Abstract
Bisphenol A (BPA) and di(2-ethylhexyl) phthalate (DEHP) are endocrine-disrupting chemicals (EDCs) used in a wide variety of industrial products as plasticizers. Exposure to EDCs, particularly in mixtures, in prenatal and early postnatal periods may lead to unwanted effects and can cause both developmental and reproductive problems. In this study, we aimed to determine the individual and combined effects of prenatal and lactational exposure to BPA and/or DEHP on testicular histology, apoptosis, and autophagic proteins. Pregnant Sprague-Dawley rats (n = 3) were divided into four groups (control, BPA (50 mg/kg/day), DEHP (30 mg/kg/day), and BPA (50 mg/kg/day) + DEHP (30 mg/kg/day)) and dosed by oral gavage during pregnancy and lactation. The male offspring (n = 6) from each group were chosen randomly, and their testicular examinations were performed on the twelfth week. The results showed that fetal and neonatal exposure to BPA and DEHP could lead to significant testicular histopathological alterations and cause increases in apoptosis markers (as evidenced by increases in caspase 3 and caspase 8 levels; increased TUNEL-positive spermatogonia and TUNEL-positive testicular apoptotic cells) and autophagic proteins (as evidenced by increased LC3 and Beclin levels and decreased p62 levels) in testicular tissue. We can suggest that EDCs cause more dramatic changes in both testicular structure and cell death when there is combined exposure.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Endocrine-disrupting chemicals (EDCs) are exogenous substances. They can be found naturally or they can be man-made. These chemicals can affect the synthesis, secretion, transport, metabolism, elimination, receptor binding, and natural homeostasis of hormones, leading to reproductive and developmental problems (Environmental Protection Agency 2017).
Synthetic EDCs consist a wide range of chemical compounds, and they are mainly used as plasticizers, industrial solvents, lubricants, or pesticides. Some EDCs, like phthalates and bisphenols, are also used in tablet coatings (Diamanti-Kandarakis et al. 2009). These chemicals can highly be present in industrial areas where they can contaminate the soil and groundwater as mixtures. Their mixtures can accumulate in the food chain, and they can cause abundant human exposure (Diamanti-Kandarakis et al. 2009; Janesick et al. 2011). Occupational exposures in various industrial fields are of particular importance and can lead to very important consequences. Their use of cosmetic products, toys, coating materials, plastic food containers, bottles, baby products, blood transfusion and dialysis bags, medical supplies, and various household chemicals are other important sources of individual exposures to EDCs (Diamanti-Kandarakis et al. 2009). Moreover, natural EDCs are present in different edible plants, like soya, garlic, parsley, or apple. However, due to short half-lives of these compounds, they do not present any harm to human health unless they are consumed in high amounts (Dickerson and Gore, 2007).
Bisphenol A (BPA) and phthalates are widely known EDCs to which human exposure is inevitable. Phthalates are used to increase the elasticity and softness of plastic materials. Due to their widespread use, phthalates are produced at very high quantities (David 2000; Yaghjyan et al. 2016). They are not covalently bound to plastic material and can be released to the environment, leading to abundant exposures. Humans can be exposed to these compounds through oral, inhalation, and dermal routes (Koch and Calafat 2009).
Phthalates are suggested to have anti-androgenic potential (Erkekoglu et al. 2011). Their target organ within the human body is the testis, with Sertoli and Leydig cells being the most affected cell types (Erkekoğlu et al. 2010). They are suggested to be at least one of the underlying factors in many diseases, including gynecomastia, premature thelarche, precocious puberty, and thyroid disorders (Dong et al. 2019; Liu et al. 2018; Durmaz et al. 2018). Di(2-ethylhexyl) phthalate (DEHP) is the most widely used phthalate derivative, and it is suggested to be a reprotoxicant (Erkekoğlu et al. 2010; Rowdhwal and Chen 2018; Erkekoglu et al. 2014a, b). DEHP may lead to both apoptosis and autophagy in testis, particularly in germ cells (Barakat et al. 2017).
Bisphenol A is an estrogen-like EDC and one of the most produced chemical substances in the world today. Diet and water can be the main exposure routes (Careghini et al. 2015). On the other hand, BPA can also be taken to the body through the lungs and the skin (Vandenberg et al. 2007). Moreover, occupational exposure (automobile, electronic product, sports equipment, and CD and DVD coatings) consists of another exposure route to workers (Fleisch et al. 2010; Rubin 2011). BPA is also suggested to affect male reproductive system, leading to lower sperm counts and alterations in testicular histology (Cariati et al. 2019; Minguez-Alarcon et al. 2016). This bisphenol derivative is also suggested to cause different types of programmed cell death in testis tissue (Zhang et al. 2017; Jiang et al. 2018).
Exposure to EDCs in different periods of life can cause serious adverse effects, particularly on the developing reproductive system (Diamanti-Kandarakis et al. 2009). Several researches on EDCs have shown that their effects in critical periods of development, including prenatal life and early childhood, may be more pronounced and may lead to serious consequences. However, studies that examine the effects of prenatal and lactational exposures are extremely limited (Swanson et al. 2009; Ferguson et al. 2014; de Boo and Harding 2006). Prenatal combined exposure to DEHP and BPA (0.1, 1, and 10 mg/kg/day, both) caused a dose-dependent decrease in sex ratio (male/female) in mice at postnatal day (PND) 15. In addition, a dose-dependent decrease in testicular weight, sperm count, testicular anti-Mullerian hormone (AMH), follicle-stimulating hormone (FSH), progesterone, and testosterone. These unwanted effects stayed persistent even at PND 42 in the sexually mature animals (Xi et al. 2011a, b). DEHP exposure was also suggested to inhibit luteal function via dysregulation of platelet endothelial cell adhesion molecule (CD31) and prostaglandin F2α in pregnant mice (Guo et al. 2015).
There are several studies in literature that show the individual effects of phthalates or BPA on a developing male reproductive system. However, studies on combined exposures are limited, and they do not present a comprehensive evaluation. It is anticipated that the adverse effects after combined exposure may have exacerbated consequences. Concerning all the available data, the present study aimed to evaluate the effects of prenatal and lactational exposure to BPA and/or DEHP on prepubertal rat testis. Histopathologic and apoptotic changes in testicular tissue were investigated, and autophagy marker proteins were also evaluated.
Materials and methods
Chemicals and reagents
Di(2-ethylhexyl) phthalate and BPA were obtained from Sigma-Aldrich (St. Louis, Missouri, USA). Commercial caspase 3 and caspase 8 kits and “In Situ Cell Death Detection Kit” were purchased from MyBioSource (San Diego, CA) and Roche (Mannheim, Germany), respectively. Commercial ELISA kits for detection autophagic biomarkers p62, human microtubule-associated proteins 1A/1B light chain 3B (MAP1LC3B), and Beclin-1 were purchased from Enzo Biochem (New York, NY), MyBioSource (San Diego, CA), and Aviva Systems Biology (San Diego, CA), respectively.
Animals and dosing regimen
Di(2-ethylhexyl) phthalate was prepared in corn oil. BPA was first dissolved in 1 mL 96% ethanol (6.25 mg/mL) and then diluted with corn oil. The doses were selected as lowest observed adverse effect levels (LOAEL) on the male reproductive system for DEHP (30 mg/kg/day) and for BPA (50 mg/kg/day) (Agency for Toxic Substances and Disease Registry 2002; Shelby 2006; Tomza-Marciniak et al. 2018; European Food Safety Authority 2007).
