Abstract
Purpose
In this study, we investigated the effects of maternal transfer of bisphenol A (BPA) and di(2-ethylhexyl) phthalate (DEHP) during gestational and weaning periods on gonadal development of male offspring.
Methods
Pregnant CD-1 mice were administered by gavages in corn oil with 0.1, 1, or 10 mg/kg/day of BPA and DEHP from gestational days (GD1–21) to the weaning period (postnatal days (PND) 1–21).
Results
Our data indicated that the exposure significantly reduced the male-to-female sex ratio and the sizes of the gonads of male pups as recorded at PND15. The testes of the perinatally exposed male pups were developed less and the expression levels of testicular anti-mullerian hormone, androgen receptor, cyclin A, and StAR were significantly lesser than the control male pups. The less developed testes were accompanied with significant reductions in the expression levels of Gnrh and Fsh at the hypothalamic–pituitary levels. The negative effects were found to be persistent in the sexually mature pups at PND42.
Conclusion
Our data reveal that the maternal transfer of BPA and DEHP may impose negative influence on the development and functions of the reproductive system of male pups.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
Endocrine disrupting chemicals (EDC) are defined as any exogenous agents that can interfere with the synthesis, metabolism, and action of endogenous hormones (Phillips et al. 2008; Phillips and Foster 2008). They can affect the hormonal system via (but not limited to) estrogenic, androgenic, anti-androgenic, and anti-thyroid mechanisms. With the benefit of hindsight, EDCs have been shown to impose long-term effects on animal health and development (Anway et al. 2005; Dolinoy et al. 2007; Leranth et al. 2008). Exposure to EDCs is one of the possible causes to explain reproductive problem worldwide as a positive association of body pollutant burdens with reproductive dysfunction has been reported in wildlife, animal, and human studies (Crinnion 2009; Grellier et al. 2010; Lyche et al. 2009; Ma 2009; Wong and Cheng 2011; Yiee and Baskin 2010). Adverse biological effects to male reproductive function were firstly reported in wild animals, whereas accidental exposure to estrogenic pollutants caused feminization or changes in reproductive behavior in the animals (Vos et al. 2000). For example, in 1980s, the adult male alligators exposed to agricultural wastes in Apopka Lake, produced low testosterone levels and presented micro-penis and disorganized testes (Guillette et al. 1994; 1995; Guillette and Guillette 1996). Other reports also highlighted the patho-physiological consequences of chemical exposures in affecting reproductive functions in mammals, birds, amphibian, and fishes (Aravindakshan et al. 2004; Barnhoorn et al. 2004; De Guise et al. 1994; Fry 1995; Hayes et al. 2003; Jobling et al. 2002; Mansfield and Land 2002; Oskam et al. 2003). The observed effects are thought to be elicited by EDCs via the modulations of the effect and metabolism of male and female sex hormones. The molecular targets of EDCs probably are (but not limited to) the sex hormone receptors and the process of steroidogenesis; those are highly conserved in metazoan (McLachlan 1993; Thornton et al. 2003; Zhang et al. 2011). Therefore, this general observation along vertebrate wildlife has highlighted the severity of the widespread presence of EDCs in the environment as well as their similar effects to humans.
The two well-known EDCs, bisphenol A (BPA) and di(2-ethylhexyl)phthalate (DEHP), have been wildly used in the production of plastics (Thompson et al. 2009). They have been involved in many high volume products, including food packaging, personal care, cosmetics, toys, and computers (Andrady and Neal 2009), and are ubiquitous in environment (Oehlmann et al. 2009; Teuten et al. 2009). Both BPA and some phthalates have been identified to exert negative effects, particularly on male reproductive functions in both in vitro and in vivo animal studies (Habert et al. 2009; Richter et al. 2007; Scott et al. 2009; Song et al. 2002; Talsness et al. 2009; Witorsch and Thomas 2010) as well as in human epidemiological studies (Galloway et al. 2010; Meeker 2010; Meeker et al. 2010). The co-existence of BPA and phthalates are commonly identified in urine samples of pregnant women (Becker et al. 2009; Meeker et al. 2009; Swan et al. 2005; 2010; Ye et al. 2008; 2009). This observation implies that the maternal transfer of both BPA and phthalates may impose potential health risk to the developing embryo, fetus, and the neonate. Potential health impacts of BPA and phthalate exposures to pregnant women are of increasing concern (Becker et al. 2009; Meeker et al. 2009; Swan et al. 2005; Teuten et al. 2009; vom Saal et al. 2007; Ye et al. 2008; 2009). In this study, we investigated the effect of in utero and lactational exposure to both BPA and DEHP on the birth sex ratio as well as the development and function of the reproductive system in the male pups. Our data reported a negative dose–response relationship between the exposure and testicular development in the animals.
2 Materials and methods
2.1 Chemicals and experimental animals
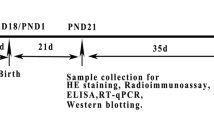
BPA (purity >99.5%) and DEHP (purity >99.0%) were purchased from Sigma-Aldrich, USA. All experimental animals were housed and handled in accordance with Guidelines and Regulations in Hong Kong Baptist University. Male and female mice (CD-1 strain, 8 weeks of age) were purchased from the Laboratory Animal Service Center of Chinese University of Hong Kong (Hong Kong, China). The entire study was conducted in triplicate with mice that were received in three separate batches. The animals were acclimated for 1 week to check any apparent abnormalities. The mice were housed in polypropylene cages with sterilized bedding and were maintained under controlled temperature (23 ± 1°C) and humidity (55 ± 5%) with a 24 h light–dark cycle (06:00–18:00, the light was on). The mice were given ad libitum access to standard rodent food Rodentdiet 5002 (Labdiet, IN, USA) and water (in glass bottles). Mice were bred, and female mice were checked for vaginal plugs the following morning. The presence of a vaginal plug defined gestational day 0. Each copulated mouse (F 0) was housed individually and was randomly assigned to one of four groups with approximately five pregnant mice per group. The mice were weighed and gavaged in the morning with (a) corn oil (group 1), (b) 0.1 mg BPA + 0.1 mg DEHP/kg/day (group 2), (c) 1 mg BPA + 1 mg DEHP/kg/day (group 3), or (d) 10 mg BPA + 10 mg DEHP/kg/day (group 4) in corn oil. The dams were exposed beginning on gestational day 1 until the end of the weaning period (postnatal day, PND 21). Individual dams were checked for birth at least twice a day, and the day when pups were first observed was designated as PND 0. All the pups were weaned. After weaning (from PND 21 to PND 42), the pups were housed individually and given ad libitum access to the standard rodent food and water (in glass bottles).
