Abstract
Chronic HIV-1 infection commonly affects behavioral, cognitive, and motor functions in the infected human host and is commonly referred to as HIV-1-associated neurocognitive disorders (HAND). This occurs, in measure, as a consequence of ingress of leukocytes into brain perivascular regions. Such cells facilitate viral infection and disease by eliciting blood–brain barrier and neuronal network dysfunctions. Previous works demonstrated that the endocannabinoid system modulates neuroimmunity and as such neuronal and glial functions. Herein, we investigated CB2R receptor expression in murine HIV-1 encephalitis (HIVE) and the abilities of a highly selective CB2R agonist, Gp1a, to modulate disease. HIV-1-infected human monocyte-derived macrophages were injected into the caudate and putamen of immunodeficient mice reconstituted with human peripheral blood lymphocytes (hu-PBL/HIVE). Brains of hu-PBL/HIVE mice showed microglial activation and increased expression of CB2R, but not CB1R or GPR55. Gp1a substantively reduced infiltration of human cells into the mouse brain and reduced HLA DQ activation. Gp1a down modulated CCR5 expression on human cells in the spleen with an increase in Fas ligand expression. Our results support the notion that CB2 receptor agonists may be a viable therapeutic candidate for HAND.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Endocannabinoids are linked to a broad range of neurologic, infectious, and inflammatory diseases (Ashton and Glass 2007; Basavarajappa et al. 2009; Leonelli et al. 2009; Patel et al. 2010). As a consequence, the G-protein coupled cannabinoid (CB) receptors, endocannabinoid (eCB) transport, as well as its metabolizing enzymes can modulate receptor-mediated signaling and may be useful in therapeutic interventions (Cabral and Griffin-Thomas 2008; Klein and Newton 2007; Pacher et al. 2006). Recent studies have implicated dysregulation of the eCB in nervous system inflammatory neurodegeneration (Benito et al. 2007a; Centonze et al. 2007; Correa et al. 2007). The eCBs, principally anandamide and 2-arachidonoylglycerol, are a class of lipid messengers that modulate a range of physiological processes in and outside the nervous system through CB1R, CB2R, and the GPR55 orphan receptor (Mackie and Stella 2006). Plant-derived cannabinoids or synthetic cannabinoids are immunomodulatory molecules that act through CB1R and CB2R. Activation of CB1 receptors has a psychotropic effect, but activation of CB2 receptors alone does not (Piomelli 2005). The distribution pattern of levels of CB2R messenger RNA displays major variation in blood cell populations with a rank order of B lymphocytes > natural killer cells > monocytes > polymorphonuclear neutrophils > CD8+ and CD4+ T lymphocytes (Galiegue et al. 1995). Studies on CB2 receptor knockout mice show clear linkages between CB2R immune cell functions for microbial infection, cell chemotaxis, and apoptosis (Buckley 2008). Modulation by either CB2 receptor agonists or antagonists/inverse agonists might be of therapeutic benefit (Arevalo-Martin et al. 2008; Gonsiorek et al. 2006; Lombard et al. 2007; Onaivi 2009).
In neuroinflammatory and neurodegenerative diseases such as multiple sclerosis, Alzheimer’s (AD) and Parkinson’s diseases, and HIV-1-associated neurocognitive disorders (HAND), neuroimmune regulation has gained recent acclaim as a viable therapeutic approach (Benito et al. 2007b; Kreitzer and Stella 2009; Lastres-Becker et al. 2005; Price et al. 2009; Shoemaker et al. 2007; Steffens et al. 2005; Thakur et al. 2009). Indeed, synthetic cannabinoids, through activation of CB2R, suppress immunity in various animal models including experimental autoimmune encephalomyelitis (Berrendero et al. 2001; Mestre et al. 2009). There is increasing evidence that the CB2R, in addition to its linkage to immune cell activities at peripheral sites, plays a functionally relevant role in the immune cells of central nervous system (CNS) including primarily microglia (Ashton and Glass 2007; Cabral et al. 2008; Stella 2009). Alterations in the components of the eCB system occur in brains of simian immunodeficiency virus (SIV)-infected macaques (Benito et al. 2005). In addition, CB2 receptor expression in activated microglial cells is associated with deposits of beta amyloid peptide in AD (Benito et al. 2003). The inducible nature of CB2 receptors under neuroinflammatory conditions makes them attractive targets for novel therapeutic approaches.
The present study was conducted to ascertain the mechanisms and benefits of targeting CB2R-mediated signaling for HAND using a murine model of human disease. Immunodeficient mice reconstituted with human peripheral blood lymphocytes (hu-PBL) are valuable tools to study HIV-1 disease [reviewed in (Pearson et al. 2008; Van Duyne et al. 2009)]. In this regard, changes in CB receptor expression were studied in murine HIV-1 encephalitis (HIVE). Hu-PBL were reconstituted into immunodeficient Balb/c-Rag−/−γc−/− mice and HIV-1-infected human monocyte-derived macrophages (MDM) were injected into the brain to induce viral encephalitis (hu-PBL/HIVE mice) (Poluektova et al. 2004a, b, 2002). Increased CB2 receptor expression was found in the brains of hu-PBL/HIVE mice. To examine whether CB2R agonists could modulate neuroinflammation in HIVE, Gp1a (N-(piperidin-1-yl)-1-(2,4-dichlorophenyl)-1,4-dihydro-6-methylindeno[1,2-c]pyrazole-3-carboxamide), a new and highly selective synthetic CB2R agonist, was tested. Its biological activity expressed in K 1 values is 0.037 and 363 nM for CB2 and CB1 receptors, respectively (Murineddu et al. 2006). In hu-PBL/HIVE mice, Gp1a reduced the levels of immune activation, infiltration of human cells into the brain and downregulated neuroinflammation in HIVE. These results, taken together, warrant further investigation for Gp1a and other CB2R agonists as an adjunctive therapy for HAND.
Materials and methods
Cells and viruses
Monocytes and PBL were obtained from HIV-1, HIV-2, and hepatitis B seronegative donor leukopaks and were purified by countercurrent centrifugal elutriation. Monocytes were cultivated in DMEM (Sigma-Aldrich, St. Louis, MO, USA) supplemented with 10% heat-inactivated pooled human serum, 1% glutamine, 50 mg/ml gentamicin, 10 mg/ml ciprofloxacin (Sigma-Aldrich), and 1,000 U/ml highly purified recombinant human macrophage MCSF (generous gift from Genetics Institute, Cambridge, MA, USA). After 7 days in culture, MDM were infected with HIV-1ADA at a multiplicity of infection of 0.01.