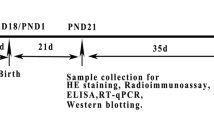
All animals were supplied from Gazi University Experimental Animals Laboratory (Ankara, Turkey). Pregnant Sprague-Dawley rats were divided randomly to four groups (n = 3/group): Controls received corn oil; BPA group received 50 mg/kg/day BPA; DEHP group received 30 mg/kg/day DEHP; and BPA + DEHP group received 50 mg/kg/day BPA and 30 mg/kg/day DEHP through 6–21 gestational days and lactation period by intra-gastric lavage. Male offspring (n = 6/group) from each mother were fed until the end of the twelfth postnatal week. They were then weighed and euthanized (cardiac exsanguination under deep anesthesia) and the testes were removed. The testes were weighed and relative testis weights were later calculated. The animals were treated humanely and with regard for alleviation of suffering, and the study was approved by Gazi University Ethical Committee (G.U.ET-15.066).
Body and organ weights
On the first day of birth, both female and male offspring were weighed. At the end of the lactation period, the male offspring were randomly selected, and the study groups were formed. The male pups were weighed again.
Histopathology and electron microscopy
The left testis was divided into two pieces. One piece was used for histopathological evaluation, and the other piece was used for electron microscopy (EM). The first piece was fixed in Bouin’s fixation solution for 18 h, and tissue samples were taken on a Leica TP 1020 light microscope tissue processor (Wetzlar, Germany). In order to provide dehydration, samples were passed through graded alcohols on the tissue processor. All specimens were embedded in paraffin, and 5-μm sections were cut from the obtained paraffin blocks. These sections were left overnight at 60 °C and were then deparaffinized with xylene. The dehydration step was repeated. Sections were stained with hematoxylin and eosin. The sections were examined under a Leica DM6000B microscope (Wetzlar, Germany) and images were taken by a Leica DC490 digital camera (Wetzlar, Germany) (Saadeldin et al. 2011).
The second piece of the fresh tissue sample was fixed in 2.5% glutaraldehyde solution in phosphate buffer, (pH 7.4, 4 h) and was post-fixed in 1% osmium tetroxide solution (in 0.1 M phosphate buffer) for 1 h. Later, tissue samples were washed with phosphate buffer and samples were dehydrated in a graded series of alcohols. Samples were treated with propylene oxide and embedded in Araldite/Epon812, and heat polymerization was performed. Sections were cut using a microtome (Leica Ultracut R); semi-thin sections were stained with methylene blue–azure II and examined using an electron microscope (Leica DM6000B, Wetzlar, Germany) with a Leica DC490 digital camera. Ultrathin sections were double-stained with uranyl acetate and lead citrate (Leica EM AC20).
TUNEL assay
Terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) assay has been designed to detect apoptotic cells that undergo extensive DNA degradation during the late stages of apoptosis. The method is based on the ability of terminal deoxynucleotidyl transferase (TdT) to label blunt ends of double-stranded DNA breaks independent of a template. Apoptotic cells in testis tissues were identified by a TUNEL assay using In Situ Cell Death Detection Kit-POD according to manufacturers’ instructions. Briefly, paraffin sections of testis (5 μm in thickness) were deparaffinized, rehydrated, and washed twice in PBS for 5 min. Then the slides were incubated with 3% H2O2 for 10 min in order to block endogen peroxidase activity. After washing in PBS three times for 5 min, the slides were incubated with permeabilization solution (0.1% Triton X-100 in 0.1% sodium citrate) for 8 min at 4 °C and later washed twice with PBS for 5 min. The labeling reaction was performed by using 50 μl TUNEL reagent for each sample, except the negative control, in which reagent without enzyme was added and incubated for 1 h at 37 °C. Following PBS washings, the slides were incubated with converter reagent for 30 min at 37 °C. After washing, the slides were incubated with 3,3′-diaminobenzidine (DAB) solution for 10 min for labeling of cells containing DNA strand breaks. In tissue samples, TUNEL-positive stained cells were counted in 20 seminiferous tubules with light microscope (DM6000B, Wetzlar, Germany) at × 400 magnification, and the average number of apoptotic spermatogenic cells was determined for the study groups.
Testis tissue preparation for the determination of apoptotic and autophagic proteins
The right testes were removed and washed in saline and deionized water. The testes were frozen immediately in liquid nitrogen and kept at − 80 °C. For the determination of apoptotic and autophagic proteins, testes were homogenized in a homogenization buffer (10 mM Tris/1 mM DTPA/1 mM PMSF) by using a Teflon-glass homogenizer. The homogenates were then centrifuged at 2000×g at 4 °C for 10 min. The supernatants were used in the determination of apoptotic and autophagic proteins.
Caspase 3 and caspase 8 levels
Both caspase 3 and caspase 8 levels were measured by commercial kits using sandwich ELISA method. The well plates within the kits were pre-coated with either caspase 3 or caspase 8 antibodies. The biotin conjugated anti-CASP3 or anti-CASP8 antibodies were used as “detection antibodies.” The standards or test samples were added to wells. Plates were incubated at 37 °C for 90 min, later discarded, and washed, and biotin-conjugated detection antibodies were added. The plates were incubated at 37 °C for 60 min and washed, and HRP-streptavidin conjugate (SABC) was later added. The plates were again incubated at 37°C for 30 min and washed. 3′,5,5′-Tetramethylbenzidine (TMB) substrate was added, and plates were incubated in the dark at 37°C for 30 min for color development. Later, stop solution was added, and the absorbance was read at 450 nm. Both of the kits had high sensitivity and specificity to caspase 3 or caspase 8.
Autophagy biomarkers
Testicular p62, human microtubule-associated proteins 1A/1B light chain 3B (MAP1LC3B), and Beclin-1 levels were measured as autophagy biomarkers. These biomarkers were measured with commercial ELISA kits based on standard sandwich ELISA method.
Statistical analysis
The results were expressed as mean ± standard deviation (SD). The differences among the groups were evaluated with Kruskal–Wallis one-way analysis of variance, followed by Mann–Whitney U test using a Statistical Package for Social Sciences Program (SPSS) version 17.0. p values < 0.05 were considered statistically significant.
Results
Body weights
The average body weights of the pups on the first day of birth are given in Table 1. The birth weights of the pups in the DEHP group were significantly higher than that of the control (8%) and BPA (10%) groups. In addition, the birth weights of the BPA + DEHP group were found to be higher than that of the control (12%) and DEHP (4%) groups (p < 0.05, both).
At the end of the lactation period, the mean body weight of male pups was significantly higher in DEHP (13%) and BPA + DEHP (6%) groups compared with that of the control group. The mean body weights of male offspring in BPA (19%) group were significantly decreased vs. that of the control (Table 1).
Testis and epididymis weights
The mean testis and epididymis, and relative testis and epididymis weights of the study groups were given in Table 2. There were no significant differences in testis and epididymis weights between the study groups and control; however, the DEHP group had significantly higher testis weights vs. the BPA group.
Relative testis weights of all the study groups were not different than that of the control; however, the DEHP group had significantly higher relative testis weights vs. the BPA group. On the other hand, relative epididymis weights of all the study groups were higher than that of the control (p < 0.05, all; 5% in BPA, 18% in DEHP, and 23% in BPA + DEHP).
Histopathology
In the control group, seminiferous tubule sections and interstitial tissue between seminiferous tubules were in normal morphological organization within the testis sections surrounded by a connective tissue called “tunica albuginea” externally. The seminiferous tubules containing myoid cells from the outside were wrapped with a fine connective tissue called “tunica propria.” There were not any degenerations in the spermatogenetic cells and Sertoli cells. No abnormalities were observed in spermatogenesis and spermiogenesis (Fig. 1 a and b).