2.2 Measurement of physical parameters and sampling procedures
Body mass of dams and neonates were measured by an electronic balance (Shimadzu, Kyoto, Japan). Perinatal mortality and number of pups per dam were recorded during the experimental period. Records of anogenital distance (AGD) of each pup were measured at PND 5 and were recorded every 5 days over a period of 15 days. Individual AGD (millimeters) was determined using a caliper to measure the distance between base of the genital papilla and proximal end of the anal opening with the pup held in a supine position, tail extended (Manno 2008). Chromosomal sex was confirmed by polymerase chain reaction (PCR) of the genomic DNA. Sex of the pups was also determined by visual inspection of the reproductive system (ovaries versus testes, vagina/penis) on PND 15. The male pups were sacrificed by cervical dislocation in the morning of PND 15 or PND 42. Blood samples were collected by cardiocenthesis, and serum samples were obtained. Serum concentrations of follicle stimulating hormone (FSH), progesterone (P4), and testosterone (T) were measured using ELISA. Hypothalami, pituitaries, and testes were collected for real-time PCR assay to determine the expression levels of reproduction-related hormones, reproduction-receptors, reproduction-binding proteins, and/or steroidogenic enzymes.
2.3 Determination of chromosomal sex
Mouse tail DNA was extracted with commercially available DNeasy Blood & Tissue Kit (Qiagen, Valencia, CA, USA), using the manufacturer’s protocols, and was eluted with 100 μl of elution buffer. The concentrations of the extracted DNA were evaluated by OD260 absorbance (NanoDrop 1000 Spectrophotometers, Thermo Scientific Inc., Waltham, MA). Primers specific for Sry 5′-TCATGAGACTGCCAACCACAG-3′, 5′-CATGACCACCACCACCACC AA-3′ and Myog 5′-TTACGTCCATCGTGGACAGC-3′, 5′-TGGGCTGGGTGTTAGTCTTA-3′ were synthesized (Koopman et al. 1991; McClive and Sinclair 2001). Standard PCR reactions with a final volume of 20 μl, containing 100 ng DNA, 0.5U Platinum® Taq DNA polymerase (Invitrogen), 0.25 μM each primer set, 0.25 mM dNTP mix, and 1.5 mM MgCl2 were run in a MJ Research PTC-200 Thermo Cycler (MJ, USA). Amplicons were analyzed on 1% agarose gels stained with ethidium bromide.
2.4 Evaluation of total epididymal sperm count
Epididymis was surgically removed from the sexually mature mice (PND 42) and was minced in 600 μl KSOM-3% bovine serum albumin for 20 min at 37°C to release the sperm into the medium. The total epididymal sperm count was assessed using a hemocytometer with a serial dilution of sperm suspension.
2.5 Quantification for gonadotropin and steroid hormones
Serum concentrations of FSH were quantified by a clinical DELFIA® time-resolved fluoroimmunoassay kit (PerkinElmer, Turku, Finland). The assay was run as outlined in the protocol supplied by PerkinElmer. Two hundred microliters of the reaction buffer and 25 μl sample/standard were added into the wells and were incubated at room temperature (RT) for 3 h. The wells were rinsed two times with the washing buffer and were added with 200 μl of tracer dilution buffer followed by 30 min incubation without shaking at RT. The wells were rinsed six times with the washing buffer, and then, 200 μl of enhance buffer were added followed by a 5-min incubation. The signal was measured using Eu-enhanced assay method by Victor® X4 2030 Multiplate reader, PerkinElmer. T and P4 in sera were quantified using ELISA kits (MP Biomedicals, OH, USA). Total 100 μl working hormone–HRP conjugate reagent, 50 μl rabbit anti-hormone reagent, and 25 μl sample/standard were added in wells and incubated at 37°C for 90 min. The wells were rinsed five times with distilled water and mixed with 100 μl TMB solution followed by 20 min incubation at room temperature. The reaction was then stopped by 1 N HCl solution, and the absorbance was read at 450 nm within 15 min.
2.6 Real-time PCR
Gene expression levels were measured by quantitative real-time polymerase chain reaction. Primers were synthesized (Table 1), and PCR products were cloned into pCRII-TOPO (Invitrogen, Carlsbad, CA) and were subjected to dideoxy sequencing for verification. Total RNA was extracted by TRIzol Reagent (Invitrogen, Paisley, UK) according to the manufacturer’s instructions. Real-time PCR was conducted for mRNA quantification. Briefly, 150 ng total RNA was reverse-transcribed using High Capacity cDNA synthesis Kit (Applied Biosystems, USA). Quantified standards (104–108) and sample cDNA were analyzed by StepOne real-time PCR detection system (Applied Biosystems, USA) using Fast SYBR®Green Master mix (Applied Biosystems, USA). The copy number for each sample was calculated, and the data were normalized using the expression level of Gapdh mRNA. The PCR conditions were 95°C for 20 s and 40 cycles of 95°C for 3 s, 56°C for 10 s, and 72°C for 30 s. Fluorescent signals were captured at 72°C; the occurrence of primer dimers and secondary products was inspected using melting curve analysis. All apparatus was treated with diethyl pyrocarbonate and autoclaved. The copy number of the transcripts was calculated in reference to the parallel amplifications of known concentrations of the respective cloned PCR fragments. Standard curves were constructed and amplification efficiencies were found to be between 0.9 and 0.95. Based on melting curve analysis, there was no primer dimer or secondary product formed. Control amplifications were done either without RT or without RNA.
2.7 Western blot
For Western blotting, samples were homogenized in sodium dodecyl sulfate (SDS) lysis buffer (2% SDS and 25% glycerol in 125 mM Tris/HCl (pH 6.8)) and subjected to electrophoresis in 10% polyacrylamide gels. Gels were blotted onto PVDF membranes (PerkinElmer Life Sciences). Western blotting was conducted using goat polyclonal antibody for anti-müllerian hormone (AMH) (1:100, Santa Cruz, USA), rabbit polyclonal antibodies for steroidogenic acute regulatory protein (StAR, 1:500, Santa Cruz), CYP11a1 (1:1000, Chemicon USA), and CYP19a1 (1:300, Abcam, UK), followed by an incubation with horseradish peroxidase-conjugated anti-goat (Santa Cruz) or anti-rabbit antibody (Bio-Rad). Specific bands were visualized with chemiluminescent reagents (Western-lightening Plus, PerkinElmer Life Sciences). Blots were then washed in PBS–0.5% Tween20 and re-probed with rabbit polyclonal antibodies for β-actin (Sigma, USA).
