Abstract
Studies identifying the relative importance of multiple ecological processes in macroinvertebrate communities in urban lakes at a basin scale are rare. In this study, 14 urban lakes in the Taihu Lake Basin were selected to explore the relative importance of environmental filtering and spatial processes in the assembly of macroinvertebrate communities. Our findings revealed significant spatiotemporal variations in macroinvertebrate communities, both between lakes and across seasons. We found that environmental filtering exerted a greater influence on taxonomic total beta diversity and its individual components (species turnover and nestedness) compared to spatial processes. Key environmental variables such as water depth, water temperature, total dissolved solids, chlorophyll a, and lake surface area were found to be crucial in shaping macroinvertebrate communities within these urban lakes. The observed high spatial heterogeneity in environmental conditions, along with intermediate basin areas, good connectivity and short distances between lakes, and the high dispersal ability of dominant taxa, likely contributed to the dominance of environmental filtering in macroinvertebrate community assembly. Our study contributes to a better understanding of the underlying mechanisms governing macroinvertebrate community assembly in urban lakes, thereby providing valuable insights for studies on community ecology and water environmental protection in urban lakes.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Understanding patterns of community assembly is the focus of community ecology (Chase 2003). Both deterministic and stochastic theories have been employed to explain community assembly patterns (Chase and Myers 2011). Deterministic theory (e.g., niche-based theory) assumes that local processes, including environmental filtering and biotic interactions, dominant patterns of community assembly. In contrast, stochastic theory (e.g., neutral theory) emphasizes that chance colonization, random extinction, and ecological drift contribute to community assembly (Chase and Myers 2011). Furthermore, metacommunity ecology theory, which integrates local (e.g., environmental filtering and biotic interactions) and regional (e.g., dispersal and ecological drift) processes, was developed to understand patterns in community assembly (Chase et al. 2020; Heino 2011; Leibold et al. 2004). In freshwater ecosystems, the metacommunity ecology theory has been applied to explore the relative importance of environmental filtering and spatial processes on the patterns of community assembly (Heino 2011; Heino and Tolonen 2017; Jamoneau et al. 2018). Macroinvertebrates play an important role in sediment mixing, nutrient cycling, and energy flow within aquatic ecosystems by accelerating detrital decomposition (van de Bund et al. 1994; Wallace and Webster 1996) and transferring nutrient to overlying open waters (Covich et al. 1999). In freshwater ecosystems, studies on macroinvertebrate communities and the mechanisms underlying macroinvertebrate community assembly are important for understanding ecosystem structure and function, as well as for water environment conservation and protection (Makumbe et al. 2022).
The metacommunity ecology theory has commonly been utilized to explore the patterns of macroinvertebrate community assembly. The relative importance of environmental filtering and spatial processes in macroinvertebrate communities has been found to vary across different types of water bodies, including streams (Heino and Mykrä 2008), rivers (Qin et al. 2022), urban rivers (Liu et al. 2021), lakes (Cai et al. 2017; Li et al. 2022), and ponds (Hill et al. 2017, 2019). Previous studies have shown that environmental filtering typically dominates macroinvertebrate communities. Physical water indices (e.g., water depth and temperature) have been identified as primary variables associated with environmental filtering (Li et al. 2021; Stewart and Schriever 2023), while water body area also plays a significant role (Hill et al. 2019; Stewart and Schriever 2023). In addition to the above local-scale environmental variables, catchment-scale environmental variables (e.g., variables related to land use) have been found to be dominant variables in environmental filtering (Liu et al. 2016). Variables related to habitat heterogeneity (e.g., macrophyte cover) are also considered important variables to environmental filtering (Hill et al. 2017, 2019). Except for environmental filtering, the contribution of spatial processes to macroinvertebrate communities can be equal to or even greater than that of environmental filtering (Liu et al. 2021; Cai et al. 2017). In terms of biodiversity, the relative importance of environmental variables (e.g., water temperature, dissolved oxygen, total phosphorus, total nitrogen, nitrate nitrogen, aquatic macrophytes coverage, and chlorophyll a) and spatial factors on the three dimensions of α diversity (i.e., taxonomic, functional, and phylogenetic diversities) has been found to vary (Li et al. 2019a, 2020; Jiang et al. 2022). The relative importance of different ecological processes also varies across different dimensions of β diversity and their different components (turnover and nestedness) (Hill et al. 2017; Hill et al. 2019; Li et al. 2021; Stewart and Schriever 2023).
Most of the aforementioned studies have focused on understanding patterns of macroinvertebrate community assembly within individual lakes (Cai et al. 2017; Li et al. 2022). However, conclusions drawn from single-lake studies are often considered individual examples, and the mechanisms underlying macroinvertebrate community assembly within a basin under the same climate remain unclear, especially in urban lakes subject to extensive human activity. For example, studies on Taihu Lake have indicated that the relative importance of spatial processes on macroinvertebrate community assembly outweighs that of environmental variables (Cai et al. 2017). However, at the basin scale, environmental variables were found to have a greater impact on the three-dimensional α diversity of macroinvertebrate communities compared to spatial factors (Jiang et al. 2022). In addition, it is still uncertain whether the metacommunity ecology theory can effectively explain macroinvertebrate community assembly in urban lakes, which have experienced serious eutrophication in recent decades (Tian et al. 2020; Chen et al. 2023). In the present study, we selected 14 urban lakes within the Taihu Lake Basin to explore the patterns of macroinvertebrate community assembly. The aims of this study were (1) to explore the spatiotemporal differences in macroinvertebrate communities within urban lakes and (2) to quantify the relative importance of environmental filtering and spatial processes on macroinvertebrate communities. Our finding will improve our understanding of the mechanisms driving macroinvertebrate community assembly, thereby providing valuable insights for studies on community ecology and water environmental protection in urban lakes.
