Abstract
Community assembly provides a conceptual foundation for understanding the processes that determine which and how many species live in a particular locality. Evidence suggests that community assembly often leads to a single stable equilibrium, such that the conditions of the environment and interspecific interactions determine which species will exist there. In such cases, regions of local communities with similar environmental conditions should have similar community composition. Other evidence suggests that community assembly can lead to multiple stable equilibria. Thus, the resulting community depends on the assembly history, even when all species have access to the community. In these cases, a region of local communities with similar environmental conditions can be very dissimilar in their community composition. Both regional and local factors should determine the patterns by which communities assemble, and the resultant degree of similarity or dissimilarity among localities with similar environments. A single equilibrium in more likely to be realized in systems with small regional species pools, high rates of connectance, low productivity and high disturbance. Multiple stable equilibria are more likely in systems with large regional species pools, low rates of connectance, high productivity and low disturbance. I illustrate preliminary evidence for these predictions from an observational study of small pond communities, and show important effects on community similarity, as well as on local and regional species richness.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The processes that determine the patterns of the number and composition of co-occurring species have been central to community ecology for decades (Gleason 1927; Clements 1938; Samuels and Drake 1997; Belyea and Lancaster 1999; Weiher and Keddy 1999). In 1975, Jared Diamond proposed that community composition was characterized by a series of 'Assembly Rules', which could be predicted by a few key variables, such as the size of the species pool, the abiotic environment, and interspecific interactions. However, in some cases, Diamond observed that community composition varied among sites that seemed similar in these key variables, and suggested that the timing of species invasions could lead to multiple stable equilibria (Diamond 1975).
The pattern of community assembly remains controversial. One view holds that there is a one-to-one match between environment and community. In this case, regardless of the historical order in which species invade, if all species have access to a given community, composition should converge towards a single configuration in localities with similar environmental conditions. Communities that differ in environmental conditions, on the other hand, will have divergent community structure (Fig. 1a). Even when the history in which species invade a community is highly variable, theoretical models (e.g., Law and Morton 1996) and controlled experiments (e.g., Neill 1975; Tilman et al. 1986; Sommer 1991) often find little or no effect of history. The alternative view holds that different historical sequences of species entering a locality can lead to different final community composition, even when the environment in each locality is similar and all species have access to the locality (Fig. 1b). This effect, known as multiple stable equilibria, can also be predicted from a variety of theoretical models (e.g., Luh and Pimm 1993; Law 1999) and has been found in several experiments (e.g., Robinson and Dickerson 1988; Drake 1991; Samuels and Drake 1997).
Schematic representation of community assembly when there is a single stable equilibrium for each of several different environments. Local diversity (α) and composition is the result of the regional species pool (γ) and the environmental filter. Differences in species composition from site to site (β-diversity) are the result of different environmental filters. B Schematic representation of community assembly when there are multiple stable equilibria from different assembly histories; environmental conditions are identical. Local diversity (α) and composition is the result of the regional species pool (γ) and the order in which species enter a community. Differences in species composition from site to site (β-diversity) are the result of different invasion sequences
Interestingly, the debate about community assembly dates back at least to the discourse between Clements and Gleason about plant succession (see Booth and Larson 1999; Young et al. 2001). Clements (e.g., 1938) viewed plant community structure as highly deterministic, with a single stable climax community determined by environmental conditions. Gleason (e.g., 1927), in developing his counterpoint to Clements, utilized concepts that were more aligned with the notion that the history of species invasions could dramatically influence the final configuration of that community. For example, Gleason (1927) wrote: "only the chances of seed dispersal have determined the allocation of species to different pools, but in the course of three or four years, each pool has a different appearance although the environment, aside from the reaction of the various species, is precisely the same for each."
Twenty years ago, Connell and Sousa (1983) extensively reviewed the evidence for multiple stable equilibria in natural communities, suggesting that such evidence was rare or nonexistent (see also Sousa and Connell 1985). More recent reviews have been equivocal (Samuels and Drake 1997; Belyea and Lancaster 1999). As such, I begin this paper by summarizing the current state of evidence for multiple stable equilibria in natural communities. Because the mechanisms by which communities assemble will affect the divergence in species composition among communities in a region of similar environmental conditions (β-diversity), community assembly will also influence the relationship between local species diversity (α-diversity) and the total number of species that occur in a region (γ−diversity), where γ=αβ (Whittaker 1972) or γ=α+β (Veech et al. 2002). I suggest that understanding the patterns of community assembly, and whether communities exist as a single or multiple stable equilibria, will be central to our understanding of the patterns of species composition and diversity that are at the heart of community ecology. Thus, I also develop a qualitative set of predictions on when communities should be more or less likely to exhibit multiple stable equilibria, and be regionally similar, with particular reference to regional and local environmental factors. Finally, I evaluate those predictions with observational data from freshwater pond communities.
Empirical evidence of community assembly
The most straightforward way to evaluate the occurrence of multiple stable equilibria is to determine if similar (or different) communities develop on similar sites (McCune and Allen 1985; Jenkins and Bukiema 1998). To demonstrate that multiple stable equilibria occur among different communities, two conditions must be met. First, the initial abiotic environmental conditions must be identical (Connell and Sousa 1983; Law 1999; Young et al. 2001). Because species can alter the abiotic conditions of their environment (e.g., Jones et al. 1995), multiple stable equilibria can only be detected if the initial environmental conditions are known. Second, all species from the regional species pool must have repeated access to the community. Thus, experimental tests require: (1) many replicates of local communities that vary only in the order in which species enter a community; and (2) a long enough time period for communities to approach some sort of equilibrium or limit cycle.