2.8 Statistical analysis
Statistical evaluations were conducted by use of SPSS16. All data were tested to be normally distributed and independent by using the Normal Plots in SPSS, and Shapiro–Wilk significance were over 0.05, which means the assumptions were valid. Differences between treatment groups and corresponding control groups were tested for statistical significance by analysis of variance, followed by Duncan’s multiple range test (significance at p < 0.05) SPSS16. Data are presented as the mean ± SEM. Groups were considered significantly different if P < 0.05.
3 Results
3.1 Assessment of body mass, litter size, sex ratio, testicular weights, and epididymal sperm counts
There were no statistical significant differences in the body masses of the maternal mice (during the gestational period), litter sizes per dam (Fig. 1a), and perinatal mortality rate per dam of F1 pups (data not shown). We observed a dose-dependent decrease in the male-to-female ratio per dam with an increasing dosage of BPA + DEHP treatment (Fig. 1b). The sex ratio decreased from 1.19 ± 0.21 in the control group to 0.64 ± 0.16 at the highest dose (10 mg/kg/day) of the treatment group. The pups were of similar body weight among the control and the exposed groups and showed no noticeable change in the anogenital distance (Electronic supplementary material Fig S1). The weights of the testes of the pups at PND 15 (Fig. 2a) and PND 42 (Fig. 2b) were decreased in 10, 1, and/or 0.1 mg/kg/day of the BPA + DEHP treatment groups as compared with the controls.
Effects of perinatal exposure to 0.1, 1, or 10 mg BPA + DEHP/kg/day on testicular weights and epididymal sperm count of the male pups (n ≥ 6 per group). Significant reductions in testicular weights were observed in the pups of the treatment groups at a PND15 and b PND 42. Epididymal sperm count (c) at PND42. Significant reductions of the sperm counts were observed at 1 and 10 mg BPA + DEHP/kg/day exposed groups
The average sperm count per epididymis was found to be 34.0 ± 5.72 million/ml at PND 42 in the control pups. As shown in Fig. 2c, significant decreases of sperm counts were observed in the male pups of 1 and 10 mg BPA + DEHP/kg/day treatment groups. The average sperm count was found to be 31.0 ± 4.16, 21.3 ± 6.76, and 21.7 ± 2.76 million/ml respectively in the pups of the treatment groups of 0.1, 1, and 10 mg/kg/day. The percentage decrease of epididymal sperm counts in BPA + DEHP treatments were 8.82%, 37.3%, and 35.9%, respectively, as compared with the control group.
3.2 Steroid hormones or reproductive hormones
Serum FSH and steroid hormones (P4 and T) of the male pups on PND 15 were below the detection limits of the ELISA kits. On PND 42, the serum levels of FSH were significantly reduced in the male pups of all BPA + DEHP-treated groups (Fig. 3a). Although there was no noticeable change in the serum P4 levels (Fig. 3b), serum T levels were significantly reduced in the treatment groups (Fig. 3c).
Effects of perinatal exposure to 0.1, 1, or 10 mg BPA + DEHP/kg/day on blood serum levels of follicle stimulating hormone (FSH), progesterone (P 4 ), and testosterone (T) of the male pups at PND42 (n ≥ 6 per group). a Significant reductions of serum FSH were detected in the BPA + DEHP-exposed groups. b No significant change in the serum levels of P4 was observed. c Serum T levels were considerably less in the BPA + DEHP-exposed groups
3.3 Gene expression levels along the hypothalamic–pituitary–gonadal axis of the male pups at PND15
The gene expression levels of the reproduction-related hormones, reproduction-receptors, reproduction-binding proteins, and steroidogenic enzymes were measured along the hypothalamic–pituitary–gonadal (HPG) axis. On PND 15, mRNA expression levels of Gnrh in the male pups exposed to 1 and 10 mg/kg/day of BPA + DEHP were significantly reduced (Fig. 4a).
Effects of perinatal exposure to 0.1, 1, or 10 mg BPA + DEHP/kg/day on the levels of gene expressions at the hypothalamic–pituitary–gonadal axis of the male pups at PND15 (n ≥ 5 per group). a At the hypothalamic level, significant reductions in the transcript levels of Gnrh were observed in the BPA + DEHP-exposed groups. b At the pituitary level, marked reductions of the mRNA levels of Fsh and Prl were observed. c At the testicular levels, considerable decreases in the expression levels of androgen receptor (Ar), anti-mullerian hormone (Amh), cyclinA1, StAR, but an increase in the levels of Cyp19a were detected (left panel). On the right panel, Western blot data shows similar changes in AMH, StAR, and CYP19a at the protein expression levels
At the pituitary levels, the transcript levels of Fsh and Prl were notably reduced in the male pups of BPA + DEHP treatment groups (Fig. 4b). The expression levels of other pituitary hormones (Lh, Tsh, Gh) and Gnrh receptor (Gnrhr) were comparable with the control.
In the testes of the pups, the expression levels of androgen receptor (Ar), anti-mullerian hormone (Amh), and cyclin A1 were significantly reduced in the 1 and 10 mg BPA + DEHP/kg/day-treated groups (Fig. 4c, left panel). The transcript levels of the steroidogenic enzymes StAR was decreased while Cyp19a was increased. Western blot data demonstrated significant reductions of AMH, StAR, but an increase of CYP19a (Fig. 4c, right panel) in the treatment groups.
3.4 Gene expression levels along the HPG axis of the male pups at PND42
On PND 42, no significant changes in the expression levels of the hypothalamic hormones and receptor were observed in the male pups (Fig. 5a). However, the transcript levels of Fsh and Tsh were significantly reduced in the BPA + DEHP-exposed pups (Fig. 5b).
Effects of perinatal exposure to 0.1, 1, or 10 mg BPA + DEHP/kg/day on the levels of gene expressions at the hypothalamic–pituitary–gonadal axis of the male pups at PND42 (n ≥ 5 per group). a At the hypothalamic level, no significant changes in the transcript levels of Kiss-1, Gnrh, and Gpr45 were noted. b At the pituitary level, marked reductions in the mRNA levels of Fsh and Tsh were detected. c At the testicular levels, considerable decreases in the expression levels of androgen receptor (Ar), androgen binding protein (Abp), StAR, but an increase in the levels of Cyp19a were detected (left panel). On the right panel, Western blot data shows similar changes in STAR and CYP19a at the protein expression levels
In the testes of BPA + DEHP-exposed male pups, the transcript levels of Ar and androgen binding protein (Abp) were significantly reduced in 10 and/or 1 mg/kg/day of BPA + DEHP treatment groups (Fig. 5c, left panel). The testicular expression levels of StAR were downregulated while CYP19a expressions were upregulated in 1 and 10 mg BPA + DEHP/kg/day treatments. Western blot data illustrated significant reductions of StAR but an increase of CYP19a (Fig. 5c, right panel).