Materials and methods
Study area and sampling sites
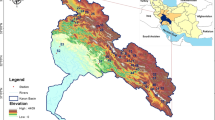
The Taihu Lake Basin (119°3′1″ –121°54′26″E and 30°7′19″–32°14′56″N) is located in the core area of the Yangtze River Delta region, which is densely covered by lakes and rivers. The total area of the basin is approximately 36,895 km2 (Huang et al. 2015), and it is situated in a typical subtropical monsoon climate zone with a high degree of urbanization (Xu et al. 2016). In this study, we focused on 14 urban lakes within the basin, all of which have experienced severe eutrophication. Three to seven sampling sites were selected in each lake according to their morphology and surface area, with Google Earth used to determine the longitude and latitude of the sampling sites. Detailed information on the sampled lakes and sites is presented in Fig. 1 and Table S1. A total of 246 samples were collected quarterly from December 2018 to September 2019 (refer to Table S2 for detailed information).
Macroinvertebrate sampling and handling
We collected surface sediment using a 0.0625 m2 modified Peterson grab at each sampling site. All collected materials were then poured through a 0.25 mm aperture-sized mesh for sieving in the field. The remaining materials were temporarily stored in transparent plastic self-sealing bags and then poured into white enamel pans, and macroinvertebrates were picked out using ophthalmic tweezers and placed in 60 mL plastic bottles. A formaldehyde solution was added to the bottles to preserve the macroinvertebrate samples. Macroinvertebrates were identified to the lowest possible taxonomic level (species or genus, occasionally family) using a stereomicroscope (Nikon Corporation, SMZ1500, Tokyo Metropolis, Japan) and a general light microscope (Olympus Corporation, CX21, Tokyo Metropolis, Japan). The references used for identifying the specimens are listed in Supplementary Material, Sect. 1. For each sample, we counted each taxon, absorbed the body surface fixative with absorbent paper, and placed an individual of each taxon on an electronic balance (precision 0.0001 g) (Sartorius Corporation, BSA124S, Gottingen, Germany) for weighing. Finally, we transformed the abundance (ind/m2) and biomass (g/m2) of each taxon at each sampling site based on the sampling area.
Environmental variable collection and measurement
Water depth (WD) and Secchi depth (SD) were measured using a Secchi disc. Water temperature (WT), conductivity (Cond), total dissolved solids (TDS), dissolved oxygen (DO), pH, oxidation–reduction potential (ORP), and turbidity (Turb) were measured at each sampling site using a YSI exo2 multiparameter water quality analyzer. We collected a 5 L water sample from 0.5 m below the water surface and 0.5 m above the surface sediment with a 5 L transparent plexiglass water collector at each sampling site and mixed the water samples. The mixed water samples were frozen and transported to the laboratory. In the laboratory, the chemical oxygen demand (CODcr), total nitrogen (TN), total phosphorus (TP), ammonia nitrogen (NH + 4-N), nitrate nitrogen (NO- 3-N), orthophosphate (PO3-4-P), and chlorophyll a (Chla) of the mixed water samples were measured following previously described methods (Huang et al. 1999). In addition, we collected data on the surface areas of these urban lakes.
Spatial factors calculation
First, we used the longitude and latitude coordinates of the sampling sites to calculate the Cartesian coordinates of corresponding sampling sites with the function “geoXY” from the SoDA package (Chambers 2020; Vincenty 1975). Subsequently, we constructed distance-based Moran’s eigenvector maps (DbMEMs) based on Cartesian coordinates to obtain spatial factors at the basin scale. DbMEMs can be used to study spatial relationships in biological communities (Dray et al. 2012) and to identify spatial factors exhibiting significant positive spatial correlations (at the 5% significance level) (Lai 2020). We obtained eight spatial factors (MEMs) with positive spatial correlations in the urban lakes of the Taihu Lake Basin and named them as integers from MEM1 to MEM8. The DbMEM analysis was conducted with the function “dbmem” in the adespatial package (Legendre and Legendre 2012; Dray et al. 2022) in R, version 4.0.5 (R Core Team 2021).
Data analysis
The Kruskal–Wallis nonparametric test, the least significant difference (LSD) multiple comparison method of one-way analysis of variance (one-way ANOVA), permutational multivariate analysis of variance (PERMANOVA), and permutational analysis of multivariate dispersions (PERMDISP) were used to explore spatiotemporal differences in environmental variables and macroinvertebrate communities. One-way ANOVA and Kruskal–Wallis nonparametric tests were conducted using SPSS 26.0 software (International Business Machines Corporation, 26.0, Amonk, The United States of America). PERMANOVA and PERMDISP were performed using the vegan package (Oksanen et al. 2022) in R, version 4.0.5 (R Core Team 2021). Furthermore, to explore the relative importance of environmental filtering and spatial processes on macroinvertebrate communities, we calculated and partitioned the β diversity of macroinvertebrate communities. Subsequently, we conducted principal coordinate analysis, redundancy analysis, and variation partitioning (Legendre and Legendre 2012) using the ape and vegan packages (Oksanen et al. 2022; Paradis and Schliep 2019) in R, version 4.0.5 (R Core Team 2021). Detailed data analysis procedures are provided in Supplementary Material, Sect. 2.
