Summary
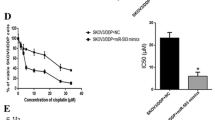
Resistance to chemotherapy is a major obstacle for the effective treatment of advanced ovarian cancer. The mechanism of chemoresistance is still poorly understood. Recently, more and more evidence showed microRNAs (miRNAs) modulated many key molecules and pathways involved in chemotherapy. microRNA-106a (miR-106a) has been implicated in many cancers, but its role in ovarian cancer and drug resistance still remains unexplored. This study was to investigate whether miR-106a mediated resistance of the ovarian cancer cell line A2780 to the chemotherapeutic agent cisplatin (DDP). The different levels of miR-106a in A2780 cells and their resistant variant A2780/DDP cells were identified by using real-time PCR. MTT assay and flow cytometry were used to analyze the effect of miR-106a on cisplatin resistance of these paired cells. Real-time PCR, Western blotting and luciferase reporter assay were applied to explore whether Mcl-1 was a target of miR-106a. As compared to A2780 cells, the expression of miR-106a was down-regulated in the cisplatin resistant cell line A2780/DDP. Moreover, knockdown of miR-106a dramatically decreased antiproliferative effects and apoptosis induced by cisplatin in A2780 cells, while overexpression of miR-106a significantly increased antiproliferative effects and apoptosis induced by cisplatin in A2780/DDP cells. Furthermore, miR-106a inhibited cell survival and cisplatin resistance through downregulating the expression of Mcl-1. Mcl-1 was a direct target of miR-106a. These results suggest that miR-106a may provide a novel mechanism for understanding cisplatin resistance in ovarian cancer by modulating Mcl-1.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Legge F, Ferrandina G, Salutari V, et al. Biological characterization of ovarian cancer: prognostic and therapeutic implications. Ann Oncol, 2005,16(Suppl 4):iv95–iv101
van der Burg ME, van Lent M, Buyse M, et al, The effect of debulking surgery after induction chemotherapy on the prognosis in advanced epithelial ovarian cancer. Gynecological Cancer Cooperative Group of the European Organization for Research and Treatment of Cancer. N Engl J Med, 1995,332(10):629–634
Cannistra SA. Cancer of the ovary. N Engl J Med, 2004,351(24):2519–2529
Zhang C, Yao Z, Zhu M, et al. Inhibitory effects of microRNA-34a on cell migration and invasion of invasive urothelial bladder carcinoma by targeting Notch1. J Huazhong Univ Sci Technol [Med Sci], 2012,32(3): 375–382
Cortes-Sempere M, Ibanez de Caceres I. microRNAs as novel epigenetic biomarkers for human cancer. Clin Transl Oncol, 2011,13(6):357–362
Pinato DJ, Graham J, Gabra H, et al. Evolving concepts in the management of drug resistant ovarian cancer: dose dense chemotherapy and the reversal of clinical platinum resistance. Cancer Treat Rev, 2013,39(2):153–160
Eitan R, Kushnir M, Lithwick-Yanai G, et al. Tumor microRNA expression patterns associated with resistance to platinum based chemotherapy and survival in ovarian cancer patients. Gynecol Oncol, 2009,114(2):253–259
Koster R, di Pietro A, Timmer-Bosscha H, et al. Cytoplasmic p21 expression levels determine cisplatin resistance in human testicular cancer. J Clin Invest, 2010,120(10):3594–3605
Dylla L, Jedlicka P. Growth-promoting role of the miR-106a approximately 363 cluster in Ewing sarcoma. PLoS One, 2013,8(4):e63032
Timoneda O, Balcells I, Cordoba S, et al. Determination of reference microRNAs for relative quantification in porcine tissues. PLoS One, 2012,7(9):e44413
Yang X, Zheng F, Xing H. The relationship between cytochrome C and the cisplatin resistance by targeting the activity of caspase-3 in ovarian cancer. Zhonghua Zhongliu Zazhi (Chinese), 2002,24(6):544–547
