Abstract
Introduction
Recent advances in thoracoscopic surgery have made it possible to perform esophagectomy with conventional lymphadenectomy (paraesophageal and subcarinal lymph node dissection) using minimally invasive techniques. However, minimally invasive esophagectomy (MIE) combined with extensive lymphadenectomy along the recurrent laryngeal nerves (RLN) has remained technically challenging for thoracic surgeons. The aim of this study was to examine the safety and efficacy of extensive lymphadenectomy when compared to conventional lymphadenectomy during MIE.
Methods
We retrospectively reviewed data from a cohort of 147 consecutive patients who underwent MIE for esophageal cancer (EC) over a 3-year period at our institution. During thoracoscopic esophagectomy, extensive lymphadenectomy along the RLN was performed on 76 patients from June 2009 to December 2010 (group A), while 71 patients underwent conventional lymphadenectomy from June 2008 to May 2009 (group B) and were enrolled as historical controls. Clinical characteristics including patient demographics, operation features, and the rate and type of complications were recorded for both groups. The number of dissected lymph nodes and the number of patients with nodes positive for cancer on histological examination were determined for both groups. Statistical analysis was used to identify differences between the two groups.
Results
All patients underwent thoracoscopic esophagectomy without conversion to open thoracotomy. Patient demographics and operation features were similar between the two groups. Of the 76 patients that underwent extensive lymphadenectomy there were 13 patients (17.11%) who were RLN positive, which resulted in upstaging of TNM in 5 patients (6.58%). The overall incidence of postoperative complications (42.10% versus 39.47%, p = 0.742) and permanent recurrent laryngeal nerve palsy (1.32% versus 0%, p = 0.517) was similar between the two groups.
Conclusions
Extensive mediastinal lymphadenectomy during minimally invasive esophagectomy is a feasible procedure for EC patients. It is technically safe and oncologically adequate in experienced hands, and improves the accuracy of tumor staging. Further study is required to discuss its long-term prognostic value for esophagus cancer patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The technical feasibility of minimally invasive esophagectomy (MIE) has been well recognized over the past decade;1–4 however, questions over the adequacy of tumor and lymph node clearance have been raised with the development of thoracoscopic techniques.5,6 Previously, high frequency of tumor dissemination to lymph nodes along the RLN was reported in open thoracotomy,7 and survival benefits were suggested when extended lymphadenectomy was performed on these patients.8 Despite these findings, Fang et al. has suggested that extensive lymphadenectomy is technically demanding and associated with a higher complication rate of 41.7% to 75.3%.9 Striking the right balance between performing extensive lymphadenectomy during MIE without increasing complication rates significantly remains a challenge.
In an earlier study from 1996, Akaishi and associates investigated thoracoscopic radical mediastinal lymphadenectomy and total esophagectomy on animals and selected patients. However, the study was non-comparative and was based on a small sample of 39 cases.10 Since then, only a few studies have been published on extensive lymphadenectomy during MIE. The promising results of two recent studies by Noshiro (2010) and Puntambekar (2010) on extended lymph node dissection during MIE11,12 have led to our research on the subject.
We started MIE on esophageal cancer (EC) patients at our institute in 2005, and our initial study has shown a better quality of life in patients that underwent MIE compared to conventional surgery.13 Based on our experience, we herein present our procedure of extensive mediastinal lymphadenectomy for EC patients, and review its safety and efficacy in comparison to conventional lymphadenectomy during MIE.
Patients and Methods
Patients
A total of 202 cases of EC were referred to Dr. Lijie Tan's clinic at the Zhongshan Hospital, Fudan University from June 2008 to December 2010. Patients were clinically staged using endoscopy, biopsy results, abdominal-thoracic computed tomography (CT), and endoscopic ultrasound. MIE was performed on clinically staged T1-3N0M0 tumors, and neoadjuvant chemotherapy was administered in all other cases. In our study, the inclusion criteria were as follows: (a) age <75 years; (b) patients who did not receive neoadjuvant therapy; (c) patients with no previous history of cancer; (d) patients with no previous history of neck or chest surgery, and (e) patients with an ASA score of I–III.
All operations were conducted thoracolaparoscopically. Extensive lymphadenectomy, which included lymph node clearance along both recurrent laryngeal nerves (RLN), in addition to subcarinal and paraesophageal nodes, was performed on 76 patients from June 2009 to December 2010 (group A). The remaining 71 patients underwent conventional lymphadenectomy (lymphadenectomy along subcarinal and paraesophageal regions) from June 2008 to May 2009 and were enrolled as historical controls (group B). Patient demographics are summarized in Table 1.