Representative histological sections of the testis of rats (H&E). a Light micrograph of the seminiferous tubules from the control group. Leydig cells (black arrow) between seminiferous tubules, Sertoli cells (white arrow), and the cells of spermatogenetic series (primary spermatocytes (arrow head), early spermatid (blue arrow), and late spermatid (yellow arrow)) in the seminiferous tubule wall are seen with normal histological appearance (Hematoxylin-Eosin × 200). b Light micrograph of seminiferous tubule from the control group. Dashed line separates the spermatogenic cells at different stages. Late spermatids with elongated and condensed nucleus (black arrow) and primary spermatocytes (white arrow) are observed in the process of spermatogenesis in the seminiferous tubule wall, and tails of the spermium (asterisk) are seen in the lumen at higher magnification. (Hematoxylin-Eosin × 400). c Light micrograph of seminiferous tubules from the BPA group. Leydig cells (black arrow) among the seminiferous tubules are observed in normal histological appearance. The spermatogenic cells (white arrow) are separated from the basal lamina in some of the areas of the seminiferous tubule wall (Hematoxylin-Eosin × 200). d Light micrograph of seminiferous tubule from the BPA group. Sertoli cells (white arrow) are seen on the basal lamina in the wall of the seminiferous tubule. Occasionally, the loss of spermatogonium (black arrow) and empty spaces (arrowhead) are observed near these cells at higher magnification. (Hematoxylin-Eosin × 400). e Light micrograph of seminiferous tubules from the DEHP group. In interstitial area Leydig cells (arrow), flat nucleated fibroblasts, and collagen fibers are seen in the DEHP-exposed group. Separation was observed between the cells in the wall of the seminiferous tubule (Hematoxylin-Eosin × 200). f Light micrograph of seminiferous tubule from the DEHP group. Separation (arrowhead) is seen between primary spermatocytes and also between primary spermatocytes and advanced spermatogenetic serial cells. Cell debris (arrow) is observed in the lumen of the seminiferous tubule in the same group (Hematoxylin-Eosin × 200). g Light micrograph of seminiferous tubules from the BPA + DEHP group. Large gaps (white arrow) were observed between the spermatogenetic cells in the cross sections of the samples belonging to the group exposed BPA + DEHP. Leydig cells (black arrow) are seen in the interstitial area (Hematoxylin-Eosin × 200). h Light micrograph of seminiferous tubule from the BPA + DEHP group. Gaps between the primer spermatocytes and the basal lamina and neighboring cells (white arrow) were observed at higher magnification. The loss of cells in the advanced stage of differentiation in the seminiferous tubule epithelium (black arrow) was observed (Hematoxylin-Eosin × 400)
In BPA group, the integrity of the epithelium was impaired in some seminiferous tubules. Primer spermatocytes were separated from the basal lamina in some regions (Fig. 1 c). Spermatogonia (located immediately adjacent to Sertoli cells in the basal lamina layer of the seminiferous tubules) were not observed in some areas of the tubules (Fig.1 d).
Significant segregations were observed in the samples of the DEHP group indicating that the intercellular connections between the Sertoli cells and the spermatogonia were impaired on the basis of seminiferous tubules. Separations were also observed among the cells of the spermatogenetic series that were differentiating on the walls of the seminiferous tubules. In the lumen of some seminiferous tubules, round-shaped spermatogenic cells were spilled, and cell debris was observed in the lumen (Fig. 1e).
In BPA + DEHP group, testis tissues maintained their presence in the interstitial tissue between the seminiferous tubules by preserving the normal histological structure of the Leydig cells. In this group, large gaps were observed between the cells of the spermatogenetic series in the affected seminiferous tubules (Fig. 1 f). In addition, we observed that the loss of the cells in the advanced differentiation stages caused a decrease in the thickness of epithelia (Fig. 1 g). Spermatogenesis and spermiogenesis were both impaired (Fig. 1 h).
Electron microscopy
Sertoli cells with indented nucleus lying over the basement membrane were observed in the control group. Spermatogonium and spermatocytes belonging to different stages of maturation, such as primary spermatocytes and spermatids, were observed in the neighboring region of Sertoli cells (Fig. 2 a).
Transmission electron micrographs of seminiferous tubules of experimental groups. a Sertoli cells with indented nucleus and different stages of spermatogonial cells in the surrounding in control. b Degenerated cells (arrow) in BPA group. c Lipid droplets, lysosomes, and vacuoles (arrow) in the cytoplasm of Sertoli cells and two primary spermatocyte connected by cytoplasmic bridges in DEHP group. d Degeneration in spermatogenesis with pyknotic spermatogenetic cells (arrow) in BPA + DEHP; S: Sertoli cell, Sp: spermatogonium, 1 sp: primary spermatocyte, Spd: spermatid, Scale bar: 2 μm
In BPA group, spermatogenesis was disrupted, and degenerated cells with condensed nuclei were observed instead of spermatogonium and spermatocytes between the Sertoli cells in the basal and luminal compartment of affected seminiferous tubules. The cytoplasmic remnants of spermatogenetic cells were observed in the wall of seminiferous tubules (Fig. 2 b).
Spermatogenetic cells and Sertoli cells were present in the DEHP group. Lipid droplets, lysosomes, and vacuoles were observed in the cytoplasm of Sertoli cells in the DEHP group (Fig. 2 c).
Spermatogenesis was more affected in the BPA + DEHP group when compared with that of the BPA or DEHP groups. Degeneration in spermatogenesis with pyknosis and chromatin condensation in the spermatogenetic cells was observed, and the nuclei of spermatogenetic cells were faded and could not be distinguished in this group (Fig. 2 d).
TUNEL assay
The number of apoptotic spermatogenic cells (Table 3) was increased statistically in BPA, DEHP, and BPA + DEHP treated groups compared to that of the control (p < 0.05). The mean number of apoptotic cells was the highest in the BPA + DEHP group and was statistically higher than both DEHP and BPA groups (p < 0.05). The images of the seminiferous tubule sections are presented in Fig. 3 a, b, c, and d. Increased TUNEL-positive spermatogonia and TUNEL-positive apoptotic cells were detected in all study groups compared with that of the control group.
The representative images showing the TUNEL labeling of the seminiferous tubule sections of experimental groups. a TUNEL-positive apoptotic cells were observed in brown (arrow) in the control group. (× 400). b The number of TUNEL-positive spermatogonium (arrow) was more in BPA than that of the control group (× 400). c An increased number of TUNEL-positive apoptotic spermatogonial cells were observed in DEHP compared with that of the control group (× 400). d Intensive TUNEL-positive apoptotic cells were observed in the BPA + DEHP group. (× 400)
Caspase 3 and caspase 8 levels
There were no statistically significant differences between testicular caspase 3 levels (Fig. 4 a) due to high standard deviation within the groups (p < 0.05). However, caspase 3 levels of BPA (29.5%), DEHP (23.71%), and BPA + DEHP groups (56.5%) were found to be higher than that of the control. In DEHP (15.9%) and BPA + DEHP (12.8%) groups, caspase-8 levels (Fig. 4 b) were found significantly higher than that of the control group (p < 0.05).
Autophagy biomarkers
LC3 levels (Fig. 5 a) of the testicular tissue were found to be increased with BPA (63%, p > 0.05), DEHP (81%, p < 0.05), and BPA + DEHP (72% increase, p < 0.05) groups vs. that of the control. Testis Beclin levels (Fig. 5 b) increased in BPA (10%, p > 0.05), DEHP (61%, p < 0.05), and BPA + DEHP (40%, p < 0.05) groups when compared with that of the control. Testis p62 levels (Fig. 5 c) were decreased in BPA (34%, p > 0.05), DEHP (46%, p > 0.05), and BPA + DEHP groups (91%, p < 0.05) were lower than that of the control group. The insignificant changes in testis p62 levels in BPA and DEHP groups were due to the high variation within these study groups.