Despite the above restrictions, many studies have empirically evaluated community assembly concepts (reviewed in Samuels and Drake 1997; Young et al. 2001). Some communities have been observed to converge to a similar species composition, even when the order in which species enter a community is experimentally or naturally varied. These studies include highly controlled laboratory experiments (Neill 1975; Tilman et al. 1986; Sommer 1991), field experiments (Mitchley and Grubb 1986; Weiher and Keddy 1995) and broad-scale observations of natural systems (Kodric-Brown and Brown 1993; Wilson et al. 2000). Many other studies have suggested that even under similar environmental conditions, very different communities can develop as a result of variation in the timing and sequence of species invasions (but see, for methodological issues, Connell and Sousa 1983; Sousa and Connell 1985; Grover and Lawton 1994). These studies also range from controlled laboratory experiments (e.g., Gilpin et al. 1986; Robinson and Dickerson 1987; Robinson and Edgemon 1988; Drake 1991; Drake et al. 1993; Lawler 1993), field experiments (Quinn and Robinson 1987; Jenkins and Buikema 1998; Chase 2003a) and observations of natural systems (Cole 1983; McCune and Allen 1985; Petraitis and Latham 1999). Thus, the general conclusion from reading the literature seems to be that in some cases, communities converge to a single equilibrium, regardless of invasion order, whereas in other cases, communities achieve multiple stable equilibria.
Interestingly, variable results have even been found within the same study under different environmental conditions. In a nutrient manipulation experiment, Inouye and Tilman (1995) found that replicate plots that differed in initial herbaceous plant composition converged towards a similar community composition in a newly established (i.e., recently disturbed) old-field, but diverged in community composition in a native savanna (i.e., less disturbed). Likewise, Drake (1991) showed that larger microcosm ecosystems showed more prominent historical effects on final community composition than did smaller microcosm ecosystems.
A framework for synthesis
"...invasion order is more influential where immigration rates are relatively low...than it is where dispersal from outside sources is high..." (Robinson and Edgemon 1988, pp 1410).
"It appears to us that an unproductive, stressed community...[is] likely to be deterministic...and not prone to control by historical events" (Booth and Larson 1999, pp 216).
Sometimes history matters, creating multiple stable equilibria, and sometimes it does not. The interesting question, then, and the focus of the remainder of this article, is whether we can identify the ecological features of a system that influence whether community assembly is primarily deterministic, and leads to a single stable equilibria, or whether stochastic events such as the timing of the arrival of species into a community, leads to multiple stable equilibria.
The mechanisms of community assembly will strongly influence the relationships between species diversity at local and regional spatial scales, as well as the degree of similarity among local communities in a region, and thus the occurrence of multiple stable equilibria. Below, I outline how the regional and local conditions should influence the relative importance of history versus environment in determining local species composition, and thus the patterns of similarity in community composition among localities within regions. In Table 1, I present a summary of the predictions discussed below. Specifically, I discuss the conditions where multiple stable equilibria are more or less likely to occur, how this will alter patterns of β-diversity among environmentally similar local communities within a region, and how this will alter the scaling relationship between local (α) and regional (γ) diversity among those communities.
Regional factors
Size of the regional species pool
In general, multiple stable equilibria should exist, and be more likely to be expressed, if the regional species pool is large (Law and Morton 1993) because it is likely that there are more species in the regional species pool that can coexist in any local community, and that many species in the regional species pool possess similar traits. In this case, initial invasion by one species is more likely to preclude invasion by other similar species, creating multiple stable equilibria. On the other hand, when the regional species pool is small, and similar to the number of species that can coexist locally, the history of species invasions will have a lower probability of determining the final community composition, and local communities will be relatively similar. This leads to a relatively simple prediction: regional (γ) diversity, by definition, will be higher in areas with large species pools, and if local (α) diversity is limited by environmental conditions and thus does not vary among similar sites, then β-diversity also must increase as the size of the regional species pool increases.
Rate of dispersal within the region
The proximity among localities within a region, as well as other landscape attributes (e.g., barriers), can influence dispersal rates. Dispersal rates, in turn, can increase the similarity among local communities by decreasing the likelihood that potential multiple equilibria will be achieved. This prediction arises from two related processes: (1) early invading species do not have the time to grow to high enough densities to dominate and preclude invasion by subsequently invading species before those other species enter the community (Robinson and Edgemon 1988; Lockwood et al. 1997); and (2) increased rates of connection among local communities with multiple stable equilibria can cause all but one of those to be eliminated; this occurs because every population is assumed to have a finite extinction probability, and with high dispersal rates among patches, one species will eventually overcome the other through a random walk unless environmental heterogeneity is present (Amarasekare 2000; Yu and Wilson 2001).