The study was approved by the hospital ethics committee, and a waiver for individual patient consent for this retrospective study was also obtained.
Surgical Technique
Analgesia and Anesthesia
All patients had a combination of epidural and general anesthesia and were provided with patient controlled analgesia postoperatively. Patients were intubated with a left-sided double-lumen endotracheal tube to accomplish selective deflation of the right lung during the thoracic phase of the operation. The cuff of the endotracheal tube was deflated temporarily when performing extensive lymphadenectomy along the left RLN.
Position and Ports
Patients were placed in the prone position, with the right side slightly raised up during the operation. A 10-mm camera port trocar was placed as the camera port at the seventh intercostal space (ICS) along the midaxillary line. A 5-mm port was placed at the fourth ICS along the mid axillary line, and the second-5 mm port was placed at the eighth ICS along the posterior axillary line to insert the harmonic scalpel (Ethicon Endosurgery, Cincinnati, OH, USA).
Surgery
The operation was divided into three stages: the thoracic, abdominal, and cervical stages. (1) The thoracic stage included thoracoscopic mobilization of the esophagus and mediastinal lymphadenectomy. During the surgery, the surgeon stood on the right side of the patient, and a high-definition video monitor was set up on the opposite end. Carbon dioxide pneumothorax was achieved at a pressure of 6–8 mmHg. Before dissection, the mediastinal pleura was inspected to assess the mobility of the tumor and its invasion into surrounding structures. Once radical surgery was guaranteed, the azygous vein would be doubly ligated and then transected. The esophageal arteries were divided by ultrasonic blade and the thoracic duct was carefully preserved. Circumferential mobilization of the esophagus, with all surrounding lymph nodes, periesophageal tissue, and the mediastinal pleura was performed from the diaphragmatic reflection to the thoracic inlet and the entire subcarinal lymph node packet was removed en bloc. The thoracic stage was completed by placement of an intercostal drain and closure of the thoracic incisions. (2) The abdominal and cervical stages were the same in both groups and performed with all patients in the supine position. The abdominal stage commenced with dissection of the gastro-hepatic ligament at the lesser curvature of the stomach and exposure of the right crus of the diaphragm. The left gastric artery was then ligated by hem-o-lock and dissected. The gastric tube was cut along the greater curve of the stomach and the right gastroepiploic arcade was carefully preserved. The short gastric vessels were divided by ultrasonic shear coagulation. Following gastric mobilization, lymph node dissection along the celiac and gastric vessels was performed. A jejunal feeding tube was conventionally placed during the abdominal procedure. (3) The cervical procedure included mobilization of cervical esophagus and esophago-gastric anastomosis. Side–side stapled, circular stapling (Ethicon Endosurgery, Cicinatti, OH, USA), or hand-sewing techniques were applied for anastomosis after the gastric tube was pulled through esophageal bed route. The surgery concluded with closure of the cervical and abdominal incisions in layers.
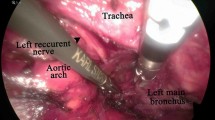
Extensive lymphadenectomy was done in all patients undergoing surgery from 2009. The surgical procedure was the same as described for group B with the addition of lymphadenectomy along the right and left recurrent laryngeal nerves. Before dissection, the mediastinal pleura was conventionally opened to make sure the tumor was resectable, and lymphadenectomy along the right RLN was performed before mobilization of the thoracic esophagus. The right RLN was identified and exposed by tracing the branches of the right vagus nerve under the right subclavian artery. The lymph nodes surrounding the right RLN were dissected by electronic hook (see Fig. 1). In order to dissect the nodes along the left RLN, the cuff of the double-lumen endotracheal tube was deflated temporarily and the trachea was rolled back, to the right and ventrally. A grasper holding small gauze was used to explore the left aspect of the trachea and the left main bronchus. The left RLN was usually found embedded in the connective tissue between the trachea and aortic arch. The lymph nodes along the left RLN were then carefully dissected (see Fig. 2). Lymph nodes from different mediastinal station were collected in separate specimen bags for further histological examination.
Data Collection and Statistics Analysis
Clinical data including patient demographics, operation features, and retrieved mediastinal lymph nodes of both groups were collected from the clinical database of the Thoracic Division at the Zhongshan Hospital, Fudan University. Medical charts were reviewed to identify complications as per the Society of Thoracic Surgeons (STS) National Database.
