Abstract
Background
Data evaluating the financial implications of volume-based referral are lacking. This study sought to compare in-hospital costs for pancreatic surgery by annual hospital volume.
Methods
Eleven thousand and eighty-one patients aged ≥18 years undergoing an elective pancreatic resection for cancer were identified using the Nationwide Inpatient Sample 2002–2011. Multivariable regression analysis was performed to compare length-of-stay (LOS), postoperative morbidity and mortality, failure-to-rescue (FTR), and inpatient costs by annual hospital volume group.
Results
Patients undergoing surgery at high-volume hospitals (HVH) demonstrated 23% lower odds (odds ratio [OR] = 0.77, 95% confidence interval [95%CI] 0.63–0.95) of developing a postoperative complication, 59% lower odds of experiencing an LOS > 14 days (OR = 0.41, 95%CI 0.34–0.50), 51% lower odds of postoperative mortality (OR = 0.49, 95%CI 0.34–0.71), and 47% lower odds of FTR (OR = 0.53, 95%CI 0.37–0.76; all p<0.05). The overall mean in-hospital cost was $39,012 (SD = $15,214) with minimal differences observed across hospital volume groups. Rather, postoperative complications (no complication vs. complication $26,686 [SD = $5762] vs. $44,633 [SD = $11,637]) and FTR (rescue vs. FTR $42,413 [SD = $8481] vs. $69,546 [SD = $13,131]) were determinant of higher in-hospital costs. While this pattern was observed at all hospital volume groups, costs varied minimally between hospital volume groups after this stratification.
Conclusions
Annual hospital surgical volume was not associated with in-hospital costs among patients undergoing pancreatic surgery.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The volume-outcome relationship was initially described in 1979 by Luft and colleagues.1 In their seminal work, the authors demonstrated an association between increasing hospital volume and improved postoperative mortality.1 Since then, a large body of literature has similarly described an inverse relationship between hospital volume and postoperative clinical outcomes, resulting in the regionalization of complex, high-risk operations to high-volume hospitals.2–6 Although the volume-outcome relationship is well-established and has remained consistent over time, the financial implications associated with the volume-based referral of patients remain largely unexplored.7–11 Referral to high-volume hospitals may be associated with a higher costs of care given the greater resource utilization associated with caring for more complex patients as well as delivering a higher quality of care.10,11 In particular, these costs may include the costs associated with maintaining intensivist-staffed ICUs, high nurse-to-bed ratios, and advanced technologies and specialist services among others.10 Alternatively, referral to high-volume hospitals may be associated with lower costs via subsidies achieved through economies of scale or via potential savings achieved through the delivery of higher quality care and prevention of costly postoperative complications.11–13
Pancreatic surgery represents a complex, high-risk operation which is frequently associated with a high postoperative morbidity and mortality.14,15 Often the only treatment option for patients with pancreatic disease, patients undergoing pancreatic resection have been targeted for volume-based referral to high-volume hospitals in favor of improved postoperative outcomes.16–18 In a recent systematic review and meta-analysis assessing the volume-outcome relationship in pancreatic surgery, Gooiker and colleagues reported that pancreatic surgery performed at high-volume hospitals was associated with a 68% lower odds of postoperative mortality.17 Similarly, van Heek et al., in their pooled analysis, reported that patients undergoing surgery at high-volume hospitals were associated with a 24% lower risk of postoperative mortality.18 In contrast to the well-established volume-outcome relationship for pancreatic surgery, limited data exist evaluating the relationship between hospital costs and referral to high-volume centers. Current studies are limited to Medicare patients aged >65 years, or state-specific analyses, and therefore are not generalizable to all patient populations /hospitals.19,20 Given the need for nationally representative estimates for the potential financial implications of volume based referral, the objective of the current study was to examine the relationship between inpatient costs and annual hospital volume using a nationally representative sample of patients undergoing a pancreatic resection.