4 Discussion
Considerable numbers of studies have reported the negative effects of BPA or DEHP on the reproductive health of wildlife, experimental animals, and humans (Halden 2010; Main et al. 2010; Smith et al. 2011). However, the combined effects of the exposure to both BPA and DEHP are not known. Animals are typically exposed to mixtures of chemicals over long periods of time, leading to declining reproductive health. There is a pressing need for studying the effects of chemicals in mixture, which is more realistic in nature to illustrate if the observed phenotype/phenomenon is comparable to humans. Therefore, in this study, the effects of the chemical mixtures BPA and DEHP on the reproductive health of developing fetus were investigated. Using pregnant mouse model, the effects of perinatal BPA + DEHP (0.1, 1, and 10 mg/kg/day) exposure on gonadal development of male offspring were reported. Our data indicated that the exposure caused a significant reduction in male-to-female sex ratio of the offspring. Significant reductions in testicular weights and/or epididymal sperm counts were identified in the immature (PND 15) and sexually mature (PND 42) pups. Mechanistic information on the molecular basis for the observed phenotypes was deciphered. Marked reductions in the expression levels of reproductive hormones, androgen receptor-signaling, and steroidogenic enzymes at the hypothalamic–pituitary-gonadal axis were revealed.
In mammals, sex development in embryonic stage depends on a delicate balance between male and female sex determining pathways (Piprek 2010; Schlessinger et al. 2010; Veitia 2010). It is generally believed that the development of the ovary from genital ridges is a default mechanism while the development of testis depends on the activity of Y chromosome testis-determining gene (Sry) and its downstream/associated factors (i.e., SRY-box containing gene 9 (Sox9), Dmrt1, prostaglandin D synthase, Amh, and testosterone) (Cool and Capel 2009; Ferguson-Smith 2007; Piprek 2009; Sim et al. 2008). The spatiotemporal action of SRY to switch the supporting cells of genital ridges from the female to male pathway is essential and should be undertaken within a critical programming time window (Hiramatsu et al. 2009). Disruption of early steps in male pathway would lead to maldevelopment of testes or an engagement of ovary development. In human epidemiological study, a significant reduction in the ratio of “male birth to total number of birth” was recorded in the Aamjiwnaang First Nation community (areas close to industrial areas) in Canada (Mackenzie et al. 2005). The incidences of low male-to-female sex ratio at birth were also reported in Seveso, Italy, (Mocarelli et al. 2000), the Austrian chloracne cohort (Moshammer and Neuberger 2000), and the victims in the Yucheng oil disaster, Taiwan (del Rio Gomez et al. 2002). Possible explanation for the change in sex ratio at birth has not been elucidated. The highly female-biased sex ratio at birth was probably due to the exposure of EDCs at early age of pregnancy (Koopman 2010; Mocarelli et al. 1996). Moreover, the etiology of this phenomenon is poorly understood (Sharpe 2006). In the present study, a low male-to-female sex ratio of the pups was observed with increasing dosages of the exposure during gestational and lactation periods. Both visual inspections of the reproductive system and chromosomal sex determination verified the sex identity of the individual pups, indicating the effects of the exposure, did not engage ovary development in the male pups. Since the paternal mice were not exposed to BPA + DEHP while the maternal mice were exposed only after copulation, the effects of the chemical pollutants on the relative abundance and activity of X- and Y-chromosome sperms should be at bare minimum. Accordingly, this observation leads to a presumption that the chemical exposure might favor implantation of embryo with XX chromosomes and/or hinder the further development of embryo with XY chromosomes. The presumption might not be unreasonable as male and female pre-implantation mammalian embryos have sexually dimorphism gene expression (Bermejo-Alvarez et al. 2011). These molecular differences may lead to sex-selective embryo loss and sex-specific epigenetic responses to environmental hazards (Bermejo-Alvarez et al. 2011; Gutierrez-Adan et al. 1999; Gutierrez-Adan et al. 2001).
Maternal–fetal transfer of BPA or DEHP has been reported, suggesting the direct effects of the compounds and/or their metabolites on the developing fetus (Mose et al. 2007; Tanaka et al. 2010). During gestational and weaning periods, the presence of BPA and DEHP in maternal/fetal blood and milk may impose estrogenic and anti-androgenic effects on the pups. In this study, the testes of the exposed male pups were significantly smaller, accompanied with significant lesser expression levels of testicular Amh, Ar, Abp, and cyclin A; those are critical for proper development and function of testes (Chang et al. 2004). This might associate with the change in the expression levels of the steroidogenic enzymes StAR and Cyp19a1 and the significant reduction in the size of testes. The effects of the exposure were consistently demonstrated at the hypothalamic–pituitary level, whereas significant reductions in the mRNA expression levels of Gnrh and Fsh were observed. These changes are in agreement with reports describing changes in the reproductive functions when either BPA or DEHP were administrated to the animals. Prenatal and postnatal BPA exposures were reported to interfere with hormone synthesis (i.e., kisspeptin, GnRH, and gonadotrophin) in hypothalamic–pituitary axis (Nakamura et al. 2010; Wei et al. 2011). Other possible BPA-mediated effect included its anti-androgenic property to inhibit androgen-induced AR transcriptional activity (Bonefeld-Jorgensen et al. 2007; Kruger et al. 2008; Lee et al. 2003). In other studies, prenatal phthalate exposure was found to associate with cryptorchidism, disordered anatomical features in sex differentiation (Fisher et al. 2003; Foster 2006; Mahood et al. 2007; Welsh et al. 2008), reduced Sertoli cell proliferation, and steroidogenesis in fetal/neonatal rats (Culty et al. 2008; Dostal et al. 1988; Li et al. 2000). Moreover, the novelty of the present study illustrated that perinatal co-exposure to BPA and phthalates alters sex differentiation and reduce the masculinization process for testicular development in the male pups.
