Abstract
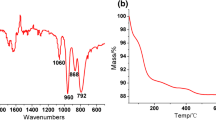
Pyridine(Py)-modified Keggin-type vanadium-substituted heteropoly acids (Py n PMo10V2O40, n=1 to 5) were prepared by a precipitation method as organic/inorganic hybrid catalysts for direct hydroxylation of benzene to phenol in a pressured batch reactor and their structures were detected by FT-IR. Among various catalysts, Py3PMo10V2O40 exhibits the highest catalytic activity (yield of phenol, 11.5%), without observing the formation of catechol, hydroquinone and benzoquinone in the reaction with 80 vol% aqueous acetic acid, molecular oxygen and ascorbic acid used as the solvent, oxidant and reducing reagent, respectively. Influences of reaction temperature, reaction time, oxygen pressure, amount of ascorbic acid and catalyst on yield of phenol were investigated to obtain the optimal reaction conditions for phenol formation. Pyridine can greatly promote the catalytic activity of the Py-free catalyst (H5PMo10V2O40), mostly because the organic π electrons in the hybrid catalyst may extend their conjugation to the inorganic framework of heteropoly acid and dramatically modify the redox properties, at the same time, pyridine adsorbed on heteropoly acids can promote the effect of “pseudo-liquid phase”, thus accounting for the enhancement of phenol yield.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Niwa S, Eswaramoorthy M, Nair J, Raj A, Itoh N, Shoji H, Namba T, Mizukami F. A one-step conversion of benzene to phenol with a palladium membrane. Science, 2002, 295: 105–107
Shiju N, Fiddy S, Sonntag O, Stockenhuber M, Sankar G. Selective oxidation of benzene to phenol over FeAlPO catalysts using nitrous oxide as oxidant. Chem Commun, 2006, (47): 4955–4957
Zhang F M, Guo M P, Ge H Q, Wang J. Hydroxylation of benzene with hydrogen peroxide over highly efficient molybdovanadophos phoric heteropoly acid catalysts. Chin J Chem Eng, 2007, 15: 895–898
Ge H Q, Leng Y, Zhou C J, Wang J. Direct hydroxylation of benzene to phenol with molecular oxygen over phase transfer catalysts: Cyclodextrins complexes with vanadium-substituted heteropoly acids. Catal Lett, 2008, 124: 324–329
Kuznetsova N I, Kuznetsova L I, Likholobov V A, Pez G P. Hydroxylation of benzene with oxygen and hydrogen over catalysts containing Group VIII metals and heteropoly compounds. Catal Today, 2005, 99: 193–198
Tani M, Sakamoto T, Mita S, Sakaguchi S, Ishii Y. Hydroxylation of benzene to phenol under air and carbon monoxide catalyzed by molybdovanadophosphoric acid. Angew Chem Int Ed, 2005, 44: 2586–2588
Bahidsky M, Hronec M. Direct hydroxylation of aromatics over copper-calcium-phosphates in the gas phase. Catal Today, 2005, 99: 187–192
Sumimoto S, Tanaka C, Yamaguchi S T, Ichihashi Y, Nishiyama S, Tsuruya S. Zinc powder as an effective reducing reagent during liquid- phase oxidation of benzene to phenol using molecular oxygen over V-substituted heteropoly acid catalysts. Ind Eng Chem Res, 2006, 45: 7444–7450
Yang Y Y, Xu L, Gao G G, Li FY, Qiu Y F, Qu X S, Liu H. Transition-metal (Mn-II and Co-II) complexes with the heteropolymolybdate fragment [(AsMo9O33)-Mo-V](7-): Crystal structures, electrochemical and magnetic properties. Eur J Inorg Chem, 2007, (17): 2500–2505
Zhang F Q, Zhang X M, Wu H S, Jiao H J. Structural and electronic properties of hetero-transition-metal keggin anions: A DFT study of alpha/beta-[XW12O40](n-) (X=Cr-VI, V-V, Ti-IV, Fe-III, Co-III, Ni-III, Co-II, and Zn-II) relative stability. J Phys Chem A, 2007, 111: 159–166
San Felices L, Vitoria P, Gutierrez-Zorrilla J M, Lezama L, Reinoso S. Hybrid inorganic-metalorganic compounds containing copper(II)-monosubstituted Keggin polyanions and polymeric copper(I) complexes. Inorg Chem, 2006, 45: 7748–7757
Tang Y, Zhang J. Direct oxidation of benzene to phenol catalyzed by vanadium substituted heteropolymolybdic acid. Trans Metal Chem, 2006, 31: 299–305