Data collected was recorded in Microsoft EXCEL for further processing. All statistical analyses were performed using SPSS version 17.0. The Student's t test and chi-square test was used to compare categorical variables as appropriate, whereas the Mann–Whitney Test was used to compare continuous variables. A two-sided p value of less than 0.05 was considered to be statistically significant.
Results
Patient Demographics
In our cohort, there was a male predominance with 102 male and 45 female patients. The mean age at time of surgery was 60.1 ± 8.7 years. No significant difference in age (p = 0.247) or gender (p = 0.792) was found between the two groups. According to the UICC (Union for International Cancer Control) esophageal cancer TNM staging system (sixth Edition, 1997), there were 6 cases of pTis (4.08%), 36 cases of pT1 (24.49%), 43 cases of pT2 (29.25%), and 62 cases of pT3 (42.18%) in the study. The depth of tumor invasion was statistically comparable between the two groups (p = 0.651). There was no difference in tumor location (p = 0.096) and histological type (p = 0.836) between the two groups.
Operation Features
All 147 patients underwent thoracoscopic esophagectomy without conversion to open thoracotomy. When comparing operation characteristics, the duration of the thoracic stage was longer in group B (99 ± 32 min) than group A (89 ± 32 min), although this was found to be of no statistical difference (p = 0.064). No significant difference was found in the length of stay, volume of blood loss, or rate of blood transfusion between the two groups. The operation features are listed in Table 1.
Mortality and Morbidity
No patient died during the operation procedure in either group. Complications were observed in a total of 60 patients in our study. In group B, one patient developed stump leakage of the gastric conduit on the eighth post operative day and died two weeks later because of septic shock and multi-organ failure. A second operation was performed on one patient in group A to control bleeding from the thoracic cavity. Operative mortality was not observed in group A.
Anastomotic leakage was the most common complication in both groups seen in 14 patients in group B and 16 patients in group A (p = 0.841). All of these patients were treated conservatively with drainage and nutritional support. Nine patients developed acute RLN palsy after surgery, and seven of these cases were from group A (7/9 77.78%). At 6 months' follow-up, permanent RLN palsy persisted in one patient from group A. Both cases of acute RLN palsy had resolved in group B at 6 months follow-up. The incidence of permanent RLN palsy was similar between the two groups (1 from group A and 0 from group B, p = 0.517). The overall complication rate was 40.08% and this was found to be of no statistical difference between the two groups (see Table 2).
Mediastinal Lymph Nodes
The average number of retrieved lymph nodes from mediastinum was 13.2 ± 6.5 in group A and 9.1 ± 4.3 in group B, which was significantly higher in the extensive lymphadenectomy group (p < 0.001). The frequency of metastasis per patient was close between the two groups (25.00% versus 22.54%, p = 0.726). Of the 76 patients that underwent extensive lymphadenectomy, 13 patients were found to be RLN positive (17.11%). According to the TNM stage criteria, five patients were upstaged (6.58%) from N0 to N1 by extensive mediastinal lymphadenectomy. The details of the retrieved mediastinal lymph nodes are listed in Table 3.
Discussion
Unlike the western world, esophageal squamous cell cancer (ESCC) is more common in Asian countries. The prevalence of the squamous cell subtype of EC in some regions of China and Japan is reported to be over 90%.14,15 Furthermore, squamous histology has been shown as an independent predictor for positive RLN nodes on multivariate analysis.16 EC patients are therefore at a higher risk of recurrent laryngeal node involvement in the Far East, and this necessitates extensive lymphadenectomy in these populations. In our study, 139 patients (94.56%) had ESCC and 17.1% of these patients had RLN-positive nodes.
Our study is limited by its retrospective design and the use of historical controls. Historical controls were used for comparison because the incidence of tumor invasion to the superior mediastinal lymph nodes was considered higher than attributable to chance alone. A randomized trial could therefore not be ethically justified since patients in the control group would have residual tumor deposits and therefore poor prognosis. To minimize technique biases, all surgeries were performed consecutively under the supervision of the same senior surgeon and consistency in technique was further verified by similar durations of the abdominal and cervical stages in both groups. We do however recognize the impact of the “learning curve” effect on the outcomes of patients in group A since they represent the latter half of this sequential study. It could therefore be argued that improvements in surgical technique that come with experience are therefore inevitable. However, the extensive previous experience of the supervising surgeon would hopefully minimize this effect.
