Abstract
Ionic liquids that are not that “green” to many organisms have recently been identified. This study examined the subchronic toxicity of the ionic liquid 1-octyl-3-methylimidazolium hexafluorophosphate ([omim]PF6) to earthworms (Eisenia fetida). Earthworms were exposed for a 28-day period (sampled on days 7, 14, 21, and 28) at concentrations of 0, 5, 10, 20, and 40 mg/kg. The levels of reactive oxygen species (ROS), antioxidant enzymes (superoxide dismutase (SOD), catalase (CAT), guaiacol peroxidase (POD)), detoxifying enzyme (glutathione S-transferase (GST)), lipid peroxidation, and DNA damage were measured. ROS significantly accumulated in all the treatment groups; the maximum ROS content was 51.9 % higher than the control at 40 mg/kg [omim]PF6 on day 28. Increased SOD activities attenuated over the time of exposure, while the CAT activities of the treatment groups were similar to the controls, except on day 14. Furthermore, the activities of POD and GST were stimulated. Lipid peroxidation in earthworms was not apparent at 5 and 10 mg/kg [omim]PF6 but was quite obvious at 40 mg/kg [omim]PF6. In addition, DNA damage was dose- and time-dependent. In conclusion, [omim]PF6 caused oxidative stress and genotoxicity in earthworms.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Ionic liquids (ILs) have been a “green” substitute for traditional solvents in technological and scientific applications. They are a class of chemicals consisting of an organic cation that is typically a bulky molecule, such as imidazolium, pyridinium, or alkylammonium and typical anions, which are often weakly coordinating halogen anions, such as chlorine, tetrafluoroborate, hexafluorophosphate, or bromide (Bubalo et al. 2014; Earle and Seddon 2000). These solvents possess a number of unique characteristics, such as negligible vapor pressure, high thermal stability, and excellent solvation ability (Moosavi and Daneshvar 2014). Due to these excellent properties, ILs have been claimed to be environmentally benign and have a wide range of use in various industries (Lawes et al. 2010; Li et al. 2013; MacFarlane et al. 2010; Ueno et al. 2008; Ueno and Watanabe 2011).
ILs are unlikely to be air contaminants, but when they leach from landfill or via effluents, their final destinations are water and soil. Previous studies have shown that some ILs have strong lethal effects on test organisms and that their toxicity is equal or even higher than some traditional organic solvents (Wu et al. 2013; Tsarpali and Dailianis 2015). Most research has focused on ILs in aquatic organisms, enzymes, cells, and plants (Bubalo et al. 2014; Liu et al. 2014; Liu et al. 2015; Tsarpali et al. 2015). Tsarpali et al. (2015) studied the cytotoxic, oxidative, and genotoxic effects of [bmim][BF4] and [omim][BF4] on the marine mussel Mytilus galloprovincialis at different concentrations. Deng et al. (2015) investigated the toxicity of [C4mim][Cl] toward the alga Scenedesmus quadricauda over a range of concentrations (0.1–1.0 mM). Furthermore, Li et al. (2010) and Luo et al. (2009, 2010) evaluated the toxicity of 1-methyl-3-octylimidazolium bromide ([C8mim]Br) to earthworms. According to their results, the 7- and the 14-day LC50 of [C8mim]Br were 206.8 and 159.4 mg/kg of artificial soil, respectively. [C8mim]Br was also indirectly shown to induce the formation of reactive oxygen species (ROS) and then harm the earthworms.
Earthworms account for 60–80 % of the total soil biomass in the ecosystem, and they play a critical role in the system. The Organization for Economic Co-operation and Development (OECD) has used the earthworm (Eisenia fetida) as a bioindicator for testing the toxicity of chemicals.
Little information of the biochemical responses and DNA damage in earthworms exposed to [omim]PF6 is available. Therefore, in the present study, the ROS content, antioxidant enzymes (superoxide dismutase (SOD), catalase (CAT), and guaiacol peroxidase (POD)), detoxifying enzyme (glutathione S-transferase (GST)), malondialdehyde (MDA), and comet assay were determined at sublethal dose of [omim]PF6 during a 28-day period. Recently, Guo et al. (2015) studied the content change of [C8mim]Cl in artificial soil. According to that study, on day 28, the [C8mim]Cl concentration in 5, 10, 20, and 40 mg/kg [C8mim]Cl-exposed treatments changed 1.2, 2.3, 4.2, and 4.4 %, respectively, compared to the initial content. Moreover, Stolte et al. (2011) have showed that the [PF6]− was recalcitrant to biodegradation, while only the elongated alkyl side chain (C8) can be degraded. Therefore, the content change in [omim]PF6 may be similar to that of [C8mim]Cl.