Discussion
Several studies have shown that the most significant toxic effects of phthalates are on the male reproductive system and normal developmental processes. Phthalates were suggested to cause decreases in testosterone levels and anogenital distance. Moreover, other problems like testicular anomalies, undescended testis, and testicular atrophy can arise from exposure to DEHP at early stages of life (Gray et al. 2006; Talsness et al. 2009; Wei et al. 2012). Studies on the effects of prenatal and postnatal DEHP exposure showed that this particular phthalate derivative can lead to male reproductive system disorders, intrauterine deaths, and testicular atrophy (Carbone et al. 2010; Saillenfait et al. 2011). Exposure to DEHP 0, 0.05, 5, and 500 mg/kg/day (in diet, from gestational day 1 until the end of lactation) caused long-term pituitary-gonadal axis disruption in male and female mice offspring. DEHP caused a dose-dependent decrease in blastocyst rate and cleavage rate of males and decreased both sperm count and live sperm in the viable offspring along with dose-dependent decreases in CYP19A1 protein levels (Pocar et al. 2012). Gestational exposure to BPA, on the other hand, is suggested to cause perturbed reproduction. However, a clear consensus is not yet reached whether prenatal BPA exposure in humans results in direct adverse effects on male genital development and reproduction or not (Williams et al. 2014). Although there are many regulations in order to reduce human exposure and environmental contamination of both phthalates and bisphenols, humans are still highly exposed to these chemicals starting from early stages of life (Barrios-Estrada et al. 2018; Bilal et al. 2019). Morever, several antioxidants have been investigated in order to mitigate the toxic effects of these EDCs in different systems, and some of these substances were found to be protective against their toxicity (Erkekoglu et al. 2010, 2014a, b; Mohammed et al. 2020).
In vitro, in vivo, and human studies have shown that BPA may also have a wide variety of adverse effects on human health. The major effects are suggested to be reproductive and developmental problems, due to estrogenic effects of BPA. In addition, BPA was proposed to cause metabolic diseases (diabetes and obesity), endocrine disorders (premature thelarche, puberty precox), thyroid hormone disorders, and oxidative stress in different organs. BPA may also have teratogenic effects like low birth weight, brain damage, and behavior disorders, and BPA exposure may be one of the underlying causes of autism (Rochester 2013; Muhamad et al. 2016; Guida et al. 2015; Stein et al. 2015; Chitra et al. 2003; Bindhumol et al. 2003; Kabuto et al. 2004; Meeker et al. 2010; Melzer et al. 2011; Lassen et al. 2014; Gramec Skledar and Peterlin Masic 2016; Usman and Ahmad 2016).
In the histopathological studies, the integrity of the epithelium was impaired in some seminiferous tubules, and the primary spermatocytes were separated from basal lamina in some regions in BPA-exposed animals. Spermatogonia, which are immediately adjacent to Sertoli cells in the normal structure basal compartment of seminiferous tubules, were not found in some areas of the seminiferous tubules in BPA group. Several studies have shown that prenatal BPA exposure can cause various disorders in the rodent testis during adulthood. These conditions were suggested to be impaired spermatogenesis, the presence of immature spermatogenic cells in the testis tubule lumen, the presence of large gaps between Sertoli cells, and tubular lumen expansions (Kass et al. 2015; Ma et al. 2017). These results are consistent with our histopathology results due to prenatal BPA exposure. In another study conducted in newborn male rats, BPA was administered subcutaneously (0.01, 0.1, and 5 mg/kg bw), and spermatogenesis deteriorated due to BPA exposure (Xie et al. 2016). On the contrary, there are also studies that propose that maternal BPA exposure does not cause histopathologic changes in the testicular tissue of the offspring (Xi et al. 2011a, b; LaRocca et al. 2011).
In the DEHP group, there were clear separations between the Sertoli cells and the spermatogonia, indicating that the intercellular connections were disrupted in the basal lamina of the seminiferous tubules. Separations were also observed among the cells of the spermatogenic series that were differentiating on the wall of the seminiferous tubules. In the lumen of some seminiferous tubules, round-shaped spermatogenic cells of the early phase of spermium differentiation were spilled, and cell debris was observed in the lumen. Akingbemi et al. (2001) suggested that the effects of DEHP on Leydig cells were reversible even if rats were exposed to this particular phthalate for a long period of time. There are also several studies showing that maternal DEHP exposure causes histopathologic changes in testis tissue and deterioration in spermatogenesis and that these changes can be transmitted to later generations (Borch et al. 2006; Christiansen et al. 2010; Li et al. 2013; Andrade et al. 2006; Jones et al. 2015; Doyle et al. 2013). In a previously study, Erkekoglu et al. (2011) aimed to detect toxic effects of DEHP on rat male reproductive system. Adult male Sprague-Dawley rats were separated into 6 groups. DEHP-treated groups received 1 g DEHP/kg dose by gavage during the last 10 days of the feeding period. Reproductive toxicity and anti-androgenic effects of DEHP were evidenced by disturbed testicular histology and spermatogenesis.
In the combined exposure group, we observed that the normal histological structure of Leydig cells in interstitial tissue between seminiferous tubules was preserved. However, large vacuoles were present between the cells of the spermatogenic series in the affected seminiferous tubules. In addition, the thickness of the epithelium decreased with the loss of advanced cells of the differentiation in seminiferous tubule epithelium along with impairments in spermatogenesis and spermiogenesis. Therefore, due to histopathologic changes in seminiferous tubules and Sertoli cells, we may suggest that the number and morphology of the sperms might decrease. There is one study in literature that suggests DEHP or dibutyl phthalate (DBP) exposures or DEHP plus DBP exposures may lead to histopathological alterations in rat testis. However, there is not any study in literature that evaluates the testicular effects of prenatal BPA plus DEHP exposure (Howdeshell et al. 2007; Manikkam et al. 2013). We can also propose that early-life combined exposure to BPA and DEHP might impair male infertility.
Our electron microscopy results showed that both BPA and DEHP exposure caused deterioration in testicular tissue. Spermatogenesis was affected within all of the study groups, particularly in the combined exposure group. In a recent study by Qin et al. (2018), 5-week-old Sprague-Dawley rats were exposed to different doses (0 mg/kg, 100 mg/kg, 500 mg/kg, and 1500 mg/kg) of DEHP for 6 weeks. In the high dose (1500 mg/kg) group, DEHP induced the formation of vacuoles in the Sertoli cells and abnormal proliferation of Leydig cells. Moreover, DEHP also caused the impairment of different stages of spermatogenic cells in seminiferous tubules. In another study, pregnant rats were exposed to DEHP (125, 250, and 500 mg/kg bw/day) orally between gestational days 7 and 18. The electron microscopy results revealed that histopathological changes were observed in fetal testis at high doses but not in the lowest dose. However, the postnatal observations revealed that all of the study groups showed testicular abnormalities after 5 and 10 weeks following birth (Shirota et al. 2005).
In a study by Vijaykumar et al. (2017), the researchers evaluated the histopathological effects of oral BPA administration at different doses (2.5, 12.5, and 25 μg/kg bw/day, for 70 days) on testicular tissue in 16 adult male monkeys. Histopathological evaluation and transmission electron microscopy results showed that sloughing of germ cells into the lumen was present along with the increases in interstitial space and vacuolation of Sertoli cell cytoplasm. Moreover, several degenerative effects on the basement membrane, Sertoli cells, Leydig cells, and other developing germ cells were observed in the 12.5 and 25 μg/kg bw/day groups when compared with that of the control.