Zhang J, Tang Y, Li G Y, Hu C. Room temperature direct oxidation of benzene to phenol using hydrogen peroxide in the presence of vanadium-substituted heteropolymolybdates. Appl Catal A, 2005, 278: 251–261
Liu Y Y, Murata K, Inaba M. Liquid-phase oxidation of benzene to phenol by molecular oxygen over transition metal substituted polyoxometalate compounds. Catal Commun, 2005, 6: 679–683
Saha P K, Dutta B, Jana S, Bera R, Saha S, Okamoto K, Koner S. Immobilization of a copper-Schiff base complex in a Y-zeolite matrix: Preparation, chromogenic behavior and catalytic oxidation. Polyhedron, 2007, 26: 563–571
Bigi F, Corradini A, Quarantelli C, Sartori G. Silica-bound decatungstates as heterogeneous catalysts for H2O2 activation in selective sulfide oxidation. J Catal, 2007, 250: 222–230
Neumann R, Khenkin A M. Molecular oxygen and oxidation catalysis by phosphovanadomolybdates. Chem Commun, 2006, (24): 2529–2538
San Felices L, Vitoria P, Gutierrez-Zorrilla J M, Reinoso S, Etxebarria J, Lezama L. A novel hybrid inorganic-metalorganic compound based on a polymeric polyoxometalate and a copper complex: Synthesis, crystal structure and topological studies. Chem Eur J, 2004, 10: 5138–5146
Xu B B, Peng Z H, Wei Y G, Powell D R. Polyoxometalates covalently bonded with terpyridine ligands. Chem Commun, 2003, (20): 2562–2563
Lu M, Wei Y G, Xu B B, Cheung C F C, Peng Z H, Powell D R. Hybrid molecular dumbbells: Bridging polyoxometalate clusters with an organic pi-conjugated rod. Angew Chem Int Ed, 2002, 41: 1566–1568
Takahashi K, Okuhara T, Misono M. Phase transition-like phenomenon observed for pseudo-liquid phase of heteropoly acid catalysis. Chem Lett, 1985, (6): 841–842
Misono M, Okuhara T, Ichiki T, Arai T, Kanda Y. Pseudo-liquid behavior of heteropoly compound catalysts. Unusual pressure dependences of the rate and selectivity for ethanol dehydration. J Am Chem Soc, 1987, 109: 5535–5536
Misono M. Heterogeneous catalysis by heteropoly compounds of molybdenum and tungsten. Catal Rev Sci Eng, 1987, 29: 269–321
Wang J, Lin Z, Han S Y, Eum M, Lee C W. 12-tungstophosphoric acid supported on dealuminated USY as a catalyst for hydroisomerization of n-heptane. J Ind Eng Chem, 2003, 9: 281–286
Vimont A, Travert A, Binet C, Pichon C, Mialane P, Secheresse F, Lavalley J C. Relationship between infrared spectra and stoichiometry of pyridine-H3PW12O40 salts using a new TGA-infrared coupling. J Catal, 2006, 241: 221–224
Li W, Oshihara K, Ueda W. Catalytic performance for propane selective oxidation and surface properties of 12-molybdophosphoric acid treated with pyridine. Appl Catal A, 1999, 182: 357–363
Song I K, Kaba M S, Barteau M A. STM investigation of pyridine interaction with heteropoly acid monolayers. J Phys Chem, 1996, 100: 17528–17534
Wang J M, Yan L, Qian G, Lv G M, Li G X, Suo J S, Wang X L. Pyridine-Keggin heteropoly compounds as catalyst for hydroxylation of phenol using hydrogen peroxide as oxidant. React Kinet Catal Lett, 2007, 91: 111–118
Yamaguchi S, Sumimoto S, Ichihashi Y, Nishiyama S, Tsuruya S. Liquid-phase oxidation of benzene to phenol over V-substituted heteropolyacid catalysts. Ind Eng Chem Res, 2005, 44: 1–7
Ishida M A, Masumoto Y, Hamada R, Nishiyama S, Tsuruya S, Masai M. Liquid-phase oxygenation of benzene over supported vanadium catalysts. J Chem Soc-Perkin Trans 2, 1999, (4): 847–853
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by the National Natural Science Foundation of China (Grant Nos. 20476046 and 20776069) and the “Qinglan” Project of Jiangsu Province for Young Researchers
Rights and permissions
About this article
Cite this article
Ge, H., Leng, Y., Zhang, F. et al. Pyridine-H5PMo10V2O40 hybrid catalysts for liquid-phase hydroxylation of benzene to phenol with molecular oxygen. Sci. China Ser. B-Chem. 52, 1264–1269 (2009). https://doi.org/10.1007/s11426-009-0055-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-009-0055-6