Kyrgiou M, Salanti G, Pavlidis N, et al. Survival benefits with diverse chemotherapy regimens for ovarian cancer: meta-analysis of multiple treatments. J Natl Cancer Inst, 2006,98(22):1655–1663
Ferrandina G, Zannoni GF, Martinelli E, et al. Class III beta-tubulin overexpression is a marker of poor clinical outcome in advanced ovarian cancer patients. Clin Cancer Res, 2006,12(9):2774–2779
Kong F, Sun C, Wang Z, et al. miR-125b confers resistance of ovarian cancer cells to cisplatin by targeting pro-apoptotic Bcl-2 antagonist killer 1. J Huazhong Univ Sci Technol [Med Sci], 2011,31(4):543–549
Yang N, Kaur S, Volinia S, et al. MicroRNA microarray identifies Let-7i as a novel biomarker and therapeutic target in human epithelial ovarian cancer. Cancer Res, 2008,68(24):10307–10314
Yang L, Li N, Wang H, et al. Altered microRNA expression in cisplatin-resistant ovarian cancer cells and upregulation of miR-130a associated with MDR1/ P-glycoprotein-mediated drug resistance. Oncol Rep, 2012,28(2):592–600
Cochrane DR, Spoelstra NS, Howe EN, et al. MicroRNA-200c mitigates invasiveness and restores sensitivity to microtubule-targeting chemotherapeutic agents. Mol Cancer Ther, 2009,8(5):1055–1066
Cheng W, Liu T, Wan X, et al. MicroRNA-199a targets CD44 to suppress the tumorigenicity and multidrug resistance of ovarian cancer-initiating cells. FEBS J, 2012,279(11):2047–2059
Ambros V. MicroRNA pathways in flies and worms: growth, death, fat, stress, and timing. Cell, 2003,113(6): 673–676
Catela Ivkovic T, Aralica G, Cacev T, et al. miR-106a overexpression and pRB downregulation in sporadic colorectal cancer. Exp Mol Pathol, 2013,94(1):148–154
Schee K, Boye K, Abrahamsen TW, et al. Clinical relevance of microRNA miR-21, miR-31, miR-92a, miR-101, miR-106a and miR-145 in colorectal cancer. BMC Cancer, 2012,12:505
Wang Z, Liu M, Zhu H, et al. miR-106a is frequently upregulated in gastric cancer and inhibits the extrinsic apoptotic pathway by targeting FAS. Mol Carcinog, 2013,52(8):634–646
Zhao S, Yang G, Mu Y, et al. MiR-106a is an independent prognostic marker in patients with glioblastoma. Neuro Oncol, 2013,15(6):707–717
Poruchynsky MS, Giannakakou P, Ward Y, et al. Accompanying protein alterations in malignant cells with a microtubule-polymerizing drug-resistance phenotype and a primary resistance mechanism. Biochem Pharmacol, 2001,62(11):1469–1480
Simonin K, Brotin E, Dufort S, et al. Mcl-1 is an important determinant of the apoptotic response to the BH3-mimetic molecule HA14-1 in cisplatin-resistant ovarian carcinoma cells. Mol Cancer Ther, 2009,8(11): 3162–3170
Yuan Z, Cao K, Lin C, et al. The p53 upregulated modulator of apoptosis (PUMA) chemosensitizes intrinsically resistant ovarian cancer cells to cisplatin by lowering the threshold set by Bcl-x(L) and Mcl-1. Mol Med, 2011,17(11-12):1262–1274
Simonin K, N’Diaye M, Lheureux S, et al. Platinum compounds sensitize ovarian carcinoma cells to ABT-737 by modulation of the Mcl-1/Noxa axis. Apoptosis, 2013,18(4):492–508
Shigemasa K, Katoh O, Shiroyama Y, et al. Increased MCL-1 expression is associated with poor prognosis in ovarian carcinomas. Jpn J Cancer Res, 2002,93(5): 542–550
Author information
Authors and Affiliations
Corresponding author
Additional information
This project was supported by the Youth Innovation Foundation of the First Affiliated Hospital of Zhengzhou University.
Rights and permissions
About this article
Cite this article
Rao, Ym., Shi, Hr., Ji, M. et al. MiR-106a targets Mcl-1 to suppress cisplatin resistance of ovarian cancer A2780 cells. J. Huazhong Univ. Sci. Technol. [Med. Sci.] 33, 567–572 (2013). https://doi.org/10.1007/s11596-013-1160-5
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-013-1160-5