Apoptosis is a type of programmed cell death in which a hemostatic mechanism works for the maintenance of healthy cell populations during different stages of life. All of the exposure groups (BPA, DEHP, and BPA plus DEHP) showed a statistically significant increase in apoptotic spermatogenic cell count compared with that of the control (p < 0.05). The mean number of apoptotic cells was the highest in the BPA plus DEHP-exposed group, and the number of apoptotic cells in the combined exposure group was significantly higher than both DEHP- and BPA-exposed groups. There are studies in literature that suggest exposure to BPA in prenatal period caused increases in apoptosis in testicular tissue (the doses and treatment periods of these studies were given above). However, some studies suggest the contrary (Ma et al. 2017; LaRocca et al. 2011). Manikkam et al. (2013) found that maternal exposure (gestational day 8–14) to high-dose mixture of BPA (50 mg/kg bw/day), DEHP (750 mg/kg bw/day), and DBP (66 mg/kg bw/day) and lower-dose mixture (one-half dose as plastics group) caused significant increases in the number of apoptotic spermatogenic cells in the F3 generation; however, apoptotic cell number decreased in F1 generation. In another study, BPA administration to adult Sprague-Dawley rats resulted in an increase in the number of apoptotic cells in the testicular tissue vs. control at doses of 100 and 200 mg/kg bw/day for 4 weeks, while no significant change was observed in the dose of 50 mg /kg bw/day (Wang et al. 2014). In another study, BPA was administered subcutaneously (0.01, 0.1, and 5 mg/kg) to neonatal male rats on postnatal day (PND) 1 to 21. Significant increases in germ cell apoptosis was observed compared the control (Xie et al. 2016). Moreover, BPA exposure has also been shown to induce apoptosis in primary culture of Sertoli cells which were isolated from 18- to 22-day-old Sprague-Dawley rats at low doses (50, 70 μM) in vitro (Qi et al. 2014). In a study by Murphy et al. (2014), MEHP was administered to young rats (PND 21, 28, and 35) orally. Researchers found that exposure to MEHP triggered apoptosis in germ cells. Therefore, we can suggest that the effects of plasticizers on testicular and germ cell apoptosis can be dose-dependent, and application period can be a major factor in the induction of testicular cell death.
Caspases are proteolytic enzymes that play essential roles in both intrinsic (caspase 9 and caspase 3) and extrinsic (caspase 8) pathways of apoptosis. There are different types of caspases with different tasks. These enzymes can be divided into 3 groups as initiators (caspase 2, 8, 9, and 10), effectors or executioners (caspase 3, 6, and 7), and inflammatory caspases (caspase 1, 4, and 5) (Gordeziani et al. 2017). In our study, there were no statistically significant differences in testicular caspase 3 levels between the groups. However, caspase 3 levels of BPA + DEHP group were found to be 56.5% higher than that of the control but still not markedly different. This can be due to high standard deviation and low number of animals within the study groups. On the other hand, testicular caspase 8 levels in DEHP and BPA + DEHP group were found to be significantly higher than that of the control group (p < 0.05), indicating that DEHP and combined exposures trigger the extrinsic apoptotic pathway. In a study by Borch et al. (2005), pregnant rats were orally exposed to DEHP (300, 750 mg/kg/day) or DEHP (750 mg/kg/day) in combination with di(2-ethylhexyl) adipate (DEHA, 400 mg/kg/day). Their male offspring also received the same treatments during lactation period between the 21st and 26th PND. Testicular caspase 3 levels showed no significant changes vs. control. In another study, oral single-dose (400 mg/kg body weight) of MEHP was administered to 28-day-old male Wistar rats. After 3 and 12 h, the activity of testicular caspase 3 increased significantly (Dalgaard et al. 2001). In a study performed on adult male Sprague-Dawley rats, BPA (50, 100, and 200 mg/kg) was administered orally for 4 weeks. There were increases in both caspase 3 and 9 levels in testis compared with that of control (Wang et al. 2014). Liu et al. (2013) conducted a study on ICR rats which were exposed to BPA in water (10, 100, and 1000 nmol/L) during pregnancy and lactation. Caspase 3 expressions in testes were found to be increased in male pups at 21st PND. These studies showed that the effects of BPA and DEHP exposure on caspases can be controversial, most possibly resulting from the differences between the doses, exposure periods, and animal models.
Autophagy is another type of programmed cell death. Targeted cytoplasmic constituents as well as long-lasting proteins, damaged organelles, microorganisms, and viruses are isolated from the rest of the cell within a double-membraned vesicle which is called “autophagosome”. Eventually, the autophagosome fuses with lysosomes, and later, these contents are degraded by lysosomal enzymes and recycled. Autophagic cell death occurs in four steps: triggering, vesicle nucleation, vesicle elongation, and fusion-degradation phase. It can be activated by different conditions, such as food starvation and high levels of intracellular oxidative stress (Yu et al. 2018). Numerous proteins play roles in the regulation of autophagy. Beclin 1 takes part on the vesicle nucleation phase of autophagy by forming the isolation membrane, a double-membrane structure that engulfs cytoplasmic material to form the autophagosome (Yu et al. 2018). p62 is an autophagy substrate that is used as a reporter of autophagic activity. The intracellular levels of p62 are dependent on transcriptional regulation and posttranslational autophagic degradation. Autophagy activation reduces the expression of p62. Collectively, p62 relates to the formation of ubiquitin-positive inclusions and binds LC3II to facilitate autophagic degradation (Liu et al. 2016). LC3 is a protein having an important role in the vesicle elongation phase. During autophagy, the autophagosomes involve cytoplasmic components (cytosolic proteins and organelles). Simultaneously, the cytosolic form of LC3 (LC3-I) is combined with phosphatidylethanolamine to generate the LC3-phosphatidylethanolamine conjugate (LC3-II). LC3-II is bound to the autophagosome membrane and takes part in phagophore expansion as well as cargo recognition (Tanida et al. 2008). In our study, we observed that testicular LC3 levels of BPA group (63% increase, p > 0.05), DEHP group (81% increase, p < 0.05), and BPA + DEHP group (72% increase, p < 0.05) were higher than that of the control group. In addition, Beclin 1 levels were found to be increased with BPA (10%, p > 0.05), DEHP (61%, p < 0.05), and BPA plus DEHP (40% increase, p < 0.05) exposure. Testis p62 levels were decreased in BPA plus DEHP group (91%) vs. control and vs. significantly lower than that of the control group and single exposure groups (p < 0.05). According to our results, prenatal and lactational exposures to BPA, DEHP, and BPA + DEHP can induce autophagy at the LOAEL doses of these EDCs. In a recent work, prepubertal Sprague-Dawley rats were exposed to different low doses of BPA and nanophenol separately or jointly for 4 weeks. The results indicate that the joint exposure induced excessive apoptosis and autophagy in the testes, as proved by a series of characteristics such as chromatin condensation and autophagosome formation (Su et al. 2018). Su et al. (2018) demonstrated that exposure to BPA (100, 200, 300, 400, 500, and 600 μM) decreased the cell viability in goat Sertoli Cells (gSCs) in a dose-dependent manner, and all results indicated that the viability of gSCs was disrupted after BPA treatment. The authors suggested that BPA induced both apoptosis and autophagy. The possible suggested mechanisms for the loss in cell viability were suggested to be high levels of intracellular ROS production and alterations in mitochondrial membrane potential (Zhang et al. 2017). In another study by Quan et al. (2017), male Sprague-Dawley rats (5 weeks) were exposed to different concentrations (0, 2, 10, and 50 mg kg/bw) of BPA for 10 days by intraperitoneal injection. The researchers found that BPA exposure led to both apoptosis and autophagy in rat testis. Male mice were exposed with different doses of DEHP (0, 100, 200, or 400 mg/kg/day) orally for 21 days in a study by Sun et al. (2018). They found that DEHP significantly induced autophagy in the testicular tissues and led to increases in LC3-II, Atg5, and Beclin-1 proteins. Another recent study by Yi et al. (2018) showed that developmental exposure to DEHP (0, 250 and 500 mg/kg/day, i.g., from postnatal day 1 to 35) simultaneously increased the number of autophagosomes and the protein expressions of autophagy markers LC3 and p62 in rat testes, suggesting that the accumulated autophagosomes raised from impaired the degradation process in autophagy. The results of these studies support our findings. However, there is no study in literature evaluating the effects of combined exposure to BPA and DEHP on autophagy.