Methods
Data Sources and Patient Population
The current study utilized data from the Nationwide Inpatient Sample (NIS) from 2002 to 2011. Maintained by the Agency for Healthcare Research and Quality (AHRQ), the NIS represents the largest inpatient, all-payer claims based database in the USA.21 Using a stratified sampling methodology, the NIS is a 20% representative sample of all inpatient admissions in the United States.21 Patients undergoing a pancreatic resection were identified using relevant International Classification of Disease, Ninth Revision (ICD-9-CM) procedure codes (“52.51,” “52.52,” “52.53,” “52.59,” “52.6,” and “52.7”). To ensure the comparability of our patient cohort, only patients ≥18 years and patients undergoing surgery for a primary diagnosis of pancreatic cancer (ICD-9-CM diagnosis codes “157,” “157.0,” “157.1,” “157.2,” “157.3,” “157.4,” “157.8,” “157.9,” and “197.8”) were included in our final study population; patients undergoing non-elective surgery, and patients transferred in from another hospital were excluded from the final study cohort (Supplemental Fig. 1). Given the change in sampling methodology for patients included in the NIS following 2011, the current analysis included only patients who underwent pancreatic surgery before 2012.21 As this study utilized deidentified data, patient consent was not required and this study was approved by the Johns Hopkins University Institutional Review Board.
For each patient, baseline characteristics including patient age, sex, race, insurance status, and socioeconomic status (defined using the median household income for the patient’s zip code) were recorded. Preoperative patient comorbidity was classified using the Charlson Comorbidity Index (CCI), and patients were categorized into three groups based on their CCI score; CCI = 2, CCI = 3–6, and CCI >6.22 Additionally, hospital level characteristics including hospital bed size, hospital region, hospital teaching status, and hospital urban vs. rural location were recorded for each patient. For each hospital, an annual hospital volume was calculated as the sum of all pancreatic resections performed for each year of available data. Hospitals were then categorized into one of three volume groups; low-volume hospitals (1–8 pancreatic resections/year), intermediate-volume hospitals (9–30 pancreatic resections/year), and high-volume hospitals (≥31 pancreatic resections/year). These cutoffs were selected such that approximately a third of all pancreatic resections were performed within each hospital volume strata as previously described.3,19
Clinical Outcomes: Postoperative Morbidity, Length-of-Stay, and Postoperative Mortality
Clinical outcomes of interest for the current study included postoperative morbidity, hospital length-of-stay (LOS) for the index admission, in-hospital mortality, and failure-to-rescue. Postoperative morbidity was defined as the development of one or more postoperative complications defined using a previously validated list of conditions including pneumonia, respiratory failure, acute myocardial infarction, stroke, venous thromboembolism (deep venous thrombosis and pulmonary embolism), renal failure, gastrointestinal bleeding, surgical site infections, sepsis, and wound dehiscence.23,24 LOS was reported and analyzed as a continuous variable, and was also examined as a binary variable by dichotomizing LOS at the 75th percentile to represent a prolonged/extended LOS (eLOS).25 This method has been previously validated for use in health services research and has been shown to be associated with resource use and variations in surgical quality between and within hospitals, and is comparable to previous studies evaluating outcomes for pancreatic surgery.25,26 Postoperative mortality was defined by any inpatient mortality, while failure-to-rescue was calculated as the proportion of patients who died following the development of one or more postoperative complication.24
Financial Outcomes: Total In-hospital Costs
Total in-hospital costs were estimated from total in-hospital charges for the index admission using a method previously defined and validated by the AHRQ.27 Total in-hospital charges are gathered and reported directly from submitted claims. To account for differences and variations in reporting, submitted charges undergo a review by the AHRQ to ensure consistency of results and so as to exclude large outliers.27 Specifically, all values are reported to the nearest dollar with total charges less than or equal to zero being set to missing. Further, outliers as defined by total charges between $25 and $1.0 million, for years 1998 to 2006; between $100 and $1.5 million, for years 2007 to 2010; and between $100 and $5.0 million, for years 2011 and later, are removed/not reported.27 In general, these charges do not include professional fees and non-covered charges. In the instance where the source provides total charges with professional fees, the professional fees are removed from the charge during processing by the AHRQ.27
Total in-hospital charges as reported by the AHRQ were first inflation adjusted using the US Department of Labor inflation calculator and reported to 2016 dollars.28 Next, inflation adjusted charges were multiplied by hospital specific cost-to-charge ratios to calculate the total in-hospital cost as previously described by the AHRQ.27 Cost-to-charge ratios represent the ratio between the cost and charge and are calculated using data obtained from the CMS Healthcare Cost Reporting Information System (HCRIS) reports which include data on hospital charges, net revenues, expenses, and payer mix for each hospital.29
Statistical Analysis
Categorical variables were reported as whole numbers with percentages and compared using Pearson’s chi-squared test. Continuous variables were reported as means with standard deviations (SD) or as medians with corresponding interquartile range (IQR) and compared using a one-way analysis of variance (ANOVA) with Bonferroni correction or the Kruskal-Wallis test, as appropriate. Multivariable logistic regression analysis was performed to explore the relationship between hospital volume and binary postoperative clinical outcomes (postoperative morbidity, eLOS, postoperative mortality, and failure-to-rescue). All multivariable logistic regression analyses adjusted for patient age, sex, race, CCI score, insurance, socioeconomic status, use of minimally invasive surgery, type of pancreatic resection, hospital region, and hospital location. Results from multivariable logistic regression analyses were reported as odds ratios (OR) with corresponding 95% confidence intervals (95%CI).