Because of difficult exposure and higher risk of injury to the nerves, lymphadenectomy along the recurrent laryngeal nerves has remained a challenge during open thoracotomy.17 It is for these reasons that the lymph node clearance in the cervical field has usually been omitted in the past at many centers. In our study, we have successfully retrieved lymph nodes along both recurrent laryngeal nerves during MIE and our results suggest that there is no significant increase in the rate of RLN injury. Similar results were presented by Puntambekar and associates,12 where the duration of surgery was approximately 60 to 120 min with a low volume of blood loss and a low rate of conversion to open thoracotomy. Given the magnified view of the surgical field, MIE has made it easier to perform extensive mediastinal lymphadenectomy in a confined operating space.
Although the operations performed in our study were extensive, whether they were of the same adequacy and efficacy as traditional surgery was worth discussion. In our study, an average of 13.2 ± 6.5 mediastinal lymph nodes were retrieved in 89 ± 32 min in group A. Similar numbers were obtained but took much longer time periods for retrieval in open thoracotomy.18 The highest level of lymph node dissection in our study was similar to that done at the upper limit of thoracic lymphadenectomy in classical three-field lymphadenectomy.19 In our experience, when performing lymph node clearance along the right RLN, it was easy to reach the inferior border of the right thyroid gland, and this facilitated the subsequent cervical stage of lymphadenectomy. The percentage of patients with positive mediastinal lymph nodes was 35.52% in group A. Our results are nearly identical to those reported by three-field dissection in open thoracotomy by Kitagawa and associates.20 Of the 76 patients in group A that underwent extensive lymph node clearance, 13 patients had positive RLN nodes. The percentage positive rate was even higher in tumors that were invading the muscularis propria (T2) and advential (T3) layers (18.5%) or located in the upper third of the esophagus (50%, two out of four patients). These results are similar to those reported by Matsubara and associates after open thoracotomy.17 Furthermore, according to the TNM stage criteria, five patients in group A were upstaged from N0 to N1 by extensive lymphadenectomy, which meant that the accuracy of tumor staging was improved by 6.58% from this procedure. Since patients with pN1 were conventionally assigned to chemotherapy that pN0 patients were not suggested to, we believe that an extensive lymphadenectomy could result in the difference in postoperative treatment, and potentially, the long-term survival on esophagus cancer patients. The extent of dissection, duration of surgery, and rate of lymph node invasion found in our cohort showed that extensive thoracoscopic lymphadenectomy was technically efficient and oncologically adequate in comparison with open thoracotomy.
In our study, a total of 60 patients developed complications with one inpatient death in group B. The complication rate was not significantly different between the two groups (39.44% versus 42.11% p = 0.742). The anastomotic leakage ranked the first in the complication table (20.41%). Similar findings were reported in the early publications. We hypothesized that an extensive lymphadenectomy that was performed along RLN would not lead to an increased ratio of anastomotic leakage since it was reported to be associated with the reduced blood supply to the gastric tube.21 This was confirmed in the close ratio of anastomotic leakage between the two groups (21.05% versus 19.72% p = 0.742).
In this report, the incidence of RLN injury is in contrast to reported RLN injury rates of 14.0% to 45.3% in open thoracotomy22 and comparable to rates of 2.6% to 36% during thoracoscopic procedures.1,23 We feel that the incidence of RLN palsy was lower in our study because of adequate exposure and identification of both recurrent laryngeal nerves prior to lymph node dissection. This avoided accidental injury to the RLN and allowed safer dissection in this field. In compared with group B, extensive lymphadenectomy along bilateral RLN led to a threefold increase in the postoperative hoarseness in group A (9.21% versus 2.82%). Although it was found to be of no statistical difference (p = 0.168), this could be owed to a relatively small population of the study. Luckily, most of these cases are likely due to neuropraxia of the RLN during extensive lymph node dissection and this was seen to resolve at 6-months follow-up with only one patient in group A having a persistent RLN palsy. We recognize that acute RLN injury could predispose these patients to a higher risk of pulmonary complications in the immediate postoperative period.24 However, the incidence of pulmonary complications were comparable even when extensive lymphadenectomy was performed (6.58% versus 8.45%, p = 0.759). This could be explained by enteral feeding through the jejuna tube, which decreased the chance of aspiration from oral intake after surgery. We therefore conclude that although extensive lymphadenectomy along the recurrent laryngeal nerves resulted in a higher rate of acute RLN palsy in our patients, most of these cases resolved without increased risks of associated complications, and the rate of permanent RLN injury was not different between the two groups.