Hu-PBL/HIVE mice
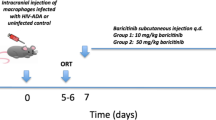
Balb/c-Rag2−/−γc−/− mice were obtained from the Central Institute of Experimental Animals (Dr. Mamoru Ito, Kawasaki, Japan) and were bred and maintained under specific pathogen-free conditions in accordance with the ethical guidelines for the care of laboratory animals at the University of Nebraska Medical Center and National Institutes of Health. Four-week-old animals were injected intraperitoneally (i.p.) with human PBL (20 × 106 cells/mouse). On day 7 after PBL reconstitution, HIV-1ADA-infected MDM (3 × 105 cells in 5 ml) were injected intracranially (i.c.) to produce hu-PBL/HIVE mice (Fig. 1a). Sham-operated mice with media injected i.c. were controls. PBL-reconstituted mice without intracranial injections of HIV-1-infected cells were included in the immunological analyses (hu-PBL mice). Animals were sacrificed at day 21 after reconstitution. Mice with limited PBL engraftment (<10% of human cells in mouse spleen) were excluded from the analyses. Three individual experiments with cells from different donors were performed. Brain tissues were used for immunohistochemistry or for RNA extractions.
hu-PBL/HIVE mouse model and scheme of the experiment. a Balb/c-Rag−/−γc−/− mice were reconstituted intraperitoneally with human PBL isolated from leukopaks. Monocytes isolated from the same leukopak were differentiated into macrophages, infected with HIV-1ADA and injected into the brain caudate putamen intracranially on day 7 using stereotactic equipment. From the day when mice are injected intracranially, they were fed (per os) with Gp1a mixed in almond paste for 14 days and then sacrificed to collect brain, blood, and spleen. For RT-PCR on brain, a 2-mm slice on ipsilateral hemisphere containing the injection area was collected for RNA extraction. The corresponding contralateral area was also used for comparison. b Immunohistology of various tissue sections isolated from a mouse reconstituted with hu-PBL for 21 days stained for human CD45
Drug treatment
Gp1a was obtained from Tocris Bioscience (Ellisville, MO, USA) and thoroughly mixed in almond paste to orally feed mice at a concentration of 1 mg/kg body weight/day. Animals were fed the drug from day 7 after PBL reconstitution or from day 1 of i.c. injections with HIV-1-infected MDM until sacrifice. The duration of drug treatment was 14 days (Fig. 1a). Control animals received vehicle alone.
FACS analysis of splenocytes
Spleens were extracted from the mice at sacrifice and crushed through a 40-μm cell strainer to obtain single cell suspensions. Splenocytes thus isolated were stained for human cells using antibodies to CD45, CD4, CD8, CD95, CD178, and CCR5. Appropriate isotype controls were used, and all antibodies were obtained from BD Pharmingen (San Diego, CA, USA). Cells were analyzed using BD LSR II with BD FACS Diva software (BD Immunocytometry Systems, Mountain View, CA, USA).
Viral load analysis
The levels of viral RNA copies per milliliter in plasma were analyzed using automated COBAS Amplicor System (Roche Molecular Diagnostics, Basel, Switzerland) with detection limit of 50 viral RNA copies/ml. Mouse plasma samples (20 μl each) were diluted to 500 μl with normal human serum for assay use, which makes the detection limit 1,250 copies/ml. HIV-1 infection was confirmed by virologic and histologic examinations.
Immunocytochemistry
Brain tissues were collected on day 14 after i.c. injection, fixed in 4% phosphate-buffered paraformaldehyde, and embedded in paraffin for later use. Blocks were cut to identify the injection site. For each mouse, 30–100 serial (5-μm-thick) sections were cut to include human MDM injection site, and three to seven slides (10 sections apart) were analyzed. Brain sections were deparaffinized with xylene and hydrated in gradient alcohols. Immunohistochemical staining followed previously published procedures for human CD45 (1/50; BD PharMingen), mouse glial fibrillary acidic protein (GFAP; 1/3,000, DAKO, Carpinteria, CA, USA), Iba-1, and HIV-1 p24 (DAKO) (Poluektova et al. 2004a). The Vectastain Elite ABC kit (Vector Laboratories, Burlingame, CA, USA) and the DAKO EnVision polymer-based system developed the immunolabeling tests. All paraffin-embedded sections were counterstained with Mayer’s hematoxylin. Deletion of primary Ab or mouse IgG served as controls. Tissue examination was performed with an Eclipse 800 microscope (Nikon, Melville, NY, USA).
Real time RT-PCR
Total RNA from brain sections was extracted with TRIzol (Invitrogen, Carlsbad, CA, USA). RNA was reverse transcribed with random hexamers, and real-time quantitative polymerase chain reaction (PCR) was performed with complementary DNA using an ABI PRISM 7000 sequence detector (Applied Biosystems, Foster City, CA, USA). Taqman assays-on-demand were used to determine the expression of HLA-DQ for human cells, mouse GFAP, tumor necrosis factor-α (TNF-α), interleukin-6 (IL-6), IL-1β, and inducible nitric oxide synthase (iNOS). HIV-1gag expression was determined using PCR assays (Cota et al. 2000). All PCR reagents were obtained from Applied Biosystems. Gene expression was normalized to glyceraldehyde 3-phosphate dehydrogenase and used as an endogenous control.
Statistical analysis
Data were analyzed using Excel with Student’s t test for comparisons and ANOVA. A value of p < 0.05 was considered statistically significant. All results are presented as the mean ± SD.
Results
Hu-PBL/HIVE mice and cannabinoid receptor expression
The hu-PBL/HIVE mouse model allows the investigation of neuroinflammation, effector function of T cells, and HIV-1 replication as it would occur in an infected human host (Poluektova et al. 2002, 2004a, b). Balb/c-Rag2–/–γc–/– mice were transplanted with human PBL into the peritoneal cavity followed by i.c. injection of HIV-1-infected human MDM. Figure 1a illustrates the steps involved in the model generation and the experimental scheme. Mice were treated with Gp1a from day 7, when the mice were injected i.c. with HIV-1-infected MDM, and continued for 14 days. Human lymphocytes reconstitute mouse lymph nodes, spleen, and other tissues including the liver, lung, and intestine (Fig. 1b). When HIV-1 MDM are placed in the brain, lymphocytes readily ingress and proliferate in association with HIV-1-infected human MDM (Poluektova et al. 2004a). At the time of sacrifice, brains collected were either processed for histological evaluation or RNA extraction.