To examine the association between hospital volume and total in-hospital costs, exploratory data analysis was first performed to examine the underlying distribution of financial data. As total in-hospital costs were right-skewed, financial data were log-transformed to obtain a more symmetrical distribution. Log-transformed costs were then entered into a multivariable linear regression analysis that adjusted for patient characteristics including patient age; sex; race; preoperative comorbidity defined by the CCI, insurance status, and income quartile; and operative characteristics including the type of pancreatic resection and the operative approach (open vs. MIS resection). Given differences in reimbursement methodologies and therefore costs, our multivariable model also accounted for hospital level characteristics including hospital region, and hospital teaching status. Additionally, given that postoperative outcomes may also affect hospital costs, our final model also adjusted for hospital LOS, in-hospital mortality, and the development of one or more postoperative complication. Results from the multivariable linear regression were then used to calculate covariate adjusted, log-transformed costs which in turn were then exponentiated to calculate covariate adjusted costs reported to the nearest dollar. Robust standard errors were used for all multivariable analyses to account for clustering of patients within individual hospitals.
To adjust for any potential differences in clinical and financial outcomes by the type of surgical resection, additional sensitivity analyses were performed among patients who underwent an open pancreaticoduodenectomy. Comparable analyses could not be performed for other types of pancreatic resections given inadequate statistical power due to a limited number of events. For all analyses, statistical significance was defined as p < 0.05. All analyses were performed using STATA version 14.0 statistical software (StataCorp, College Station, TX).
Results
Patient and Hospital Characteristics by Hospital Volume Group
A total of 11,081 patients were identified who met inclusion criteria. The median age of all patients was 66 years (IQR 57–73) with an equal number of male and female patients included in the final study population (Table 1). Approximately two thirds of the patient population were Caucasian (n = 7355, 66.4%), while African-American patients and non-white Hispanics comprised 6.3% (n = 697) and 5.3% (n = 586) of the patient cohort, respectively. Comorbidities were commonly observed with 51.4% (n = 5693) of patients presenting with a CCI score between 3 and 6. Medicare was the most common payor accounting for 50.1% (n = 5550) of patients followed by private payors (n = 4592, 41.5%) and Medicaid (n = 471, 4.3%). A majority of patients underwent a pancreaticoduodenectomy (n = 7290, 65.8%) while a distal pancreatectomy was performed in 24.6% of patients (n = 2721); minimally invasive surgery was performed in 7.6% (n = 844) of patients.
Approximately, one third of patients underwent a pancreatic resection at either a low-, an intermediate-, or a high-volume hospital (Supplemental Fig. 2). In contrast, 84.0% (n = 1540) of hospitals were categorized as low-volume hospitals, while 12.4% (n = 228) and 3.6% (n = 66) of hospitals were categorized as either intermediate- or high-volume hospitals, respectively. Of note, a significant increase in the regionalization of care was observed over the study time period with the number of patients undergoing surgery at a low-volume hospital decreasing from 47.3% (n = 1195) in the time period between 2002 and 2004 to 23.6% (n = 1026) in the time period between 2009 and 2011 (Supplemental Fig. 3). Conversely, the proportion of patients undergoing surgery at a high-volume hospital increased from 19.2% (n = 486) between 2002 and 2004 to 44.0% (n = 1911) between 2009 and 2011.
While patient age, sex, and insurance status were comparable between the three hospital volume groups, several differences in patient and operative characteristics were observed across these strata. Specifically, patients undergoing surgery at a high-volume hospital were proportionally more likely to be Caucasian (low vs. intermediate vs. high 59.7 vs. 65.0 vs. 74.7%; p < 0.001), more likely to undergo a pancreaticoduodenectomy (60.4 vs. 67.9 vs. 69.3%; p < 0.001), and were proportionally more likely to undergo surgery via a minimally invasive approach (5.0 vs. 6.2 vs. 11.8%; p < 0.001). Similarly, differences in hospital level characteristics were also observed across the three volume groups with low-volume hospital being more likely to be located in a rural setting (4.0 vs. 3.4 vs. 0.0%) and being designated as a non-teaching hospital (40.8 vs. 7.5 vs. 1.4%; p < 0.001) compared with intermediate- and high-volume hospitals.