Conclusion
The results of this study revealed that early-life exposure to EDCs may cause adverse effects on male reproductive system in the later stages of life. These adverse effects are suggested to be testicular histopathological changes, apoptosis, and autophagy. Although there are studies in literature that suggest the reprotoxic effects of BPA and/or DEHP, this study further contributes to literature by comparing the prenatal and postnatal cytotoxic, reprotoxic, and developmental effects of BPA + DEHP to individual exposures to BPA and DEHP. Further studies are needed to examine the combined impact of different plasticizers on the male reproductive system in order to elucidate the underlying mechanisms. As inter-species changes make it difficult to understand the effects of these chemicals on humans, more studies on different species as well as in vitro research are needed in order to understand the outcomes of combined exposures.
References
Agency for Toxic Substances and Disease Registry (ATSDR) (2002) Toxicological profile for di(2-ethylhexyl) phthalate (DEHP). https://www.atsdr.cdc.gov/toxprofiles/tp.asp?id=684&tid=65. Accessed 5 June 2019
Akingbemi BT, Youker RT, Sottas CM, Ge R, Katz E, Klinefelter GR, Zirkin BR, Hardy MP (2001) Modulation of rat Leydig cell steroidogenic function by di(2 ethylhexyl)phthalate. Biol Reprod 65:1252–1259. https://doi.org/10.1095/biolreprod65.4.1252
Andrade AJ, Grande SW, Talsness CE, Grote K, Golombiewski A, Sterner-Kock A, Chahoud I (2006) A dose-response study following in utero and lactational exposure to di-(2 ethylhexyl) phthalate (DEHP): effects on androgenic status, developmental landmarks and testicular histology in male offspring rats. Toxicology 225:64–74
Barakat R, Lin PP, Rattan S, Brehm E, Canisso IF, Abosalum ME, Flaws JA, Hess R, Ko C (2017) Prenatal exposure to DEHP induces premature reproductive senescence in male mice. Toxicol Sci 156:96–108
Barrios-Estrada C, Rostro-Alanis MJ, Parra AL, Belleville MP, Sanchez-Marcano J, Iqbal HMN, Parra-Saldívar R (2018) Potentialities of active membranes with immobilized laccase for bisphenol A degradation. Int J Biol Macromol 108:837–844
Bilal M, Iqbal HMN, Barceló D (2019) Mitigation of bisphenol A using an array of laccase-based robust bio catalytic cues - a review. Sci Total Environ 689:160–177
Bindhumol V, Chitra KC, Mathur PP (2003) Bisphenol A induces reactive oxygen species generation in the liver of male rats. Toxicology 188:117–124
Borch J, Dalgaard M, Ladefoged O (2005) Early testicular effects in rats perinatally exposed to DEHP in combination with DEHA-apoptosis assessment and immunohistochemical studies. Reprod Toxicol 19:517–525
Borch J, Metzdorff SB, Vinggaard AM, Brokken L, Dalgaard M (2006) Mechanisms underlying the anti-androgenic effects of diethylhexyl phthalate in fetal rat testis. Toxicology 223:144–155
Carbone S, Szwarcfarb B, Ponzo O, Reynoso R, Cardoso N, Deguiz L, Moguilevsky JA, Scacchi P (2010) Impact of gestational and lactational phthalate exposure on hypothalamic content of amino acid neurotransmitters and FSH secretion in peripubertal male rats. Neurotoxicology 31:747–751
Careghini A, Mastorgio AF, Saponaro S, Sezenna E (2015) Bisphenol A, nonylphenols, benzophenones, and benzotriazoles in soils, groundwater, surface water, sediments, and food: a review. Environ Sci Pollut Res Int 22:5711–5741
Cariati F, Nadja D, Francesca B, Stefania I, Giacomo G, Rossella T (2019) Bisphenol a: an emerging threat to male fertility. Reprod Biol Endocrinol 17:6. https://doi.org/10.1186/s12958-018-0447-6
Chitra KC, Latchoumycandane C, Mathur PP (2003) Induction of oxidative stress by bisphenol A in the epididymal sperm of rats. Toxicology 185:119–127
Christiansen S, Boberg J, Axelstad M, Dalgaard M, Vinggaard AM, Metzdorff SB, Hass U (2010) Low-dose perinatal exposure to di(2-ethylhexyl) phthalate induces anti-androgenic effects in male rats. Reprod Toxicol 30:313–321
Dalgaard M, Nellemann C, Lam HR, Sørensen IK, Ladefoged O (2001) The acute effects of mono(2-ethylhexyl) phthalate (MEHP) on testes of prepubertal Wistar rats. Toxicol Lett 122:69–79
David RM (2000) Exposure to phthalate esters. Environ Health Perspect 108:A440
de Boo HA, Harding JE (2006) The developmental origins of adult disease (Barker) hypothesis. Aust N Z J Obstet Gynaecol 46:4–14
Diamanti-Kandarakis E, Bourguignon JP, Giudice LC, Hauser R, Prins GS, Soto AM, Zoeller RT, Gore AC (2009) Endocrine-disrupting chemicals: an Endocrine Society scientific statement. Endocr Rev 30:293–342
Dickerson SM, Gore AC (2007) Estrogenic environmental endocrine-disrupting chemical effects on reproductive neuroendocrine function and dysfunction across the life cycle. Rev Endocr Metab Disord 8:143–159
Dong J, Cong Z, You M, Fu Y, Wang Y, Wang Y, Fu H, Wei L, Chen J (2019) Effects of perinatal di (2-ethylhexyl) phthalate exposure on thyroid function in rat offspring. Environ Toxicol Pharmacol 67:53–60
Doyle TJ, Bowman JL, Windell VL, McLean DJ, Kim KH (2013) Transgenerational effects of di-(2-ethylhexyl) phthalate on testicular germ cell associations and spermatogonial stem cells in mice. Biol Reprod 88:112–115
Durmaz E, Erkekoglu P, Asci A, Akçurin S, Bircan İ, Kocer-Gumusel B (2018) Urinary phthalate metabolite concentrations in girls with premature thelarche. Environ Toxicol Pharmacol 59:172–181
Environmental Protection Agency (2017) What is endocrine disruption? https://www.epa.gov/endocrine-disruption/what-endocrine-disruption. Accessed 5 Mar 2019
Erkekoğlu P, Rachidi W, De Rosa V, Giray B, Favier A, Hincal F (2010) Protective effect of selenium supplementation on the genotoxicity of di(2-ethylhexyl) phthalate and mono(2 ethylhexyl) phthalate treatment in LNCaP cells. Free Radic Biol Med 49:559–566
Erkekoglu P, Zeybek ND, Giray B, Asan E, Arnaud J, Hincal F (2011) Reproductive toxicity of di(2-ethylhexyl) phthalate in selenium supplemented and selenium-deficient rats. Drug Chem Toxicol 34:379–389
Erkekoglu P, Giray B, Rachidi W, Hininger-Favier I, Roussel AM, Favier A, Hincal F (2014a) Effects of di(2-ethylhexyl) phthalate on testicular oxidant/antioxidant status in selenium-deficient and selenium-supplemented rats. Environ Toxicol 29(98):107