In utero exposure to BPA or some phthalates was reported to impose long-lasting suppressive effects on male fertility in rodents and primates (Culty et al. 2008; Kobayashi et al. 2010; Meeker et al. 2009; Salian et al. 2009). It is probably due to the irreversible disruption at the hypothalamic–pituitary–gonadal axis in mouse offspring upon BPA (Wei et al. 2011) or DEHP exposure (Pocar et al. 2012). This prompted us to investigate the effects of perinatal BPA + DEHP on the fertility of the sexually mature pups on PND42. The negative effects of perinatal BPA + DEHP exposure on the development and function of the male reproductive system were found to be persistent. Significant lesser testicular weight, epididymal sperm counts, and serum T levels were observed in the sexually mature pups. Tanaka and coworkers reported the effect of BPA exposure on the reduction of serum T production (Tanaka et al. 2006). BPA-mediated modulatory effects on T production were found to be related to the downregulation of some steroidogenic enzymes (i.e., 3β-HSD, CYP17a1, and 17β-HSD) and the disruption of estradiol metabolism (Zhang et al. 2011). Occupational exposure to phthalate was also reported to cause significant reduction in serum testosterone levels (Pan et al. 2006). In the present study, the mRNA expression levels of CYP17a1 were comparable among the control and the BPA + DEHP-exposed groups. However, the significant reductions in the expression levels of the rate-limiting steroidogenic enzyme StAR were consistently measured in the male pups at PND 15 and 42. In addition, the upregulations of the transcript levels of CYP19a were observed in the testes of the exposed male pups. These changes probably caused the reduction of T production in the testes. It is a fact that the production of gonadal steroid hormones is regulated by the feedback mechanism along the hypothalamic–pituitary axis, while the release of GnRH and gonadotropin (Gn) regulates the development and function of testes (Amory and Bremner 2003). An investigation of the effects of BPA + DEHP exposure on hypothalamic–pituitary axis of the sexually mature male pups was necessary. Our data indicated that there were significant reductions in the mRNA levels of Fsh and thyroid-stimulating hormone (Tsh) in the pituitary as well as serum FSH levels in the sexually mature male pups. The low serum levels of FSH and T in the exposed male pups caused the reduction of sperm production (Ruwanpura et al. 2010). A marked reduction of TSH expression in pituitary, which may lead to lower testicular weight and reduction in serum T levels (Kumar et al. 2007; Lagu et al. 2005). Collectively, our observation supports the notion that perinatal chemical exposure may affect the formation of the neuronal circuitry at the hypothalamic–pituitary axis and so with the related functions (Yeung et al. 2011), leading to reproductive impairment at adult stage.
5 Conclusion
In the present study, the no-observed adverse effect level (NOAEL) is found to be lesser, or at the low (0.1 mg/kg/day) dosage of the exposure. For the dose translation from mice to human, the NOAEL is divided by the uncertainty factor 100 and the calculated value is 1 μg/kg/day (Speijers 1999). According to the information from US Environmental Protection Agency, the human allowable daily intake levels for BPA and DEPH are 50 and 20 μg/kg/day, respectively (US Environmental Protection Agency (USEPA) 1993). Therefore, the daily oral intake doses used in the present study are environmentally relevant. The present study does not compare the effects by using single (BPA or DEHP) and combined exposure (BPA + DEHP). The major reason is on the lack of the mechanistic data of these individual agents in the literatures. The current understanding of the endocrine-disrupting mechanisms of BPA or DEHP is incomplete. Without these fundamental data, it is difficult to assume their cohort action would be additive, synergistic, or even counteractive. More importantly, this study aimed to identify the physiological consequences, in a realistic situation on the perinatal co-exposure to both BPA and DEHP. Our data reveal that the maternal transfer of BPA and DEHP may impose an issue on the development and functions of the reproductive system in male pups. The findings of this study certainly warrant further investigation on the molecular and cellular targets of BPA and/or DEHP.
References
Amory JK, Bremner WJ (2003) Regulation of testicular function in men: implications for male hormonal contraceptive development. J Steroid Biochem Mol Biol 85(2–5):357–361
Andrady AL, Neal MA (2009) Applications and societal benefits of plastics. Philos Trans R Soc Lond B Biol Sci 364(1526):1977–1984
Anway MD, Cupp AS, Uzumcu M, Skinner MK (2005) Epigenetic transgenerational actions of endocrine disruptors and male fertility. Science 308(5727):1466–1469
Aravindakshan J, Paquet V, Gregory M, Dufresne J, Fournier M, Marcogliese DJ, Cyr DG (2004) Consequences of xenoestrogen exposure on male reproductive function in spottail shiners (Notropis hudsonius). Toxicol Sci 78(1):156–165
Barnhoorn IE, Bornman MS, Pieterse GM, van Vuren JH (2004) Histological evidence of intersex in feral sharptooth catfish (Clarias gariepinus) from an estrogen-polluted water source in Gauteng. South Africa Environ Toxicol 19(6):603–608
Becker K, Goen T, Seiwert M, Conrad A, Pick-Fuss H, Muller J, Wittassek M, Schulz C, Kolossa-Gehring M (2009) GerES IV: phthalate metabolites and bisphenol A in urine of German children. Int J Hyg Environ Health 212(6):685–692
Bermejo-Alvarez P, Rizos D, Lonergan P, Gutierrez-Adan A (2011) Transcriptional sexual dimorphism during preimplantation embryo development and its consequences for developmental competence and adult health and disease. Reproduction 141(6):801–808
Bonefeld-Jorgensen EC, Long M, Hofmeister MV, Vinggaard AM (2007) Endocrine-disrupting potential of bisphenol A, bisphenol A dimethacrylate, 4-n-nonylphenol, and 4-n-octylphenol in vitro: new data and a brief review. Environ Health Perspect 115(1):69–76
