Collecting Techniques and Practice in Crop Genebank between China and United States of America (2014DFG31860). The authors would like to acknowledge Gayle M Volk (National Center for Genetic Resources Preservation, United States Department of Agriculture, Fort Collins, Colorado, USA) for assistance in revisions of the manuscript.
Author information
Authors and Affiliations
Contributions
Jin-Mei Zhang designed and performed research, analyzed data, and wrote the manuscript. XX, GKY, JJH, and BH participated in the acquisition of the original data and the analysis of the data. XXL and DJ participated in the revision of the manuscript. XLC designed research and revised the manuscript. All the authors have read and approved the final manuscript.
Corresponding author
Additional information
Editor: Barbara Reed
Rights and permissions
About this article
Cite this article
Zhang, JM., Lu, XX., Xin, X. et al. Cryopreservation of Citrus anthers in the National Crop Genebank of China. In Vitro Cell.Dev.Biol.-Plant 53, 318–327 (2017). https://doi.org/10.1007/s11627-017-9848-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11627-017-9848-z