Abstract
Poyang Lake is the largest freshwater lake in China and an important drinking water source. Since the year 2000, toxic cyanobacteria have been observed frequently in Poyang Lake. In the present study, spatial and seasonal variations of microcystins (MCs; MC-RR, MC-YR, and MC-LR) in water column were examined monthly from January to December (except the months of March, May, and November) in 2013, by using ultra-high-performance liquid chromatography-electrospray ionization tandem triple quadrupole/mass spectrometry (UPLC-MS/MS). MC-RR was the most dominant variant, followed by MC-LR, while MC-YR was detected in low concentration. Total MC concentrations (intracellular + extracellular MCs) ranged from 1.26 to 9916.05 ng/L, with an average of 469.99 ng/L, and only 3.14% (6 out of 192 samples) of the water samples contained MC concentrations that exceeded the drinking water guideline level of 1 μg/L for MC-LR proposed by the World Health Organization (WHO). MC concentrations in water column showed obvious seasonal variations in Poyang Lake. Intracellular and extracellular MCs were both at a low level from January to April but increased quickly from June to August and decreased dramatically thereafter. Intracellular MCs exhibited similar spatial distribution pattern with extracellular MCs. Both intracellular and extracellular MC concentrations in eastern bays and around Songmen Mountain of Poyang Lake were higher than other regions. Intracellular MC concentrations were positively correlated with Chl a (r = 046, P < 0.01), pH (r = 0.25, P < 0.01), cyanobacterial biomass (r = 0.40, P < 0.01), and temperature (r = 0.36, P < 0.01) but negatively correlated with TN (r = − 0.28, P < 0.01), suggesting that TN, cyanobacteria biomass, pH, and temperature might be regulating factors for MC production in Poyang Lake.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The frequent occurrence of cyanobacteria blooms in freshwater has become a serious problem worldwide (Brookes and Carey 2011). Several species of cyanobacteria (e.g., Micorcystis, Planktothrix, Oscillatoria, and Anabaena) can produce an impressive range of toxins including neurotoxins, hepatotoxins, cytotoxins, and lipopolysaccharide endotoxins (Metcalf and Codd 2012). Among these cyanotoxins, microcystins (MCs) are the most common and dangerous groups in freshwater (Chorus and Bartram 1999). Until now, more than 90 MC variants have been recognized (Pantelic et al. 2013).
Toxicological studies indicated that MCs mediate their toxicity by uptake into hepatocytes via a carrier-mediated transport system, and then inhibits the activity of serine/threonine protein phosphatases 1 and 2A (Falconer et al. 1981). This inhibition leads to protein phosphorylation imbalance, followed by the disruption of the liver cytoskeleton, finally resulting in massive hepatic hemorrhage and thus the cause of death (Pacagadhi and Balasubramanian 2013). Exposure to MCs represents potential health risks to aquatic organisms and humans (Malbrouck and Kestemont 2006). Previous studies have evidenced that the high incidence of primary liver cancer in the eastern region of China is attributed to MC contaminations in drinking water (Yu 1995). In addition, MCs were identified in the serum from fishermen at Lake Chaohu, China (Chen et al. 2009).
Many research have reported that occurrences of MCs in natural environment are highly variable due to the tremendous differences of cyanobacterial biomass in freshwater with different tropic status and environmental factors (Dai et al. 2016; Sinang et al. 2013; Wu et al. 2006), and these relationships between MCs and environmental parameters are sometimes different or even contradictory (Dai et al. 2016; Sinang et al. 2013; Salmaso et al. 2014; Zhang et al. 2015; Singh et al. 2015).
This might be due to the complicated interactions of various environmental factors in different water bodies. And thus, environmental drivers of MC variability are needed to study extensively, to explore the mechanism of MC occurrence in the field.
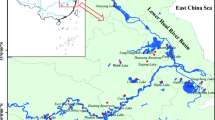
Poyang Lake, the largest freshwater lake, as well as a typical Yangtze-connected lake in China, is located in northern Jiangxi province. Five main tributaries (Ganjiang River, Xiushui River, Raohe River, Xinjiang River, and Fuhe River) drain into the lake from the south and freely exchanges water with the Yangtze River in Hukou (Fig. 1). The water level varies largely in different seasons, depending on the balance between Poyang Lake and Yangtze River (Shankman et al. 2006). In recent years, the environment of Poyang Lake has undergone great changes due to the rapid development of economy and increased human activity, of which eutrophication is the most serious (Liao et al. 2017). Meanwhile, MCs were found in Poyang Lake many years ago (Sui et al. 2007; Wu et al. 2006; Zhang et al. 2015). Due to the special geographical conditions and unique hydrological regime, studies on seasonal and spatial variations of MCs and its relationship with driving factors are essential.