If multiple stable equilibria are less likely with higher rates of dispersal, then β-diversity will decline with increasing connectance among localities. Similar patterns are also expected if β-diversity is determined by heterogeneity in environmental conditions among localities, rather than multiple stable equilibria (Loreau and Mouquet 1999; Amarasekare and Nisbet 2001; Mouquet and Loreau 2002). As a result, regional (γ) diversity should generally decrease with higher rates of dispersal. Predictions for local (α) diversity are somewhat more complex. I suggest that if adjacent communities connected by dispersal reside in multiple stable equilibria, then moderate dispersal can enhance local diversity through processes akin to the source-sink models of heterogeneous environments (Mouquet and Loreau 2002). This is because if each species dominates a different locality, and can preclude invasion by the other, the locality it dominates will act as a source, and the other locality, a sink, even if the environmental conditions among the localities are identical (see, e.g., Amarasekare 1998, for similar phenomena in single species models). From low to intermediate rates of dispersal, local (α) diversity should increase with dispersal rates, because species can persist as sink populations in localities that they otherwise would be eliminated. From intermediate to high rates of dispersal, local (α) diversity decreases with dispersal, because species that are adept at colonizing open patches can eliminate less adept species from the regional species pool (Mouquet and Loreau 2002).
Local factors
The environment itself can influence whether or not multiple stable equilibria are likely to occur. Here, I build upon a theme developed by Booth and Larson (1999) that multiple stable equilibria will be more common in local habitats that have relatively benign environmental conditions, and that a single equilibrium will be more common in harsh environmental conditions. To illustrate these general predictions, I discuss two specific types of environmental factors that vary from benign to harsh, productivity and disturbance.
Primary production
Primary productivity is often considered to be a primary determinant of species composition, coexistence, and diversity (e.g., Rosenzweig 1995). In a verbal model, Booth and Larson (1999) suggested that multiple stable equilibria would be more likely in high than in low productivity environments. However, models consisting only of a few species sometimes predict that multiple stable equilibria could be present at intermediate levels of primary productivity whereas a single equilibrium should exist at low and high productivity. Furthermore, this effect should only occur when species have stronger relative interspecific effects than intraspecific effects, such as in food webs where prey species are size-structured or intraguild predators are present (e.g., Chase 1999; Diehl and Feissel 2000). In several, but not all cases, Chase and Leibold (2003) showed that adding more species into these types of models can recapture the verbal intuition of Booth and Larson (1999). For example, in a food web model where prey species share a common resource and a common predator, and there are trade-offs among species abilities to compete for resources and to resist or tolerate predators (sensu Holt et al. 1994), the interaction between resource competition and apparent competition mediated through predators can lead to more frequent multiple stable equilibria at high than at low productivity (Chase and Leibold 2003). In low productivity regions, only a limited number species — i.e., those tolerant of low resources — can persist, and multiple stable equilibria are not expected. However, as productivity increases, more species can potentially persist, than do so at any given equilibrium. Here, early invading species can alter the environment by either reducing resources through consumption or by increasing the density of predators (if the invader is a prey species), which influences the potential for later invaders to establish, and thus can create multiple stable equilibria. Interestingly, a similar effect does not occur when species compete for resources with no predators present (Chase and Leibold 2003).
If higher productivity localities have more multiple stable equilibria, and are thus more dissimilar than lower productivity localities, I also predict that the productivity-diversity relationship will depend on spatial scale. At local scales, a wide variety of theoretical and empirical evidence suggests that the relationship is often unimodal (hump-shaped), first rising, and then declining with productivity (Rosenzweig 1995). However, because more productive communities are predicted to be more dissimilar in species composition, regional diversity should increase with productivity, even if local diversity declines. Such scale-dependence may be typical in nature (Mittelbach et al. 2001; Chase and Leibold 2002).
Rate of disturbance
I define disturbance as an abiotic event (e.g., fire, flooding, drought) that kills (influencing death rates) or significantly stresses (influencing birth rates) a significant proportion of the individuals of at least some species in the regional pool. Based on this definition, I predict that localities with higher rates of disturbance are less likely to exhibit multiple stable equilibria than localities with lower rates of disturbance. This prediction arises from three related effects. First, only a subset of the species in the regional species pool can persist in localities with high rates of disturbance, whereas most species could persist in localities with low rates of disturbance in the absence of interspecific interactions. Because fewer species can persist in disturbed regions, the probability of multiple stable equilibria declines. Second, frequent disturbance will prolong the time it takes for an early invading species to achieve a high density, or if it does achieve high density, will reduce their numbers. Third, species with traits that allow colonization following habitat disturbance, or with traits that make them resistant to disturbance in the first place, often are poorer competitors due to trade-offs (e.g., Tilman 1994; Yu and Wilson 2001). Because those species are the ones expected in disturbed habitats, they will be less likely to preclude establishment by other species through interspecific interactions.
At local spatial scales, diversity is often expected to have a unimodal (hump-shaped) relationship with diversity highest at intermediate levels of disturbance; an effect known as the 'intermediate disturbance hypothesis' (Connell 1978). At regional spatial scales, the influence of disturbance on diversity will depend on the magnitude of difference between local and regional diversity. If regional diversity is much higher than local diversity then the influence of disturbance on local diversity through the intermediate disturbance hypothesis will have a negligible effect on regional diversity as compared to the expected decrease in regional diversity with increasing similarity among localities (low β-diversity). Such scale-dependence in the disturbance-diversity relationship may be a primary reason behind the mixed results observed from empirical studies, such as those reviewed by MacKay and Currie (2001). Alternatively, if regional and local diversity are more similar, then the effect of disturbance on regional diversity is less straightforward. There will be a positive effect on regional diversity owing to the intermediate disturbance effect at local scales, but a negative effect on regional diversity owing to reduced among-locality compositional differences (β-diversity); the final result will depend on the balance between those factors.