Chang C, Chen YT, Yeh SD, Xu Q, Wang RS, Guillou F, Lardy H, Yeh S (2004) Infertility with defective spermatogenesis and hypotestosteronemia in male mice lacking the androgen receptor in Sertoli cells. PNAS 101:6876–6881
Cool J, Capel B (2009) Mixed signals: development of the testis. Semin Reprod Med 27(1):5–13
Crinnion WJ (2009) Maternal levels of xenobiotics that affect fetal development and childhood health. Altern Med Rev 14(3):212–222
Culty M, Thuillier R, Li W, Wang Y, Martinez-Arguelles DB, Benjamin CG, Triantafilou KM, Zirkin BR, Papadopoulos V (2008) In utero exposure to di-(2-ethylhexyl) phthalate exerts both short-term and long-lasting suppressive effects on testosterone production in the rat. Biol Reprod 78(6):1018–1028
De Guise S, Lagace A, Beland P (1994) True hermaphroditism in a St. Lawrence beluga whale (Delphinapterus leucas). J Wildl Dis 30(2):287–290
del Rio Gomez I, Marshall T, Tsai P, Shao YS, Guo YL (2002) Number of boys born to men exposed to polychlorinated byphenyls. Lancet 360(9327):143–144
Dolinoy DC, Huang D, Jirtle RL (2007) Maternal nutrient supplementation counteracts bisphenol A-induced DNA hypomethylation in early development. Proc Natl Acad Sci U S A 104(32):13056–13061
Dostal LA, Chapin RE, Stefanski SA, Harris MW, Schwetz BA (1988) Testicular toxicity and reduced Sertoli cell numbers in neonatal rats by di(2-ethylhexyl)phthalate and the recovery of fertility as adults. Toxicol Appl Pharmacol 95(1):104–121
Ferguson-Smith M (2007) The evolution of sex chromosomes and sex determination in vertebrates and the key role of DMRT1. Sex Dev 1(1):2–11
Fisher JS, Macpherson S, Marchetti N, Sharpe RM (2003) Human ‘testicular dysgenesis syndrome’: a possible model using in-utero exposure of the rat to dibutyl phthalate. Hum Reprod 18(7):1383–1394
Foster PM (2006) Disruption of reproductive development in male rat offspring following in utero exposure to phthalate esters. Int J Androl 29(1):140–147
Fry DM (1995) Reproductive effects in birds exposed to pesticides and industrial chemicals. Environ Health Perspect 103(Suppl 7):165–171
Galloway T, Cipelli R, Guralnik J, Ferrucci L, Bandinelli S, Corsi AM, Money C, McCormack P, Melzer D (2010) Daily bisphenol A excretion and associations with sex hormone concentrations: results from the InCHIANTI adult population study. Environ Health Perspect 118(11):1603–1608
Grellier J, Bennett J, Patelarou E, Smith RB, Toledano MB, Rushton L, Briggs DJ, Nieuwenhuijsen MJ (2010) Exposure to disinfection by-products, fetal growth, and prematurity: a systematic review and meta-analysis. Epidemiology 21(3):300–313
Guillette LJ Jr, Guillette EA (1996) Environmental contaminants and reproductive abnormalities in wildlife: implications for public health? Toxicol Ind Health 12(3–4):537–550
Guillette LJ Jr, Gross TS, Masson GR, Matter JM, Percival HF, Woodward AR (1994) Developmental abnormalities of the gonad and abnormal sex hormone concentrations in juvenile alligators from contaminated and control lakes in Florida. Environ Health Perspect 102(8):680–688
Guillette LJ Jr, Gross TS, Gross DA, Rooney AA, Percival HF (1995) Gonadal steroidogenesis in vitro from juvenile alligators obtained from contaminated or control lakes. Environ Health Perspect 103(Suppl 4):31–36
Gutierrez-Adan A, Perez G, Granados J, Garde JJ, Perez-Guzman M, Pintado B, De La FJ (1999) Relationship between sex ratio and time of insemination according to both time of ovulation and maturational state of oocyte. Zygote 7(1):37–43
Gutierrez-Adan A, Granados J, Pintado B, De La FJ (2001) Influence of glucose on the sex ratio of bovine IVM/IVF embryos cultured in vitro. Reprod Fertil Dev 13(5–6):361–365
Habert R, Muczynski V, Lehraiki A, Lambrot R, Lecureuil C, Levacher C, Coffigny H, Pairault C, Moison D, Frydman R, Rouiller-Fabre V (2009) Adverse effects of endocrine disruptors on the foetal testis development: focus on the phthalates. Folia Histochem Cytobiol 47(5):S67–S74
Halden RU (2010) Plastics and health risks. Annu Rev Public Health 31:179-194
Hayes T, Haston K, Tsui M, Hoang A, Haeffele C, Vonk A (2003) Atrazine-induced hermaphroditism at 0.1 ppb in American leopard frogs (Rana pipiens): laboratory and field evidence. Environ Health Perspect 111(4):568–575
Hiramatsu R, Matoba S, Kanai-Azuma M, Tsunekawa N, Katoh-Fukui Y, Kurohmaru M, Morohashi K, Wilhelm D, Koopman P, Kanai Y (2009) A critical time window of Sry action in gonadal sex determination in mice. Development 136(1):129–138
Jobling S, Coey S, Whitmore JG, Kime DE, Van Look KJ, McAllister BG, Beresford N, Henshaw AC, Brighty G, Tyler CR, Sumpter JP (2002) Wild intersex roach (Rutilus rutilus) have reduced fertility. Biol Reprod 67(2):515–524
Kobayashi K, Ohtani K, Kubota H, Miyagawa M (2010) Dietary exposure to low doses of bisphenol A: effects on reproduction and development in two generations of C57BL/6J mice. Congenit Anom (Kyoto) 50(3):159–170
Koopman P (2010) The delicate balance between male and female sex determining pathways: potential for disruption of early steps in sexual development. Int J Androl 33(2):252–258
Koopman P, Gubbay J, Vivian N, Goodfellow P, Lovell-Badge R (1991) Male development of chromosomally female mice transgenic for Sry. Nature 351(6322):117–121
Kruger T, Long M, Bonefeld-Jorgensen EC (2008) Plastic components affect the activation of the aryl hydrocarbon and the androgen receptor. Toxicology 246(2–3):112–123
Kumar A, Mohanty BP, Rani L (2007) Secretion of testicular steroids and gonadotrophins in hypothyroidism. Andrologia 39(6):253–260
Lagu SK, Bhavsar NG, Sharma RK, Ramachandran AV (2005) Neonatal hypothyroidism-induced changes in rat testis size, dependence on temperature. Neuro Endocrinol Lett 26(6):780–788
Lee HJ, Chattopadhyay S, Gong EY, Ahn RS, Lee K (2003) Antiandrogenic effects of bisphenol A and nonylphenol on the function of androgen receptor. Toxicol Sci 75(1):40–46
Leranth C, Hajszan T, Szigeti-Buck K, Bober J, MacLusky NJ (2008) Bisphenol A prevents the synaptogenic response to estradiol in hippocampus and prefrontal cortex of ovariectomized nonhuman primates. Proc Natl Acad Sci U S A 105(37):14187–14191