Results
Environmental variables exhibited significant differences among the urban lakes and seasons
The results of the Kruskal–Wallis nonparametric test indicated that all environmental variables showed significant differences (P < 0.01) among the lakes, including WD, SD, Cond, TDS, DO, pH, ORP, Turb, COD, TN, TP, NH + 4-N, NO- 3-N, PO3-4-P, and Chla, except WT (P > 0.05) (refer to Table S3 for detailed information). The Kruskal–Wallis nonparametric test and LSD multiple comparison indicated significant differences among the seasons for most environmental variables (P < 0.01), including SD, Cond, TDS, DO, pH, ORP, Turb, COD, TN, TP, NH + 4-N, NO- 3-N, and Chla. In addition, the PO3-4-P levels showed a significant difference (P < 0.05). Only WD showed no significant differences (P > 0.05) (refer to Table S3 for detailed information). A detailed description of the environmental variables of the urban lakes is presented in Table S4.
The results of PERMANOVA (based on Euclidean distance, permutations = 999) indicated that environmental variables showed significant differences among both lakes and seasons (refer to Table 1 for detailed information). Furthermore, PERMDISP (based on Euclidean distance, permutations = 999) indicated that, for environmental variables, the dispersion of samples showed significant differences among lakes, whereas centroids showed significant differences among seasons (refer to Table 2 for detailed information). Results of PERMANOVA associated with PERMDISP indicated that the environmental variables were significantly different between lakes and seasons. The above statistical analyses indicated that environmental variables, such as SD, Cond, TDS, DO, pH, ORP, Turb, COD, TN, TP, NH + 4-N, NO- 3-N, and PO3-4-P, had significant differences among both lakes and seasons in the 14 urban lakes.
Macroinvertebrate communities exhibited significant differences among the urban lakes and seasons
A total of 64 taxa were identified from 246 samples collected during the four seasons across the 14 urban lakes. Aquatic insects were the most abundant group (22 taxa), followed by Gastropoda and Bivalvia (11 taxa each). The average total density of the macroinvertebrate community ranged from 462.10 to 5762.67 ind/m2 among lakes, with the highest average density observed in aquatic insects (ranging from 213.52 to 1860.60 ind/m2 among lakes) (refer to Fig. 2a and Table S5 for detailed information). Likewise, the average total biomass of the macroinvertebrate communities ranged from 1.69 to 893.47 g/m2. Oligochaeta and aquatic insects had the highest biomass (0.09 to 0.94 g/m2 and 0.37 to 11.79 g/m2, respectively), except for Mollusca (refer to Fig. 2b and Table S5 for detailed information). The results of the Kruskal–Wallis nonparametric test indicated that the total density and biomass of the macroinvertebrate community, the density and biomass of each group, and species richness (S) were significantly different (P < 0.01) among lakes, as were the Shannon–Wiener index (H'), Simpson index (D), and Pielou evenness index (J) (P < 0.05) (refer to Table S6 for detailed information). However, the Kruskal–Wallis nonparametric test and LSD multiple comparisons indicated that only the density and biomass of aquatic insects showed significant differences (P < 0.01) among seasons (refer to Table S6 for detailed information).
The results of the calculation and partition of β diversity (based on the Sørensen dissimilarity index) indicated that βsør ranged from 0.68 to 0.91, βsim ranged from 0.53 to 0.86, and βsne ranged from 0.04 to 0.18 (refer to Table 3 for detailed information). Compared with studies on streams (Jamoneau et al. 2018; Li et al. 2021), ponds (Hill et al. 2017), and lakes (Cortés-Guzmán and Alcocer 2022), the total β diversity of this study was high both at the basin and lake scales, which indicated that the macroinvertebrate community exhibited marked spatiotemporal differences at both the basin and lake scales. The total β diversity (βsør) was dominated by the species turnover component (βsim) and hardly explained by the nestedness component (βsne) at both the basin and lake scales, suggesting significant species turnover occurred, whereas nestedness varied little among the urban lakes (refer to Table 3 for detailed information).
In addition, the results of PERMANOVA (based on Bray–Curtis distance, permutations = 999) indicated that the macroinvertebrate communities were significantly different among lakes and seasons, as was the lake × season interaction (refer to Table 1 for detailed information). Furthermore, PERMDISP (based on Bray–Curtis distance, permutations = 999) results showed significant differences for the dispersion of samples among lakes, whereas the centroids of each season showed significant differences among seasons (refer to Table 2 for detailed information). Overall, the above results indicated that the macroinvertebrate community differed significantly among lakes and seasons.
Relative importance of environmental filtering and spatial processes on macroinvertebrate community
According to the redundancy analysis results, five environmental variables (WD, WT, TDS, Chla, and lake surface area) and three spatial factors (MEM1, MEM2, and MEM3) were significantly associated with total β diversity (βsør) through forward selection (based on P < 0.05, permutations = 999, see Lai 2020). The results of variation partitioning indicated that environmental filtering and spatial processes jointly explained 23.3% of the total β diversity (βsør), with environmental filtering and spatial processes individually explaining 11.3% and 4.1%, respectively (refer to Fig. 3a for detailed information). The relative importance of environmental filtering on the total β diversity (βsør) of macroinvertebrate communities exceeded that of the spatial processes.
