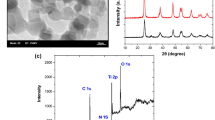
Abstract
In this study, the catalytic activity of decatungstate (W10O324−) supported on mesoporous TiO2 nanoparticle assemblies (DT-MTA) was compared with that of homogeneous [Bu4N]4W10O32 catalysts under mild conditions. Our experiments showed that both catalytic systems achieve exceptionally high activity and selectivity under UV-visible light oxidation of various para-substituted aryl alcohols, using molecular oxygen as a “green” oxidant. The chemoselective transformation of aryl alcohols into the corresponding ketones was investigated with gas chromatography (GC) and NMR spectroscopy. Product analysis and kinetic results also indicated that these photooxidation reactions proceed via both electron transfer (ET) and hydrogen atom transfer (HAT) mechanisms over the DT-MTA catalyst, with the former one as the predominant, whereas a HAT route was adopted to account for the decatungstate homogeneous catalyzed reactions.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
R. A. Sheldon and J. K. Kochi, Metal-Catalyzed Oxidations of Organic Compounds, Academic Press, New York, 1981
R. A. Sheldon and I. W. C. E. Arends, Catal. Metal Complexes, 2003, vol. 26, ch. 3, p. 123
R. A. Sheldon, I. W. C. E. Arends and A., Dijksman, Catal. Today, 2000 57 157
R., Neumann, Prog. Inorg. Chem., 1998 47 317
N. Mizuno and M., Misono, Chem. Rev., 1998 98 199
R. Neumann and M., Levin, J. Org. Chem., 1991 56 5707
R. Neumann, A. M. Khenkin and I., Vigderganz, Chem. - Eur. J., 2000 6 875
R. Ben-Daniel, P. Alsters and R., Neumann, J. Org. Chem., 2001 66 8650
M. A. Khenkin, J. W. L. Shimon and R., Neumann, Inorg. Chem., 2003 42 3331
D. Ravelli, D. Dondi, M. Fagnoni and A., Albini, Chem. Soc. Rev., 2009 38 1999
Selected reviews
M. Zhang, C. Chen, W. Ma and J., Zhao, Angew. Chem., Int. Ed., 2008 47 9730
G. Palmisano, V. Augugliaro, M. Pagliaro and L., Palmisano, Chem. Commun., 2007 3425
C. C. Chen, W. H. Ma and J. C., Zhao, Chem. Soc. Rev., 2010 39 4206
A. L. Linsebigler, G. Q. Lu and J. T., Yates, Chem. Rev., 1995 95 735
M. R. Hoffmann, S. T. Martin, W. Choi and D. W., Bahnemann, Chem. Rev., 1995 95 69
V. Augugliaro and L., Palmisano, ChemSusChem, 2010 3 1135
A. Kubacka, M. Fernández-García and G. Colón Chem. Rev., 2012 112 1555
X. Chen and S. S., Mao, Chem. Rev., 2007 107 2891
J. C. Yu, G. Li, X. Wang, X. Hu, C. W. Leung and Z., Zhang, Chem. Commun., 2006 2717
R. Asahi, T. Morikawa, T. Ohwaki, K. Aoki and Y., Taga, Science, 2001 293 269
O. S. Mohamed, A. M. Gaber and A. A. Abdel-Wahab J. Photochem. Photobiol., A, 2002 148 205
T. Mitkina, C. Stanglmair, W. Setzer, M. Gruber, H. Kisch and B., Konig, Org. Biomol. Chem., 2012 10 3556
A. Maldotti and A., Molinari, Top. Curr. Chem., 2011 303 185
M. Fagnoni, D. Dondi, D. Ravelli and A., Albini, Chem. Rev., 2007 107 2725
Special issues are devoted to polyoxometalates
C. L. Hill, ed., Chem. Rev., 1998, 98, 1
C. L. Hill, ed., J. Mol. Catal. A: Chem., 2007, 262, 1
Selected reviews in polyoxometalates catalysis