Postoperative Clinical Outcomes and the Volume-Outcome Relationship
Among all patients, 37.9% (n = 4197) of patients developed one or more postoperative complication with postoperative morbidity being highest at low-volume hospitals compared with intermediate and high-volume hospitals (41.3% vs. 38.6% vs. 33.7%; p < 0.001; Table 2). After adjusting for patient and hospital level characteristics, patients undergoing surgery at a high-volume hospital demonstrated 23% lower odds (OR = 0.77, 95%CI 0.63–0.95; p = 0.014) of developing a postoperative complication compared with patients undergoing surgery at a low-volume hospital (Table 3; Supplementary Table 1). A similar volume-outcome relationship was also observed when evaluating LOS, and postoperative mortality. Specifically, the proportion of patients experiencing an eLOS (LOS > 14 days 31.4 vs. 24.3% vs. 17.4%; p < 0.001), postoperative mortality (5.2 vs. 3.1 vs. 1.9%; p < 0.001), and failure-to-rescue (11.1 vs. 7.1 vs. 5.4%; p < 0.001) were all observed to decrease with an increase in annual hospital volume. This effect was also observed on multivariable analysis with patients undergoing surgery at a high-volume hospital demonstrating 59% lower odds of experiencing an eLOS (OR = 0.41, 95%CI 0.34–0.50; p < 0.001; Supplemental Table 2), 51% lower odds of postoperative mortality (OR = 0.49, 95%CI 0.34–0.71; p < 0.001; Supplemental Table 3), and 47% lower odds of failure-to-rescue (OR = 0.53, 95%CI 0.37–0.76; p = 0.001; Supplemental Table 4). On a sensitivity analysis among patients undergoing an open pancreaticoduodenectomy, a similar volume-outcome relationship was also observed with patients undergoing an open pancreaticoduodenectomy at a high-volume hospital demonstrating a 28% lower odds of developing a postoperative complication (OR = 0.72, 95%CI 0.57–0.92), 65% lower odds of experiencing an eLOS (OR = 0.35, 95%CI 0.28–0.44), a 63% lower odds of dying during the inpatient admission (OR = 0.37, 95%CI 0.25–0.55), and a 50% lower odds of failure-to-rescue (OR = 0.50, 95%CI 0.32–0.77) compared with patients undergoing surgery at a low-volume hospital (Supplementary Table 6).
Total Hospital Costs by Hospital Volume Group
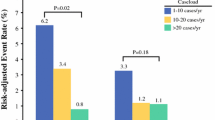
The mean covariate-adjusted inpatient cost for all patients was $39,012 (SD = $15,214). While average adjusted costs were observed to be only slightly higher at low-volume hospitals compared to high-volume hospitals ($40,276 [SD = $16,648] vs. $38,969 [SD = $13,687], ∆ = −3.2%; Supplemental Table 5), the average adjusted costs were almost twofold higher among patients who developed a postoperative complication compared with those who did not (no complication vs. complication $26,686 [SD = $5762] vs. $44,633 [SD = $11,637], ∆ = +67.3%; p < 0.001; Supplemental Fig. 4); this pattern was consistently observed across all hospital volume groups (Fig. 1). Interestingly, despite this stratification by the development of a postoperative complication, minimal differences in the average adjusted costs were observed across hospital volume groups.
To further understand the relationship between inpatient costs, hospital volume and postoperative complications, patients were stratified according to whether they were rescued following a postoperative complication. Among all patients who developed a postoperative complication, patients who were rescued demonstrated markedly lower covariate-adjusted costs compared with patients who died following a postoperative complication, which is failure-to-rescue (rescue vs. failure-to-rescue $42,413 [SD = $8481] vs. $69,546 [SD = $13,131], ∆ = +64.0%; p < 0.001, Fig. 2). Of note, while this trend was observed at all hospital volume groups, inpatient costs varied minimally between hospital volume groups after this stratification. Furthermore, a similar pattern in covariate-adjusted costs was also observed on sensitivity analysis performed among patients who underwent an open pancreaticoduodenectomy with the development of a postoperative complication and failure-to-rescue being strongly associated with higher covariate-adjusted costs (Supplementary Table 7).