The aims of the present study are to (a) examine seasonal and spatial variations of MCs in Poyang Lake and (b) determine the potential environmental drivers of MC variability.
Materials and methods
Study area
Poyang Lake (28°4′–29°46′N, 115°49′–116°46′E) lies on the southern bank of the middle and lower reaches of the Yangtze River. The lake is an important source for drinking water, as well as water for irrigation, aquaculture, and the industries. This lake serves all the main functions of an ecosystem, including provisioning, supporting, and regulating environmental functions as well as providing cultural and recreational functions for local residents (Li et al. 2009). The floodplains are inundated and form a large lake with an inundation area reaching > 4000 km2 during the wet season from April to September (Shankman et al. 2006). During the dry season from October to March, the inundation area of the lake can shrink to < 1000 km2 to form a narrow, meandering channel.
Reagents and standards
MC-RR and MC-LR standards were purchased from Taiwan Algal Science Inc. (Taiwan, China), and MC-YR standard was purchased from Alexis Biochemical Corporation (Lausen, Switzerland). Methanol, formic acid, and ammonium acetate were of HPLC grade. Methanol was obtained from Tedia company Inc. (OH, USA), formic acid was purchased from Aladdin Industrial Corporation (Shanghai, China), and ammonium acetate was obtained from Alfa Aesar (MA, USA). Other reagents used were of analytical grade.
Sample collection
Thirty-four sampling stations (numbered 1–34) were established according to the geographic characteristics of Poyang Lake (Fig. 1). According to the variation of water level in Poyang Lake and the occurrence of cyanobacteria in this lake, field samples were collected from January to December (except months of March, May, and November) 2013. Sites 1–15 were sampled all the studied months, while water samples of other sites (sites 16–34) were collected from June to August during the rainy season. Each sample was a mixture collected from the top (0–0.5 m surface water) and the bottom (0–0.5 m over sediment) of the water column with a 5-L Schindler sampler.
Water quality parameters
In the field, transparency was measured by a Secchi disk, and the water depth was measured using a digital ultrasonic echo-sounder (Umwet and Wissenschafts Technik, Mondsee, Austria). Water temperature, conductivity, turbidity, and pH were determined using a five-star portable multi-parameter measuring instrument (Orion, CO, USA). Chlorophyll a (Chl a) was analyzed by using hot-ethanol methods (Chen and Gao 2000). Chemical oxygen demand (CODMn), total nitrogen (TN), and total phosphorus (TP) contents were analyzed according to APHA (1998).
Phytoplankton
One-liter integrated water sample for phytoplankton was preserved with 1% acidified Lugol’s iodine solution and concentrated to 30 mL after sedimentation for 48 h (Chen et al. 2003). After mixing, 0.1 mL concentrated samples were counted directly under ×400 magnification. Taxonomic identification was made according to Hu et al. (1979) and biomass was estimated from approximate geometric volumes of each taxon, assuming that 1 mm3 equals 10−6 μg fresh weight (Shei et al. 1993). The geometric dimensions were measured on 10–30 individuals for each dominant species.
MC extraction and analysis
MC extraction and analysis were conducted according to the method of Zhang et al. (2015). The limit of detection (LOD) for the MC-RR, MC-YR, and MC-LR were 0.04, 0.03, and 0.02 ng/mL, respectively.
Recovery experiment
Milli-Q water was filtered with GF/C membrane and used as blank matrix. One-liter blank water was spiked with mixed standards of MCs (MC-RR, MC-YR, MC-LR) solution to a final concentration of 10 ng/L. Samples were enriched, purified, and analyzed according to the method of Zhang et al. (2015). Recovery experiments were carried out in triplicate, and the recovery and the relative standard deviation of the analytical method were calculated.
Statistical analysis
The values below the detection limit can be represented in the dataset by half of the detection limit, while the values for the target compounds that were not detected in the samples were set by zero.
