Abstract
Diatoms hold great promise as potential sources of biofuel production. In the present study, the biomass and lipid production in the marine diatom Navicula phyllepta, isolated from Cochin estuary, India and identified as a potential biodiesel feedstock, were optimized using Plackett-Burman (PB) statistical experimental design followed by central composite design (CCD) and response surface methodology (RSM). The growth analyses of the isolate in different nitrogen sources, salinities and five different enriched sea water media showed the best growth in the cheapest medium with minimum components using urea as nitrogen source at salinity between 25 and 40 g kg−1. Plackett-Burman experimental analyses for screening urea, sodium metasilicate, sodium dihydrogen phosphate, ferric chloride, salinity, temperature, pH and agitation influencing lipid and biomass production showed that silicate and temperature had a positive coefficient on biomass production, and temperature had a significant positive coefficient, while urea and phosphate showed a negative coefficient on lipid content. A 24 factorial central composite design (FCCD) was used to optimize the concentration of the factors selected. The optimized media resulted in 1.62-fold increase (64%) in biomass (1.2 ± 0.08 g L−1) and 1.2-fold increase (22%) in estimated total lipid production (0.11 ± 0.003 g L−1) compared to original media within 12 days of culturing. A significantly higher biomass and lipid production in the optimized medium demands further development of a two-stage strategy of biomass production followed by induction of high lipid production under nutrient limitation or varying culture conditions for large-scale production of biodiesel from the marine diatom.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Marine microalgae have been proved to be one of the most promising candidates for biofuel production. Reports on biofuel production from microalgae have been focussing more on green algae as they are easily being mass produced and more related to terrestrial plants. Recently, research on diatoms has started gaining momentum in the biofuel arena as they are found to be rich source of neutral lipids (Hildebrand et al. 2012; Levitan et al. 2014; D’Ippolito et al. 2015). Effective commercialisation of microalgae-based biofuel is limited for lack of technical and economic feasibility studies in mass production, biomass harvesting and downstream processing. Research on marine diatoms for biofuel applications is advantageous for use in large-scale raceway ponds due to its ability to tolerate a wide range of salinity fluctuations; the actual use may be limited until conditions are optimized for diatom cell growth and lipid accumulation (Fields et al. 2014). A number of factors are involved in the economically feasible culturing of microalgae, such as optimum light, nutrient availability and temperature, to yield high lipid content and growth rate (Araujo et al. 2011; Huang et al. 2013). Optimisation of micronutrients in the growth medium is an important requirement in establishing a sustainable production system of microalgae. The conventional one-factor-at-a-time approach does not take into account the interactions between nutrients (Song et al. 2012). Therefore, optimisation of bioprocess using statistical tools is the best option to understand multi-factorial interactions in the production of biomass and targeted products from algal biomass.
Response surface methodology is a collection of mathematical and statistical techniques widely used for designing experiments, building models and determining optimum conditions of several factors influencing a mechanism (Ghadge and Raheman 2006; Acıkel et al. 2010; Said and Amin 2016). It helps in screening the key factors rapidly from multiple factors, which can avoid the defects brought by single-factor optimisation (Zhang et al. 2012; Qin et al. 2013). Most of the reports related to the statistical optimisation of microalgal media for biodiesel production are mainly on green algae (Azma et al. 2011; Chen et al. 2014; Jia et al. 2014; Yang et al. 2014a, b; Fawzy 2017). The research using response surface methodology (RSM) for improving the value of biomass and lipid production towards biofuel production by one-stage culture in diatoms has been scarcely reported. As differences among species and strains of the same genus exist, it is imperative to optimize the medium for each organism in order to obtain the maximum response.
Navicula phyllepta MACC8 isolated from Cochin estuary in the west coast of India was identified as a potential biofuel feedstock based on its biomass and lipid productivities and fatty acid methyl ester composition (Sabu et al. 2017). It is a commonly reported benthic diatom from brackish and marine sediments (Clavero et al. 2000; Sabbe et al. 2003). N. phyllepta is ubiquitous in nature, suggesting its adaptation capability across a wide range of environmental conditions such as salinity, emersion time and temperature (Witkowski et al. 2000; Sabbe et al. 2003). The lipids and fatty acids of some Navicula species have already been investigated (Mansour et al. 2005; Duong et al. 2015; Joseph et al. 2016), and the lipid quality indicated the potential of this microalga to be used as a feedstock for biodiesel production (Matsumoto et al. 2010; Sanjay et al. 2013). In addition, the characteristics of the cells of Navicula sp. to settle to the bottom of the vessel or to adhere to the surface of photobioreactors (non-suspended, membrane-based cultivation) (Liu et al. 2013; Bilad et al. 2014; Katarzyna et al. 2015) can be helpful in the easy harvesting the biomass, reducing positively the cost of biofuel production.
The present study was undertaken to identify and evaluate the effect of the different growth factors on biomass and lipid production in the oleaginous microalga N. phyllepta MACC8 identified as a biofuel feedstock and to develop an economic growth medium by optimizing the significant factors using statistical modelling.
Materials and methods
Microalgal culture
Pennate diatom N. phyllepta MACC8 (KC178569) was isolated from brackish waters of Cochin estuary (9° 55′ 35″ N, 96° 17′ 53″ E), India and maintained at the Culture Collection of National Centre for Aquatic Animal Health. The strain was grown in F/2 medium (Guillard 1975) at 26–28 °C under 27 μmol m−2 s−1 with 16:8 light and dark photoperiods.
Light and transmission electron microscopy studies on N. phyllepta MACC8
Algal cells were collected by centrifuging at 4000×g for 5 min. A small drop of the cell pellet was mounted on a clean glass slide with a cover and viewed under oil immersion under a light microscope (Olympus CH20iBIMF, India).
The cultures were observed under transmission electron microscope for studying the ultrastructure of the cell during lipid accumulating stationary stage. A sample of 5 mL microalgal cells was harvested during stationary phase (18–20 days) and washed in 1× phosphate buffer solution (PBS) two to three times. The cells were centrifuged at 8000×g, supernatant was discarded and 500 μL of 2.5% glutaraldehyde was added and incubated at 4 °C. The cells were harvested after 12 h, washed in 1× PBS two to three times and 200 μL of 2% osmium tetroxide was added and incubated at 4 °C for 4 h or more till the culture got stained black/dark brown. The cells were harvested and washed with 1× PBS two to three times. The cells were dehydrated with a graded series of acetone and embedded in epoxy resin. The embedded specimen was cut into ultrathin sections and stained with uranyl acetate and lead citrate (Lewis and Knight 1977). Transmission electron micrograph was recorded using TECNAI 200 TEM (FEI, Electron Optics, USA) at All India Institute of Medical Sciences (AIIMS), Delhi.