Erkekoglu P, Zeybek ND, Koçer-Giray B, Rachidi W, Kızılgün M, Hininger-Favier I, Favier A, Asan E, Hincal F (2014b) The effects of di(2-ethylhexyl) phthalate on rat liver in relation to selenium status. Int J Exp Pathol 95:64–77
European Food Safety Authority (2007) Opinion of the Scientific Panel on food additives, flavourings, processing aids and materials in contact with food (AFC) related to 2,2-bıs(4 Hydroxyphenyl)Propane. https://www.efsa.europa.eu/en/efsajournal/pub/428. Accessed 5 June 2019
Ferguson KK, Peterson KE, Lee JM, Mercado-García A, Blank-Goldenberg C, Téllez Rojo MM, Meeker JD (2014) Prenatal and peripubertal phthalates and bisphenol A in relation to sex hormones and puberty in boys. Reprod Toxicol 47:70–76
Fleisch AF, Sheffield PE, Chinn C, Edelstein BL, Landrigan PJ (2010) Bisphenol A and related compounds in dental materials. Pediatrics 126:760–768
Gordeziani M, Adamia G, Khatisashvili G, Gigolashvili G (2017) Programmed cell self liquidation (apoptosis). Ann Agrarian Sci 15:148–154
Gramec Skledar D, Peterlin Masic L (2016) Bisphenol A and its analogs: do their metabolites have endocrine activity? Environ Toxicol Pharmacol 47:182–199
Gray LE Jr, Wilson VS, Stoker T, Lambright C, Furr J, Noriega N, Howdeshell K, Ankley GT, Guillette L (2006) Adverse effects of environmental antiandrogens and androgens on reproductive development in mammals. Int J Androl 29:96–108
Guida M, Troisi J, Ciccone C, Granozio G, Cosimato C, Di Spiezio SA, Ferrara C, Guida M, Nappi C, Zullo F, Di Carlo C (2015) Bisphenol A and congenital developmental defects in humans. Mutat Res 774:33–39
Guo M, Lai L, Zong T, Lin Y, Yang B, Zhang L, Li M, Kuang H (2015) Exposure to di(2-ethylhexyl) phthalate inhibits luteal function via dysregulation of CD31 and prostaglandin F2alpha in pregnant mice. Reprod Biol Endocrinol 3(13):11
Howdeshell KL, Furr J, Lambright CR, Rider CV, Wilson VS, Gray LE (2007) Cumulative effects of dibutyl phthalate and diethylhexyl phthalate on male rat reproductive tract development: altered fetal steroid hormones and genes. Toxicol Sci 99:190–202
Janesick A, Blumberg B, Heindel JJ (2011) Endocrine disrupting chemicals and disease susceptibility. J Steroid Biochem Mol Biol 127:204–215
Jiang X, Yin L, Zhang N, Han F, Liu WB, Zhang X, Chen HQ, Cao J, Liu JY (2018) Bisphenol A induced male germ cell apoptosis via IFNbeta-XAF1-XIAP pathway in adult mice. Toxicol Appl Pharmacol 355:247–256
Jones S, Boisvert A, Francois S, Zhang L, Culty M (2015) In utero exposure to Di-(2 Ethylhexyl) phthalate induces testicular effects in neonatal rats that are antagonized by genistein cotreatment. Biol Reprod 93(4):92
Kabuto H, Amakawa M, Shishibori T (2004) Exposure to bisphenol A during embryonic/fetal life and infancy increases oxidative injury and causes underdevelopment of the brain and testis in mice. Life Sci 74:2931–2940
Kass L, Durando M, Altamirano GA, Manfroni-Ghibaudo GE, Luque EH, Muñoz-de Toro M (2015) Prenatal Bisphenol a exposure delays the development of the male rat mammary gland. Reprod Toxicol 54:37–46
Koch HM, Calafat AM (2009) Human body burdens of chemicals used in plastic manufacture. Philos Trans R Soc Lond Ser B Biol Sci 364:2063–2078
LaRocca J, Boyajian A, Brown C, Smith SD, Hixon M (2011) Effects of in utero exposure to bisphenol A or diethylstilbestrol on the adult male reproductive system. Birth Defects Res B Dev Reprod Toxicol 92:526–533
Lassen TH, Frederiksen H, Jensen TK, Petersen JH, Joensen UN, Main KM, Skakkebaek NE, Juul A, Jørgensen N, Andersson AM (2014) Urinary bisphenol A levels in young men: association with reproductive hormones and semen quality. Environ Health Perspect 122:478–484
Li M, Qiu L, Zhang Y, Hua Y, Tu S, He Y et al (2013) Dose-related effect by maternal exposure to di-(2-ethylhexyl) phthalate plasticizer on inducing hypospadiac male rats. Environ Toxicol Pharmacol 35:55–60
Liu XL, Xıao-Yu C, Zhı-Cheng W, Tong S, Huna Z (2013) Effects of exposure to bisphenol A during pregnancy and lactation on the testicular morphology and caspase-3 protein expression of ICR pups. Biomed Rep 1:420–424
Liu WJ, Ye L, Huang WF, Guo LJ, Xu ZG, Yang C, Liu HF (2016) p62 links the autophagy pathway and the ubiqutin-proteasome system upon ubiquitinated protein degradation. Cell Mol Biol Lett 21:29. https://doi.org/10.1186/s11658-016-0031-z
Liu T, Wang Y, Yang M, Shao P, Duan L, Li M, Zhu M, Yang J, Jiang J (2018) Di-(2 ethylhexyl) phthalate induces precocious puberty in adolescent female rats. Iran J Basic Med Sci 21:848–855
Ma S, Shi W, Wang X, Song P, Zhong X (2017) Bisphenol A exposure during pregnancy alters the mortality and levels of reproductive hormones and genes in offspring mice. Biomed Res Int 2017:1–8
Manikkam M, Tracey R, Guerrero-Bosagna C, Skinner MK (2013) Plastics derived endocrine disruptors (BPA, DEHP and DBP) induce epigenetic transgenerational inheritance of obesity, reproductive disease and sperm epimutations. PLoS One 8(1):e55387
Meeker JD, Calafat AM, Hauser R (2010) Urinary bisphenol A concentrations in relation to serum thyroid and reproductive hormone levels in men from an infertility clinic. Environ Sci Technol 44:1458–1463
Melzer D, Harries L, Cipelli R, Henley W, Money C, McCormack P, Young A, Guralnik J, Ferrucci L, Bandinelli S, Corsi AM, Galloway T (2011) Bisphenol A exposure is associated with in vivo estrogenic gene expression in adults. Environ Health Perspect 119:1788–1793
Minguez-Alarcon L, Hauser R, Gaskins AJ (2016) Effects of bisphenol A on male and couple reproductive health: a review. Fertil Steril 106:864–870
Mohammed ET, Hashem KS, Ahmed AE, Aly MT, Aleya L, Abdel-Daim MM (2020) Ginger extract ameliorates bisphenol A (BPA)-induced disruption in thyroid hormones synthesis and metabolism: involvement of Nrf-2/HO-1 pathway. Sci Total Environ 703:134664
Muhamad MS, Salim MR, Lau WJ, Yusop Z (2016) A review on bisphenol A occurrences, health effects and treatment process via membrane technology for drinking water. Environ Sci Pollut Res Int 23:11549–11567
Murphy CJ, Stermer AR, Richburg JH, Stermer AR, Richburg JH (2014) Age- and species dependent infiltration of macrophages into the testis of rats and mice exposed to mono-(2-ethylhexyl) phthalate (MEHP). Biol Reprod 18:1–11