The SPSS (Chicago, IL, USA) for Windows (Ver 13.0) statistical software was used for all analyses. The spatial distribution maps of extracellular and intracellular MCs were constructed using Surfer 8.0 (Golden, Greensboro, NC, USA).
Results
Recovery test
The mean recoveries (n = 3) of MC-RR, MC-YR, and MC-LR were 78.21% (ranging from 74.25 to 84.10%), 90.25% (ranging from 85.75 to 94.38%), and 96.57% (ranging from 92.40 to 98.32%), respectively. The relative standard deviations (RSDs) of MC-RR, MC-YR, and MC-LR were 5.98, 6.12, and 6.97%, respectively.
Environmental parameters
The annual mean and ranges of the physicochemical and biological variables of Poyang Lake in 2013 are presented in Table 1. Water depths varied from 0.60 to 19.34 m with an average of 5.53 m. Water temperature varied from 3.37 to 33.70 °C with an average of 22.41 °C. pH, Secchi depth, conductivity, Chl a, CODMn, and turbidity ranged from 6.97 to 8.59, from 0.10 to 1.50 m, from 60.30 to 388.00 μs/cm, from 0.15 to 34.04 μg/L, from 1.10 to 4.82 mg/L, and from 8.20 to 543.30 NTU, respectively, and the average values of these parameters were 7.71, 0.43 m, 149.19 μs/cm, 5.51 μg/L, 2.26 mg/L, and 74.85 NTU, respectively. In this study, average concentrations of nutrient salts, including TN and TP, were 1.82 and 0.09 mg/L, respectively, which ranged from 0.17 to 7.79 mg/L and from 0.02 to 0.48 mg/L, respectively. Cyanobacterial biomass ranged from 0.00 to 8.07 mg/L, with an average value of 0.45 mg/L.
MC concentrations
Three variants (MC-RR, MC-YR, and MC-LR) were identified in water samples of Poyang Lake, among which MC-RR and MC-LR were the main components, while MC-YR was detected in low concentrations (Table 2). Intracellular MC-RR, MC-YR, and MC-LR ranged from below detectable limit to 8067.09 ng/L, from below detectable limit to 30.63 ng/L, and from below detectable limit to 1975.07 ng/L, respectively, and the average values were 372.19, 2.41, and 70.27 ng/L, respectively. Dissolved MC-RR, MC-YR, and MC-LR ranged from below detectable limit to162.37 ng/L, from below detectable limit to 2.71 ng/L, and from below detectable limit to 44.24 ng/L, respectively, and with mean values of 21.05, 0.32, and 7.38 ng/L, respectively. Total MC concentrations (intracellular MCs + extracellular MCs) ranged between 1.26 and 9916.05 ng/L, with an average of 469.99 ng/L.
Seasonal variations
Seasonal variations of intracellular and extracellular MCs are demonstrated in Fig. 2. Intracellular and extracellular MCs showed similar seasonal changes. Intracellular and extracellular MCs were both at a low level from January to April but increased quickly from June to August while decreased dramatically thereafter. The highest intracellular and extracellular MC concentrations (9871.50 and 166.81 ng/L, respectively) were observed in August, and the lowest values were found in February.
Spatial distributions
Both intracellular and extracellular MC concentrations (MC-RR + MC-YR + MC-LR) showed great spatial distributions in Poyang Lake during wet season, and MCs in June, July, and August exhibited a similar spatial distribution pattern, and thus, spatial distribution maps of MCs in Poyang Lake were depicted according to the data collected from August, due to the maximum MC value occurrence in this month. The highest extracellular (166.81 ng/L) and intracellular MC (9871.50 ng/L) concentrations were found at sites 31 and 28, respectively (Fig. 3), and these two sites were located in eastern bays. Generally, intracellular and extracellular MCs showed a similar spatial distribution pattern, namely higher MC concentrations that appeared in eastern bays and around Songmen Mountain of Poyang Lake, while lower concentrations occurred in the west, center, and north region (Fig. 3).
Relationship between MC concentrations and environmental factors
As showed in Table 3, intracellular MC concentrations were positively correlated with Chl a (r = 046, P < 0.01), pH (r = 0.25, P < 0.01), cyanobacterial biomass (r = 0.40, P < 0.01), and temperature (r = 0.36, P < 0.01) but negatively correlated with TN(r = − 0.28, P < 0.01). No significant correlations were observed between intracellular MC concentrations and TP (P > 0.05), turbidity (P > 0.05), and CODMn (P > 0.05).