Brain sections were stained for human cells (CD45), HIV-1 p24, murine astrocytes (GFAP), and macrophage/microglial cells (Iba-1, species non-specific). Representative sections are shown in Fig. 2. Serial sections containing the injected site are shown containing both implanted HIV-1-infected MDM and infiltrated human lymphocytes. Sections stained for Iba-1 demonstrate microglial nodule formation around the injection line as a result of increased microglial activation. Gp1a treatment shows reduced intensity of Iba-1 staining, but GFAP was found to be similar in both groups. Quantitative analysis of cannabinoid receptors, human cell presence, the levels of infection, and inflammation was performed by RT-PCR.
Immunohistology of brain sections. Paraffin-embedded 5-μm sections were stained for human CD45 to detect human cells, HIV-1 p24 for infected cells, glial markers Iba-1 for microglial activation, and GFAP for astrogliosis. CD45 stained both injected human macrophages and infiltrated peripheral lymphocytes. Bigger cells are macrophages (red arrow) and smaller/brighter cells (black arrow) are lymphocytes. Iba-1 stained both injected macrophages and activated mouse microglia surrounding the injection line. Microglial nodules (arrow heads) were observed to a higher extent in the non-treated group. GFAP intensity was found to be similar
Cannabinoid receptor expression
RNA extracted from the brains collected at 14 days after i.c. injections were analyzed by real-time RT-PCR for CB1 and CB2 receptors and GPR55. CB1 receptors and GPR55 are widely expressed in the brain, and we could not find significant changes in the expression of CB1R and GPR55 in hu-PBL/HIVE mouse brain when compared to sham-injected controls. However, CB2 receptor expression was significantly higher (fourfold, p < 0.005) in injected hemispheres of hu-PBL/HIVE mice compared to their contralateral hemispheres and also to that of sham-injected mice (Fig. 3). Based on these results, we next tested whether CB2 receptor agonists could be effective as neuroimmune modulators in our murine HIVE model of human disease.
Cannabinoid receptor expression in hu-PBL/HIVE mice brains. CB1R, CB2R, and GPR55 expressions in brains were analyzed by RT-PCR. Only CB2R was found to be upregulated in the ipsilateral hemisphere (ih) containing the injected site compared to its contralateral hemisphere (ch) and to the sham-operated mice brains. Results are expressed as mean±SD and #p < 0.05 compared to HIVE ih
Human cells and HIV-1 viral load in the brain
Brains were analyzed by RT-PCR for human cells (HLA-DQ gene) and HIV-1 infection (HIV-1gag). HLA-DQ expression was significantly reduced in both ipsilateral hemispheres (containing injection site) and contralateral hemispheres of Gp1a-treated animals compared to non-treated controls [tenfold reduction (Fig. 4a)]. This decrease in HLA-DQ could account for both reduced numbers of infiltrated cells as well as reduced expression of HLA-DQ with decrease in activation. HIV-1 gag expression in brains was reduced with Gp1a but did not reach significant levels (49 ± 36 vs 20.6 ± 12, p = 0.058). Peripheral viral load measured in the sera of non-treated animals was similar to that of Gp1a-treated animals (Fig. 4b).
Human cells and HIV-1 infection in brain. a RT-PCR analyses of brains from hu-PBL/HIVE mice showed a significant decrease in HLA-DQ expression in the Gp1a-treated group when compared to the corresponding hemispheres from the non-treated group. HIV-1 gag expression was reduced with Gp1a but did not reach significance. Results are expressed as mean±SD and #p < 0.05 compared to HIVE ih. b Viral load in peripheral blood was measured using Roche COBAS Amplicor system and the viral RNA copy numbers were not significantly different between the two groups. Results are expressed as mean±SD
Brain inflammation
Expression of inflammation markers and inflammatory cytokines in the brains was determined by real-time RT-PCR. Figure 5 shows the expression levels of Mac-1 (CD11b) for microglial activation, iNOS, and TNF-α in the brains of Gp1a-treated and non-treated HIVE animals. A significant decrease in Mac-1 and TNF-α expression in the ipsilateral hemispheres of Gp1a-treated brains was observed compared to non-treated controls (twofold decrease, p < 0.05). Infiltration of human cells into the brain induced significant levels of iNOS expression, and Gp1a treatment did not reduce iNOS expression. GFAP as a marker of astrogliosis and the proinflammatory cytokines including IL-6 and IL-1B were also measured and were not affected significantly by Gp1a treatment (data not shown).
RT-PCR for brain inflammatory markers. Mac-1 (CD11b), TNf-α, and iNOS expressions were determined by real-time PCR with the RNA obtained from ipsilateral (ih) and contralateral hemispheres (ch) of hu-PBL/HIVE mice treated or not treated with Gp1a. Mac-1 and TNF-α were found to be significantly reduced with Gp1a treatment. Results are expressed as mean±SD and #p < 0.05 compared to HIVE ih
Gp1a and survival
Human PBL reconstitution in immunodeficient mice has been shown to induce graft-versus-host (GVH) disease due to the activation of human cells reacting to the mouse environment. Almost 50% of animals died from the non-treated group, while 100% survival was observed in the treated group (Fig. 6). Gp1a treatment significantly reduced GVH-induced deaths.
Survival of hu-PBL reconstituted mice with Gp1a. Hu-PBL reconstitution induces GVH in some animals. Gp1a treatment dramatically reduced the deaths by GVH. One hundred percent survival was seen in Gp1a-treated group compared to ∼50% survival rate in the non-treated groups. Results from one out of three individual experiments are shown. N = 20 per group
CD4/CD8 T-cell numbers and FasL expression
Morphologically, spleens from Gp1a-treated animals were two times smaller than those of non-treated animals. Flow cytometric analyses on splenocytes showed that the total number of human cells (CD45+) was slightly reduced in Gp1a-treated non-infected hu-PBL mice compared to controls (Fig. 7). Significant reduction in the total number of human cells was observed with HIV infection (hu-PBL vs HIVE, 70.8 ± 3.3% and 43.1 ± 6.1%, respectively). Remarkable changes in CD4/CD8 ratios were found with Gp1a treatment in hu-PBL mice. Normally, with PBL reconstitution in immunodeficient mice, CD8+ cells are found at greater proportion than CD4+ cells (CD4/CD8 ratio 0.7 ± 0.1). Gp1a treatment normalized the ratio to 1.0 ± 0.3, by increasing the number of CD4+ cells (31.7 ± 5.04% to 37.5 ± 5%, p < 0.05) and by reducing the number of CD8+ cells (48 ± 2 to 35 ± 12). In HIVE animals, the number of CD4+ cells was decreased with infection and the CD4/CD8 ratios fell to 0.5 ± 0.2. Gp1a treatment in HIVE mice increased the CD4/CD8 ratio to 0.7 ± 0.2, but the rise was not significant compared to the non-treated group. To explore the mechanisms of Gp1a-mediated changes in populations of human cells, we studied lymphocyte surface expression of Fas (CD95) and Fas ligand (FasL, CD178). CB2R agonists were shown to induce apoptosis (Lombard et al. 2007), and the Fas/FasL pathway contributes to the deletion of activated T cells. CD95 expression was similar throughout the groups; however, CD178 was found to be differentially expressed on both CD4 and CD8 positive T cells with Gp1a treatment (Fig. 4a). Gp1a increased CD178 expression on CD4+ cells in hu-PBL mice while the increase in Gp1a-treated HIVE mice was not significantly higher than non-treated HIVE mice. In both infected and non-infected mice, CD178 expression on CD8+ cells was significantly increased with Gp1a.