Variation partitioning of the macroinvertebrate community attributed to environmental filtering (Env) and spatial processes (Spa) across the 14 urban lakes in the Taihu Lake Basin: a total beta diversity (βsør), b species turnover component (βsim), and c nestedness component (βsne). Values represent the adjusted R2 values. *** represents P < 0.001 based on 999 permutation test, ** represents P < 0.005 based on 999 permutation test
In the species turnover component (βsim), five environmental variables (WD, WT, TDS, Chla, and lake surface area) and four spatial factors (MEM1, MEM2, MEM3, and MEM6) were significantly associated through forward selection (based on P < 0.05, permutations = 999, see Lai 2020). The results of variation partitioning indicated that the total explained variation by environmental filtering and spatial processes in βsim was 24.1%, of which environmental filtering and spatial processes explained 11.0% and 6.2%, respectively (refer to Fig. 3b for detailed information). Once again, environmental filtering exhibited a greater relative importance in driving βsim.
In the nestedness component (βsne), six environmental variables (WD, WT, TDS, NO- 3-N, Chla, and lake surface area) and four spatial factors (MEM1, MEM2, MEM3, and MEM6) were significantly associated through forward selection (based on P < 0.05, permutations = 999, see Lai 2020). The results of variation partitioning indicated that the total explained variation by environmental filtering and spatial processes for the nestedness component was 12.2%, of which environmental filtering and spatial processes explained 6.7% and 2.5%, respectively (refer to Fig. 3c for detailed information). Once again, environmental filtering exerted a stronger influence on βsne.
In summary, for total β diversity (βsør) and its different components (βsim and βsne) in macroinvertebrate communities, the contributions of environmental filtering exceeded those of spatial processes. The primary environmental variables influencing macroinvertebrate communities in these urban lakes were WD, WT, TDS, Chla, and lake surface area, as determined by forward selection (based on P < 0.05, permutations = 999, see Lai 2020).
Discussion
Roles of environmental filtering and spatial processes on the macroinvertebrate community in urban lakes
In our study, significant spatiotemporal differences were observed in macroinvertebrate communities across the urban lakes. We found that environmental filtering played a dominant role over spatial processes in structuring the taxonomic total β diversity (βsør), as well as the turnover (βsim) and nestedness (βsne) components of macroinvertebrate communities. This finding is consistent with the results observed in streams (Heino and Mykrä 2008), rivers (Li et al. 2019a; Liu et al. 2016), and ponds (Hill et al. 2017, 2019). Specifically, several key environmental variables — WD, WT, TDS, Chla, and lake surface area — significantly influenced macroinvertebrate communities in these urban lakes. Many studies have highlighted the importance of WD, WT, and Chla in affecting macroinvertebrate communities (Burlakova et al. 2022; Chen et al. 2018; Pires et al. 2021). Chla, for example, is associated with trophic status and strongly regulates macroinvertebrate communities (Burlakova et al. 2022; Dou et al. 2021; Li et al. 2019b). Our analyses, including Kruskal–Wallis nonparametric tests, LSD multiple comparison, PERMANOVA, and PERMDISP, revealed significant spatiotemporal differences in almost all detected environmental variables (refer to Table S3 and Table S4 for detailed information). This suggests that environmental variables provided high spatial and temporal heterogeneity in habitat, which likely influenced macroinvertebrate communities. The heterogeneity of environmental variables is reported to be a key driver of variation in β diversity and its turnover and nestedness components (Cottenie et al. 2003; Cottenie and De Meester 2004; Viana et al. 2016). The wide range of environmental variables observed in our study provides ample opportunities for environmental filtering to operate, thereby shaping macroinvertebrate community structure (Li et al. 2019a; Liu et al. 2016; Qin et al. 2022). In addition, we observed that the lake surface area significantly influenced macroinvertebrate communities in these urban lakes, which aligns with the results of previous studies (Hill et al. 2017, 2019). Large habitat areas can increase habitat heterogeneity, providing diverse niches for macroinvertebrate (Heino 2000). Environmental variables associated with habitat space and complexity have been shown to contribute significantly to macroinvertebrate community assembly (Hill et al. 2019). The highly heterogeneous lake surface area in our study (refer to Fig. 1 and Table S1 for detailed information) likely promoted environmental filtering as the dominant process shaping macroinvertebrate communities.
In contrast, when the contribution of spatial processes reached (Liu et al. 2021) or outweighed that of environmental variables on the macroinvertebrate community (Cai et al. 2017; Li et al. 2021; Qin et al. 2022), a high abundance of pollution-tolerant taxa (e.g., Oligochaeta and Chironomidae) and habitat homogenization were considered possible reasons. The high abundance of pollution-tolerant taxa and habitat homogenization could weaken the responses of communities to environmental variables (Heino and Tolonen 2017; Liu et al. 2021).