Discussion
Although the well-established volume-outcome relationship for pancreatic surgery has led to policy makers and healthcare administrators to advocate for the regionalization of surgery, limited data exist evaluating the financial implications of volume-based referral.14,17,18 In the current study, we sought to compare inpatient hospital costs relative to hospital volume for patients undergoing pancreatic surgery for a primary diagnosis of cancer. In our analysis of 11,081 patients undergoing pancreatic surgery, we observed that high-volume hospitals were associated with improved in-hospital clinical outcomes in the form of improved postoperative morbidity, a shorter length-of-stay, a lower failure-to-rescue rate, and consequently a lower in-hospital mortality. In contrast, covariate-adjusted costs were observed to be comparable across all hospital volume groups. Rather, the development of a postoperative complication and failure to be rescued following the postoperative complication was associated with a higher in-hospital cost following pancreatic surgery. Interestingly, in a stratified analysis, while differences in costs were observed by the development of a postoperative complication and failure-to-rescue, in-hospital costs remained comparable across all hospital volume groups.
Results of the current study are consistent with previous literature evaluating the volume-outcome relationship for patients undergoing pancreatic surgery. Specifically, a recent series of systematic reviews and meta-analyses demonstrated that high-volume hospitals were associated with a 24–68% lower mortality compared with low-volume hospitals.17,18 Similarly, previous research from our group has also demonstrated that high-volume hospitals are associated with a lower odds of failure-to-rescue and therefore an improved postoperative mortality.30 Collectively, while these data have supported calls for the regionalization of pancreatic surgery, the financial consequences of such policies remain largely undetermined. High-volume hospitals may achieve the observed improved surgical outcomes at significantly higher costs due to greater utilization of resources.10 In turn, these costs may be passed on to payors or patients resulting in an overall increase in healthcare spending due to volume based referral of patients. Alternatively, referral to high-volume hospitals may be able to decrease the overall costs of care via quality improvement and prevention of costly postoperative complications or via subsidies achieved through economies of scale.10,31,32 Interestingly, in the current study, we observed no clinically significant differences in the covariate-adjusted in-hospital costs between high-, low-, and intermediate-volume hospitals with in-hospital costs varying by 3.0–6.0% across hospital volume groups. Consistent with our findings, Nathan et al. reported comparable costs between low- and high-volume hospitals in their analysis of Medicare enrollees undergoing pancreatic surgery for cancer.19 Furthermore, Ho and colleagues in their statewide analysis of patients undergoing pancreatic surgery also reported no differences in costs between hospital volume groups.20 Taken together with previous reports, results of the current study suggest that although the volume based referral may result in improved postoperative outcomes, the improved clinical outcomes may not translate to significant cost savings as a result of volume based referral strategies.
In the current study, rather than hospital volume, other postoperative clinical factors were associated with an increase in in-hospital costs following pancreatic surgery. For example, the development of a postoperative complication was associated with a 67.3% in the total in-hospital cost. These findings are consistent with previous research from our own group as well as others demonstrating a strong association between postoperative complications and a higher in-hospital cost.12,13,33,34 Collectively, results of the current study reiterate the need to further refine processes of care so as to identify and/or prevent postoperative complication in a timely manner, examples of which may include the implementation of standardized practices and pathways.35,36 Perhaps more interestingly, we also observed significant differences in hospital costs when stratified by whether or not patients were rescued following a postoperative complication. Of note, patients who were rescued following the development of a postoperative complication demonstrated 64% lower costs than those patients who died following a postoperative complication—an effect that was uniformly observed across all hospital volume strata. Although hospitals are increasingly benchmarked and reimbursed on their ability to deliver high-quality care, limited data exist evaluating the financial impact of how well a hospital can manage patients who develop a postoperative complication. Results of the current study add to a limited body of evidence demonstrating an association between the failure-to-rescue and hospital costs, and are consistent with a recent report by Pradarelli and colleagues demonstrating wide variation in hospital payments by the ability of hospitals to rescue Medicare patients undergoing surgery.37,38 In their study, the authors reported wide variation in in-patient and postdischarge related payments and demonstrated that significant cost savings can be achieved by referring patients to hospitals demonstrating low costs for failure-to-rescue.37 Although the authors did not explicitly evaluate hospital characteristics associated with a lower cost of rescue, in our analysis, no clinically significant differences in in-hospital costs were observed relative to the hospital volume suggesting that although high-volume hospitals may achieve improved clinical outcomes, limited if any, cost savings would be achieved via volume-based referral to high-volume hospitals.37 As we move towards an era of value based payment and large, horizontal hospital networks, it remains to be seen whether limiting complex, high-risk operations to high-volume hospitals will result in the delivery of higher valued care to patients. Moving forward, further research is therefore required to better understand the microsystems within hospitals that lead to improved surgical care and to identify variations for future cost containment.