Discussion
It has been well documented that MC production was influenced by a complex suite of interacting environmental factors, such as nutrient inputs (Tao et al. 2012; Yu et al. 2014), hydrodynamics (i.e., turbulence, wind-driven currents, and stratification) (Zhou et al. 2016; Eilliott 2012), Chl a (Yu et al. 2014), cyanobacteria biomass (Zhang et al. 2015), temperature, and pH (Tao et al. 2012). In the present study, intracellular MC concentrations were positively correlated with Chl a, pH, cyanobacterial biomass, and temperature but negatively correlated with TN, and no significant correlations were found between intracellular MC concentrations and TP, conductivity, turbidity, suggesting that TN, cyanobacterial biomass, pH, and temperature might be regulating factors for MC production in Poyang Lake. The not significant correlation between MC concentrations and TP could be due to the fact that TP in Poyang Lake (annual mean of TP was 0.09 mg/L) always exceeded the minimum requirement for cyanobacteria growth of 10 μg/L (Chorus and Bartram 1999). A similar result was also found in Taihu Lake (Tao et al. 2012). Until now, there have been extensive studies on the relationship between MC content and environmental factors, while such relationship was not always similar among different water bodies (Wu et al. 2006; Yu et al. 2014; Tao et al. 2012; Duong et al. 2013). For instance, MCs were significantly correlated with Chl a, cyanobacterial biomass, and NO− 3 in 30 subtropical shallow lakes in China, but MC concentrations showed no significant correlations with water temperature, TN, and TP (Wu et al. 2006). In contrast, MCs exhibited a significant correlation with temperature but had poor relationships with other variables (i.e., relative abundance of cyanobacteria, Chl a, Microcystis, nutrients) (Duong et al. 2013). It has also been reported that conductivity, water temperature, and PH presented significant correlations with MC cellular quotas in Taihu Lake (Tao et al. 2012). However, no correlations were found between MC concentrations and all the measured variables in a Germany reservoir (Jungmann et al. 1996). These contradictory relationships can be attributed to different responses of MC-producing cyanobacteria to various environmental parameters (Duong et al. 2013).
In the present study, though intracellular MCs exhibited significant correlations with various environmental factors, the absolute value of coefficients was low (r < 0.46). This suggests that various environmental factors (i.e., hydrological parameter) which are not included in our study might be involved in the regulation of MC production. Previous studies pointed out that turbidity, water velocity, and fluctuating water level can extensively affect the phytoplankton abundance and composition in water bodies (Sharma et al. 2007; Crayton and Sommerfeld 1979), and Liu et al. (2016) revealed that hydrological variables play important roles in the cyanobacterial bloom accumulation in the Yangtze-connected eutrophic lakes of eastern China. Unlike other large shallow eutrophic lakes (i.e., Lake Taihu and Chaohu) along the Yangtze River, Poyang Lake is characterized with its connection to Yangtze River. Its size of water body (> 4000 km2 during rainy season and < 1000 km2 during dry season) undergoes great change within a year, and water velocity can reach to 1.48–2.85 m/s when the water level in Xingzi Station of Poyang Lake is below 15 m (Gu and Wan 2011). The above results indicate that hydrological parameter might play an important role in regulating MC production in Poyang Lake, and the effect of hydrological parameters on MC production in Poyang Lake is needed to be examined in our future study.
Previous studies confirmed that seasonal and spatial distributions of MCs are caused by variances of the toxic strains in cyanobacteria blooms (Chorus 2001), and water currents are regarded as the dominant variable determining phytoplankton distribution in some water bodies (Webster 1990; Liu et al. 2016). In the present study, intracellular MCs were closely correlated with cyanobacterial biomass, and extracellular and intracellular MC concentrations exhibited a similar distribution pattern, that is, the MC content in eastern bays and around Songmen Mountain region were higher than that in other regions. The higher MC concentrations in eastern bays of Poyang Lake might be due to the higher amount of toxic cyanobacteria in these bays, since Liu et al. (2016) reported that a large amount of cyanobacteria are maintained for a longer time in the eastern bays of Poyang Lake due to local clockwise water recirculation. In addition, water in the main part of Poyang Lake must flow through a narrow channel between Songmen Mountain and Duchang County to the Yangtze River, and then a relatively high amount of cyanobacterial biomass accumulated in this region around Songmen Mountain, finally resulting in higher MC concentration in this area. The spatial distribution pattern in our present study was similar with that in Poyang Lake in 2012 (Zhang et al. 2015) and can be attributed to the cyanobacteria distribution driven by water currents and wind (Liu et al. 2016). Meanwhile, in our previous study, we also found that wind speed is considered an important factor for spatial distribution of MCs in Poyang Lake (Zhang et al. 2015). The above results demonstrated that water currents and wind play important roles in spatial distributions of MCs in Poyang Lake and should be examined extensively in the future.