In our study, the spatial scale of the Taihu Lake Basin played a significant role in driving environmental filtering as the dominant process. Some studies have shown that the relative importance of variables to metacommunities may be related to the spatial extent of the study area (Mykrä et al. 2007; Bennett et al. 2010; Heino et al. 2010). The contribution of environmental filtering to the community could be expected to peak at intermediate spatial extents (e.g., from 102 to 105 km2), where dispersal rates were neither too high nor too low, and most individuals could reach optimal habitat patches (Heino 2011; Leibold et al. 2004). Given that the Taihu Lake Basin spans approximately 36,895 km2, it creates conditions conducive to environmental filtering shaping macroinvertebrate communities. In addition, isolated habitats can present dispersal barriers for many species, potentially enhancing spatial effects (Li et al. 2021). However, in our study, some of these urban lakes were connected by rivers (Fig. 1), and the predominantly plain landscape suggests relatively minimal dispersal barriers for species. Further, at the scale of the basin, the macroinvertebrate community exhibited the highest average density of aquatic insects. Specifically, Chironomidae emerged as the dominant group within this category. Apart from Chironomidae, only 4 other taxa of aquatic insects were identified, and they were rarely observed. Thus, Chironomidae emerged as the dominant group in these urban lakes (refer to Table 3 for detailed information). Notably, Chironomidae are recognized for their high dispersal capacity (they can fly as adults), implying they can effectively respond to variations in heterogeneous environmental conditions (Heino et al. 2014; Leibold et al. 2004; Soininen 2014). The geomorphological characteristics of the Taihu Lake Basin indicate that the dispersal limit for macroinvertebrates, especially chironomids, potentially attenuates spatial processes within the macroinvertebrate community. In summary, environmental filtering plays an important role in macroinvertebrate communities in these urban lakes.
Species turnover dominated β diversity in urban lakes
Our study revealed that the turnover component dominated the β diversity of macroinvertebrate communities at both lake and basin scales, which was consistent with previous studies on ponds (Hill et al. 2017), rivers (Qin et al. 2022), and lakes (Cortés-Guzmán and Alcocer 2022). The high proportion of the species turnover component in total β diversity supports the hypothesis that niche-related processes (e.g., environmental filtering) govern community dynamics (Nunes et al. 2016). Associations between environmental variables, species dispersal abilities, and the proportion of different β diversity components have been documented in previous studies (Cottenie 2005; Heino 2013; Langer et al. 2016), suggesting that high environmental heterogeneity may promote species turnover (Cortés-Guzmán and Alcocer 2022). In our study, the substantial heterogeneity of environmental variables, as indicated by the results from the Kruskal–Wallis nonparametric tests, LSD multiple comparison, PERMANOVA, and PERMDISP (refer to Table 1 and Table S3 for detailed information), likely facilitated species turnover, thus driving the predominance of this process. Furthermore, species with high dispersal abilities, such as Chironomids, which dominated the macroinvertebrate community in these urban lakes (Table 3), could further promote species turnover (Baselga 2012; Andrew et al. 2012; Dobrovolski et al. 2012; Keil et al. 2012). In addition, the short distances between the urban lakes, coupled with high environmental heterogeneity, may promote high species turnover (Cortés-Guzmán and Alcocer 2022). In our study, the distances between several of these urban lakes are relatively close (see Fig. 1), which may also contribute to species turnover.
Conclusions and study prospect
In our study, we explored the relative importance of environmental filtering and spatial processes on the taxonomic total β diversity and its different components (species turnover and nestedness) within 14 urban lakes in the Taihu Lake Basin. By conducting our research within a single catchment, we were able to mitigate the effects of climatic influences. We found that macroinvertebrate communities in these urban lakes exhibited high spatiotemporal heterogeneity. Environmental filtering emerged as the dominant force shaping the β diversity of macroinvertebrate communities, with spatial processes playing a relatively minor role. Species turnover constituted the primary driver of total β diversity, while nestedness made a lesser contribution. The high spatial heterogeneity of environmental variables, intermediate basin area, good connectivity, short distances between lakes, and the high dispersal ability of dominant taxa (e.g., Chironomidae) likely account for the dominance of environmental filtering in shaping macroinvertebrate communities. Among the environmental variables, WD, WT, TDS, Chla, and lake surface area were identified as the main environmental variables maintaining macroinvertebrate communities in these urban lakes, with relevance to trophic status and the physical status of water bodies. Furthermore, our study aids in understanding the mechanisms of macroinvertebrate community assembly in urban lakes within a single catchment, providing valuable insights for studies on community ecology and water environmental protection in urban environments.