Pocar P, Fiandanese N, Secchi C, Berrini A, Fischer B, Schmidt JS, Schaedlich K, Borromeo V (2012) Exposure to di(2-ethyl-hexyl) phthalate (DEHP) in utero and during lactation causes long-term pituitary-gonadal axis disruption in male and female mouse offspring. Endocrinology 153(2):937–948
Qi S, Fu W, Wang C, Liu C, Quan C, Kourouma A et al (2014) BPA-induced apoptosis of rat Sertoli cells through Fas/FasL and JNKs/p38 MAPK pathways. Reprod Toxicol 50:108–116
Qin X, Ma Q, Yuan J, Hu X, Tan Q, Zhang Z, Wang L, Xu X (2018) The effects of di-2 ethylhexyl phthalate on testicular ultrastructure and hormone-regulated gene expression in male rats. Toxicol Res (Camb) 7:408–414
Quan C, Wang C, Duan P, Huang W, Chen W, Tang S, Yang K (2017) Bisphenol a induces autophagy and apoptosis concurrently involving the Akt/mTOR pathway in testes of pubertal SD rats. Environ Toxicol 32:1977–1989
Rochester JR (2013) Bisphenol A and human health: a review of the literature. Reprod Toxicol 42:132–155
Rowdhwal SSS, Chen J (2018) Toxic effects of di-2-ethylhexyl phthalate: an overview. Biomed Res Int 2018:1750368–1750368
Rubin BS (2011) Bisphenol A: an endocrine disruptor with widespread exposure and multiple effects. J Steroid Biochem Mol Biol 127:27–34
Saadeldin IM, Fadel AM, Hamada MMZ, El-Badry AA (2011) Effects of exposure to 50 Hz, 1 gauss magnetic field on reproductive traits in male albino rats; hussein. Querectin alleviates zinc oxide nanoreprotoxicity in male albino rats. Acta Vet Brno 80:107–111
Saillenfait AM, Roudot AC, Gallissot F, Sabaté JP (2011) Prenatal developmental toxicity studies on di-n-heptyl and di-noctyl phthalates in Sprague-Dawley rats. Reprod Toxicol 32:268–276
Shelby MD (2006) NTP-CERHR monograph on the potential human reproductive and developmental effects of di (2-ethylhexyl) phthalate (DEHP). Ntp cerhr mon. 2006. v, vii-7, II-iii-xiii passim
Shirota M, Saito Y, Imai K, Horiuchi S, Yoshimura S, Sato M, Nagao T, Ono H, Katoh M (2005) Influence of di-(2-ethylhexyl) phthalate on fetal testicular development by oral administration to pregnant rats. J Toxicol Sci 30:175–194
Stein TP, Schluter MD, Steer RA, Guo L, Ming X (2015) Bisphenol A exposure in children with autism spectrum disorders. Autism Res 8:272–283
Su Y, Quan C, Li X, Shi Y, Duan P, Yang K (2018) Mutual promotion of apoptosis and autophagy in prepubertal rat testes induced by joint exposure of bisphenol A and nonylphenol. Environ Pollut 243:693–702
Sun Y, Shen J, Zeng L, Yang D, Shao S, Wang J, Wei J, Xiong J, Chen J (2018) Role of autophagy in di-2-ethylhexyl phthalate (DEHP)-induced apoptosis in mouse Leydig cells. Environ Pollut 243:563–572
Swanson JM, Entringer S, Buss C, Wadhwa PD (2009) Developmental origins of health and disease: environmental exposures. Semin Reprod Med 27:391–402
Talsness CE, Andrade AJ, Kuriyama SN, Taylor JA, vom Saal FS (2009) Components of plastic: experimental studies in animals and relevance for human health. Philos Trans R Soc Lond Ser B Biol Sci 364:2079–2096
Tanida I, Ueno T, Kominami E (2008) LC3 and autophagy. Methods Mol Biol 445:77–88
Tomza-Marciniak A, Stępkowska P, Kuba J, Pilarczyk B (2018) Effect of bisphenol A on reproductive processes: a review of in vitro, in vivo and epidemiological studies. J Appl Toxicol 38:51–80
Usman A, Ahmad M (2016) From BPA to its analogues: is it a safe journey? Chemosphere 158:131–142
Vandenberg LN, Hauser R, Marcus M, Olea N, Welshons WV (2007) Human exposure to bisphenol A (BPA). Reprod Toxicol 24:139–177
Vijaykumar T, Singh D, Vanage GR, Dhumal RV, Dighe VD (2017) Bisphenol A-induced ultrastructural changes in the testes of common marmoset. Indian J Med Res 146:126–137
Wang P, Luo C, Li Q, Chen S, Hu Y (2014) Mitochondrion-mediated apoptosis is involved in reproductive damage caused by BPA in male rats. Environ Toxicol Pharmacol 38:1025–1033
Wei Z, Song L, Wei J, Chen T, Chen J, Lin Y, Xia W, Xu B, Li X, Chen X, Li Y, Xu S (2012) Maternal exposure to di-(2-ethylhexyl) phthalate alters kidney development through the renin-angiotensin system in offspring. Toxicol Lett 212:212–221
Williams C, Bondesson M, Krementsov DN, Teuscher C (2014) Gestational bisphenol A exposure and testis development. Endocr Disruptors (Austin) 2(1)
Xi W, Lee CK, Yeung WS, Giesy JP, Wong MH, Zhang X, Hecker M, Wong CK (2011a) Effect of perinatal and postnatal bisphenol A exposure to the regulatory circuits at the hypothalamus-pituitary gonadal axis of CD-1 mice. Reprod Toxicol 31:409–417
Xi W, Wan HT, Zhao YG, Wong MH, Giesy JP, Wong CK (2011b) Effects of perinatal exposure to bisphenol a and di(2-ethylhexyl)-phthalate on gonadal development of male mice. Environ Sci Pollut Res Int 19(7):2515–2527
Xie M, Bu P, Li F, Lan S, Wu H, Yuan L et al (2016) Neonatal bisphenol A exposure induces meiotic arrest and apoptosis of spermatogenic cells. Oncotarget 7:10606–10615
Yaghjyan L, Ghita GL, Dumont-Driscoll M, Yost RA, Chang SH (2016) Maternal exposure to di 2-ethylhexylphthalate and adverse delivery outcomes: a systematic review. Reprod Toxicol 65:76–86
Yi W, Xiang-Liang T, Zhou Y, Liu B, Shen LJ, Long C, Tao L et al (2018) DEHP exposure destroys blood-testis barrier (BTB) integrity of immature testes through excessive ROS mediated autophagy. Genes Dis 5:263–274
Yu L, Chen Y, Tooze SA (2018) Autophagy pathway: cellular and molecular mechanisms. Autophagy 14:207–215
Zhang Y, Han L, Yang H, Pang J, Li P, Zhang G et al (2017) Bisphenol A affects cell viability involved in autophagy and apoptosis in goat testis sertoli cell. Environ Toxicol Pharmacol 55:137–147
Funding
This study was supported by Hacettepe University Research Fund, project no: THD-2017-12150.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Responsible editor: Mohamed M. Abdel-Daim
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Balci, A., Ozkemahli, G., Erkekoglu, P. et al. Histopathologic, apoptotic and autophagic, effects of prenatal bisphenol A and/or di(2-ethylhexyl) phthalate exposure on prepubertal rat testis. Environ Sci Pollut Res 27, 20104–20116 (2020). https://doi.org/10.1007/s11356-020-08274-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-020-08274-6