In the present study, intracellular MC contents in Poyang Lake were one to two orders of magnitude higher than extracellular concentrations. The mean annual total MC content in the water column of Poyang Lake was 469.99 ng/L, which is considered to be low when compared with previous studies. The MC level in our present study was comparable with those given from Erhai Lake (Yu et al. 2014) but generally lower to the MC level in Taihu Lake (Tao et al. 2012), Chaohu Lake (Yang et al. 2006), in China; Lake Vancouver in USA (Lee et al. 2015); Lake Biwa in Japan (Ozawa et al. 2005); and Kovada Lake in Turkey (Gurbuz et al. 2009).
In order to avoid potential health risk of MCs to humans, the World Health Organization (WHO) has proposed a provisional guideline level of 1 μg/L for MC-LR equivalent (MC-LReq) in drinking water (Chorus 2001). In the present study, MC-RR and MC-YR were converted to MC-LReq according to the intraperitoneal median lethal dose in mice for MC-RR, MC-YR, and MC-LR reported by Gupta et al. (2003). Though only 3.14% (6 out of 192 samples) of the water samples in Poyang Lake contained MC concentrations above the safety limit of 1 μg/L MC-LReq required for drinking water, the highest MC concentration reached as high as 3.44 μg/L MC-LReq in August. Hence, long-term monitoring of MC contamination in Poyang Lake is needed.
Conclusions
The present study shows that MCs in Poyang Lake may vary by orders of magnitude both seasonally and spatially due to changes in both cyanobacterial biomass and physicochemical/hydrological parameters. TN, pH, and temperature might be regulating factors of MC production in Poyang Lake. It seems that hydrological characteristics such as water currents play a very important role in spatial distributions of MCs. The effect of hydrological parameters on spatial and temporal variations of MCs in Poyang Lake is needed to be studied extensively in our future work.
References
American Public Health Association (APHA) (1998) Standard methods for the examination of water and waste water, 20th edn. American public Health Association, Washington, DC
Brookes JD, Carey CC (2011) Resilience to blooms. Science 334(6052):46–47. https://doi.org/10.1126/science.1207349
Chen J, Xie P, Li L, Xu J (2009) First identification of the hepatotoxic microcystins in the serum of a chronically exposed human population together with indication of hepatocellular damage. Toxicol Sci 108(1):81–89. https://doi.org/10.1093/toxsci/kfp009
Chen YW, Qin BQ, Teubner K, Dokulil MT (2003) Long-term dynamics of phytoplankton assemblages: Microcystis-domination in Lake Taihu, a large shallow lake in China. J Plankton Res 25(4):445–453. https://doi.org/10.1093/plankt/25.4.445
Chen YW,Gao XY (2000) Comparison of Two Methods for Phytoplankton Chlorophyll-a Concentration Measurement. J Lake Sci 12:185-188.https://doi.org/10.18307/2000.0215
Chorus I (2001) Cyanotoxins: occurrence, causes, consequences. Springer, Berlin. https://doi.org/10.1007/978-3-642-59514-1
Chorus I, Bartram J (1999) Toxic cyanobacteria in water: a guide to their public health consequences, monitoring, and management. E&FN Spon, London. https://doi.org/10.4324/9780203478073
Crayton WM, Sommerfeld MR (1979) Composition and abundance of phytoplankton in tributaries of the lower Colorado river, Grand Canyon region. Hydrobiologia 66:81–93
Dai RH, Wang PF, Jia PL, Zhang Y, Chu XC, Wang YF (2016) A review on factors affecting microcystins production by algae in aquatic environments. World J Microbiol Biotechnol 32(3):51. https://doi.org/10.1007/s11274-015-2003-2