The incidence of pulmonary complications in our cohort was not as high as described in previous studies.25 This was likely due to a shorter duration of the thoracic stage of surgery. The mean duration of the thoracic stage of the operation was 89 ± 32 min in group A and this was lower than the mean duration for group B (99 ± 32 min). We therefore conclude that the assumption that extensive lymphadenectomy during MIE is more time consuming than conventional lymphadenectomy is not correct. This may be attributed to the refinement of surgical techniques and on the surgeon's experience. Extensive lymphadenectomy during MIE has been shown to have a steep learning curve however it can be mastered safely and quickly under the direction of an experienced surgeon.26 Of the 76 cases we have performed, the thoracic lymphadenectomy was done before the mobilization of esophagus to avoid destroy of mediastinum structure and therefore assuring the clearance of surgical field during the lymph node dissection along right RLN. During the operation, patients were prone positioned. The esophagus was located in the up side and posterior mediastinum was pulling down by gravity, which meant bleeding would not affect the process significantly and operation continuality would be assured, so it was no wonder that two groups were close in thoracic duration even when more extensive procedure was performed.
Conclusions
In this study, extensive mediastinal lymphadenectomy during MIE was found to be safe, feasible, and more accurate for tumor staging in EC patients. The main limitation of this study was its retrospective design and the use of historical controls. Further prospective studies on the role of extensive lymphadenectomy for MIE in the form of randomized controlled trials are necessary to confirm these findings.
References
Luketich JD, Alvelo-Rivera M, Buenaventura PO, Christie NA, McCaughan JS, LiMIE VR, Schauer PR, Close JM, Fernando HC. Minimally invasive esophagectomy: outcomes in 222 patients. Ann Surg. 2003; 238: 486–494.
Smithers BM, GoMIEy DC, Martin I, Thomas JM. Comparison of the outcomes between open and minimally invasive esophagectomy. Ann Surg. 2007; 245: 232–240.
Palanivelu C, Prakash A, Senthilkumar R, Senthilnathan P, Parthasarathi R, Rajan PS, Venkatachlam S. Minimally invasive esophagectomy: thoracoscopic mobilization of the esophagus and mediastinal lymphadenectomy in prone position--experience of 130 patients. J Am Coll Surg. 2006; 203: 7–16.
Berrisford RG, Wajed SA, Sanders D, Rucklidge MW. Short-term outcomes following total minimally invasive oesophagectomy. Br J Surg. 2008; 95: 602–610.
Nagpal K, Ahmed K, Vats A, Yakoub D, James D, Ashrafian H, Darzi A, Moorthy K, Athanasiou T. Is minimally invasive surgery beneficial in the management of esophageal cancer? A meta-analysis. Surg Endosc. 2010; 24: 1621–1629.
Veeramachaneni NK, Zoole JB, Decker PA, Putnam JB Jr, Meyers BF. Lymph node analysis in esophageal resection—American College of Surgeons Oncology Group Z0060 trial. Ann Thorac Surg. 2008; 86: 418–421.
Akiyama H, Tsurumaru M, Udagawa H, Kajiyama Y. Radical lymph node dissection for cancer of the thoracic esophagus. Ann Surg. 1994; 220: 364–372.
Kang CH, Kim YT, Jeon SH, Sung SW, Kim JH. Lymphadenectomy extent is closely related to long-term survival in esophageal cancer. Eur J Cardiothorac Surg. 2007; 31: 154–60.
Fang WT, Chen WH. Current trends in extended lymph node dissection for esophageal carcinoma. Asian Cardiovasc Thorac Ann. 2009; 17: 208–213.
Akaishi T, Kaneda I, Higuchi N, Kuriya Y, Kuramoto J, Toyoda T, Wakabayashi A. Thoracoscopic en bloc total esophagectomy with radical mediastinal lymphadenectomy. J Thorac Cardiovasc Surg. 1996; 112:1533–1540.
Noshiro H, Iwasaki H, Kobayashi K, Uchiyama A, Miyasaka Y, Masatsugu T, Koike K, Miyazaki K. Lymphadenectomy along the left recurrent laryngeal nerve by a minimally invasive esophagectomy in the prone position for thoracic esophageal cancer. Surg Endosc. 2010; 24: 2965–2973.