Results of the current study should be considered with several limitations. As with all analyses using administrative datasets, results of the current study are subject to the limitations inherent to administrative data that include potential discrepancies in coding practices and a lack of granular clinical details.39 Specifically, data pertaining to preoperative patient tumor stage, tumor grade, and invasion of and/or resection of adjacent structures was not available in the current dataset. As such, we were unable to adjust for these variable in our multivariable analysis and were unable to comment on differences in patient case mix between hospital volume groups and the effect of these differences in patient case mix on in-hospital costs. As a result of this, some residual confounding may exist in the analyses, although to minimize this, we identified a homogenous study population targeted for volume-based referral and utilized previously validated ICD-9-CM codes to identify postoperative complications.23 Second, given our use of administrative data, the severity/grade of postoperative complications could not be determined. For example, patients undergoing surgery at high-volume hospital may have incurred more severe postoperative complications compared with those undergoing surgery at low-volume hospitals, thereby offsetting any potential cost-savings that may have been achieved by quality improvement at high-volume hospitals. Furthermore, given that data pertaining to the exact area of spending is not recorded within the NIS, we were unable to identify specific areas of spending between hospitals. Specifically, in our analysis, we were unable to discern whether the driver of cost was the actual surgery or the postoperative management of these patients. Lastly, given that the NIS collects data only for the inpatient admission, long-term clinical outcomes such as survival and recurrence as well as patient-reported outcomes and measures of quality-of-life could not be compared between hospital volume groups. Additionally, financial data associated with postdischarge care and subsequent admissions could not be compared across hospital volume groups; however, previous studies have demonstrated minimal differences in postdischarge/postacute care spending between hospital volume groups.19
In conclusion, in our analysis of 11,081 patients, annual hospital surgical volume was not associated with in-hospital costs for pancreatic surgery. In contrast, in-hospital costs were observed to vary by the development of a postoperative complication and the ability of a hospital to successfully rescue a patient following the development of a postoperative complication. Costs associated with postoperative complications and rescue were comparable across hospital volume groups. Although policymakers are increasingly advocating for the regionalization of complex, high-risk surgery to high-volume hospitals, it remains to be seen whether these policies will translate in cost savings for patients and health systems alike.
References
Luft HS, Bunker JP, Enthoven AC. Should Operations Be Regionalized? N Engl J Med. 1979;301(25):1364–1369.
Birkmeyer JD, Siewers AE, Finlayson EVA, et al. Hospital volume and surgical mortality in the United States. N Engl J Med. 2002;346(15):1128–1137.
Buettner S, Gani F, Amini N, et al. The relative effect of hospital and surgeon volume on failure to rescue among patients undergoing liver resection for cancer. Surgery. 2016;159(4):1004–1012.
Birkmeyer JD, Finlayson E V, Birkmeyer CM. Volume standards for high-risk surgical procedures: potential benefits of the Leapfrog initiative. Surgery. 2001;130(3):415–422.
Schrag D, Panageas KS, Riedel E, et al. Hospital and surgeon procedure volume as predictors of outcome following rectal cancer resection. Ann Surg. 2002;236(5):583–592.
Schrag D, Cramer LD, Bach PB, Cohen AM, Warren JL, Begg CB. Influence of hospital procedure volume on outcomes following surgery for colon cancer. JAMA. 2000;284(23):3028–3035. http://www.ncbi.nlm.nih.gov/pubmed/11122590. Accessed May 1, 2015.
Reames BN, Ghaferi AA, Birkmeyer JD, Dimick JB. Hospital volume and operative mortality in the modern era. Ann Surg. 2014;260(2):244–251.
Finks JF, Osborne NH, Birkmeyer JD. Trends in hospital volume and operative mortality for high-risk surgery. N Engl J Med. 2011;364(22):2128–2137.