Human cells in spleen. Splenocytes were stained for human CD45, CD4, CD8, and CD178 (FasL) surface expression by flow cytometry. a Human CD45+ cells in spleen were decreased with Gp1a in hu-PBL or hu-PBL/HIVE mice but not significantly when compared to the non-treated groups. HIV-1 infection in Hu-PBL/HIVE mice significantly reduced CD45 cell numbers compared to hu-PBL mice. CD4/CD8 ratios were significantly increased with Gp1a in hu-PBL mice. Reduction in CD4/CD8 ratio in Hu-PBL/HIVE mice compared to non-infected hu-PBL mice. CD178 expression in both CD4 and CD8 positive cells was increased to a significant extent with Gp1a treatment. Results are expressed as mean±SD and #p < 0.05 and $p < 0.05 compared to hu-PBL and HIVE groups, respectively. b. FACS scatter plots demonstrating differential expression of CD178 on CD4 and CD8 cells are shown. Gating strategy was human CD45 → CD4 → CD178 or CD45 → CD8 → CD178
CCR5 receptor expression
Given that the reconstituted human PBL in mice get significantly activated and as a result induce GVH, and the Gp1a treatment reduced GVH-induced deaths significantly, we wanted to estimate the changes in the activation levels of T cells with Gp1a treatment. Due to a significant involvement of CCR5 receptor in HIV-1 entry in human lymphocytes and macrophages and because this marker is broadly expressed on activated human memory T cells, which preferentially are eliminated during HIV-1 infection, we stained the splenocytes for CCR5. Flow cytometric analyses showed that CCR5 expression on CD4+ cells was significantly reduced with Gp1a treatment both in hu-PBL (25.9 ± 5.7 non-treated vs 16.7 ± 4.8 treated) and HIVE mice (13 ± 2.8 vs 9.7 ± 4.6) (Fig. 8). The levels of CCR5 expression on CD8+ cells was not altered significantly between treated and non-treated groups (50.5 ± 10.8% and 56 ± 6.5%, respectively).
Expression of activation markers on human cells. Splenocytes were stained for CCR5 surface expression and analyzed by FACS. a CCR5 expression on CD4+ cells was reduced significantly with Gp1a treatment both in hu-PBL and hu-PBL/HIVE mice compared to the non-treated groups. Results are expressed as mean±SD and #p < 0.05 and $p < 0.05 compared to hu-PBL and HIVE groups, respectively. b Representative flow cytometry scatter plots are shown. Gating strategy was human CD45 → CD4 → CCR5 or CD45 → CD8 → CCR5
Discussion
HAND results from neuronal injury induced by inflammatory neurotoxic factors produced from immune-competent and virus-infected perivascular macrophages and microglia (Gendelman et al. 1997, 2004). The neurotoxic factors include, but are not limited to, HIV-1 proteins such as gp120 and tat, proinflammatory cytokines, arachidonic acid and its metabolites, quinolinic acid, and glutamate (Kaul et al. 2005; Rostasy et al. 1999; Smith et al. 2001). Despite the widespread use of antiretroviral therapy, mild cognitive impairments still persist (Ances and Ellis 2007; Kaul and Lipton 2006). SIV-infected rhesus macaques demonstrate neural dysfunction in the early stages of viral infection (Marcondes et al. 2001), and these disturbances parallel T-cell ingress to the brain (Marcondes et al. 2001, 2003). Hu-PBL/HIVE mice mimics the hallmarks of human brain disease such as the presence of HIV-1-infected multinucleated giant cells, T cell infiltration, astro- and microgliosis, and neuronal loss with neuropathological observations of myelin pallor, features of advanced HIV-1 infection, and encephalitis. Increased expression of neurotoxic factors including proinflammatory cytokines and iNOS was also described (Poluektova et al. 2004a). Using mouse models of neuroAIDS, our laboratories investigated a range of immune-modulatory adjunctive therapies that can interrupt neuronal damage and attenuate macrophage activation (Eggert et al. 2009, 2010; Gorantla et al. 2007; Potula et al. 2005). We now report that the mouse model also exhibits altered CB2 receptor expression. Profound changes in the distribution pattern of cannabinoid CB2 and FAAH proteins in cortical regions of macaque brains affected SIV encephalitis (SIVE) (Benito et al. 2005). CB2 receptors were shown to be expressed abundantly in perivascular microglial cells and infiltrated lymphocytes in the brain. Interestingly, no changes in CB1 receptor and GPR55 expression were observed. CB1 receptor and GPR55 are widely expressed in the CNS (Ryberg et al. 2007). The pattern of CB1 receptor expression is unchanged in AD (Benito et al. 2003) and SIVE-infected macaque brains (Benito et al. 2005). Since our mouse model allows experimental modulation of adaptive immunity as well as microglial activation, we attempted to investigate whether CB2 receptor agonist could lead to neuroprotection in HIVE.
CB2 receptor is the main peripheral molecular target responsible for the inhibitory properties of the cannabinoids on the immune system (Docagne et al. 2008; Lunn et al. 2006, 2008). In the present study, we have evaluated the effect of CB2 receptor activation on the immune system using Gp1a, a new synthetic CB2R-selective agonist. Using the hu-PBL/HIVE mouse model, we demonstrated that Gp1a induced a strong suppressive effect on the immune system. There are several synthetic CB2 agonists available, none specific to CB2 or CB1 alone because there is 40% homology between the receptors. However, different agonists have varying levels of affinity towards the receptors. Gp1a is a highly selective CB2 agonist with a binding affinity for CB2R 1,000-fold higher than for CB1R (Murineddu et al. 2006). Ours is the first in vivo study using this agonist.