Data availability
All data included in this study are available upon request by contact with the corresponding author.
References
Andrew ME, Wulder MA, Coops NC, Baillargeon G (2012) Beta-diversity gradients of butterflies along productivity axes. Glob Ecol Biogeogr 21(3):352–364. https://doi.org/10.1111/j.1466-8238.2011.00676.x
Baselga A (2012) The relationship between species replacement, dissimilarity derived from nestedness, and nestedness. Glob Ecol Biogeogr 21(12):1223–1232. https://doi.org/10.1111/j.1466-8238.2011.00756.x
Bennett JR, Cumming BF, Ginn BK, Smol JP (2010) Broad-scale environmental response and niche conservatism in lacustrine diatom communities. Glob Ecol Biogeogr 19(5):724–732. https://doi.org/10.1111/j.1466-8238.2010.00548.x
Burlakova LE, Karatayev AY, Hrycik AR, Daniel SE, Mehler K, Rudstam LG, Watkins JM, Dermott R, Scharold J, Elgin AK, Nalepa TF (2022) Six decades of Lake Ontario ecological history according to benthos. J Gt Lakes Res 48(2):274–288. https://doi.org/10.1016/j.jglr.2021.03.006
Cai YJ, Xu H, Vilmi A, Tolonen KT, Tang XM, Qin BQ, Gong ZJ, Heino J (2017) Relative roles of spatial processes, natural factors and anthropogenic stressors in structuring a lake macroinvertebrate metacommunity. Sci Total Environ 601:1702–1711. https://doi.org/10.1016/j.scitotenv.2017.05.264
Chambers JM (2020) SoDA: functions and examples for “Software for Data Analysis”. R package version 1.0–6.1. https://CRAN.R-project.org/package=SoDA
Chase JM (2003) Community assembly: when should history matter? Oecologia 136(4):489–498. https://doi.org/10.1007/s00442-003-1311-7
Chase JM, Myers JA (2011) Disentangling the importance of ecological niches from stochastic processes across scales. Philos Trans R Soc B-Biol Sci 366(1576):2351–2363. https://doi.org/10.1098/rstb.2011.0063
Chase JM, Jeliazkov A, Ladouceur E, Viana DS (2020) Biodiversity conservation through the lens of metacommunity ecology. Ann NY Acad Sci 1469(1):86–104. https://doi.org/10.1111/nyas.14378
Chen JQ, Hu DF, Zhang CL, Ding ZF (2018) Temporal and spatial changes of macrobenthos community in the regions frequently occurring black water aggregation in Lake Taihu. Sci Rep 8:5712. https://doi.org/10.1038/s41598-018-24058-y
Chen J, Liu JR, Han SP, Su HJ, Xia WL, Wang HJ, Liu Y, Zhang L, Ke ZX, Zhang X, Tang HJ, Shen H, Tao M, Shi PL, Zhang WZ, Wang H, Zhang J, Chen YR, Rao QY, Rao X, Xie LQ, Chen FZ, Deng XW, Fan XY, He J, Chen L, Xu J, Xie P (2023) Nontraditional biomanipulation: a powerful ecotechnology to combat cyanobacterial blooms in eutrophic freshwaters. Innov Life 1(3):10038. https://doi.org/10.59717/j.xinn-life.2023.10038
Cortés-Guzmán D, Alcocer J (2022) Turnover drives high benthic macroinvertebrates’ beta diversity in a tropical Karstic Lake district. Diversity-Basel 14(4):259. https://doi.org/10.3390/d14040259
Cottenie K (2005) Integrating environmental and spatial processes in ecological community dynamics. Ecol Lett 8(11):1175–1182. https://doi.org/10.1111/j.1461-0248.2005.00820.x
Cottenie K, De Meester L (2004) Metacommunity structure: synergy of biotic interactions as selective agents and dispersal as fuel. Ecology 85(1):114–119. https://doi.org/10.1890/03-3004
Cottenie K, Michels E, Nuytten N, De Meester L (2003) Zooplankton metacommunity structure: regional vs local processes in highly interconnected ponds. Ecology 84(4):991–1000. https://doi.org/10.1890/0012-9658(2003)084[0991:ZMSRVL]2.0.CO;2
Covich AP, Palmer MA, Crowl TA (1999) The role of benthic invertebrate species in freshwater ecosystems-zoobenthic species influence energy flows and nutrient cycling. Bioscience 49(2):119–127. https://doi.org/10.2307/1313537
Dobrovolski R, Melo AS, Cassemiro FAS, Diniz-Filho JAF (2012) Climatic history and dispersal ability explain the relative importance of turnover and nestedness components of beta diversity. Glob Ecol Biogeogr 21(2):191–197. https://doi.org/10.1111/j.1466-8238.2011.00671.x
Dou QM, Du X, Cong YF, Wang L, Zhao C, Song D, Liu H, Huo TB (2021) Influence of environmental variables on macroinvertebrate community structure in Lianhuan Lake. Ecol Evol 12(2):e8553. https://doi.org/10.1002/ece3.8553
Dray S, Pélissier R, Couteron P, Fortin MJ, Legendre P, Peres-Neto PR, Bellier E, Bivand R, Blanchet FG, De Cáceres M, Dufour AB, Heegaard E, Jombart T, Munoz F, Oksanen J, Thioulouse J, Wagner HH (2012) Community ecology in the age of multivariate multiscale spatial analysis. Ecol Monogr 82(3):257–275. https://doi.org/10.1890/11-1183.1
Dray S, Bauman D, Blanchet G, Borcard D, Clappe S, Guenard G, Jombart T, Larocque G, Legendre P, Madi N, Wagner HH (2022) adespatial: multivariate multiscale spatial analysis. R package version 0.3–16. https://CRAN.R-project.org/package=adespatial
Heino J (2000) Lentic macroinvertebrate assemblage structure along gradients in spatial heterogeneity, habitat size and water chemistry. Hydrobiologia 418:229–242. https://doi.org/10.1023/A:1003969217686
Heino J (2011) A macroecological perspective of diversity patterns in the freshwater realm. Freshw Biol 56(9):1703–1722. https://doi.org/10.1111/j.1365-2427.2011.02610.x