Li LH, Jester WF Jr, Laslett AL, Orth JM (2000) A single dose of Di-(2-ethylhexyl) phthalate in neonatal rats alters gonocytes, reduces Sertoli cell proliferation, and decreases cyclin D2 expression. Toxicol Appl Pharmacol 166(3):222–229
Lyche JL, Gutleb AC, Bergman A, Eriksen GS, Murk AJ, Ropstad E, Saunders M, Skaare JU (2009) Reproductive and developmental toxicity of phthalates. J Toxicol Environ Health B Crit Rev 12(4):225–249
Ma L (2009) Endocrine disruptors in female reproductive tract development and carcinogenesis. Trends Endocrinol Metab 20(7):357–363
Mackenzie CA, Lockridge A, Keith M (2005) Declining sex ratio in a first nation community. Environ Health Perspect 113(10):1295–1298
Mahood IK, Scott HM, Brown R, Hallmark N, Walker M, Sharpe RM (2007) In utero exposure to di(n-butyl) phthalate and testicular dysgenesis: comparison of fetal and adult end points and their dose sensitivity. Environ Health Perspect 115(Suppl 1):55–61
Main KM, Skakkebaek NE, Virtanen HE, Toppari J (2010) Genital anomalies in boys and the environment. Best Pract Res Clin Endocrinol Metab 24(2):279–289
Manno FA III (2008) Measurement of the digit lengths and the anogenital distance in mice. Physiol Behav 93(1–2):364–368
Mansfield KG, Land ED (2002) Cryptorchidism in Florida panthers: prevalence, features, and influence of genetic restoration. J Wildl Dis 38(4):693–698
McClive PJ, Sinclair AH (2001) Rapid DNA extraction and PCR-sexing of mouse embryos. Mol Reprod Dev 60(2):225–226
McLachlan JA (1993) Functional toxicology: a new approach to detect biologically active xenobiotics. Environ Health Perspect 101(5):386–387
Meeker JD (2010) Exposure to environmental endocrine disrupting compounds and men’s health. Maturitas 66(3):236–241
Meeker JD, Sathyanarayana S, Swan SH (2009) Phthalates and other additives in plastics: human exposure and associated health outcomes. Philos Trans R Soc Lond B Biol Sci 364(1526):2097–2113
Meeker JD, Calafat AM, Hauser R (2010) Urinary bisphenol A concentrations in relation to serum thyroid and reproductive hormone levels in men from an infertility clinic. Environ Sci Technol 44(4):1458–1463
Mocarelli P, Brambilla P, Gerthoux PM, Patterson DG Jr, Needham LL (1996) Change in sex ratio with exposure to dioxin. Lancet 348(9024):409
Mocarelli P, Gerthoux PM, Ferrari E, Patterson DG Jr, Kieszak SM, Brambilla P, Vincoli N, Signorini S, Tramacere P, Carreri V, Sampson EJ, Turner WE, Needham LL (2000) Paternal concentrations of dioxin and sex ratio of offspring. Lancet 355(9218):1858–1863
Mose T, Mortensen GK, Hedegaard M, Knudsen LE (2007) Phthalate monesters in perfusate from a dual placenta perfusion system, the placenta tissue and umbilical cord blood. Reprod Toxicol 23(1):83–91
Moshammer H, Neuberger M (2000) Sex ratio in the children of the Austrian chloracne cohort. Lancet 356(9237):1271–1272
Nakamura D, Yanagiba Y, Duan Z, Ito Y, Okamura A, Asaeda N, Tagawa Y, Li C, Taya K, Zhang SY, Naito H, Ramdhan DH, Kamijima M, Nakajima T (2010) Bisphenol A may cause testosterone reduction by adversely affecting both testis and pituitary systems similar to estradiol. Toxicol Lett 194:16–25
Oehlmann J, Schulte-Oehlmann U, Kloas W, Jagnytsch O, Lutz I, Kusk KO, Wollenberger L, Santos EM, Paull GC, Van Look KJ, Tyler CR (2009) A critical analysis of the biological impacts of plasticizers on wildlife. Philos Trans R Soc Lond B Biol Sci 364(1526):2047–2062
Oskam IC, Ropstad E, Dahl E, Lie E, Derocher AE, Wiig O, Larsen S, Wiger R, Skaare JU (2003) Organochlorines affect the major androgenic hormone, testosterone, in male polar bears (Ursus maritimus) at Svalbard. J Toxicol Environ Health A 66(22):2119–2139
Pan G, Hanaoka T, Yoshimura M, Zhang S, Wang P, Tsukino H, Inoue K, Nakazawa H, Tsugane S, Takahashi K (2006) Decreased serum free testosterone in workers exposed to high levels of di-n-butyl phthalate (DBP) and di-2-ethylhexyl phthalate (DEHP): a cross-sectional study in China. Environ Health Perspect 114(11):1643–1648
Phillips KP, Foster WG (2008) Key developments in endocrine disrupter research and human health. J Toxicol Environ Health B Crit Rev 11(3–4):322–344
Phillips KP, Foster WG, Leiss W, Sahni V, Karyakina N, Turner MC, Kacew S, Krewski D (2008) Assessing and managing risks arising from exposure to endocrine-active chemicals. J Toxicol Environ Health B Crit Rev 11(3–4):351–372
Piprek RP (2009) Genetic mechanisms underlying male sex determination in mammals. J Appl Genet 50(4):347–360
Piprek RP (2010) Molecular and cellular machinery of gonadal differentiation in mammals. Int J Dev Biol 54(5):779–786
Pocar P, Fiandanese N, Secchi C, Berrini A, Fischer B, Schmidt JS, Schaedlich K, Borromeo V (2012) Exposure to di(2-ethyl-hexyl) phthalate (DEHP) in utero and during lactation causes long-term pituitary–gonadal axis disruption in male and female mouse offspring. Endocrinology 153(2):937–948
Richter CA, Birnbaum LS, Farabollini F, Newbold RR, Rubin BS, Talsness CE, Vandenbergh JG, Walser-Kuntz DR, vom Saal FS (2007) In vivo effects of bisphenol A in laboratory rodent studies. Reprod Toxicol 24(2):199–224
Ruwanpura SM, McLachlan RI, Meachem SJ (2010) Hormonal regulation of male germ cell development. J Endocrinol 205(2):117–131
Salian S, Doshi T, Vanage G (2009) Perinatal exposure of rats to bisphenol A affects the fertility of male offspring. Life Sci 85(21–22):742–752
Schlessinger D, Garcia-Ortiz JE, Forabosco A, Uda M, Crisponi L, Pelosi E (2010) Determination and stability of gonadal sex. J Androl 31(1):16–25
Scott HM, Mason JI, Sharpe RM (2009) Steroidogenesis in the fetal testis and its susceptibility to disruption by exogenous compounds. Endocr Rev 30(7):883–925
Sharpe RM (2006) Pathways of endocrine disruption during male sexual differentiation and masculinization. Best Pract Res Clin Endocrinol Metab 20(1):91–110
Sim H, Argentaro A, Harley VR (2008) Boys, girls and shuttling of SRY and SOX9. Trends Endocrinol Metab 19(6):213–222
Smith CA, MacDonald A, Holahan MR (2011) Acute postnatal exposure to di(2-ethylhexyl) phthalate adversely impacts hippocampal development in the male rat. Neuroscience 193:100–108