Selection of nitrogen source and salinity for high growth
Equimolar concentrations (2 mM) of ammonium chloride, sodium nitrate and urea were used as nitrogen source in the basal medium F/2 with salinity 30 g kg−1. An aliquot of 1 mL of 1 × 106 cells was inoculated in to 50 mL sterilized medium keeping the other culture conditions constant. The cell count was determined by withdrawing 1 mL of sample from the each culture flask every alternate day up to 14 days using Neubauer haemocytometer. For measuring salt tolerance, the diatom was cultured in F/2 medium (with sodium nitrate as nitrogen source) with salinities 0, 10, 20, 30, 35 and 40 g kg−1. Lower salinities were prepared by diluting sea water (30 g kg−1) with distilled water, while higher salinities were prepared by adding NaCl and measured using refractometer. Tap water was used for zero salinity. An aliquot of 1 ml of 1 × 106 cells was inoculated into 100-mL Erlenmeyer flasks containing 50 mL sterilized F/2 medium at 26–28 °C with a photoperiod of 16:8 h light/dark cycles under fluorescent white light (27 μmol m−2 s−1). Cell count and growth rate were determined every alternate days up to 14 days. The experiments were carried out in triplicate under different nitrogen sources, salinities and cultivation time, and the mean values measured at different experimental conditions (different nitrogen sources/salinities) and cultivation time were analysed by two-way analysis of variance (ANOVA).
Selection of growth medium
One millilitre culture of N. phyllepta at a cell density 1 × 106 was inoculated into 100-mL Erlenmeyer flasks containing different sterilized enriched sea water media of 30 g kg−1 such as F/2 (Guillard 1975), modified F/2 for diatoms (modified from Andersen et al. 2005), L1 medium (Guillard and Hargraves 1993), diatom artificial medium (DAM) (Gagneux-Moreaux et al. 2007) and modified sea water medium (MSWM) (Nurachman et al. 2010) at 26–28 °C with a photoperiod of 16:8 h light/dark cycles under fluorescent white light (27 μmol m−2 s−1) and cultured for 14 days. Nutritional composition of each medium is given in Table 1. Cell count was measured every alternate day and the growth rates of the isolate in different media were determined in triplicates for a period of 14 days following Guillard (1973). The significances of the differences in mean growth rate of the diatom in different culture media and cultivation time were tested using two-way ANOVA.
Enumeration of cell count using Neubauer haemocytometer
The concentration of cells was calculated following the method given by Guillard and Sieracki (2005) as follows:
Determination of specific growth rate
The specific growth rate (μ) based on cell density was calculated following the equation (Guillard 1973):
where X 0 and X 1 are densities at the starting and end of the exponential phase at days T 0 and T 1, respectively.
Plackett-Burman experimental design-based screening for significant variables
Plackett-Burman design was employed for screening eight variables such as urea, sodium metasilicate, sodium dihydrogen phosphate, ferric chloride, salinity, temperature, pH and agitation influencing lipid and biomass production. Each variable was set at a higher (+) and lower (−) value to identify which factor had significant influence on the production (Table 2). An experimental design of 12 experiments or runs was formulated for the eight factors predicted by Design Expert software version 6.0.9 (Stat-Ease Inc., Minneapolis, MN, USA) based on the range of the variables provided. The experiments were carried out in 100-mL Erlenmeyer flasks containing 50 mL MSWM medium, in an incubator shaker (Orbitek® LEIL, Scigenics Biotech, India) under illumination of 27 μmol m−2 s−1, 16:8 h light and dark photoperiods. Five percent inoculum containing 1.5 × 106 mL−1 of cells in the exponential growth phase was added to the 100 mL culture medium. The responses were measured in terms of dry weight of biomass (g L−1) and total lipid content estimates (g L−1) towards the end of exponential phase (12th day). The dry weight was estimated by harvesting 30 mL of 12 day culture at 4000×g for 3–5 min, washed with sterilized distilled water and lyophilized at − 72 °C for 12 h, and the weight was determined (Becker 1994). The total lipids were extracted from lyophilized microalgal biomass (3–30 mg) (Shenbaga Devi et al. 2012) following the modified method of Bligh and Dyer (1959), to which 0.6 mL water, 1 mL methanol and 1 mL chloroform were added. The solution was mixed for 30 s, and an additional 1 mL chloroform and 1 mL water were added and the contents of the culture tube were mixed for 30 s. The tubes were centrifuged at 3105×g for 10 min. The upper layer was withdrawn using a pipette, and the lower chloroform phase containing the extracted lipids was transferred into another culture tube; the residue was extracted twice as above, and the chloroform phases were pooled together and dried under nitrogen. Thereafter, the total lipids were measured gravimetrically, and the lipid content was estimated. The responses obtained were subjected to ANOVA, and the significant (p < 0.05) variables were optimized for their concentrations of biomass and total lipid content.
Response surface methodology
A 24 factorial central composite design (FCCD) was used to optimize the concentration of the factors selected such as sodium silicate, urea, sodium dihydrogen phosphate and temperature, keeping rest of the factors such as ferric chloride, salinity, pH and agitation speed constant. An experimental design of 30 experiments or runs was formulated using the Design Expert software 6.0.9 (Stat-Ease Inc., Minneapolis, USA). The experiments were conducted in 100-mL Erlenmeyer flasks containing 50 mL medium (pH 7, 30 g kg−1) prepared according to the design in Table 5. Five percent inoculum containing 1.5 × 106 cells mL−1 in exponential phase was added to the culture medium. The cultures were incubated in an incubator shaker at 120×g under 27 μmol m−2 s−1, 16:8 h light and dark photoperiods and the biomass (g L−1) and total lipid content (g L−1) were estimated from 30 mL sample volume at the end of 12 days. Response surface methodology 3D plots were generated to understand the interaction between different factors and to find the optimum concentration of the medium components favouring the responses. The optimized values obtained were confirmed using point prediction.
Validation of the model
The predicted responses (biomass and lipid) were experimentally validated in the optimized medium under optimized culture conditions predicted by the software at the end of the analyses by shake flask experiments, and it was compared with the un-optimized medium for understanding the effectiveness of the whole optimisation process. The experiments were carried out in triplicates.
Statistical analyses of data
The data was processed and analysed by the statistical software, Design Expert. To estimate the coefficient of regression of experimental data and to plot response surface. ANOVA was used to determine the significance of each term in the fitted equations and to estimate the goodness of fit in each case.