C. L. Hill, M. Christina and C. M. Prosser-McCartha Coord. Chem. Rev., 1995 143 407
C. L., Hill, Synlett, 1995 127
A. Hiskia, A. Mylonas and E., Papaconstantinou, Chem. Soc. Rev., 2001 30 62
A. Maldotti, A. Molinari and R., Amadelli, Chem. Rev., 2002 102 3811
M. Vazylyev, D. Sloboda-Rozner, A. Haimov, G. Maayan and R., Neumann, Top. Catal., 2005 34 93
G. S. Armatas, A. P. Katsoulidis, D. E. Petrakis and P. J., Pomonis, J. Mater. Chem., 2010 20 8631
M. D. Tzirakis, I. N. Lykakis and M., Orfanopoulos, Chem. Soc. Rev., 2009 38 2609
C., Tanielian, Coord. Chem. Rev., 1998 178-180 1165
A. Molinari, R. Amadelli, L. Andreotti and A., Maldotti, J. Chem. Soc., Dalton Trans., 1999 1203
A. Maldotti, A. Molinari, G. Varani, M. Lenarda, L. Storaro, F. Bigi, R. Maggi, A. Mazzacani and G., Sartori, J. Catal., 2002 209 210
A. Molinari, R. Amadelli, A. Mazzacani, G. Sartori and A., Maldotti, Langmuir, 2002 18 5400
A. Maldotti, R. Amadelli, I. Vitali, L. Borgatti and A., Molinari, J. Mol. Catal. A: Chem., 2003 204 703
G. S. Armatas, G. Bilis and M., Louloudi, J. Mater. Chem., 2011 21 2997
Y. Guo, C. Hu, X. Wang, Y. Wang and E., Wang, Chem. Mater., 2001 13 4058
Y. Guo, C. Hu, S. Jiang, C. Guo, Y. Yang and E., Wang, Appl. Catal., B, 2002 36 9
S. Farhadi and Z., Momeni, J. Mol. Catal. A: Chem., 2007 277 47
A. Molinari, A. Bratovcic, G. Magnacca and A., Maldotti, Dalton Trans., 2010 39 7826
F. Bigi, A. Corradini, C. Quarantelli and G., Sartori, J. Catal., 2007 250 222
A. Maldotti, A. Molinari and F., Bigi, J. Catal., 2008 253 312
L. Ni, J. Ni, Y. Lv, P. Yang and Y., Cao, Chem. Commun., 2009 2171
M. D. Tzirakis, I. N. Lykakis, G. Panagiotou, K. Bourikas, A. Lycourghiotis, C. Kordoulis and M., Orfanopoulos, J. Catal., 2007 272 178
I. Tamiolakis, I. N. Lykakis, A. P. Katsoulidis and G. S., Armatas, Chem. Commun., 2012 48 6687
I. Tamiolakis, I. N. Lykakis and G. S., Armatas, Catal. Today, 2014 DOI:10.1016/j.cattod.2014.03.047
A. Chemseddine, C. Sanchez, J. Livage, J. P. Launay and M., Fournier, Inorg. Chem., 1984 23 2609
I. N. Lykakis, S. Lestakis and M., Orfanopoulos, Tetrahedron Lett., 2003 44 6247
I. N. Lykakis, C. Tanielian, R. Seghrouchni and M., Orfanopoulos, J. Mol. Catal. A: Chem., 2007 262 176
C. Tanielian, K. Duffy and A., Jones, J. Phys. Chem. B, 1997 101 4276
D. C. Duncan and M. A., Fox, J. Phys. Chem. A, 1998 102 4559
D. C. Duncan, T. L. Netzel and C. L., Hill, Inorg. Chem., 1995 34 4640
Author information
Authors and Affiliations
Corresponding author
Additional information
Electronic supplementary information (ESI) available: Catalysts characterization, reaction time profiles, NMR data and copies of spectra. See DOI: 10.1039/c4pp00268g
Rights and permissions
About this article
Cite this article
Symeonidis, T.S., Tamiolakis, I., Armatas, G.S. et al. Green photocatalytic organic transformations by polyoxometalates vs. mesoporous TiO2 nanoparticles: selective aerobic oxidation of alcohols. Photochem Photobiol Sci 14, 563–568 (2015). https://doi.org/10.1039/c4pp00268g
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1039/c4pp00268g