Song KH, Lee K, Choi HS (2002) Endocrine disrupter bisphenol a induces orphan nuclear receptor Nur77 gene expression and steroidogenesis in mouse testicular Leydig cells. Endocrinology 143(6):2208–2215
Speijers GJ (1999) Precision of estimates of an ADI (or TDI or PTWI). Regul Toxicol Pharmacol 30(2):S87–S93
Swan SH, Main KM, Liu F, Stewart SL, Kruse RL, Calafat AM, Mao CS, Redmon JB, Ternand CL, Sullivan S, Teague JL (2005) Decrease in anogenital distance among male infants with prenatal phthalate exposure. Environ Health Perspect 113(8):1056–1061
Swan SH, Liu F, Hines M, Kruse RL, Wang C, Redmon JB, Sparks A, Weiss B (2010) Prenatal phthalate exposure and reduced masculine play in boys. Int J Androl 33(2):259–269
Talsness CE, Andrade AJ, Kuriyama SN, Taylor JA, vom Saal FS (2009) Components of plastic: experimental studies in animals and relevance for human health. Philos Trans R Soc Lond B Biol Sci 364(1526):2079–2096
Tanaka M, Nakaya S, Katayama M, Leffers H, Nozawa S, Nakazawa R, Iwamoto T, Kobayashi S (2006) Effect of prenatal exposure to bisphenol A on the serum testosterone concentration of rats at birth. Hum Exp Toxicol 25:369–373
Tanaka M, Kawamoto T, Matsumoto H (2010) Distribution of 14 C-bisphenol A in pregnant and newborn mice. Dent Mater 26(6):e181–e187
Teuten EL, Saquing JM, Knappe DR, Barlaz MA, Jonsson S, Bjorn A, Rowland SJ, Thompson RC, Galloway TS, Yamashita R, Ochi D, Watanuki Y, Moore C, Viet PH, Tana TS, Prudente M, Boonyatumanond R, Zakaria MP, Akkhavong K, Ogata Y, Hirai H, Iwasa S, Mizukawa K, Hagino Y, Imamura A, Saha M, Takada H (2009) Transport and release of chemicals from plastics to the environment and to wildlife. Philos Trans R Soc Lond B Biol Sci 364(1526):2027–2045
Thompson RC, Moore CJ, vom Saal FS, Swan SH (2009) Plastics, the environment and human health: current consensus and future trends. Philos Trans R Soc Lond B Biol Sci 364(1526):2153–2166
Thornton JW, Need E, Crews D (2003) Resurrecting the ancestral steroid receptor: ancient origin of estrogen signaling. Science 301(5640):1714–1717
US Environmental Protection Agency (USEPA) (1993) Integrated risk information system, http://www.epa.goc/iris/subst/. 0014.htm-0356.htm. 11-11-1993
Veitia RA (2010) FOXL2 versus SOX9: a lifelong “battle of the sexes”. Bioessays 32(5):375–380
vom Saal FS, Akingbemi BT, Belcher SM, Birnbaum LS, Crain DA, Eriksen M, Farabollini F, Guillette LJ Jr, Hauser R, Heindel JJ, Ho SM, Hunt PA, Iguchi T, Jobling S, Kanno J, Keri RA, Knudsen KE, Laufer H, LeBlanc GA, Marcus M, McLachlan JA, Myers JP, Nadal A, Newbold RR, Olea N, Prins GS, Richter CA, Rubin BS, Sonnenschein C, Soto AM, Talsness CE, Vandenbergh JG, Vandenberg LN, Walser-Kuntz DR, Watson CS, Welshons WV, Wetherill Y, Zoeller RT (2007) Chapel Hill bisphenol A expert panel consensus statement: integration of mechanisms, effects in animals and potential to impact human health at current levels of exposure. Reprod Toxicol 24(2):131–138
Vos JG, Dybing E, Greim HA, Ladefoged O, Lambre C, Tarazona JV, Brandt I, Vethaak AD (2000) Health effects of endocrine-disrupting chemicals on wildlife, with special reference to the European situation. Crit Rev Toxicol 30(1):71–133
Wei X, Lee CK, Yeung WS, Giesy JP, Wong MH, Zhang X, Hecker M, Wong CK (2011) Effect of perinatal and postnatal bisphenol A exposure to the regulatory circuits at the hypothalamus–pituitary–gonadal axis of CD-1 mice. Reprod Toxicol 31:409–417
Welsh M, Saunders PT, Fisken M, Scott HM, Hutchison GR, Smith LB, Sharpe RM (2008) Identification in rats of a programming window for reproductive tract masculinization, disruption of which leads to hypospadias and cryptorchidism. J Clin Invest 118(4):1479–1490
Witorsch RJ, Thomas JA (2010) Personal care products and endocrine disruption: a critical review of the literature. Crit Rev Toxicol 40(Suppl 3):1–30
Wong EW, Cheng CY (2011) Impacts of environmental toxicants on male reproductive dysfunction. Trends Pharmacol Sci 32(5):290–299
Ye X, Pierik FH, Hauser R, Duty S, Angerer J, Park MM, Burdorf A, Hofman A, Jaddoe VW, Mackenbach JP, Steegers EA, Tiemeier H, Longnecker MP (2008) Urinary metabolite concentrations of organophosphorous pesticides, bisphenol A, and phthalates among pregnant women in Rotterdam, The Netherlands: the Generation R study. Environ Res 108(2):260–267
Ye X, Pierik FH, Angerer J, Meltzer HM, Jaddoe VW, Tiemeier H, Hoppin JA, Longnecker MP (2009) Levels of metabolites of organophosphate pesticides, phthalates, and bisphenol A in pooled urine specimens from pregnant women participating in the Norwegian Mother and Child Cohort Study (MoBa). Int J Hyg Environ Health 212(5):481–491
Yeung BH, Wan HT and Wong CK (2011) Endocrine disrupting chemicals, multiple effects on testicular signaling and spermatogenesis. Spermatogenesis 1(3): 231–239
Yiee JH, Baskin LS (2010) Environmental factors in genitourinary development. J Urol 184(1):34–41
Zhang X, Chang H, Wiseman S, He Y, Higley E, Jones P, Wong CK, Al Khedhairy A, Giesy JP, Hecker M (2011) Bisphenol A disrupts steroidogenesis in human H295R cells. Toxicol Sci 121(2):320–327
Acknowledgment
This work was supported by Collaborative Research Fund (HKBU 1/CRF/08), University Grants Committee (CKC Wong). Prof. John Giesy was supported by the Canada Research Chair program and an at large Chair Professorship at the Department of Biology and Chemistry and State Key Laboratory in Marine Pollution, City University of Hong Kong.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Philippe Garrigues
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Fig. S1
Effects of perinatal exposure to 0.1, 1, or 10 mg BPA + DEHP/kg/day on the anogenital distance (AGD) in the female and male pups, measured from PND5 to PND15 (PPT 94 kb)
Rights and permissions
About this article
Cite this article
Xi, W., Wan, H.T., Zhao, Y.G. et al. Effects of perinatal exposure to bisphenol A and di(2-ethylhexyl)-phthalate on gonadal development of male mice. Environ Sci Pollut Res 19, 2515–2527 (2012). https://doi.org/10.1007/s11356-012-0827-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-012-0827-y