Results and discussions
Cell structure
Tropical marine diatoms are generally difficult to adapt to laboratory conditions (Nurachman et al. 2010), but in the present study, Navicula sp. could be cultured under laboratory conditions. This benthic diatom was isolated from the water samples collected from Cochin Estuary, India. The diatom N. phyllepta is unicellular and uni-nucleate appearing brown to slightly green in colour (Fig. 1a). The primary photosynthetic pigments are chlorophyll a and c and ß-carotene masked by fucoxanthin and xanthophylls (Kuczynska et al. 2015). The ultrastructure of the cell (Fig. 1b) showed the presence of chloroplasts at the two ends of the cell. The large spherical nucleus was attached to the chloroplasts. Tubular shaped mitochondrion was localized in the peripheral cytoplasm layer which is a characteristic of diatoms. The lipid bodies were found at the centre or periphery (Dawes 1998).
Effect of different nitrogen sources
On assessing the effect of different sources of nitrogen on the growth of the benthic diatom in F/2 medium with salinity 30 g kg−1, urea gave higher cell densities compared to sodium nitrate and ammonium chloride as nitrogen source (Fig. 2a). There were significant differences in cell densities of N. phyllepta MACC8 in different media (p = 0.0003) and between the culturing periods (p = 0.007). The growth rates of the diatom in sodium nitrate (NaNO3), urea and ammonium chloride (NH4Cl) were 0.44, 0.40 and 0.21 day−1, respectively. Similar results were reported in a study using diatom Cylindrotheca fusiformis, where NaNO3 and urea were equally good in promoting growth (Suman et al. 2012). Nitrate and urea were better nitrogen sources than ammonium salts in Phaeodactylum tricornutum (Yongmanitchai and Ward 1991), Chlorella vulgaris and Scenedesmus sp. (Crofcheck et al. 2012; Wijanarko 2011; Muthu et al. 2013). The possible explanation for this improved growth mechanism could be that the various genes involved in nitrate assimilation/acquisition in microalgae were actively expressed in the presence of nitrate and urea medium but repressed in the presence of ammonium medium (Hildebrand and Dahlin 2000; Imamura et al. 2010; McDonald et al. 2010). Most marine and fresh water microalgae can effectively use NO3−, NO2−, N2 or NH4+ as nitrogen sources but through absolutely different pathways (Glass et al. 2009). Urea with double nitrogen groups (NH2), upon degradation, results in other nitrogen sources including ammonium ion and nitrate, thus increasing the amount of nitrogen in vivo. It is also known to boost the algal growth as it acts as a complementary source of organic carbon (Saumya et al. 2016). Moreover, the use of urea in growth media stabilizes pH due to lack of ionic charge (Eustance et al. 2013). From the study, both urea and sodium nitrate were proved to be good nitrogen sources, but since urea is more cost-effective (Wijanarko 2011; Kim et al. 2016), urea-based medium was selected as a more feasible one for mass production of N. phyllepta MACC8, a potential biodiesel producer.
Effect of different salinities on diatom growth
N. phyllepta, an estuarine isolate, exhibited wide salinity tolerance by growing in all tested salinities 0, 10, 20, 30, 35 and 40 g kg−1 in F/2 medium with sodium nitrate as nitrogen source. N. phyllepta is a dominant member of communities along estuarine gradient (5–30 g kg−1) (Vanelslander et al. 2009; Smol and Stoermer 2010; Bellinger and Sigee 2015). N. phyllepta has been reported from a broad range of salinities, from electrolyte-rich freshwaters (Krammer and Lange-Bertalot 1986) to hypersaline environments with salinities up to 75 g kg−1 (Clavero et al. 2000). In the present study, the highest cell densities were obtained at salinities ranging from 20 to 40 g kg−1. There was significant difference in growth between the extreme salinities 0 and 40 g kg−1 (p < 0.05). However, there was no significant growth difference within a salinity range of 10–40 g kg−1 and cultivation time (Fig. 2b). There was proportional reduction in lag phase with increasing salinity. Growth in zero salinity showed a long lag phase of 8 days, whereas in salinities higher than 30 g kg−1, the exponential phase started immediately after fourth day. Similar studies reported that estuarine diatom Thalassiosira weissflogii had higher growth rate and better biochemical composition at salinities 25 and 30 g kg−1 (García et al. 2012). The diatom Chaetoceros calcitrans had significant growth when cultured at salinity of 30 g kg−1 (Adenan et al. 2013). Similarly, Nannochloropsis salina belonging to Eustigmatophyceae, a potential biofuel feedstock, showed significant increase in growth rate, biomass and lipid content at higher salinities in the range of 22–34 g kg−1 and higher salinities helped in controlling the invading non-targeted algae and grazers (Bartleya et al. 2013). The results of the present study showed that after a long lag phase of 8 days, the diatom showed a quiet steady growth in zero salinity, i.e. fresh water. This indicates the reason for the abundance of the species at low salinities and also imparts competitive advantage in oligohaline and mesohaline parts of the estuary (Vanelslander et al. 2009). The tolerance to low salinities is important during heavy rainfall when the salinity of sediment top layer almost reduces to fresh water conditions (Coull 1999). The growth rate was found to be the highest in the salinity range of 10–40 g kg−1 with an average rate of 0.46 day−1 with least shown in zero salinity (0.36 day−1). Hence, the isolate can be cultured in sea water (30 g kg−1), encouraging the utilisation of non-potable waters for mass production and minimising the invasion of contaminating invasive organisms.
Selection of growth medium
Of the five types of enriched sea water media of salinity 30 g kg−1 tested, such as F/2, modified F/2 for diatom (change in quantities, not final concentration), L1, DAM and MSWM, the modified sea water medium was found to be a cheaper and better medium for the stable growth of N. phyllepta. Growth studies showed that N. phyllepta MACC8 had a significant growth difference in different media tested and during different cultivation times (p < 0.05) (Fig. 2c). The highest growth rate was found in MSWM (0.48 day−1), followed by F/2 (0.41 day−1) and L1 (0.32 day−1) medium. There was no significant growth difference in modified F/2 medium with a growth rate of 0.26 day−1and DAM with 0.25 day−1. The major obstacle in the large-scale production of biodiesel from microalgae is the high cost in production and one of the factors contributing to the cost is the medium used for culturing the organism. The conventional medium used for culturing this strain contains many nutrients and trace metals which increase the overall cost of the culturing medium. An optimal media is the one which should contain the minimum quantity of nutrients to support maximum growth of the microalgae (Crofcheck et al. 2012). Different media have varying nutrient quantities that can significantly change the quantity of cell biomass and its biochemical composition during cultivation (Mandalam and Palsson 1998).