Duong TT, Le TPQ, Dao TS, Pflugmacher S, Rochelle-Newall E, Hoang TK, NT V, Ho CT, Dang DK (2013) Seasonal variation of cyanobacteria and microcystins in the Nui Coc Reservoir, northern Vietnam. J Appl Phycol 25(4):1065–1075. https://doi.org/10.1007/s10811-012-9919-9
Elliott JA (2012) Predicting the impact of changing nutrient load and temperature on the phytoplankton of England’s largest lake, Windermere. Freshw Biol 57(2):400–413. https://doi.org/10.1111/j.1365-2427.2011.02717.x
Falconer IR, Jackson ARB, Langley J (1981) Liver pathology in mice in poisoning by the blue-green alga Microcystis aeruginosa. Aust J Diol Sci 34:179–187
Gu P, Wan JB (2011) Hydrology character of Poyang Lake and its influence on water quality. Environ Pollut Control 33:15–19 (in Chinese)
Gupta N, Pant SC, Vijayarghavan R, Rao PV (2003) Comparative toxicity evaluation of cyanobacterial cyclic peptide toxin microcystin variants (LR, RR, YR) in mice. Toxicology 188(2-3):285–296. https://doi.org/10.1016/S0300-483X(03)00112-4
Gurbuz F, Metcalf JS, Karahan AG, Codd GA (2009) Analysis of dissolved microcystins in surface water samples from Kovada Lake, Turkey. Sci Total Environ 407(13):4038–4046. https://doi.org/10.1016/j.scitotenv.2009.02.039
Hu HJ, Li R, Wei YX, Zhu C, Chen J, Shi ZX (1979) Freshwater algae in China. Science Press, Shanghai (in Chinese)
Jungmann D, Ludwichowski KU, Faltin V, Benndorf J (1996) A field study to investigate environmental factors that could affect microcystin sythesis of a Microcystis population in the Bautzen reservoir. Int Rev Hydrobiol 81(4):493–501. https://doi.org/10.1002/iroh.19960810402
Lee TA, Rollwagen-Bollens G, Bollens SM (2015) Environmental influence on cyanobacteria abundance and microcystin toxin production in a shallow temperate lake. Ecotoxicol Environ Saf 114:318–325. https://doi.org/10.1016/j.ecoenv.2014.05.004
Li F, Zhen L, Huang HQ, Han P, Liu XL, Jiang LG, Wei YJ (2009) Impacts of land use functional change on WTA and economic compensation for core stakeholders: a case study in Poyang Lake. Resources Sci 31:580–589 (in Chinese)
Liao MN, Yu G, Guo Y (2017) Eutrophication in Poyang Lake (eastern China) over the last 300 years in response to changes in climate and lake biomass. PLoS One 12(1):e0169319. https://doi.org/10.1371/journal.pone.0169319
Liu X, Li YL, Liu BG, Qian KM, Chen YW, Gao JF (2016) Cyanobacteria in the complex river-connected Poyang Lake: horizontal distribution and transport. Hydrobiologia 768(1):95–110. https://doi.org/10.1007/s10750-015-2536-2
Malbrouck C, Kestemont P (2006) Effects of microcystins on fish. Environ Toxicol Chem 25(1):72–86. https://doi.org/10.1897/05-029R.1
Metcalf JS, Codd GA (2012) Cyanotoxins. In: Whitton BA (ed) Ecology of cyanobacteria II. Springer, pp 651–675. https://doi.org/10.1007/978-94-007-3855-3_24
Ozawa K, Fujioka H, Muranaka M, Yokoyama A, Katagami Y, Watanabe MF, Park HD (2005) Spatial distribution and temporal variation of Microcystis species composition and microcystin concentration in Lake Biwa. Environ Toxicol 20(3):270–276. https://doi.org/10.1002/tox.20117
Pantelic D, Svircev Z, Simeunovic J, Vidovic M, Trajkovic I (2013) Cyanotoxins: characteristics, production and degradation routes in drinking water treatment with reference to the situation in Serbia. Chemosphere 91(4):421–441. https://doi.org/10.1016/j.chemosphere.2013.01.003
Pavagadhi S, Balasubramanian R (2013) Toxicological evaluation of microcystins in aquatic fish species: current knowledge and future directions. Aquat Toxicol 142-143:1–16. https://doi.org/10.1016/j.aquatox.2013.07.010
Salmaso N, Copetti D, Cerasino L, Shams S, Capelli C, Boscaini A, Valsecchi L, Pozzoni F, Guzzellla L (2014) Variability of microcystin cell quota in metapopulations of Planktothrix rubescens: causes and implications for water management. Toxicon: Off J Int Soc Toxinol 90:82–96. https://doi.org/10.1016/j.toxicon.2014.07.022