Interactions among the factors
I have discussed some predictions about the effects of several regional and local factors on the similarity among communities within regions. All of these factors are likely to simultaneously act upon any given local community, and thus the interaction of the factors may also play a strong role. In general, the factor that constrains community similarity the most will have overriding effects on local community similarity. For example, if the size of the regional species pool is small, then this constraint will have the strongest effect on community similarity, regardless of the effects of dispersal rate, disturbance rate, or level of productivity. Likewise, if disturbance is high, then this will most strongly influence community similarity, regardless of the level of primary productivity, dispersal rate, or size of the regional species pool.
Empirical tests in ponds
Below, I illustrate some patterns of community similarity, as well as local and regional species richness from broad scale observational survey of small ponds. Individual ponds represent reasonably distinct communities, and there is considerable variation among ponds in disturbance rate, productivity and interpond distance (a surrogate for dispersal rate). I cannot directly assess whether the patterns of community compositional similarity arise because of single or multiple stable equilibria, because I do not know the history of these environments. Instead, I use these observational surveys to determine whether the patterns are consistent with the predictions outlined above. Experimental studies will be necessary to determine whether these patterns are due to the mechanisms ascribed.
Overview of sampling methods
In May to August of 1996, 1997 and 1998, I surveyed macroscopic animals in 72 ponds (also marshes and swamps) throughout southwestern Michigan, USA (Kalamazoo, Calhoun, and Barry Counties). In May to August 1998, 1999, 2000, and 2001, I surveyed 45 similar habitat types in northeastern Pennsylvania, USA (Crawford, Erie, and Mercer counties) and northwestern Ohio, USA (Ashtabula and Trumbull counties). I did not survey habitats that contained large fish, such as Centrachidae sunfishes, which are indicative of lake ecosystems, but did survey habitats with smaller fishes, amphibians, and a wide-variety of invertebrates.
In each pond, I surveyed several biotic and abiotic variables (see Chase and Leibold 2002; Chase 2003b, for additional detail). For geographical, physical, and chemical variables, I measured pH, conductivity, total nitrogen, and total phosphorus, pond area, average depth, tree canopy coverage, and proximity to other lentic habitats. I estimated in situ primary productivity by measuring algal accrual on plastic substrates covered by 0.33-mm mesh (to exclude most grazers) and placed in each pond for 2 weeks (Clesceri et al. 1998). This method correlates with other methods for estimating productivity (Clesceri et al. 1998).
I determined presence or absence of taxa by sampling with a 0.1x0.1–m wide D-net and a 1-mm mesh net. Species collected included most benthic invertebrates (e.g., Amphipoda and Mollusca), benthic and free-swimming insects (primarily Coleoptera, Diptera, Hemiptera and Odonata,), amphibians (larval Anura, larval and adult Caudata), and small fishes (primarily Cyprinidae, Umbridae, and Gasterosteridae). Qualitative results for planktonic animals (e.g., cladocera, copepoda, and rotifera) and plants (macro-algae and emergent macrophytes) are often similar (Chase and Leibold 2002).
I spread samples over the entire surface and depth of the pond. After every sweep, species were dumped into a white enamel pan, and were identified immediately visually, or with the aid of a hand lens or dissecting microscope. I ignored terrestrial and semi-terrestrial species that were often found on the surface, or on emergent plants. I continued D-net sweeps in a pond until no new groups of species were observed in ten sweeps. Of the species that were difficult to identify visually, or when multiple coexisting species were too similar to discern in the field, I collected voucher specimens, preserved them in 70% ethanol, and transported them back to the laboratory for more detailed identification. I was able to categorize approximately 50% of all individuals collected to species, and another 25% to genus, but not species. Approximately 10% of the individuals were categorized to family or a higher taxonomic level. The remaining 15% of individuals were chironomidae midges (Diptera), which I sorted into broad morphological categories. Because species presence/absence can vary temporally within and among seasons, I only included ponds in this analyses that were sampled in more than one sampling period [classified as early (May to June) or late (July to August)] and in more than one year; a taxon was classified as present in a pond if it was found in any one of those periods.
To explore the local and regional patterns of species richness and community composition, I defined an individual pond as the local scale, and defined a region as three local ponds that were nested within a watershed. Although there were often more than three ponds in a watershed, I only sampled those three, but included the other ponds in the watershed when quantifying the characteristics of the region (e.g., average distance of separation). I took care so that the three ponds chosen to represent a region were similar in total surface area (range: 1,500 m2±500m2), so as not to confound area as a factor influencing species richness or composition. I calculated local species richness as the number of species found within a single pond and regional richness as the number of species found within the three-pond region. Finally, I calculated inter-pond dissimilarity in species composition within each three-pond region as an index of β-diversity. I used species presence/absence data to calculate dissimilarity as 1-C, where C is Jaccard's index of community similarity (C). C=j/(a+b−j), where a and b are the number of species in each community, and j is the number of species that are present in both communities. Dissimilarity ranges from 1 when no species are shared between two communities to 0 when all species are shared. I averaged dissimilarity estimates from each pair-wise comparison of ponds within a three-pond region to estimate regional dissimilarity.
For each of the comparisons below, I chose a set of pond regions (watersheds with three ponds) to measure average local species richness (the average richness of the three ponds), regional species richness (the cumulative richness of the three ponds), and species compositional dissimilarity among the three ponds within a region. I compared the effects of several independent variables: average distance of separation (a surrogate of dispersal rate; e.g., Maguire 1963; Conrad et al. 1999), primary productivity, and pond permanence (a surrogate of disturbance). For each independent variable, I chose regions so that the variance in other factors that might influence species diversity and composition, were minimized. I discuss these potential confounding factors along with the results and their potential implications in each section below.