Gani F, Azoulay D, Pawlik TM. Evaluating Trends in the Volume-Outcomes Relationship Following Liver Surgery: Does Regionalization Benefit All Patients the Same? J Gastrointest Surg. November 2016.
Birkmeyer JD, Skinner JS, Wennberg DE. Will volume-based referral strategies reduce costs or just save lives? Health Aff (Millwood). 21(5):234–241. http://www.ncbi.nlm.nih.gov/pubmed/12224888. Accessed August 8, 2015.
Regenbogen SE, Gust C, Birkmeyer JD. Hospital Surgical Volume and Cost of Inpatient Surgery in the Elderly. J Am Coll Surg. 2012;215(6):758–765.
Gani F, Hundt J, Makary MA, Haider AH, Zogg CK, Pawlik TM. Financial Impact of Postoperative Complication Following Hepato-Pancreatico-Biliary Surgery for Cancer. Ann Surg Oncol. December 2015.
Eappen S, Lane BH, Rosenberg B, et al. Relationship between occurrence of surgical complications and hospital finances. JAMA. 2013;309(15):1599–1606.
Dudekula A, Munigala S, Zureikat AH, Yadav D. Operative Trends for Pancreatic Diseases in the USA: Analysis of the Nationwide Inpatient Sample from 1998-2011. J Gastrointest Surg. 2016;20(4):803–811.
Okunrintemi V, Gani F, Pawlik TM. National Trends in Postoperative Outcomes and Cost Comparing Minimally Invasive Versus Open Liver and Pancreatic Surgery. J Gastrointest Surg. 2016;20(11):1836–1843.
Datta J, Vollmer CM. Advances in Surgical Management of Pancreatic Diseases. Gastroenterol Clin North Am. 2016;45(1):129–144.
Gooiker GA, van Gijn W, Wouters MWJM, Post PN, van de Velde CJH, Tollenaar RAEM. Systematic review and meta-analysis of the volume-outcome relationship in pancreatic surgery. Br J Surg. 2011;98(4):485–494.
van Heek NT, Kuhlmann KFD, Scholten RJ, et al. Hospital volume and mortality after pancreatic resection: a systematic review and an evaluation of intervention in the Netherlands. Ann Surg. 2005;242(6):781–788, NaN-90. http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=1409869&tool=pmcentrez&rendertype=abstract. Accessed December 2, 2016.
Nathan H, Atoria CL, Bach PB, Elkin EB. Hospital volume, complications, and cost of cancer surgery in the elderly. J Clin Oncol. 2015;33(1):107–114.
Ho V, Aloia T. Hospital volume, surgeon volume, and patient costs for cancer surgery. Med Care. 2008;46(7):718–725.
HCUP Databases. Healthcare Cost and Utilization Project (HCUP). March 2017. Agency for healthcare research and quality. Rockville, MD. https://www.hcup-us.ahrq.gov/nisoverview.jsp#data. Accessed Jun 11, 2017.
Charlson ME, Pompei P, Ales KL, MacKenzie CR A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40(5):373–383. http://www.ncbi.nlm.nih.gov/pubmed/3558716. Accessed July 10, 2014.
Iezzoni LI, Daley J, Heeren T, et al. Identifying complications of care using administrative data. Med Care. 1994;32(7):700–715. http://www.ncbi.nlm.nih.gov/pubmed/8028405. Accessed December 2, 2016.
Ghaferi AA, Birkmeyer JD, Dimick JB. Complications, failure to rescue, and mortality with major inpatient surgery in medicare patients. Ann Surg. 2009;250(6):1029–1034. http://www.ncbi.nlm.nih.gov/pubmed/19953723. Accessed April 26, 2015.
Krell RW, Girotti ME, Dimick JB. Extended length of stay after surgery: complications, inefficient practice, or sick patients? JAMA Surg. 2014;149(8):815–820.
Nassour I, Wang SC, Christie A, et al. Minimally Invasive Versus Open Pancreaticoduodenectomy. Ann Surg. May 2017:1.
HCUP-US Cost-to-Charge Ratio Files. https://www.hcup-us.ahrq.gov/db/state/costtocharge.jsp. Accessed May 31, 2017.
Bureau of Labor Statistics USD of L. CPI Inflation Calculator. http://www.bls.gov/data/inflation_calculator.htm.
HCUP-US Cost-to-Charge Ratio Files. http://www.hcup-us.ahrq.gov/db/state/costtocharge.jsp. Accessed December 2, 2016.