Via the investigation and discussion of the biochemical and genotoxic effects in earthworms exposed to [omim]PF6, the present study aims to provide a scientific basis and support for further evaluating the ecotoxicity of ILs.
Materials and methods
Chemicals
The [omim]PF6 (purity 99.0 %) was purchased from the Shanghai Cheng Jie Chemical Co., Ltd. (Shanghai, China). All other reagents were of analytical grade and obtained from the Sigma Chemical Co. (St. Louis, MO, USA), the Beijing Chemical Co. (Beijing, China), and the Shanghai Sangon Biological Engineering Technology and Service Co., Ltd. (Shanghai, China).
Earthworms and artificial soil
Earthworms were obtained from the earthworm farm at Shandong Agriculture University. They were matured adults with well-developed clitella. Each weighed between 300 and 600 mg and was maintained in mixed sphagnum and cattle feces for 2 weeks. OECD and International Organization for Standardization (ISO 11268-1 1993) artificial soil was used in the toxicological tests, the composition of which (dry weight) was 70 % industrial sand, 20 % kaolin, and 10 % sphagnum peat moss. The pH of the artificial soil was adjusted to 6.0 ± 0.5 by CaCO3 (OECD 1984; ISO 11268-1 1993). The earthworms were acclimatized for 24 h in artificial soil before toxification.
Experimental procedures
Conditions and treatment
Four concentrations (5, 10, 20, and 40 mg/kg) of [omim]PF6 and a control in artificial soil (dry weight) were chosen for the subchronic toxicity tests according to Li et al.(2010). There were three replicates for each concentration. Fifteen dried and weighed earthworms were added to a beaker (1000 ml, 750 g of artificial soil) that was then covered by plastic containing holes. All containers were maintained in an incubator at 20 ± 1 °C with a light-dark cycle between 12 h/12 h and 16 h/8 h for 28 days. To maintain the worms during the exposure period, the worms in each container were fed weekly on the soil surface with 0.5g wetted cattle manure per worm (Liu et al. 2009). After 7, 14, 21, and 28 days, three earthworms were sampled from each beaker. One earthworm was used for ROS; one for the antioxidant enzymes, GST, and MDA; and one for the comet assay. Each concentration had three replicates. The earthworms were removed 12 h before testing and stored in Petri dishes on filter paper soaked with normal saline (in the dark at 20 ± 1 °C) to void gut contents.
Sample preparation and measurements
ROS production was determined according to the 2′,7′-dichlorofluorescin diacetate (DCFH-DA) method (Lawler et al. 2003; Zhang et al. 2013a). Three earthworms were homogenized in 100 mM ice-cold potassium phosphate buffer (pH 7.4) at 3000×g for 10 min. Then, the supernatants were recentrifuged at 20,000×g for 10 min (Eppendorf, Centrifuge 5804), and after the pellet was resuspended, the mitochondria suspension was obtained. All the procedures described above were carried out at 4 °C. The resuspension liquid mixed with DCFH-DA at a final concentration of 2 μM was cultivated in a water bath for 30 min (37 °C). The fluorescence of the sample was detected by a fluorescence spectrophotometer (Shimadzu, RF-5301PC) at an excitation wavelength of 488 nm and an emission wavelength of 522 nm. The protein content was determined by the Sigma Bradford method (Bradford 1976). Bovine serum albumin (BSA) was used as the standard substance. The ROS levels are expressed as the fluorescence intensity per milligram of protein.
To determine the enzyme activities, MDA content, and protein content, three earthworms were homogenized in ice-cold Tris-phosphate buffer (1:9, w/v, pH 7.5). The homogenates were centrifuged at 2500×g for 5 min and then were transferred to another centrifuge tube and centrifuged again at 3000×g at 4 °C for 10 min (Eppendorf, Centrifuge 5804). Then, the supernatants were collected and used as soon as possible. The course of enzyme extraction and storage was strictly set at 4 °C. The protein content was determined by the Sigma Bradford method (Bradford 1976). BSA was used as the standard.
SOD activity was assayed by the inhibition of the photochemical reduction of nitroblue tetrazolium (NBT) (Sun et al. 1988; Song et al. 2009). The reaction mixture consisted of 50 mM phosphate buffer (pH 7.8), 130 mM methionine, 750 μM NBT, 100 μM ethylenediaminetetraacetic acid (EDTA), 20 μM riboflavin, and 50 μl of enzyme extract. Then, all the tubes were mixed thoroughly and illuminated under a 4000-lx fluorescent lamp for 20 min. The absorbance of the reaction mixture was determined at 560 nm. One unit (U) of SOD activity was expressed as the quantity of the enzyme that inhibited half of the NBT photoreduction rate.