Habitat connectivity
-
Prediction 1: more connected communities are more similar in community composition than less connected communities
-
Prediction 2: more connected communities have higher local, but lower regional species richness than less connected communities
To examine these predictions, I chose a subset of regions that consisted of permanent ponds (those with standing water year-round) of similar intermediate levels of productivity (2–4 mg cm-2 day-1 of algal accrual), and in which the average distance of separation between ponds within a region varied. There were a total of eight three-pond regions that matched these criteria. Regional inter-pond distances averaged between <10 m to >950 m. There were no obvious differences among these regions in any other environmental variables.
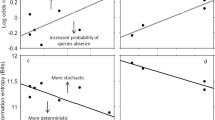
Ponds that were closer to one another, and thus more likely connected by dispersal, were also more similar to one another in composition (Fig. 2a). This pattern is qualitatively similar to that predicted if higher dispersal rates decrease the frequency of multiple stable equilibria. An alternative explanation is that regions with more distant ponds are also more different from one another in other environmental characteristic. However, none of the seven environmental variables I measured that were not directly related to the variables of interest here (e.g., water chemistry) correlated with distance of separation (P>0.5). Furthermore, the average local species richness was higher, but the regional species richness lower in regions that had ponds that were closer together (Fig. 2b). Again, this result is consistent with the theoretical predictions outlined above (see also Hastings and Gavrilets 1999; Amerasakare 2000).
The relationship between average interpond distance and regional dissimilarity. The slope of the relationship is significantly positive (regression: n=8, r 2=0.80, P<0.003). B The relationship between average interpond distance and average local and regional species richness. Average local richness is negatively related to interpond distance (regression: n=8, r 2=0.75, P<0.004), whereas regional richness is positively related to interpond distance (regression: n=8, r 2=0.45, P<0.04)
Primary productivity
-
Prediction 1: less productive regions are more similar in community composition than more productive regions
-
Prediction 2: the shape of the productivity-diversity relationship depends on spatial scale
To examine these predictions, I chose regions of permanent ponds that had similar, intermediate average distances of separation (300–600 m) among ponds with the region. There were a total of ten regions that satisfied this criterion, and the estimated rate of primary productivity ranged from <1 to >6 mg cm-2 day-1 (see also Chase and Leibold 2002). Average productivity of a region was unrelated to any of the other measured environmental variables (P>0.4). The degree of dissimilarity in community composition was positively correlated with primary productivity (Fig. 3a), as predicted. An alternative explanation for this pattern would be if the variance in productivity, or other environmental factors, increased as the mean value of productivity increased. However, average productivity was not significantly correlated with the variance in productivity or the other measured environmental factors (P>0.2) (see also Chase and Leibold 2002).
. A The relationship between primary productivity and regional dissimilarity in permanent ponds. The slope of the relationship is significantly positive (regression: n=10, r 2=0.79, P<0.001). B The relationship between primary productivity and local and regional species richness. Local richness is unimodally related to productivity (the estimated peak of the relationship from quadratic regression is significantly higher than the minimum and less than the maximum productivity values; P>0.05; Mitchell-Olds and Shaw 1987), while regional richness is positively related to productivity (regression: n=10, r 2=0.75, P<0.001). Points at the lowest two productivity levels are displaced for clarity. Based on the data from Chase and Leibold (2002); note that dissimilarity values differ from the original publication due to some graphical errors in the original presentation, and use of slightly different sets of data
To test the prediction that variation in the degree of similarity among regions along a productivity gradient would lead to scale-dependant productivity-diversity relationships, I compared the relationship between productivity and species richness at local and regional scales. At a local, among pond scale, species richness was a unimodal (hump-shaped) function of productivity, whereas regional species richness was positively linearly related to productivity (Fig. 3b) (see also Chase and Leibold 2002). This result may help to explain previous discrepancies in patterns of the productivity-diversity relationship where the pattern is typically unimodal at local scales(e.g., Rosenzweig 1995; Dodson et al. 2000; Mittelbach et al. 2001), but increasing at larger spatial scales (Currie and Paquin 1987; Currie 1991; Mittelbach et al. 2001).
Disturbance
-
Prediction 1: more disturbed regions are more similar in community composition than less disturbed regions
-
Prediction 2: the relationship between disturbance and diversity depends on spatial scale
For this analysis, I categorized ponds into three categories based on their probability of retaining standing water throughout the year: permanent, semi-permanent, or temporary (see also Wellborn et al. 1996; Williams 1996; Schneider 1997). Permanent ponds are those that did not dry for at least the 10 years previous to this study (my personal observations combined with personal communications from many sources). Semi-permanent ponds are those that dried completely once every 3–5 years during the course of the time that I monitored them. Temporary ponds are those that permanently dried every year, typically in late summer.
Defining pond permanence as a metric for disturbance may be unappealing because some species are so well adapted to these ephemeral habitats, that they actually need a pond to completely dry before they can complete their life-cycles. Indeed, this is likely to be true for any of the events that we often label as a disturbance. For example, some species of terrestrial plants require floods or fire in order to complete their life-cycles, and yet we usually term these events as disturbances. Many species that occur in temporary ponds can also live in permanent ponds when interspecific interactors are removed, when ponds are initiated, and when a rare drying event (natural or human-caused) kills off permanent pond species. In contrast species that live in permanent waters cannot persist in temporary habitats (Wellborn et al. 1996; Williams 1996; Schneider 1997). This suggests that the environmental filter for temporary ponds is harsher than that for habitats with permanent standing water, and that there are fewer species from the regional species pool that can live in temporary habitats than can live in permanent habitats.