Navicula sp. is a major biofilm producer as the cells are capable for producing transparent exopolymer particles (TEP), which are colonized by bacteria. These bacteria produce extra-cellular polysaccharides in response to the presence of phytoplankton, thereby initiating cell adhesion and cell to cell attachment (Buhmann et al. 2011; Amin et al. 2012). Such a property is very disadvantageous in terms of the mass cultivation of this diatom in any form of cultivation systems. In this present experiment, the culturing of the diatom N. phyllepta grown in the modified sea water medium helped to reduce the cell adhesion and cell aggregation compared to the conventional F/2 medium. The cells were almost homogenously suspended in the media upon agitation. Figure 3 shows the change in cell aggregation in MSWM and F/2 media. It could be due to reduced number of bacterial population in the media devoid of external addition of trace metals and vitamins as it was reported by Windler et al. (2015) that xenic cultures of benthic diatom Achnanthidium minutissimum showed visible aggregates, whereas the axenic cultures were almost suspended or less aggregated. It may be also due to that some diatom secretome inhibited the biofilm formation under the changed environmental conditions (Doghri et al. 2016). Another possible argument is that urea, a major protein denaturant, must have denatured/solubilized the cell wall proteins involved in diatom cell adhesion (Nguyen and Harvey 2001; Lee and van der Vegt 2006; Willis et al. 2013). Finally, taking all the factors discussed above into consideration, the modified sea water medium was selected for further statistical optimisation.
Screening of variables using Plackett-Burman design
Plackett-Burman design helps us to screen the important factors affecting the desired response with limited number of experiments. Table 3 shows the distribution of different variables and the responses in the study. Of the seven factors tested, urea, sodium silicate, sodium phosphate and temperature were considered as significant variables influencing the responses. The factors silicate and temperature had a positive coefficient on biomass production in N. phyllepta. In the case of total lipid content estimates, temperature had a significant positive coefficient, while urea and phosphate had a negative coefficient. The rest of the variables had no significant effects on the responses and were kept constant in further experiments. Statistical analysis was carried out at 95% confidence interval (CI) throughout, based on t test statistic. Table 4 represents the ANOVA results of the selected variables having significant effect on biomass production and estimated total lipid content. Increasing the silica concentration in the culture media enhances the cell division in diatoms, which is vital for improving algal biodiesel productivity in terms of increased biomass (Moll et al. 2014; Yang et al. 2014a, b). Most of the published studies on phytoplankton were carried out between 20 and 30 °C, and this was suitable for mass cultivation (Karthikeyan et al. 2010; Adenan et al. 2013). The results indicated that concentration of silicon and the temperature can be increased in their levels for further improving the biomass production.
Identification of the best culture medium and growth conditions using response surface methodology
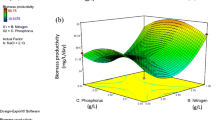
Following screening, response surface methodology using face-centred composite design was employed to understand the interactions between various nutritional and physical factors affecting biomass and lipid production. Table 5 summarizes the results of FCCD experiment of each run with results of the response. The results were analysed by standard analysis of variance (ANOVA), which gave the following quadratic equation:
where, A is sodium silicate, B is urea, C is sodium phosphate and D is temperature. The ANOVA results for biomass and estimated total lipid production (Table 6) showed that the model was significant (p < 0.05). In the case of biomass, the model F value of 57.49 implied that the model was significant. There was only a 0.01% chance that an F value this large could occur due to noise. In this case A, B, D, AB and AC were significant model terms. The predicted adjusted R 2 value 0.9006 was reasonably in agreement with the adjusted R 2 value 0.9646. The “lack of fit F value” of 2.09 implied that the lack of fit was not significant relative to the error. There was a 21.56% chance that a lack of fit F value of this large could occur. Non-significant lack of fit was good. The results showed that silicate, urea, temperature and interactive effect between silicate and urea, and silicate and phosphate were significant for biomass production. The predicted R 2 value of 0.8181 for model to predict lipid production was in fairly reasonable agreement with the adjusted R 2 of 0.9235. The model F value of 25.99 implied that the model was significant. There was only a 0.01% chance that an F value of this large could occur due to noise. Values of “Prob > F” less than 0.05 indicated that model terms were significant. In this case, B, D, AC, AD, BC, BD, CD, B 2 and D 2 are significant model terms. The lack of fit F value of 1.44 implied that the lack of fit was not significant relative to the pure error. There was a 36.07% chance that a lack of fit F value of this large could occur due to the noise. The model showed that the estimated total lipid content in the diatom was controlled by a number of individual factors such as urea, temperature and interactive factors such as silicate-phosphate, silicate-temperature, urea-phosphate, urea-temperature and phosphate-temperature. Perturbation graphs were plotted to compare the effect of all the individual factors at a particular point in the design space. The response is plotted by changing only one factor over its range, while keeping the other factors constant. The lines showing curves or steep slope are the variables showing significant effect on the responses. The silicate (A) had great influence on the biomass, whereas urea (B) and temperature (D) showed major effect on the estimated total lipid production (Fig. 4a, b). Three-dimensional response surfaces were plotted on the basis of the model equation to investigate the interactions among the variables and to determine the optimum concentration of each factor for maximum response (Figs. 5 and 6). Each figure presents the effect of two factors while the other factor was held at zero level. It was clearly seen that silicate had important role in the biomass production, whereas urea, phosphate and temperature showed an interactive effect towards the estimated total lipid production. The predicted values obtained from this model were as follows: 4.89 mM sodium metasilicate, 0.90 mM urea, 0.1 mM sodium dihydrogen phosphate and 30.8 °C temperature for giving a biomass of 1.18 g L−1, whereas 4.69 mM sodium metasilicate, 0.76 mM urea, 0.13 mM sodium dihydrogen phosphate and 25 °C temperature for the estimated total lipid production of 0.16 g L−1.