Heino J (2013) Environmental heterogeneity, dispersal mode, and co-occurrence in stream macroinvertebrates. Ecol Evol 3(2):344–355. https://doi.org/10.1002/ece3.470
Heino J, Mykrä H (2008) Control of stream insect assemblages: roles of spatial configuration and local environmental factors. Ecol Entomol 33(5):614–622. https://doi.org/10.1111/j.1365-2311.2008.01012.x
Heino J, Tolonen KT (2017) Ecological drivers of multiple facets of beta diversity in a lentic macroinvertebrate metacommunity. Limnol Oceanogr 62(6):2431–2444. https://doi.org/10.1002/lno.10577
Heino J, Bini LM, Karjalainen SM, Mykrä H, Soininen J, Vieira LCG, Diniz JAF (2010) Geographical patterns of micro-organismal community structure: are diatoms ubiquitously distributed across boreal streams? Oikos 119(1):129–137. https://doi.org/10.1111/j.1600-0706.2009.17778.x
Heino J, Melo AS, Bini LM (2014) Reconceptualising the beta diversity-environmental heterogeneity relationship in running water systems. Freshw Biol 60(2):223–235. https://doi.org/10.1111/fwb.12502
Hill MJ, Heino J, Thornhill I, Ryves DB, Wood PJ (2017) Effects of dispersal mode on the environmental and spatial correlates of nestedness and species turnover in pond communities. Oikos 126(11):1575–1585. https://doi.org/10.1111/oik.04266
Hill MJ, Heino J, White JC, Ryves DB, Wood PJ (2019) Environmental factors are primary determinants of different facets of pond macroinvertebrate alpha and beta diversity in a human-modified landscape. Biol Conserv 237:348–357. https://doi.org/10.1016/j.biocon.2019.07.015
Huang Q, Gao JF, Cai YJ, Yin HB, Gao YN, Zhao JH, Liu LZ, Huang JC (2015) Development and application of benthic macroinvertebrate-based multimetric indices for the assessment of streams and rivers in the Taihu Basin, China. Ecol Indic 48:649–659. https://doi.org/10.1016/j.ecolind.2014.09.014
Huang XF, Chen WM, Cai QM (1999) Survey, observation and analysis of lake ecology. Standard methods For Observation and Analysis in Chinese ecosystem Research Network. Series V. Standards press of China, Beijing. (in Chinese)
Jamoneau A, Passy SI, Soininen J, Leboucher T, Tison-Rosebery J (2018) Beta diversity of diatom species and ecological guilds: response to environmental and spatial mechanisms along the stream watercourse. Freshw Biol 63(1):62–73. https://doi.org/10.1111/fwb.12980
Jiang XM, Sun X, Alahuhta J, Heino J, Xie ZC (2022) Responses of multiple facets of macroinvertebrate alpha diversity to eutrophication in floodplain lakes. Environ Pollut 306:119410. https://doi.org/10.1016/j.envpol.2022.119410
Keil P, Schweiger O, Kühn I, Kunin WE, Kuussaari M, Settele J, Henle K, Brotons L, Pe’er G, Lengyel S, Moustakas A, Steinicke H, Storch D (2012) Patterns of beta diversity in Europe: the role of climate, land cover and distance across scales. J Biogeogr 39(8):1473–1486. https://doi.org/10.1111/j.1365-2699.2012.02701.x
Lai JS (2020) Numerical ecology with R, 2nd edn. Higher Education Press, Beijing (in Chinese)
Langer TA, Murry BA, Pangle KL, Uzarski DG (2016) Species turnover drives β-diversity patterns across multiple spatial and temporal scales in Great Lake Coastal Wetland Communities. Hydrobiologia 777(1):55–66. https://doi.org/10.1007/s10750-016-2762-2
Legendre P, Legendre L (2012) Numerical ecology, 3rd, English. Elsevier Science BV, Amsterdam
Leibold MA, Holyoak M, Mouquet N, Amarasekare P, Chase JM, Hoopes MF, Holt RD, Shurin JB, Law R, Tilman D, Loreau M, Gonzalez A (2004) The metacommunity concept: a framework for multi-scale community ecology. Ecol Lett 7(7):601–613. https://doi.org/10.1111/j.1461-0248.2004.00608.x
Li K, Liu XJ, Zhou Y, Xu Y, Lv Q, Ouyang S, Wu XP (2019a) Temporal and spatial changes in macrozoobenthos diversity in Poyang Lake Basin. China Ecol Evol 9(11):6353–6365. https://doi.org/10.1002/ece3.5207
Li ZF, Wang J, Liu ZY, Meng XL, Heino J, Jiang XK, Xiong X, Jiang XM, Xie ZC (2019b) Different responses of taxonomic and functional structures of stream macroinvertebrate communities to local stressors and regional factors in a subtropical biodiversity hotspot. Sci Total Environ 655:1288–1300. https://doi.org/10.1016/j.scitotenv.2018.11.222
Li ZF, Liu ZY, Heino J, Jiang XM, Wang J, Tang T, Xie ZC (2020) Discriminating the effects of local stressors from climatic factors and dispersal processes on multiple biodiversity dimensions of macroinvertebrate communities across subtropical drainage basins. Sci Total Environ 711:134750. https://doi.org/10.1016/j.scitotenv.2019.134750
Li ZF, Heino J, Liu ZY, Meng XL, Chen X, Ge YH, Xie ZC (2021) The drivers of multiple dimensions of stream macroinvertebrate beta diversity across a large montane landscape. Limnol Oceanogr 66(1):226–236. https://doi.org/10.1002/lno.11599
Li ZF, Tonkin JD, Meng XL, Liu ZY, Zhang JQ, Chen X, Xie ZC, Heino J (2022) Seasonal variation in the metacommunity structure of benthic macroinvertebrates in a large river-connected floodplain lake. Ecol Indic 136:108662. https://doi.org/10.1016/j.ecolind.2022.108662
Liu SR, Xie GX, Wang LZ, Cottenie K, Liu DX, Wang BX (2016) Different roles of environmental variables and spatial factors in structuring stream benthic diatom and macroinvertebrate in Yangtze River Delta, China. Ecol Indic 61:602–611. https://doi.org/10.1016/j.ecolind.2015.10.011