Amini N, Spolverato G, Kim Y, Pawlik TM. Trends in Hospital Volume and Failure to Rescue for Pancreatic Surgery. J Gastrointest Surg. 2015;19(9):1581–1592.
Birkmeyer JD, Gust C, Dimick JB, Birkmeyer NJO, Skinner JS. Hospital Quality and the Cost of Inpatient Surgery in the United States. Ann Surg. 2012;255(1):1–5.
Short MN, Aloia TA, Ho V. The influence of complications on the costs of complex cancer surgery. Cancer. 2014;120(7):1035–1041.
Knechtle WS, Perez SD, Medbery RL, et al. The Association Between Hospital Finances and Complications After Complex Abdominal Surgery: Deficiencies in the Current Health Care Reimbursement System and Implications for the Future. Ann Surg. 2015;262(2):273–279.
Zogg CK, Najjar P, Diaz AJR, et al. Rethinking Priorities: Cost of Complications After Elective Colectomy. Ann Surg. 2016;264(2):312–322.
Page AJ, Gani F, Crowley KT, et al. Patient outcomes and provider perceptions following implementation of a standardized perioperative care pathway for open liver resection. Br J Surg. February 2016.
Kagedan DJ, Ahmed M, Devitt KS, Wei AC. Enhanced recovery after pancreatic surgery: a systematic review of the evidence. HPB (Oxford). 2015;17(1):11–16.
Pradarelli JC, Healy MA, Osborne NH, Ghaferi AA, Dimick JB, Nathan H. Variation in Medicare Expenditures for Treating Perioperative Complications: The Cost of Rescue. JAMA Surg. October 2016:e163340.
Gani F, Pawlik TM. Assessing the Costs Associated with Volume-Based Referral for Hepatic Surgery. J Gastrointest Surg. 2016;20(5):945–952.
Nathan H, Pawlik TM. Limitations of claims and registry data in surgical oncology research. Ann Surg Oncol. 2008;15(2):415–423.
Acknowledgements
Drs. Gani and Pawlik had full access to all of the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis.
Author information
Authors and Affiliations
Contributions
1. Study, concept, and design: Gani, Johnston, Nelson-Williams, and Pawlik
2. Acquisition, analysis, and interpretation of data: Gani, Johnston, Nelson-Williams, Cerullo, Dillhoff, Schmidt, and Pawlik
3. Drafting of manuscript: Gani and Pawlik
4. Critical review of manuscript: Gani, Johnston, Nelson-Williams, Cerullo, Dillhoff, Schmidt, and Pawlik
5. Final approval for submission: Gani, Johnston, Nelson-Williams, Cerullo, Dillhoff, Schmidt, and Pawlik
Corresponding author
Ethics declarations
The current study utilized deidentified, administrative data. As such patient consent was not required and did not involve any human subjects. The study was approved by the Johns Hopkin University Institutional Review Board.
Conflicts of Interest
The authors declare that they have no conflict of interest.
Additional information
This study was presented as an oral presentation at the American College of Surgeons, Clinical Congress held in Washington D.C., between October 16 and 20, 2016.
Electronic Supplementary Material
Supplemental Figure 1
Inclusion and exclusion criteria to derive the final study population using the Nationwide Inpatient Sample 2002-2011. (GIF 94 kb)
Supplemental Figure 2
Proportion of patients undergoing surgery at low (1-8 pancreatic resections / year), intermediate (9-30 pancreatic resections / year), and high (≥31 pancreatic resections / year) volume hospitals, and the proportion of hospital categorized and low, intermediate, and high volume hospitals. (GIF 122 kb)
Supplemental Figure 3
Proportion of patients undergoing surgery at a low (1-8 pancreatic resections / year), intermediate (9-30 pancreatic resections / year), and high (≥31 pancreatic resections / year) volume hospitals over the study time periods 1, 2, and 3. (GIF 159 kb)
Supplemental Figure 4
Comparison of covariate-adjusted inpatient costs by the development of a postoperative complication and failure-to-rescue. (GIF 141 kb)
ESM 1
(DOCX 41 kb)
Rights and permissions
About this article
Cite this article
Gani, F., Johnston, F.M., Nelson-Williams, H. et al. Hospital Volume and the Costs Associated with Surgery for Pancreatic Cancer. J Gastrointest Surg 21, 1411–1419 (2017). https://doi.org/10.1007/s11605-017-3479-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11605-017-3479-x