Amorphous silica is vital for cell growth as it is essential component for frustule formation (Martin-Jézéquel et al. 2000), causing Si availability to be a key factor in the regulation of diatom growth in nature. But the concentration of silicate is critical, as higher concentrations can be inhibitory as reported by Alverson (2007). There are various studies focussing on the significance of mutual effects of nutrients and environmental stress on the growth and lipid production rather than their individual effects (Juneja et al. 2013; Spilling et al. 2015; Singh et al. 2015). Nitrogen has been already identified as limiting agent for increased lipid production in marine microalgae. This could be due to alteration in metabolic pathways (gene regulation) under stress conditions leading to lipid accumulation (Yang et al. 2013). The combined limitation of both nitrogen and phosphorus resulted in the highest lipid concentrations in P. tricornutum (Valenzuela et al. 2012, 2013), Chlamydomonas reinhardtii (Kamalanathan et al. 2015) and Chlorella minutissima (Arora et al. 2016). Increase in lipid during deprivation or limitation of nitrogen, phosphorus source or both could be due to decrease in protein synthesis, causing the excess carbon to get channelized into storage molecules such as neutral lipids (triacylglycerides) and starch (Arora et al. 2016). Temperature affects the physiological processes by changing the rate of chemical reactions and the stability of cellular components (Sandnes et al. 2005; Wagenen et al. 2012). The response of microalgal lipid content to high and low growth temperatures varies from species to species (Renaud et al. 2002; Wu et al. 2013). The results in the present study are in accordance with the results of study by Wu et al. (2013) on Monoraphidium sp., in which lipid content was the highest at 25 °C, while high biomass and lipid productivities were achieved at 30 °C. Similarly, Wah et al. (2015) stated in his study on the effect of different temperatures on the lipid profile of a benthic diatom Amphora subacutiuscula that the total lipid content was the highest at 23 °C in a range of 5–35 °C and saturated fatty acid content was more at lower temperatures. This could be the result of the adaptive mechanism of the organism at low temperatures. Also, Fakhry and El Maghraby (2015) reported that the degree of nitrogen availability in coupled effect with temperature has been identified as the critical factor for the maximal production of lipid in microalgae. In this work, the high diurnal temperature range of 25–35 °C makes N. phyllepta suitable for the outdoor culture in tropical regions; however, more outdoor experiments are needed to evaluate the feasibility.
Validation of the model
Statistical optimisation of growth medium and conditions of N. phyllepta towards high biomass and lipid production using RSM design provided the most simplest and accurate means for obtaining the most efficient medium with the best combination of interacting factors. The validation experiments for confirming the adequacy of the model designed in this study yielded a maximum biomass of 1.2 ± 0.08 g L−1, which was 1.62-fold higher (64% increase) than under un-optimized conditions (0.74 ± 0.08 g L−1). The estimated total lipid content was measured to be 0.11 ± 0.003 g L−1, which was 1.2-fold higher (22% increase) than in un-optimized conditions (0.09 ± 0.009 g L−1). A study on improved culturing conditions for green microalga Scenedesmus sp. by Yang et al. (2014a, b) showed an increase of 13.41% in biomass and 36.32% in lipid content compared to the original conditions. A report on application of factorial design of experiment for biofuel production by the haptophyte Isochrysis galbana (Chen et al. 2012) demonstrated a 3.93% increase in lipid content upon modifying the main interacting variables. The maximum predicted value of biomass (2.95 g L−1) obtained was increased by 1.3 times when compared with the original medium (2.27 g L−1) in the case of microalgae Chlorella pyrenoidosa (Yadavalli and Rao 2013). The maximum biomass attained after RSM based optimisation in microalga Desmodesmus sp. was only 0.758 g L−1 (1.3-fold higher than initial medium) at the end of 14th day of culturing (Ji et al. 2013). Cheng et al. (2013) reported that statistical optimisation of culture media in two-stage cultivation method of Chlorella protothecoides gave a biomass concentration of 1.19 g L−1 in optimized biomass production medium after 11 days of cultivation which was 1.8 times higher than that in the original medium, whereas 12.9% lipid content was obtained from the biomass in the lipid production medium, which was three times higher than that from the original medium. In this study, the amount of total lipid estimated for 1.2 g L−1 of biomass in biomass production medium was 0.132 g L−1 (lipid content 11% dcw) and 0.56 g L−1 of biomass yielded 0.11 g L−1 of estimated total lipid in lipid production medium (lipid content 19.6% dcw) at the end of 12 days. On comparison with these reports, it could be concluded that the optimisation of media components and culture conditions enhanced the biomass and total lipid production in the diatom from that of the original conditions without subjecting to any stress conditions. The results indicated that the media conditions optimized for high biomass production can be adopted for culturing the diatom, and the lipid productivity can be further augmented by subjecting the cells to stress conditions by altering the growth conditions. The present study on RSM-based optimisation of biomass and total lipid production in the newly isolated N. phyllepta identified the important parameters favouring augmented production and, thus, is the first step towards designing a two-stage cultivation method for increased biomass and lipid production in this microalga.
Conclusions
In this study, an optimized process was developed for biomass and lipid production in the lipid rich marine diatom N. phyllepta MACC8. The high growth rate of the algal cells in minimal sea water medium with minimum components and urea as nitrogen source offers a great potential for the mass production of the diatom in a cost effective process. The Plackett-Burman design- and response surface methodology-based optimisations of biomass and lipid production in N. phyllepta MACC8 resulted in 1.62-fold increase (64%) in biomass and 1.2-fold increase (22%) in estimated total lipid production. The optimized process parameters and growth medium obtained in this experiment can be further utilized for large-scale biomass production from N. phyllepta and subsequent augmented lipid production by limiting or altering the factors such as urea, silicate, phosphate and temperature as identified in the present study.