There was a significant reduction in the HLA-DQ expression, a measure of the number of human cells as well as the level of human cell activation, in Gp1a-treated brains. CB2 receptor agonists suppress leukocyte trafficking by reducing rolling and infiltration into inflamed areas (Xu et al. 2007). Activated proinflammatory Th1-directed T cells infiltrate the brain during HIV infection and could affect perivascular macrophage and microglial responses, leading to neuronal dysfunction and deficits in neural structural integrity (Gorantla et al. 2007). Diminished microglial activation and decreased proinflammatory cytokine TNF-α expression in HIVE brains due to Gp1a could be due to the reduced infiltration of activated lymphocytes into the brain, as well as a direct effect of agonist on microglia. CB2R stimulation was shown to suppress microglial activation and reduce TNF-α production in rodent microglial cells (Ehrhart et al. 2005; Facchinetti et al. 2003; Puffenbarger et al. 2000). Gp1a did not downregulate iNOS, probably because inducible nitric oxide production in brains is regulated via CB1R (Cabral et al. 2001; Waksman et al. 1999). In addition, the effect of CB2R agonist on endothelial cells could also contribute to diminished infiltration of human cells into the brain by reducing the expression of adhesion molecules required for transmigration (Burstein et al. 1992; Mestre et al. 2009). Given that Gp1a is an agonist for both mouse and human CB2R, the effect of Gp1a on mouse macrophages and endothelial cells needs to be investigated in further studies.
Significant reduction in GVH-mediated deaths with Gp1a treatment suggested an immune-suppressive effect of Gp1a. It is well known that hu-PBL mice experience severe GVH disease (Berney et al. 2001; Gorin et al. 2002; Hoffmann-Fezer et al. 1993; Huppes et al. 1994; Roychowdhury et al. 2005; Tary-Lehmann et al. 1994; Tournoy et al. 2000; Verlinden et al. 1998). Depending on the human-murine histocompatibility, hu-PBL animals readily develop GVH disease that kills mice in a time-frame from 3 to 5 weeks post transplantation (Goldman et al. 1998; Pino et al. 2010; Schneider and Gronvik 1995). The use of this model provided a unique possibility to evaluate anti-GVH activity of CB2R signaling and Gp1a was found to be a strong suppressant of GVH. FasL was found to be involved in this immunosuppressive effect of Gp1a. Increased expression of FasL on both CD4 and CD8 positive cells was observed with Gp1a. Cannabinoids are known to induce apoptosis in immune cells and inhibit their proliferation (Lombard et al. 2007). FasL acts as a prototypic death factor due to its ability to induce apoptosis in Fas (APO-1, CD95) expressing cells (Dhein et al. 1995; Ju et al. 1995). Moreover, the ligation of Fas with FasL was recognized as an important homeostatic control mechanism for maintaining appropriate numbers of T cells (Takahashi et al. 1994). Its role in the downregulation of immune responses during activation-induced cell death (AICD) is well documented (Green et al. 2003). Increased levels of FasL expression with Gp1a might be leading to AICD in GVH and thus assist with the maintenance of immune homeostasis, as observed with an increase in CD4/CD8 ratios with Gp1a. However, an increase in FasL expression paralleling an increase in CD4+ cell numbers with Gp1a treatment is contradictory. Further analysis of T-cell subsets expressing FasL with Gp1a is needed to understand exactly in which subsets of CD4+ cells FasL is expressed, to understand the involvement of FasL in CD4+ cell rescue. Taken together, our study suggests that Gp1a regulates immune homeostasis and might have therapeutic potential in tissue transplantation.
CCR5 is another marker that we studied to understand the attenuation of lymphocyte activation by Gp1a. CCR5 is expressed mainly on Th1 cells, and T cells infiltrating inflammatory sites, usually of the activated/memory type, express CCR5 (Loetscher et al. 1998; Qin et al. 1998). CCR5 is a marker for T cells associated with certain inflammatory reactions. Reduction in CCR5 expression on CD4+ cells in hu-PBL mice may contribute to the decrease in chemokine-mediated homing to the inflamed sites. During HIV-1 infection, naive CD4+ T cells exhibit increased expression of the major viral co-receptor CCR5 (Ostrowski et al. 1998). In our study, there was a decrease in CCR5 expression on CD4+ cells with HIV infection in hu-PBL/HIVE mice compared to hu-PBL mice, which may be due to the progressive depletion of activated CD4+ cells with infection. Further reduction in CCR5 expression on CD4+ cells with Gp1a in HIV-1-infected mice did not reduce peripheral viral load. The synthetic CB1/CB2 agonist WIN55,212-2 was found to suppress replication of HIV-1 in microglial cell cultures via CB2 receptors (Rock et al. 2007). Decreased expression of HIV-gag was observed in the brains of Gp1a-treated animals but was not significant. The collective effect of Gp1a, either by reduced infiltration of these cells into HIVE mouse brains, by promoting the apoptosis of these cells or by altering the cytokine profiles to Th2 responses, diminished microglial activation and decreased proinflammatory cytokine TNF-α expression in HIVE brains.
The present study serves to provide a rationale for employing immunosuppressive strategies targeting the cannabinoid system to modulate neuroinflammatory responses for therapeutic use for HAND. Cannabinoids have widespread effects on both innate and adaptive immune responses; hence, the significance of CB2 receptor as an appealing therapeutic target has risen in recent years. Its effect on immune cell motility and novel immunoregulatory role of CB2 receptor specific compounds makes this approach attractive.