From my surveys, I found four regions of temporary ponds and three regions of semi-permanent ponds that were all intermediate (between 2 and 4 mg cm-2 day-1) in productivity, with which I could compare with four regions of permanent ponds in this range of productivity. All pond regions for this analysis had an average distance of separation of intermediate values (300–600 m) so not as to confound the influence of dispersal rates. Permanent ponds were most dissimilar, temporary ponds were most similar, and semi-permanent ponds were intermediate (Fig. 4a).
The dissimilarity among ponds within regions that vary in permanence. Overall, there was an overall negative effect of decreasing permanence on regional community dissimilarity (ANOVA: MS=0.032, F 2,8=5.32, P<0.03). Bars with different letters refer to significant differences among pairwise comparisons (Tukey's hsd: P<0.05). B Local (white bars) and regional (black bars) richness among ponds in the different permanence categories. Local richness was significantly effected by pond permanence (ANOVA: MS=60.38, F 2,8=15.38, P<0.002), and was highest in the semi-permanent ponds. Regional richness was significantly effected by pond permanence (ANOVA: MS=110.38, F 2,8=24.9, P<0.0001), and was highest in permanent ponds. For local richness, bars with different letters, and for regional richness, bars with different numbers, refer to significant differences among pairwise comparisons (Tukey's hsd: P<0.05)
Because pond habitats that were more permanent were also more dissimilar in species composition, I also expect a scale-dependant relationship between permanence (disturbance) and species richness. In accord with the intermediate disturbance hypothesis (see above), the average local species richness was highest in semi-permanent ponds, and equally low in both permanent and semi-permanent ponds. However, at the regional spatial scale, species richness was highest in permanent and semi-permanent ponds and lowest in temporary ponds (Fig. 4b). This result suggests that understanding the effects of disturbance on diversity will require a consideration of scale, and may help to explain some of the discrepancy observed in studies exploring the effects of disturbance on species diversity (Mackay and Currie 2001).
Conclusions
The mechanisms of community assembly are important for determining the composition of local communities, local diversity (α-diversity), regional diversity (γ-diversity), and site-to-site variation in species composition (β-diversity). To date, however, theory, observations, and controlled experiments have not yielded general patterns or processes. Rather than argue that community assembly primarily leads to a single stable equilibria, or whether the sequence of assembly history can drive community composition towards multiple stable equilibria, I argue instead that it is more useful to ask when multiple stable equilibria should and should not be common. I suggest that a single equilibrium of community composition, and similar community composition in environmentally similar sites, should be most prominent in systems with small species pools, high levels of dispersal, low productivity, and high rates of disturbance. Alternatively, multiple stable equilibria, and dissimilar community composition in environmental similar sites, should be more prominent in systems with large species pools, low levels of dispersal, high productivity, and low rates of disturbance.
Throughout, I focus equilibrium concepts, and assume that community composition is reasonably stable. My use of the equilibrium concept need not be taken literally, and communities do not have to reside at a single unchanging equilibrium, in order for the concepts to be of value. Nevertheless, cases may exist where species composition is continually in flux (Law 1999); the concepts discussed here will be of less value for such instances. Likewise, several studies, primarily on trees in tropical forests, have examined the factors that influence community composition by estimating metrics of community similarity across spatial gradients and comparing the relative role of environmental factors and spatial proximity (dispersal limitation) in determining those patterns (e.g., Condit et al. 2002; Potts et al. 2002; Tuomisto et al. 2003). These studies have focused primarily on spatial versus environmental effects on community composition, ignoring the potential role of history leading to multiple stable equilibria. In contrast, my synthesis focused on the role of heterogeneity versus variation in the history of species invasions and ignored the potential role of spatial distribution and dispersal limitation. An important avenue for future research on patterns of community composition will be to consider all three of these factors (environment, dispersal, and invasion history) simultaneously.
Community assembly also has several applied implications. For example, successful restoration of a degraded community requires information about the processes that naturally determine community composition. If local conditions alone determine community composition, then restoration efforts should focus on returning the local condition (e.g. level of productivity, rate of disturbance) to its previous state. However, if regional processes, or a combination of local conditions and regional processes, determine community composition, then both local conditions and regional processes need to be restored to achieve the desired community (Keddy 1999; Lockwood and Pimm 1999; Young et al. 2001). In addition, many empirical studies have shown that both species diversity and species composition affect a variety of ecosystem-level properties, such as productivity and nutrient cycling (see overviews in Loreau et al. 2001; Kinzig et al. 2002). Community assembly affects the diversity and composition of local communities, and therefore might also affect the ecosystem function of those communities. However, the link between community assembly and ecosystem function remains unclear. If multiple stable equilibria arise because species are somewhat functionally redundant, then there may be little effect on ecosystem function.