References
Acıkel U, Erşan M, Açıke YS (2010) Optimization of critical medium components using response surface methodology for lipase production by Rhizopus delemar. Food Bioprod Process 88:31–39
Adenan NR, Yusoff FMD, Sheriff M (2013) Effect of salinity and temperature on growth of diatoms and green algae. J Fish Aqua Sci 8:397–404
Alverson AJ (2007) Strong purifying selection in the silicon transporters of marine and freshwater diatoms. Limnol Oceanogr 52:1420–1429
Amin SA, Parker MS, Armbrust EV (2012) Interactions between diatoms and bacteria. Microbiol Mol Biol Rev 76:667–684
Andersen RA, Berges JA, Harrison PJ, Watanabe MM (2005) Recipes for freshwater and seawater media. In: Anderson RA (ed) Algal culturing techniques. Elsevier, Amsterdam, pp 429–538
Araujo GS, Matos LJBL, Gonçalves LRB, Fernandes FAN, Farias WRL (2011) Bioprospecting for oil producing microalgal strains: evaluation of oil and biomass production for 10 microalgal strains. Bioresour Technol 102:5248–5250
Arora N, Patel A, Pruthi PA, Pruthi V (2016) Synergistic dynamics of nitrogen and phosphorous influences lipid productivity in Chlorella minutissima for biodiesel production. Bioresour Technol 213:79–87
Azma M, Mohamed MS, Mohamad R, Rahim RA, Ariff AB (2011) Improvement of medium composition for heterotrophic cultivation of green microalgae, Tetraselmis suecica, using response surface methodology. Biochem Eng J 53:187–195
Bartleya ML, Boeinga WJ, Corcorana AA, Holguinb FO, Schaubb T (2013) Effects of salinity on growth and lipid accumulation of biofuel microalga Nannochloropsis salina and invading organisms. Biomass Bioenergy 54:83–88
Becker EW (1994) Microalgae: biotechnology and microbiology. Cambridge University Press, Cambridge
Bellinger EG, Sigee DC (2015) Freshwater algae: identification, enumeration and use as bioindicators, 2nd edn. Wiley Blackwell, UK
Bilad MR, Arafat HA, Vankelecom FJ (2014) Membrane technology in microalgae cultivation and harvesting: a review. Biotechnol Adv 32:1283–1300
Bligh EG, Dyer WJ (1959) A rapid method of total lipid extraction and purification. Can J Biochem Phys 37:911–917
Buhmann M, Schleheck D, Windler M, Kroth PG (2011) Bacteria influence diatom biofilm formation. Eur J Phycol 46:80–80
Chen JJ, Li YR, Lai WL (2014) Application of experimental design methodology for optimization of biofuel production from microalgae. Biomass Bioenergy 64:11–19
Chen JJ, Li YR, Xie MZ, Chiu CY, Liao SW, Lai WL (2012) Factorial design of experiment for biofuel production by Isochrysis galbana. Int Proc Chem Biol Environ Eng 33:91–95
Cheng KC, Ren M, Ogden KL (2013) Statistical optimization of culture media for growth and lipid production of Chlorella protothecoides UTEX 250. Bioresour Technol 128:44–48
Clavero E, Hernandez-Marine M, Grimalt JO, Garcia-Pichel F (2000) Salinity tolerance of diatoms from thalassic hypersaline environments. J Phycol 36:1021–1034
Coull BC (1999) Role of meiofauna in estuarine soft-bottom habitats. Aust J Ecol 24:327–343
Crofcheck C, Xinyi E, Shea A, Montross M, Crocker M, Andrews R (2012) Influence of media composition on the growth rate of Chlorella vulgaris and Scenedesmus acutus utilized for CO2 mitigation. J Biochem Tech 4:589–594
Dawes CJ (1998) Microalgae and their communities, 2nd edn. Marine botany, John Wiley and Sons, NY, pp 168–204
D’Ippolito G, Sardo A, Paris D, Vella FM, Adelfi MG, Botte P, Gallo C, Fontana A (2015) Potential of lipid metabolism in marine diatoms for biofuel production. Biotechnol Biofuels 8:1–28
Doghri I, Lavaud J, Dufour A, Bazire A, Lannelu I, Sablé S (2016) Cell-bound exopolysaccharides from an axenic culture of the intertidal mudflat Navicula phyllepta diatom affect biofilm formation by benthic bacteria. J Appl Phycol:1–13
Duong VT, Thomas-Hall SR, Schenk PM (2015) Growth and lipid accumulation of microalgae from fluctuating brackish and sea water locations in South East Queensland-Australia. Front Plant Sci 6:359
Eustance E, Gardner RD, Moll KM, Menicucci J, Gerlach R, Peyton BM (2013) Growth, nitrogen utilization and biodiesel potential for two chlorophytes grown on ammonium, nitrate or urea. J Appl Phycol 25:1663–1677
Fakhry EM, El Maghraby DM (2015) Lipid accumulation in response to nitrogen limitation and variation of temperature in Nannochloropsis salina. Bot Stud 56:6
Fawzy MA (2017) Fatty acid characterization and biodiesel production by the marine microalga Asteromonas gracilis: statistical optimization of medium for biomass and lipid enhancement. Mar Biotechnol:1–13
Fields MW, Hise LEJ, Bell T, Gardner RD, Corredor L, Moll K, Peyton BM, Characklis GW, Gerlach R (2014) Sources and resources: importance of nutrients, resource allocation, and ecology in microalgal cultivation for lipid accumulation. Appl Microbiol Biotechnol 98:4805–4816
Gagneux-Moreaux S, Moreau C, Cosson RP (2007) Diatom artificial medium (DAM): a new artificial medium for the diatom Haslea ostrearia and other marine microalgae. J Appl Phycol 19:549–556
García N, López-Elías JA, Miranda A, Martínez-Porchas M, Huert N, García A (2012) Effect of salinity on growth and chemical composition of the diatom Thalassiosira weissflogii at three culture phases. Lat Am J Aquat Res 40:435–440
Ghadge SV, Raheman H (2006) Process optimization for biodiesel production from mahua (Madhuca indica) oil using response surface methodology. Bioresour Technol 97:379–384
Glass JB, Wolfe-Simon F, Anbar AD (2009) Coevolution of metal availability and nitrogen assimilation in cyanobacteria and algae. Geobiology 7:100–123
Guillard RRL (1973) Division rates. In: Stein (ed) Handbook of phycological methods. Vol. 1. Cambridge University Press, Cambridge, pp 289–312
Guillard RRL (1975) Culturing of phytoplankton for feeding marine invertebrates. In: Smith WL, Chanley MH (eds) Culture of marine invertebrates animals. Plenum Press, NY, pp 26–60
Guillard RRL, Hargraves PE (1993) Stichochrysis immobilis is a diatom, not a chrysophyte. Phycologia 32:234–236
Guillard RR, Sieracki MS (2005) Counting cells in cultures with the light microscope. In: Anderson RA (ed) Algal culturing techniques, 1st edn. Academic Press, Elsevier, pp 239–252