References
Ances BM, Ellis RJ (2007) Dementia and neurocognitive disorders due to HIV-1 infection. Semin Neurol 27(1):86–92
Arevalo-Martin A, Garcia-Ovejero D, Gomez O, Rubio-Araiz A, Navarro-Galve B, Guaza C, Molina-Holgado E, Molina-Holgado F (2008) CB2 cannabinoid receptors as an emerging target for demyelinating diseases: from neuroimmune interactions to cell replacement strategies. Br J Pharmacol 153(2):216–225
Ashton JC, Glass M (2007) The cannabinoid CB2 receptor as a target for inflammation-dependent neurodegeneration. Curr Neuropharmacol 5(2):73–80
Basavarajappa BS, Nixon RA, Arancio O (2009) Endocannabinoid system: emerging role from neurodevelopment to neurodegeneration. Mini Rev Med Chem 9(4):448–462
Benito C, Nunez E, Tolon RM, Carrier EJ, Rabano A, Hillard CJ, Romero J (2003) Cannabinoid CB2 receptors and fatty acid amide hydrolase are selectively overexpressed in neuritic plaque-associated glia in Alzheimer’s disease brains. J Neurosci 23(35):11136–11141
Benito C, Kim WK, Chavarria I, Hillard CJ, Mackie K, Tolon RM, Williams K, Romero J (2005) A glial endogenous cannabinoid system is upregulated in the brains of macaques with simian immunodeficiency virus-induced encephalitis. J Neurosci 25(10):2530–2536
Benito C, Nunez E, Pazos MR, Tolon RM, Romero J (2007a) The endocannabinoid system and Alzheimer’s disease. Mol Neurobiol 36(1):75–81
Benito C, Romero JP, Tolon RM, Clemente D, Docagne F, Hillard CJ, Guaza C, Romero J (2007b) Cannabinoid CB1 and CB2 receptors and fatty acid amide hydrolase are specific markers of plaque cell subtypes in human multiple sclerosis. J Neurosci 27(9):2396–2402
Berney T, Molano RD, Pileggi A, Cattan P, Li H, Ricordi C, Inverardi L (2001) Patterns of engraftment in different strains of immunodeficient mice reconstituted with human peripheral blood lymphocytes. Transplantation 72(1):133–140
Berrendero F, Sanchez A, Cabranes A, Puerta C, Ramos JA, Garcia-Merino A, Fernandez-Ruiz J (2001) Changes in cannabinoid CB(1) receptors in striatal and cortical regions of rats with experimental allergic encephalomyelitis, an animal model of multiple sclerosis. Synapse 41(3):195–202
Buckley NE (2008) The peripheral cannabinoid receptor knockout mice: an update. Br J Pharmacol 153(2):309–318
Burstein SH, Audette CA, Breuer A, Devane WA, Colodner S, Doyle SA, Mechoulam R (1992) Synthetic nonpsychotropic cannabinoids with potent antiinflammatory, analgesic, and leukocyte antiadhesion activities. J Med Chem 35(17):3135–3141
Cabral GA, Griffin-Thomas L (2008) Cannabinoids as therapeutic agents for ablating neuroinflammatory disease. Endocr Metab Immune Disord Drug Targets 8(3):159–172
Cabral GA, Harmon KN, Carlisle SJ (2001) Cannabinoid-mediated inhibition of inducible nitric oxide production by rat microglial cells: evidence for CB1 receptor participation. Adv Exp Med Biol 493:207–214
Cabral GA, Raborn ES, Griffin L, Dennis J, Marciano-Cabral F (2008) CB2 receptors in the brain: role in central immune function. Br J Pharmacol 153(2):240–251
Centonze D, Rossi S, Finazzi-Agro A, Bernardi G, Maccarrone M (2007) The (endo)cannabinoid system in multiple sclerosis and amyotrophic lateral sclerosis. Int Rev Neurobiol 82:171–186
Correa F, Docagne F, Mestre L, Loria F, Hernangomez M, Borrell J, Guaza C (2007) Cannabinoid system and neuroinflammation: implications for multiple sclerosis. Neuroimmunomodulation 14(3–4):182–187
Cota M, Mengozzi M, Vicenzi E, Panina-Bordignon P, Sinigaglia F, Transidico P, Sozzani S, Mantovani A, Poli G (2000) Selective inhibition of HIV replication in primary macrophages but not T lymphocytes by macrophage-derived chemokine. Proc Natl Acad Sci U S A 97(16):9162–9167
Dhein J, Walczak H, Baumler C, Debatin KM, Krammer PH (1995) Autocrine T-cell suicide mediated by APO-1/(Fas/CD95). Nature 373(6513):438–441
Docagne F, Mestre L, Loria F, Hernangomez M, Correa F, Guaza C (2008) Therapeutic potential of CB2 targeting in multiple sclerosis. Expert Opin Ther Targets 12(2):185–195
Eggert D, Dash PK, Serradji N, Dong CZ, Clayette P, Heymans F, Dou H, Gorantla S, Gelbard HA, Poluektova L et al (2009) Development of a platelet-activating factor antagonist for HIV-1 associated neurocognitive disorders. J Neuroimmunol 213(1-2):47–59
Eggert D, Dash PK, Gorantla S, Dou H, Schifitto G, Maggirwar SB, Dewhurst S, Poluektova L, Gelbard HA, Gendelman HE (2010) Neuroprotective activities of CEP-1347 in models of neuroAIDS. J Immunol 184(2):746–756
Ehrhart J, Obregon D, Mori T, Hou H, Sun N, Bai Y, Klein T, Fernandez F, Tan J, Shytle RD (2005) Stimulation of cannabinoid receptor 2 (CB2) suppresses microglial activation. J Neuroinflammation 2:29
Facchinetti F, Del Giudice E, Furegato S, Passarotto M, Leon A (2003) Cannabinoids ablate release of TNFalpha in rat microglial cells stimulated with lipopolysaccharide. Glia 41(2):161–168
Galiegue S, Mary S, Marchand J, Dussossoy D, Carriere D, Carayon P, Bouaboula M, Shire D, Le Fur G, Casellas P (1995) Expression of central and peripheral cannabinoid receptors in human immune tissues and leukocyte subpopulations. Eur J Biochem 232(1):54–61
Gendelman HE, Persidsky Y, Ghorpade A, Limoges J, Stins M, Fiala M, Morrisett R (1997) The neuropathogenesis of the AIDS dementia complex. Aids 11(Suppl A):35–45
Gendelman HE, Diesing S, Gelbard H, Swindells S (2004) The neuropathogenesis of HIV-1 infection. Elsevier, London, pp 95–116
Goldman JP, Blundell MP, Lopes L, Kinnon C, Di Santo JP, Thrasher AJ (1998) Enhanced human cell engraftment in mice deficient in RAG2 and the common cytokine receptor gamma chain. Br J Haematol 103(2):335–342
Gonsiorek W, Hesk D, Chen SC, Kinsley D, Fine JS, Jackson JV, Bober LA, Deno G, Bian H, Fossetta J et al (2006) Characterization of peripheral human cannabinoid receptor (hCB2) expression and pharmacology using a novel radioligand, [35S]Sch225336. J Biol Chem 281(38):28143–28151