References
Amarasekare P (1998) Interactions between local dynamics and dispersal: Insights from single species models. Theor Popul Biol 53:44–59
Amarasekare P (2000) The geometry of coexistence. Biol J Linn Soc 71:1–31
Amarasekare P, Nisbet R (2001) Spatial heterogeneity, source-sink dynamics, and the local coexistence of competing species. Am Nat 158:572–584
Belyea LR, Lancaster J (1999) Assembly rules within a contingent ecology. Oikos 86:402–416
Booth BD, Larson DW (1999) Impact of language, history, and choice of system on the study of assembly rules. In Weiher E, Keddy PA (eds), Ecological assembly rules: perspectives, advances, retreats. Cambridge University Press, Cambridge, UK, pp 206–229
Chase JM (1999) Food web effects of prey size-refugia: variable interactions and alternative stable equilibria. Am Nat 154:559–570
Chase JM (2003a) Experimental evidence for alternative stable equilibria in a benthic fond food web. Ecology Letters 6:(in press)
Chase JM (2003b) Strong and weak trophic cascades along a productivity gradient. Oikos 101:187–195
Chase JM, Leibold MA (2002) Spatial scale dictates the productivity-diversity relationship. Nature 415:427–430
Chase JM, Leibold MA (2003). Ecological niches: linking classical and contemporary approaches. University of Chicago Press, Chicago, Ill.
Clements FE (1938) Nature and structure of the climax. J Ecol 24:252–282
Clesceri LS., Greeberg AE, Eaton AD (1998) Standard methods for the examination of water and wastewater, 20th edn. American Public Health Association, Washington, DC
Cole BJ (1983) Assembly of mangrove ant communities: patterns of geographical distribution. J Anim Ecol 52:397–347
Condit R, Pitman N, Leigh Jr EG, Chave J., Terborgh J, Foster RB, Núñez P, Aguilar S, Valencia R, Villa G, Muller-Landau HC, Losos E, Hubbell SP (2002) Beta-diversity in tropical forest trees. Science 295:666–669
Connell JH (1978) Diversity in tropical rainforests and coral reefs. Science 199:1302–1310
Connell JH, Sousa WP (1983) On the evidence needed to judge ecological stability of persistence. Am Nat 1212:789–824
Conrad KF, Willson, KH, Harvey IF, Thomas CJ, Sherratt, TN (1999) Dispersal characteristics of seven odonate species in an agricultural landscape. Ecography 22:524–531
Currie DJ (1991) Energy and large-scale patterns of animal and plant species richness. Am Nat 137:27–49
Currie DJ, Paquin V (1987) Large-scale biogeographical patterns of species richness of trees. Nature 329:326–327
Diamond JM (1975) Assembly of species communities. In: Cody ML, Diamond JM (eds) Ecology and evolution of communities. Harvard University Press, Cambridge, Mass., pp 342–444
Diehl S, Feissel M (2000) Effects of enrichment on three-level food chains with omnivory. Am Nat 155:200–218
Dodson SI, Arnott SE, Cottingham KL (2000) The relationship in lake communities between primary productivity and species richness. Ecology 81:2662–2679
Drake JA (1991) Community assembly mechanics and the structure of an experimental species ensemble. Am Nat 137:1-26
Drake JA, Flum TE. Witteman GJ. Voskuil J, Hoylman AM, Creason C, Kenny DA, Huxel GR, Larue CS, Duncan JR (1993) The construction and assembly of an ecological landscape. J Anim Ecol 62:117–130
Gilpin ME, Carpenter MP, Pomerantz, MJ (1986) The assembly of a laboratory community: multi-species competition in Drosophila. In: Diamond J, Case TJ (eds) Community ecology. Harper and Row, New York, pp 23–40
Gleason HA (1927) Further views on the succession concept. Ecology 8:299–326
Grover JP, Lawton JH (1994) Experimental studies on community convergence and alternative stable states: comments on a paper by Drake et al. J Anim Ecol 63:484–487
Hastings A, Gavrilets S (1999) Global dispersal reduces local diversity. Proc R Soc Lond B 266:2067–2070
Holt RD, Grover J.,Tilman, D (1994) Simple rules for interspecific dominance in systems with exploitation and apparent competition. Am Nat 144:741–771
Inouye RS, Tilman, D (1995) Convergence and divergence of old-field vegetation after 11-year of Nitrogen addition. Ecology 76:1872–1887
Jenkins DG, Buikema Jr. AL (1998) Do similar communities develop in similar sites? A test with zooplankton structure and function. Ecol Monogr 68:421–443
Jones CG, Lawton JH, Shachak M (1995) Organisms as ecosystem engineers. Oikos 69:373–386
Keddy PA (1999) Wetland restoration: the potential for assembly rules in the serivice of conservation. Wetlands 19:242–253
Kinzig AP, Pacala S., Tilman D (eds). (2002) The functional consequences of biodiversity: empirical progress and theoretical extensions. Princeton University Press, Princeton, N.J.
Kodric-Brown A, Brown, JH. (1993) Highly structured fish communities in Australian desert springs. Ecology 74:1847–1855
Law R (1999) Theoretical aspects of community assembly. In: McGlade J (ed.) Advances in ecological theory: principles and applications. Blackwell, Oxford, pp 141–171
Law R, Morton RD (1993) Alternative permanent states of ecological communities. Ecology 74:1347–1361
Law R, Morton RD (1996) Permanence and the assembly of ecological communities. Ecology 77:762–775
Lawler SP (1993) Direct and indirect effects in microcosm communities of protists. Oecologia 93:184–190
Lockwood JL, Pimm SL (1999) When does restoration succeed? In: Weiher E, Keddy PA (eds) Ecological assembly rules: perspectives, advances, retreats. Cambridge University Press, Cambridge, UK, pp 363–392
Lockwood JL, Powell RD, Nott MP, Pimm SL (1997) Assembling ecological communities in time and space. Oikos 80:549–553
Loreau M, Mouquet N (1999) Immigration and the maintenance of local species diversity. Am Nat 154:427–440
Loreau M. et al. (2001) Biodiversity and ecosystem functioning: current knowledge and future challenges. Science 294:804–808
Luh HK, Pimm SL (1993) The assembly of ecological communities: a minimalist approach. J Anim Ecol 62:749–765
Mackay RK, Currie DJ (2001) The diversity-disturbance relationship: is it generally strong and peaked? Ecology 82:3479–3492
Maguire B (1963) The passive dispersal of small aquatic organisms and their colonization of isolated bodies of water. Ecol Monogr 33:61–185.