Hildebrand M, Dahlin K (2000) Nitrate transporter genes from the diatom Cylindrotheca fusiformis (Bacillariophyceae): mRNA levels controlled by nitrogen source and by the cell cycle. J Phycol 36:702–713
Hildebrand M, Davis AK, Smith SR, Traller JC, Abbriano R (2012) The place of diatoms in the biofuels industry. Biofuels 3:221–240
Huang X, Huang Z, Wen W, Yan J (2013) Effects of nitrogen supplementation of the culture medium on the growth, total lipid content and fatty acid profiles of three microalgae (Tetraselmis subcordiformis, Nannochloropsis oculata and Pavlova viridis). J Appl Phycol 25:129–137
Imamura S, Terashita M, Ohnuma M, Maruyama S, Minoda A, Weber AP, Inouye T, Sekine Y, Fujita Y, Omata T, Tanaka K (2010) Nitrate assimilatory genes and their transcriptional regulation in a unicellular red alga Cyanidioschyzon merolae: genetic evidence for nitrite reduction by a sulfite reductase-like enzyme. Plant Cell Physiol 51(5):707–717
Ji F, Hao R, Liu Y, Li G, Zhou Y, Dong R (2013) Isolation of a novel microalgae strain Desmodesmus sp. and optimization of environmental factors for its biomass production. Bioresour Technol 148:249–254
Jia Z, Liu Y, Daroch M, Geng S, Cheng JJ (2014) Screening, growth medium optimisation and heterotrophic cultivation of microalgae for biodiesel production. Appl Biochem Biotechnol 173:1667–1679
Joseph MM, Renjith KR, John G, Nair SM, Chandramohanakumar N (2016) Biodiesel prospective of five diatom strains using growth parameters and fatty acid profiles. Biofuels:1–9
Juneja A, Ceballos RM, Murthy GS (2013) Effects of environmental factors and nutrient availability on the biochemical composition of algae for biofuels production: a review. Energies 6:4607–4638
Kamalanathan M, Pierangelini M, Shearman LA, Gleadow R, Beardall J (2015) Impacts of nitrogen and phosphorus starvation on the physiology of Chlamydomonas reinhardtii. J Appl Phycol 28:1509–1520
Karthikeyan P, Jayasudha S, Sampathkumar P, Manimaran K, Santhoshkumar C, Ashokkumar S, Ashokprabu V (2010) Effect of industrial effluent on the growth of marine diatom, Chaetoceros simplex (Ostenfeld, 1901). J Appl Sci Environ Manag 14:35–37
Kim G, Mujtaba G, Lee K (2016) Effects of nitrogen sources on cell growth and biochemical composition of marine chlorophyte Tetraselmis sp. for lipid production. Algae 31:257–266
Krammer K, Lange-Bertalot H (1986) Bacillariophyceae. 1. Teil: Naviculaceae. In: Ettl H, Gerloff J,Heynig H, Mollenhaurer D (eds) Süsswasserflora von Mitteleuropa, Band 2/1. VEB G. Fischer, Jena, pp 876
Kuczynska P, Jemiola-Rzeminska M, Strzalka K (2015) Photosynthetic pigments in diatoms. Mar Drugs 13:5847–5881
Katarzyna L, Sai G, Singh OA (2015) Non-enclosure methods for non-suspended microalgae cultivation: literature review and research needs. Renew Sust Energ Rev 42:1418–1427
Lee ME, van der Vegt NF (2006) Does urea denature hydrophobic interactions? J Am Chem Soc 128:4948–4949
Levitan O, Dinamarca J, Hochman G, Paul G. Falkowski (2014) Diatoms: a fossil fuel of the future. Trends Biotechnol 32:117–124
Lewis PR, Knight DP (1977) Staining methods for sectioned material. In: Glauert AM (ed) Practical methods in electron microscopy: Vol. 5. Elsevier, North Holland
Liu T, Wanga J, Hub Q, Chenga P, Jia B, Liua J, Chena Y, Zhanga W, Chena X, Chena L, Gaoa L, Jia C, Wanga H (2013) Attached cultivation technology for microalgae for efficient biomass feedstock production. Bioresour Technol 127:216–222
Mandalam RK, Palsson B (1998) Elemental balancing of biomass and medium composition enhances growth capacity in high density Chlorella vulgaris cultures. Biotechnol Bioeng 59:605–611
Mansour MP, Frampton DMF, Nichols PD, Volkman JK, Blackburn SI (2005) Lipid and fatty acid yield of nine stationary-phase microalgae: applications and unusual C24–C28 polyunsaturated fatty acids. J Appl Phycol 17:287–300
Martin-Jézéquel V, Hildebrand M, Brzezinski MA (2000) Silicon metabolism in diatoms: implications for growth. J Phycol 36:821–840
Matsumoto M, Sugiyama H, Maeda Y, Sato R, Tanaka T, Matsunaga T (2010) Marine diatom, Navicula sp. strain JPCC DA0580 and marine green alga, Chlorella sp. strain NKG400014 as potential sources for biodiesel production. Appl Biochem Biotechnol 161:483–490
McDonald SM, Plant JN, Worden AZ (2010) The mixed lineage nature of nitrogen transport and assimilation in marine eukaryotic phytoplankton: a case study of Micromonas. Mol Biol Evol 27:2268–2283
Moll KM, Gardner RD, Eustance EO, Gerlach R, Peyton BM (2014) Combining multiple nutrient stresses and bicarbonate addition to promote lipid accumulation in diatoms. Algal Res 5:7–15
Muthu A, Agarwal A, Arya MC, Ahmed Z (2013) Influence of nitrogen sources on biomass productivity of microalgae Scenedesmus bijugatus. Bioresour Technol 131:246–249
Nguyen RT, Harvey HR (2001) Preservation of protein in marine systems: hydrophobic and other non-covalent associations as major stabilizing forces. Geochim Cosmochim Acta 65:1467–1480
Nurachman Z, Panggabean LMG, Anita S (2010) Screening of local marine microalgae for biodiesel production. SEAMEO-SEARCA, SEARCA Agriculture & Development Paper Series 3
Qin JZ, Song FF, Qiu YF, Li XX, Guan X (2013) Optimization of the medium composition of a biphasic production system for mycelial growth and spore production of Aschersonia placenta using response surface methodology. J Invertebr Pathol 112:108–115
Renaud SM, Thinh LV, Lambrinidis G, Parry DL (2002) Effect of temperature on growth, chemical composition and fatty acid composition of tropical Australian microalgae grown in batch cultures. Aquaculture 211:195–214
Sabbe K, Verleyen E, Hodgson DA, Vanhoutte K, Vyverman W (2003) Benthic diatom flora of freshwater and saline lakes in the Larsemann Hills and Rauer Islands, East Antarctica. Antarct Sci 15:227–248
Sabu S, Singh ISB, Joseph V (2017) Molecular identification and comparative evaluation of tropical marine microalgae for biodiesel production. Mar Biotechnol. https://doi.org/10.1007/s10126-017-9754-8