Puntambekar SP, Agarwal GA, Joshi SN, Rayate NV, Sathe RM, Patil AM. Thoracolaparoscopy in the lateral position for esophageal cancer: the experience of a single institution with 112 consecutive patients. Surg Endosc. 2010; 24: 2407–2414.
Wang H, Tan L, Feng M, Zhang Y, Wang Q. Comparison of the short-term health-related quality of life in patients with esophageal cancer with different routes of gastric tube reconstruction after minimally invasive esophagectomy. Qual Life Res. 2011; 20:179–189.
Isono K, Sato H, Nakayama K. Results of a nationwide study on three-field lymph node dissection of esophageal cancer. Oncology. 1991; 48: 411–420.
He Z, Zhao Y, Guo C, Liu Y, Sun M, Liu F, Wang X, Guo F, Chen K, Gao L, Ning T, Pan Y, Li Y, Zhang S, Lu C, Wang Z, Cai H, Ke Y. Prevalence and risk factors for esophageal squamous cell cancer and precursor lesions in Anyang, China: a population-based endoscopic survey. Br J Cancer. 2010; 103:1085–1088.
Stiles BM, Mirza F, Port JL, Lee PC, Paul S, Christos P, Altorki NK. Predictors of cervical and recurrent laryngeal lymph node metastases from esophageal cancer. Ann Thorac Surg. 2010; 90: 1805–1811.
Matsubara T, Ueda M, Nagao N, Takahashi T, Nakajima T, Nishi M. Cervicothoracic approach for total mesoesophageal dissection in cancer of the thoracic esophagus. J Am Coll Surg. 1998; 187: 238–245.
Tachibana M, Kinugasa S, Yoshimura H, Shibakita M, Tonomoto Y, Dhar DK, Nagasue N. Clinical outcomes of extended esophagectomy with three-field lymph node dissection for esophageal squamous cell carcinoma. Am J Surg. 2005; 189: 98–109.
Lerut T, De Leyn P, Coosemans W, Van Raemdonck D, Scheys I, LeSaffre E. Surgical strategies in esophageal carcinoma with emphasis on radical lymphadenectomy. Ann Surg. 1992; 216: 583–589.
Ando N, Ozawa S, Kitagawa Y, Shinozawa Y, Kitajima M. Improvement in the results of surgical treatment of advanced squamous esophageal carcinoma during 15 consecutive years. Ann Surg. 2000; 232: 225–232.
Junemann-Ramirez M, Awan MY, Khan ZM, Rahamim JS. Anastomotic leakage post-esophagogastrectomy for esophageal carcinoma: retrospective analysis of predictive factors, management and influence on long term survival in a high volume centre. Eur J Cardiothorac Surg. 2005; 27: 3–7.
Natsugoe S, Okumura H, Matsumoto M, Ishigami S, Owaki T, Nakano S, Aikou T. Reconstruction of recurrent laryngeal nerve with involvement by metastatic node in esophageal cancer. Ann Thorac Surg. 2005; 79: 1886–1889.
Dexter SPL, Martin IG, McMahonz MJ. Radical thoracoscopic esophagectomy for cancer. Surg Endosc. 1996 10:147–151.
Fang W, Kato H, Tachimori Y, Igaki H, Sato H, Daiko H. Analysis of pulmonary complications after three-field lymph node dissection for esophageal cancer. Ann Thorac Surg. 2003; 76: 903–8.
Zingg U, Smithers BM, GoMIEy DC, Smith G, Aly A, Clough A, Esterman AJ, Jamieson GG, Watson DI. Factors associated with postoperative pulmonary morbidity after esophagectomy for cancer. Ann Surg Oncol. 2011; 18: 1460–1468.
Ninomiya I, Osugi H, Tomizawa N, Fujimura T, Kayahara M, Takamura H, Fushida S, Oyama K, Nakagawara H, Makino I, Ohta T. Learning of thoracoscopic radical esophagectomy: how can the learning curve be made short and flat? Dis Esophagus. 2010; 23: 618–626.
Acknowledgments
We thank Mr. Jianfeng Luo for the statistical support of this work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shen, Y., Zhang, Y., Tan, L. et al. Extensive Mediastinal Lymphadenectomy During Minimally Invasive Esophagectomy: Optimal Results from a Single Center. J Gastrointest Surg 16, 715–721 (2012). https://doi.org/10.1007/s11605-012-1824-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11605-012-1824-7