Gorantla S, Liu J, Sneller H, Dou H, Holguin A, Smith L, Ikezu T, Volsky DJ, Poluektova L, Gendelman HE (2007) Copolymer-1 induces adaptive immune anti-inflammatory glial and neuroprotective responses in a murine model of HIV-1 encephalitis. J Immunol 179(7):4345–4356
Gorin NC, Piantadosi S, Stull M, Bonte H, Wingard JR, Civin C (2002) Increased risk of lethal graft-versus-host disease-like syndrome after transplantation into NOD/SCID mice of human mobilized peripheral blood stem cells, as compared to bone marrow or cord blood. J Hematother Stem Cell Res 11(2):277–292
Green DR, Droin N, Pinkoski M (2003) Activation-induced cell death in T cells. Immunol Rev 193:70–81
Hoffmann-Fezer G, Gall C, Zengerle U, Kranz B, Thierfelder S (1993) Immunohistology and immunocytology of human T-cell chimerism and graft-versus-host disease in SCID mice. Blood 81(12):3440–3448
Huppes W, Fickenscher H, tHart BA, Fleckenstein B (1994) Cytokine dependence of human to mouse graft-versus-host disease. Scand J Immunol 40(1):26–36
Ju ST, Panka DJ, Cui H, Ettinger R, el-Khatib M, Sherr DH, Stanger BZ, Marshak-Rothstein A (1995) Fas(CD95)/FasL interactions required for programmed cell death after T-cell activation. Nature 373(6513):444–448
Kaul M, Lipton SA (2006) Mechanisms of neuronal injury and death in HIV-1 associated dementia. Curr HIV Res 4(3):307–318
Kaul M, Zheng J, Okamoto S, Gendelman HE, Lipton SA (2005) HIV-1 infection and AIDS: consequences for the central nervous system. Cell Death Differ 12(Suppl 1):878–892
Klein TW, Newton CA (2007) Therapeutic potential of cannabinoid-based drugs. Adv Exp Med Biol 601:395–413
Kreitzer FR, Stella N (2009) The therapeutic potential of novel cannabinoid receptors. Pharmacol Ther 122(2):83–96
Lastres-Becker I, Molina-Holgado F, Ramos JA, Mechoulam R, Fernandez-Ruiz J (2005) Cannabinoids provide neuroprotection against 6-hydroxydopamine toxicity in vivo and in vitro: relevance to Parkinson’s disease. Neurobiol Dis 19(1–2):96–107
Leonelli M, Torrao AS, Britto LR (2009) Unconventional neurotransmitters, neurodegeneration and neuroprotection. Braz J Med Biol Res 42(1):68–75
Loetscher P, Uguccioni M, Bordoli L, Baggiolini M, Moser B, Chizzolini C, Dayer JM (1998) CCR5 is characteristic of Th1 lymphocytes. Nature 391(6665):344–345
Lombard C, Nagarkatti M, Nagarkatti P (2007) CB2 cannabinoid receptor agonist, JWH-015, triggers apoptosis in immune cells: potential role for CB2-selective ligands as immunosuppressive agents. Clin Immunol 122(3):259–270
Lunn CA, Reich EP, Bober L (2006) Targeting the CB2 receptor for immune modulation. Expert Opin Ther Targets 10(5):653–663
Lunn CA, Reich EP, Fine JS, Lavey B, Kozlowski JA, Hipkin RW, Lundell DJ, Bober L (2008) Biology and therapeutic potential of cannabinoid CB2 receptor inverse agonists. Br J Pharmacol 153(2):226–239
Mackie K, Stella N (2006) Cannabinoid receptors and endocannabinoids: evidence for new players. AAPS J 8(2):E298–E306
Marcondes MCG, Burudi EME, Huitron-Resendiz S, Sanchez-Alavez M, Watry D, Zandonatti M, Henriksen SJ, Fox HS (2001) Highly activated CD8+ T cells in the brain correlate with early central nervous system dysfunction in simian immunodeficiency virus infection. J Immunol 167(9):5429–5438
Marcondes MC, Phillipson CA, Fox HS (2003) Distinct clonal repertoire of brain CD8+ cells in simian immunodeficiency virus infection. AIDS 17(11):1605–1611
Mestre L, Docagne F, Correa F, Loria F, Hernangomez M, Borrell J, Guaza C (2009) A cannabinoid agonist interferes with the progression of a chronic model of multiple sclerosis by downregulating adhesion molecules. Mol Cell Neurosci 40(2):258–266
Murineddu G, Lazzari P, Ruiu S, Sanna A, Loriga G, Manca I, Falzoi M, Dessi C, Curzu MM, Chelucci G et al (2006) Tricyclic pyrazoles. 4. Synthesis and biological evaluation of analogues of the robust and selective CB2 cannabinoid ligand 1-(2′, 4′-dichlorophenyl)-6-methyl-N-piperidin-1-yl-1, 4-dihydroindeno[1, 2-c ]pyrazole-3-carboxamide. J Med Chem 49(25):7502–7512
Onaivi ES (2009) Cannabinoid receptors in brain: pharmacogenetics, neuropharmacology, neurotoxicology, and potential therapeutic applications. Int Rev Neurobiol 88:335–369
Ostrowski MA, Justement SJ, Catanzaro A, Hallahan CA, Ehler LA, Mizell SB, Kumar PN, Mican JA, Chun TW, Fauci AS (1998) Expression of chemokine receptors CXCR4 and CCR5 in HIV-1-infected and uninfected individuals. J Immunol 161(6):3195–3201
Pacher P, Batkai S, Kunos G (2006) The endocannabinoid system as an emerging target of pharmacotherapy. Pharmacol Rev 58(3):389–462
Patel KD, Davison JS, Pittman QJ, Sharkey KA (2010) Cannabinoid CB(2) receptors in health and disease. Curr Med Chem 17(14):1394–1410
Pearson T, Greiner DL, Shultz LD (2008) Humanized SCID mouse models for biomedical research. Curr Top Microbiol Immunol 324:25–51
Pino S, Brehm MA, Covassin-Barberis L, King M, Gott B, Chase TH, Wagner J, Burzenski L, Foreman O, Greiner DL et al (2010) Development of novel major histocompatibility complex class I and class II-deficient NOD-SCID IL2R gamma chain knockout mice for modeling human xenogeneic graft-versus-host disease. Methods Mol Biol 602:105–117
Piomelli D (2005) The endocannabinoid system: a drug discovery perspective. Curr Opin Investig Drugs 6(7):672–679
Poluektova LY, Munn DH, Persidsky Y, Gendelman HE (2002) Generation of cytotoxic T cells against virus-infected human brain macrophages in a murine model of HIV-1 encephalitis. J Immunol 168(8):3941–3949
Poluektova L, Gorantla S, Faraci J, Birusingh K, Dou H, Gendelman HE (2004a) Neuroregulatory events follow adaptive immune-mediated elimination of HIV-1-infected macrophages: studies in a murine model of viral encephalitis. J Immunol 172(12):7610–7617