Shankman D, Keim BD, Song J (2006) Flood frequency in China’s Poyang Lake region: trends and teleconnections. Int J Climatol 26(9):1255–1266. https://doi.org/10.1002/joc.1307
Sharma A, Sharma RC, Anthwal A (2007) Monitoring phytoplanktonic diversity in the hill stream Chandrabhaga of Garhwal Himalaya. Life Sci J 4:80–84
Shei P, Lin W, Wang S, Liu JK (1993) Plankton and seston structure in a shallow, eutrophics subtropical Chinese Lake. Arch Hydrobiol 129:199–220
Sinang SC, Reichwaldt ES, Ghadouani A (2013) Spatial and temporal variability in the relationship between cyanobacterial biomass and microcystins. Environ Monit Assess 185(8):6379–6395. https://doi.org/10.1007/s10661-012-3031-0
Singh S, Rai PK, Chau R, Ravi AK, Neilan BA, Asthana RK (2015) Temporal variations in microcystin-producing cells and microcystin concentrations in two fresh water ponds. Water Res 69:131–142. https://doi.org/10.1016/j.watres.2014.11.015
Sui HX, Xu HB, Yan WX, Chen Y (2007) Pollution status of microcystin in Dianshan Lake and Poyang Lake in China. J Environ Health 24:136–138 (in Chinese)
Tao M, Xie P, Chen J, Qin BQ, Zhang DW, Niu Y, Zhang M, Wang Q, Wu LY (2012) Use of a generalized additive model to investigate key abiotic factors affecting microcystin cellular quotas in heavy bloom areas of Lake Taihu. PLoS One 7(2):e32020. https://doi.org/10.1371/journal.pone.0032020
Webster LT (1990) Effect of wind on the distribution of phytoplankton cells in lakes. Limnol Oceanogr 35(5):989–1001. https://doi.org/10.4319/lo.1990.35.5.0989
Wu SK, Xie P, Liang GD, Wang SB, Liang XM (2006) Relationships between microcystins and environmental parameters in 30 subtropical shallow lakes along the Yangtze River, China. Freshw Biol 51:2309–2319
Yang H, Xie P, Xu J, Zheng L, Deng D, Zhou Q, Wu S (2006) Seasonal variation of microcystin concentration in Lake Chaohu, a shallow subtropical lake in the People’s Republic of China. Bull Environ Contam Toxicol 77(3):367–374. https://doi.org/10.1007/s00128-006-1075-y
Yu GL, Jiang YG, Song GF, Tan WH, Zhu ML, LRH (2014) Variation of Microcystis and microcystins coupling nitrogen and phosphorus nutrients in Lake Erhai, a drinking-water source in Southwest Plateau, China. Environ Sci Pollut Res 21(16):9887–9898. https://doi.org/10.1007/s11356-014-2937-1
Yu SZ (1995) Primary prevention of hepatocellular carcinoma. J Gastroenterol Hepatol 10:674–682
Zhang DW, Liao QG, Zhang L, Wang DG, Luo LG, Chen YW, Zhong JY, Liu JT (2015) Occurrence and spatial distributions of microcystins in Poyang Lake, the largest freshwater lake in China. Ecotoxicology 24(1):19–28
Zhou J, Qin BQ, Han XX, Zhu L (2016) Turbulence increases the risk of microcystin exposure in a eutrophic lake (Lake Taihu) during cyanobacterial bloom periods. Harmful Algae 55:213–220. https://doi.org/10.1016/j.hal.2016.03.016
Acknowledgments
Special thanks are due to the Lake Poyang Laboratory for Wetland Ecosystem Research (PLWER) for providing the foundation for the experiment.
Funding
This study was financially supported by the National Natural Science Foundations of China (Grant 31360133).
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Vitor Manuel Oliveira Vasconcelos
Rights and permissions
About this article
Cite this article
Zhang, L., Liu, J., Zhang, D. et al. Seasonal and spatial variations of microcystins in Poyang Lake, the largest freshwater lake in China. Environ Sci Pollut Res 25, 6300–6307 (2018). https://doi.org/10.1007/s11356-017-0967-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-017-0967-1