Liu ZY, Zhou TT, Cui YD, Li ZF, Wang WM, Chen YS, Xie ZC (2021) Environmental filtering and spatial processes equally contributed to macroinvertebrate metacommunity dynamics in the highly urbanized river networks in Shenzhen, South China. Ecol Process 10(1):23. https://doi.org/10.1186/s13717-021-00297-2
Makumbe P, Kanda A, Chinjani T (2022) The relationship between benthic macroinvertebrate assemblages and water quality parameters in the Sanyati Basin, Lake Kariba, Zimbabwe. Sci World J 2022:5800286. https://doi.org/10.1155/2022/5800286
Mykrä H, Heino J, Muotka T (2007) Scale-related patterns in the spatial and environmental components of stream macroinvertebrate assemblage variation. Glob Ecol Biogeogr 16(2):149–159. https://doi.org/10.1111/j.1466-8238.2006.00272.x
Nunes CA, Braga RF, Figueira JEC, Neves FD, Fernandes GW (2016) Dung beetles along a tropical altitudinal gradient: environmental filtering on taxonomic and functional diversity. PLoS One 11(6):e0157442. https://doi.org/10.1371/journal.pone.0157442
Oksanen J, Simpson GL, Blanchet FG, Kindt R, Legendre P, Minchin PR, O'Hara RB, Solymos P, Stevens MHH, Szoecs E, Wagner H, Barbour M, Bedward M, Bolker B, Borcard D, Carvalho G, Chirico M, De Caceres M, Durand S, Evangelista HBA, FitzJohn R, Friendly M, Furneaux B, Hannigan G, Hill MO, Lahti L, McGlinn D, Ouellette M-H, Cunha ER, Smith T, Stier A, Ter Braak CJF, Weedon J (2022) vegan: community ecology package. R package version 2.6–2. https://CRAN.R-project.org/package=vegan
Paradis E, Schliep K (2019) ape 5.0: an environment for modern phylogenetics and evolutionary analyses in R. Bioinformatics 35(3):526–528. https://doi.org/10.1093/bioinformatics/bty633
Pires MM, Bieger L, Boelter T, Stenert C, Maltchik L (2021) Spatiotemporal assembly patterns of macroinvertebrate metacommunity structure in subtropical wetlands with different hydroperiods. Int Rev Hydrobiol 106(5–6):239–248. https://doi.org/10.1002/iroh.202002072
Qin CY, Ge YF, Gao J, Zhou SL, Yu J, Wang BX, Datry AT (2022) Ecological drivers of macroinvertebrate metacommunity assembly in a subtropical river basin in the Yangtze River Delta, China. Sci Total Environ 837:155687. https://doi.org/10.1016/j.scitotenv.2022.155687
R Core Team (2021) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. URL https://www.R-project.org/
Soininen J (2014) A quantitative analysis of species sorting across organisms and ecosystems. Ecology 95(12):3284–3292. https://doi.org/10.1890/13-2228.1
Stewart NA, Schriever TA (2023) Local environmental conditions influence species replacement in Great Lakes interdunal wetland macroinvertebrate communities. Freshw Biol 68(1):46–60. https://doi.org/10.1111/fwb.14008
Tian W, Yang ZS, Shao KQ, Pan H, Hu Y, Bai CR, Jiang XY, Gao G, Pan XM (2020) Effect of a comprehensive improvement project on water quality in urban lakes: a case study of water quality variation in Lihu Lake over the past 30 years. Environ Sci 41(1):183–193. https://doi.org/10.13227/j.hjkx.201906022. (in Chinese)
van de Bund WJ, Goedkoop W, Johnson RK (1994) Effects of deposit-feeder activity on bacterial production and abundance in profundal lake sediment. J N Am Benthol Soc 13(4):532–539. https://doi.org/10.2307/1467849
Viana DS, Figuerola J, Schwenk K, Manca M, Hobaek A, Mjelde M, Preston CD, Gornall RJ, Croft JM, King RA, Green AJ, Santamaria L (2016) Assembly mechanisms determining high species turnover in aquatic communities over regional and continental scales. Ecography 39(3):281–288. https://doi.org/10.1111/ecog.01231
Vincenty T (1975) Direct and inverse solutions of geodesics on the ellipsoid with application of nested equations. Surv Rev 23:88–94. https://doi.org/10.1179/sre.1975.23.176.88
Wallace JB, Webster JR (1996) The role of macroinvertebrates in stream ecosystem function. Ann Rev Entomol 41:115–139. https://doi.org/10.1146/annurev.en.41.010196.000555
Xu XB, Yang GS, Tan Y, Zhuang QL, Li HP, Wan RR, Su WZ, Zhang J (2016) Ecological risk assessment of ecosystem services in the Taihu Lake Basin of China from 1985 to 2020. Sci Total Environ 554:7–16. https://doi.org/10.1016/j.scitotenv.2016.02.120
Funding
This study was supported by the National Key Research and Development Program of China (2022YFC3204103) and the National Natural Science Foundation of China (92251304).
Author information
Authors and Affiliations
Contributions
Wenze Lu: conceptualization, data curation, formal analysis, investigation, methodology, visualization, writing—original draft. Xuwei Deng, Ping Xie, and Jun Chen: project administration, supervision, writing—review and editing. Zixin Zhao: formal analysis. Ren Ren and Xiao Rao: data curation, investigation.
Corresponding author
Ethics declarations
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
This article has not been published before; that it is not under consideration for publication elsewhere; that it is publication has been approved by all co-authors.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Thomas Hein
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lu, W., Deng, X., Zhao, Z. et al. Influence of environmental filtering and spatial processes on macroinvertebrate community in urban lakes in the Taihu Lake Basin, China. Environ Sci Pollut Res 31, 37010–37019 (2024). https://doi.org/10.1007/s11356-024-33694-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-024-33694-z