Said KA, Amin MA (2016) Overview on the response surface methodology (RSM) in extraction processes. J Appl Sci Process Eng 2:8–17
Sandnes JM, Kallqvist T, Wenner D, Gislerod HR (2005) Combined influence of light and temperature on growth rates of Nannochloropsis oceanica: linking cellular responses to large-scale biomass production. J Appl Phycol 17:515–525
Sanjay KR, Nagendra PMN, Anupama S, Yashaswi BR, Deepak B (2013) Isolation of diatom Navicula cryptocephala and characterization of oil extracted for biodiesel production. Afr J Environ Sci Technol 7:41–48
Saumya D, Kannan DC, Dhawan V (2016) Understanding urea assimilation and its effect on lipid production and fatty acid composition of Scenedesmus sp. SOJ Biochem 2:7
Shenbaga Devi A, Santhanam P, Rekha V, Ananth S, Prasath BB, Nandakumar R, Jeyanthi S, Kumar SD (2012) Culture and biofuel producing efficacy of marine microalgae Dunaliella salina and Nannochloropsis sp. J Algal Biomass Utln 3:38–44
Singh P, Guldhe A, Kumari S, Rawat I, Bux F (2015) Investigation of combined effect of nitrogen, phosphorus and iron on lipid productivity of microalgae Ankistrodesmus falcatus KJ671624 using response surface methodology. Biochem Eng J 94:22–29
Smol JP, Stoermer EF (2010) The diatoms: applications for the environmental and earth sciences, 2nd edn. Cambridge University Press, Cambridge
Song L, Qin JG, Su S, Xu J, Clarke S, Shan Y (2012) Micronutrient requirements for growth and hydrocarbon production in the oil producing green alga Botryococcus braunii (Chlorophyta). PLoS ONE 7:e41459. https://doi.org/10.1371/journal.pone.0041459
Spilling K, Ylostalo P, Simis S, Seppala J (2015) Interaction effects of light, temperature and nutrient limitations (n, p and si) on growth, stoichiometry and photosynthetic parameters of the cold-water diatom Chaetoceros wighamii. PLoS ONE 10:e0126308. https://doi.org/10.1371/journal.pone.0126308
Suman KK, Devi T, Sarma UK, Nittala S (2012) Culture medium optimization and lipid profiling of Cylindrotheca, a lipid- and polyunsaturated fatty acid-rich pennate diatom and potential source of eicosapentaenoic acid. Bot Mar 55:1–11
Valenzuela J, Carlson RP, Gerlach R, Cooksey KE, Peyton BM, Bothner B, Fields MW (2013) Nutrient re-supplementation arrests bio-oil accumulation in Phaeodactylum tricornutum. Appl Microbiol Biotechnol 97:7049–7059
Valenzuela J, Mazurie A, Carlson RP, Gerlach R, Cooksey KE, Peyton BM, Fields MW (2012) Potential role of multiple carbon fixation pathways during lipid accumulation in Phaeodactylum tricornutum. Biotechnol Biofuels 5:1–17
Vanelslander B, Créach V, Vanormelingen P, Ernst A, Chepurnov VA, Sahan E, Muyzer G, Stal LJ, Vyverman W, Sabbe K (2009) Ecological differentiation between sympatric pseudocryptic species in the estuarine benthic diatom Navicula phyllepta (Bacillariophyceae). J Phycol 45:1278–1289
Wagenen JV, Miller TW, Hobbs S, Hook P, Crowe B, Huesemann M (2012) Effects of light and temperature on fatty acid production in Nannochloropsis salina. Energies 5:731–740
Wah NB, Ahmad AL, Chieh DC, Hwai AT (2015) Changes in lipid profiles of a tropical benthic diatom in different cultivation temperature. Asian J Appl Sci Eng 4:91–101
Wijanarko A (2011) Effect of the presence of substituted urea and also ammonia as nitrogen source in cultivation medium on Chlorella’s lipid content. In: Shaukat SS (ed) Progress in biomass and bioenergy production. InTech, pp 273–282
Windler M, Leinweber K, Bartulos CR, Philipp B, Kroth PG (2015) Biofilm and capsule formation of the diatom Achnanthidium minutissimum are affected by a bacterium. J Phycol 51:343–355
Willis A, Chiovitti A, Dugdale TM, Wetherbee R (2013) Characterization of the extracellular matrix of Phaeodactylum tricornutum (Bacillariophyceae): structure, composition, and adhesive characteristics. J Phycol 49:937–949
Witkowski A, Lange-Bertalot H, Metzeltin D (2000) Diatom flora of marine coasts I. Iconogr Diatomol 7:1–925
Wu LF, Chen PC, Lee CM (2013) The effects of nitrogen sources and temperature on cell growth and lipid accumulation of microalgae. Int Biodeter Biodegr 85:506–510
Yadavalli R, Rao R (2013) Response surface methodological approach to optimize process parameters for the biomass production of Chlorella pyrenoidosa. Int J Biotechnol Res 1:37–48
Yang ZK, Niu YF, Ma YH, Xue J, Zhang MH, Yang WD, Liu JS, Lu SH, Guan Y, Li HY (2013) Molecular and cellular mechanisms of neutral lipid accumulation in diatom following nitrogen deprivation. Biotechnol Biofuels 6:67
Yang F, Long L, Sun X, Wu H, Li T, Xiang W (2014a) Optimization of medium using response surface methodology for lipid production by Scenedesmus sp. Mar Drugs 12:1245–1257
Yang M, Zhao W, Xie X (2014b) Effects of nitrogen, phosphorus, iron and silicon on growth of five species of marine benthic diatoms. Acta Ecol Sin 34:311–319
Yongmanitchai W, Ward OP (1991) Growth of and omega-3 fatty acid production by Phaeodactylum tricornutum under different culture conditions. Appl Environ Microbiol 57:419–425
Zhang J, Fu D, Xu Y, Liu C (2012) Optimization of parameters on photocatalytic degradation of chloramphenicol using TiO2 as photocatalyst by response surface methodology. J Environ Sci 22:1281–1289
Acknowledgements
The authors acknowledge Dr. Sunitha Poulose and Dr. Sareen Sarah John for supporting analyses using Design Expert Software.
Funding
The authors acknowledge the University Grants Commission, Government of India for the financial support under the major research grant (File No. 41-568/2012 (SR)),
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Responsible editor: Santiago V. Luis
Rights and permissions
About this article
Cite this article
Sabu, S., Singh, I.S.B. & Joseph, V. Optimisation of critical medium components and culture conditions for enhanced biomass and lipid production in the oleaginous diatom Navicula phyllepta: a statistical approach. Environ Sci Pollut Res 24, 26763–26777 (2017). https://doi.org/10.1007/s11356-017-0274-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-017-0274-x