McCune B, Allen TFH (1985) Will similar forests develop on similar sites? Can J Bot. 63:367–376
Mitchell-Olds T, Shaw RG (1987) Regression analysis of natural selection: statistical inference and biological interpretation. Evolution 41:1149–1161
Mitchley J, Grubb PJ (1986) Control of relative abundance of perennials in chalk grassland in southern England I. Constance of rank order and results of pot- and field-experiments on the role of interference. J Ecol 74:1139–1166
Mittelbach GG, Steiner CF, Scheiner, SM. Gross KL, Reynolds HL Waide RB, Dodson SI, Gough L (2001) What is the observed relationship between species richness and productivity? Ecology 82:2381–2396
Mouquet N, Loreau M (2002) Coexistence in metacommunities: the regional similarity hypothesis. Am Nat 149:420–426
Neill WE (1975) Experimental studies of microcrustacean competition, community composition and efficiency of resource utilization. Ecology 56:809–826
Petraitis PS, Latham RE (1999) The importance of scale in testing the origins of alternative community states. Ecology 80:429–442
Potts MD., Ashton PS, Kaufman LS, Plotkin JB (2002) Habitat patterns in tropical rainforests: a comparison of 105 plots in northwest Borneo. Ecology 83:2782–2797
Quinn JF, Robinson GR (1987) The effects of experimental subdivision on flowering plant diversity in a California annual grassland. J Ecol 75:837–855
Robinson JV, Dickerson JE (1987) Does invasion sequence affect community structure? Ecology 68:587–595
Robinson JV, Edgemon MA (1988) An experimental evaluation of the effect of invasion history on community structure. Ecology 69:1410–1417
Rosenzweig ML (1995) Species diversity in space and time. Cambridge University Press, Cambridge, UK
Samuels CL, Drake JA (1997) Divergent perspectives on community convergence. Trends Ecol Evol 12:427–432
Schneider DW (1997) Predation and food web structure along a habitat duration gradient. Oecologia 110:567–575
Sommer U (1991) Convergent succession of phytoplankton in microcosms with different inoculum species composition. Oecologia 87:171–179
Sousa WP, Connell JH (1985) Further comments on the evidence for multiple stable points in natural communities. Am Nat 125:612–615
Tilman D (1994) Competition and biodiversity in spatially structured habitats. Ecology 75:2-16
Tilman D, Kiesling R, Sterner R, Kilham S, Johnson FA (1986) Green, bluegreen and diatom algae: taxonomic differences in competitive ability for phosphorus, silicon, and nitrogen. Arch Hydrobiol 106:473–485
Tuomisto H, Ruokolainen K, Yli-Halla M (2003) Dispersal, environment, and floristic variation of western Amazonian forests. Science 299:241–244
Veech JA, Summerville KS, Crist TO, Gering JC (2002) The additive partitioning of species diversity: recent review of an old idea. Oikos 99:3-9
Weiher E, Keddy P (1995) The assembly of experimental wetland plant communities. Oikos 73:323–335
Weiher E, Keddy P (1999) Assembly rules as general constraints on community composition. In: Weiher E, Keddy P (eds) Ecological assembly rules: perspectives, advances, retreats. Cambridge University Press, Cambridge, UK, pp 251–271
Wellborn GA, Skelly DK, Werner EE (1996) Mechanisms creating community structure across a freshwater habitat gradient. Annu Rev Ecol Syst 27:337–363
Whittaker RH (1972) Evolution and the measurement of species diversity. Taxon 21:213–251
Williams DD (1996) Environmental constraints in temporary fresh waters and their consequences for the insect fauna. J North Am Benthol Soc 15:634–650
Wilson JB, Steel ME, Dood BJ, Andreson I, Ullmann, Bannister P (2000) A test of community reassembly using the exotic communities of New Zealand roadsides in comparison to British roadsides. J Ecol 88:757–764
Young TP, Chase JM, Huddleston RD (2001) Community succession and assembly: comparing, contrasting, and combining paradigms in the context of ecological restoration. Ecol Rest 19:5-18
Yu DW, Wilson HB (2001) The competition-colonization trade-off is dead; Long live the competition-colonization trade-off. Am Nat 158:49–63
Acknowledgments
This paper is based on a talk presented at the 'Metacommunities' symposium of the Ecology Society of America annual meeting organized by M. Holyoak and R. Holt. I thank C. Osenberg for encouraging me to write this article, M. Leibold, J. Shurin, and especially T. Knight for providing many important insights and comments on the ideas presented herein, and S. Diehl, C. Osenberg, and an anonymous reviewer for careful reviews that helped me to clarify a number of issues. Funding was provided by the University of California-Davis, the University of Pittsburgh, and the National Science Foundation (DEB 97–01120, 01–08118, 02–41080).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chase, J.M. Community assembly: when should history matter?. Oecologia 136, 489–498 (2003). https://doi.org/10.1007/s00442-003-1311-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-003-1311-7