Poluektova LY, Gorantla S, Gendelman HE (2004b) Studies of adaptive immunity in a murine model of HIV-1 encephalitis. In: Gendelman HE, Grant I, Lipton S, Swindells S (eds) Neurology of AIDS. Oxford University Press, Oxford
Potula R, Poluektova L, Knipe B, Chrastil J, Heilman D, Dou H, Takikawa O, Munn DH, Gendelman HE, Persidsky Y (2005) Inhibition of indoleamine 2, 3-dioxygenase (IDO) enhances elimination of virus-infected macrophages in an animal model of HIV-1 encephalitis. Blood 106(7):2382–2390
Price DA, Martinez AA, Seillier A, Koek W, Acosta Y, Fernandez E, Strong R, Lutz B, Marsicano G, Roberts JL et al (2009) WIN55, 212-2, a cannabinoid receptor agonist, protects against nigrostriatal cell loss in the 1-methyl-4-phenyl-1, 2, 3, 6-tetrahydropyridine mouse model of Parkinson’s disease. Eur J Neurosci 29(11):2177–2186
Puffenbarger RA, Boothe AC, Cabral GA (2000) Cannabinoids inhibit LPS-inducible cytokine mRNA expression in rat microglial cells. Glia 29(1):58–69
Qin S, Rottman JB, Myers P, Kassam N, Weinblatt M, Loetscher M, Koch AE, Moser B, Mackay CR (1998) The chemokine receptors CXCR3 and CCR5 mark subsets of T cells associated with certain inflammatory reactions. J Clin Invest 101(4):746–754
Rock RB, Gekker G, Hu S, Sheng WS, Cabral GA, Martin BR, Peterson PK (2007) WIN55, 212-2-mediated inhibition of HIV-1 expression in microglial cells: involvement of cannabinoid receptors. J Neuroimmune Pharmacol 2(2):178–183
Rostasy K, Monti L, Yiannoutsos C, Kneissl M, Bell J, Kemper TL, Hedreen JC, Navia BA (1999) Human immunodeficiency virus infection, inducible nitric oxide synthase expression, and microglial activation: pathogenetic relationship to the acquired immunodeficiency syndrome dementia complex. Ann Neurol 46(2):207–216
Roychowdhury S, Blaser BW, Freud AG, Katz K, Bhatt D, Ferketich AK, Bergdall V, Kusewitt D, Baiocchi RA, Caligiuri MA (2005) IL-15 but not IL-2 rapidly induces lethal xenogeneic graft-versus-host disease. Blood 106(7):2433–2435
Ryberg E, Larsson N, Sjogren S, Hjorth S, Hermansson NO, Leonova J, Elebring T, Nilsson K, Drmota T, Greasley PJ (2007) The orphan receptor GPR55 is a novel cannabinoid receptor. Br J Pharmacol 152(7):1092–1101
Schneider MK, Gronvik KO (1995) Acute graft-versus-host reaction in SCID mice leads to an abnormal expansion of CD8+ V beta 14+ and a broad inactivation of donor T cells followed by a host-restricted tolerance and a normalization of the TCR V beta repertoire in the chronic phase. Scand J Immunol 41(4):373–383
Shoemaker JL, Seely KA, Reed RL, Crow JP, Prather PL (2007) The CB2 cannabinoid agonist AM-1241 prolongs survival in a transgenic mouse model of amyotrophic lateral sclerosis when initiated at symptom onset. J Neurochem 101(1):87–98
Smith DG, Guillemin GJ, Pemberton L, Kerr S, Nath A, Smythe GA, Brew BJ (2001) Quinolinic acid is produced by macrophages stimulated by platelet activating factor, Nef and Tat. J NeuroVirol 7(1):56–60
Steffens S, Veillard NR, Arnaud C, Pelli G, Burger F, Staub C, Karsak M, Zimmer A, Frossard JL, Mach F (2005) Low dose oral cannabinoid therapy reduces progression of atherosclerosis in mice. Nature 434(7034):782–786
Stella N (2009) Endocannabinoid signaling in microglial cells. Neuropharmacology 56(Suppl 1):244–253
Takahashi T, Tanaka M, Brannan CI, Jenkins NA, Copeland NG, Suda T, Nagata S (1994) Generalized lymphoproliferative disease in mice, caused by a point mutation in the Fas ligand. Cell 76(6):969–976
Tary-Lehmann M, Lehmann PV, Schols D, Roncarolo MG, Saxon A (1994) Anti-SCID mouse reactivity shapes the human CD4+ T cell repertoire in hu-PBL-SCID chimeras. J Exp Med 180(5):1817–1827
Thakur GA, Tichkule R, Bajaj S, Makriyannis A (2009) Latest advances in cannabinoid receptor agonists. Expert Opin Ther Pat 19(12):1647–1673
Tournoy KG, Depraetere S, Pauwels RA, Leroux-Roels GG (2000) Mouse strain and conditioning regimen determine survival and function of human leucocytes in immunodeficient mice. Clin Exp Immunol 119(1):231–239
Van Duyne R, Pedati C, Guendel I, Carpio L, Kehn-Hall K, Saifuddin M, Kashanchi F (2009) The utilization of humanized mouse models for the study of human retroviral infections. Retrovirology 6(1):76
Verlinden SF, Mulder AH, de Leeuw JP, van Bekkum DW (1998) T lymphocytes determine the development of xeno GVHD and of human hemopoiesis in NOD/SCID mice following human umbilical cord blood transplantation. Stem Cells 16(Suppl 1):205–217
Waksman Y, Olson JM, Carlisle SJ, Cabral GA (1999) The central cannabinoid receptor (CB1) mediates inhibition of nitric oxide production by rat microglial cells. J Pharmacol Exp Ther 288(3):1357–1366
Xu H, Cheng CL, Chen M, Manivannan A, Cabay L, Pertwee RG, Coutts A, Forrester JV (2007) Anti-inflammatory property of the cannabinoid receptor-2-selective agonist JWH-133 in a rodent model of autoimmune uveoretinitis. J Leukoc Biol 82(3):532–541
Acknowledgments
Charles Kusinsky, Meghan Michalak, and Victoria Smith at the University of Nebraska Medical Center, Omaha, NE, are thanked for their help with the FACS analyses. We thank Jillian Braun, summer undergraduate student, for her assistance in data analyses. We thank Dr. Mamoru Ito at Central Institute of Experimental Animals, Kawasaki, Japan for providing Balb/c-Rag2−/−γc−/− mice. We also thank Robin Taylor of the University of Nebraska Medical Center for administrative assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gorantla, S., Makarov, E., Roy, D. et al. Immunoregulation of a CB2 Receptor Agonist in a Murine Model of NeuroAIDS. J Neuroimmune Pharmacol 5, 456–468 (2010). https://doi.org/10.1007/s11481-010-9225-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11481-010-9225-8