CAT activity was determined by assessing the decrease of H2O2 absorbance at 240 nm based on Song et al. (2009). First, the H2O2-phosphate buffer ii was prepared, which included 160 μl H2O2 (30 % w/v) and 100 ml of phosphate buffer i (0.067 M, pH 7.0). Ten microliters of enzyme extract and 3 ml of phosphate buffer i were added to the reference cell. The sample cell contained 10 μl of enzyme extract and 3 ml of H2O2-phosphate buffer ii. The absorbance of H2O2-phosphate buffer ii was approximately 0.500 at 240 nm. The CAT activity was calculated as the ultraviolet absorption decrease from the decomposition H2O2 by CAT at 250 nm in unit time.
The POD activity was determined following Song et al. (2009). Guaiacol can be oxidized to dark brown by H2O2. The reaction mixture contained 50 ml of potassium phosphate buffer (100 mM, pH 6.0), 19 ml of 30 % H2O2, and 28 ml of guaiacol. The supernatant (20 μl) was added to a 3-ml reaction mixture as sample; the potassium phosphate buffer (100 mM, pH 6.0) containing the reaction mixture served as the control. The absorbance at 470 nm for 3 min was determined.
The GST activity measurement was carried out according to Habig et al. (1974) and Zhu et al. (2011) by measuring the rate of absorbance increase of the combination of GSH and 1-chloro-2,4-dinitro-benzene (CDNB). The control contained 0.2 ml of enzyme extract, 2.6 ml of phosphate buffer (0.1 M sodium phosphate buffer, pH 7.5, containing 1 mM ethylenediaminetetraacetic acid (EDTA), 0.1 mM dithiothreitol (DTT), and 0.1 mM phenylmethylsulfonyl fluoride (PMSF)), and 0.2 ml of CDNB (15 mM). The sample contained 0.2 ml of enzyme extract, 2.4 ml of phosphate buffer, 0.2 ml of CDNB (15 mM), and 0.2 ml of glutathione (15 mM). The changes in absorbance were recorded at 340 nm for 3 min. A millimolar extinction coefficient of 9.6/mM/cm was used to obtain the activity measurements.
The MDA content was measured by the thiobarbituric acid assay (Han et al. 2014). An enzyme sample (0.2 ml) was mixed with the reaction mixture (0.2 ml of 8.1 % SDS, 1.5 ml of 20 % acetic acid, pH 3.5, 1 % of TBA, and 1 ml of deionized water) and then was incubated at 90 °C for 1 h. The mixtures were allowed to cool down and were centrifuged at 3000 rpm for 15 min (Eppendorf, Centrifuge 5804). The MDA content in the supernatant was determined by measuring the absorbance at 532 nm. The molar extinction coefficient was 1.56 × 105/M/cm.
Single-cell gel electrophoresis (SCGE) was performed according to Singh et al. (1988) and Song et al. (2009) with slight modifications. Three earthworms from every treatment secreted coelomocytes spontaneously in the cell extraction. The cells were collected by centrifugation at 5315 rpm for 10 min (Eppendorf, Centrifuge 5804) and then were suspended in phosphate-buffered saline (PBS) at 4 °C for SCGE. SCGE was performed based on the methods of Singh et al. (1988) and Song et al. (2009) with slight modifications. An appropriate amount of cell suspension mixed with 500 μl of 0.5 % low melting agar (LMA) in PBS at 37 °C and 80 μl of the mixture was pipetted onto a fully solidified precoated layer of 100 μl 0.8 % normal melting agar (NMA). After solidification, a third layer of 85 μl LMA was added and covered with a coverslip before it solidified. The microslides were immerged into analysis solution (2.5 M NaCl, 10 mM Tris, 100 mM Na2EDTA (pH 10.0), 1 % Na sarcosinate, 10 % dimethyl sulfoxide (DMSO), and 1 % Triton X-100) for 1 h. After cleanup, they were incubated in 500 ml Na2EDTA and 500 ml NaOH for 20 min before electrophoresis at 25 V (300 mA) for 15 min. The microslides were then neutralized at 0.4 M Tris-HCl, dehydrated in ethanol at 4 °C for an hour, and stained with 40 μl ethidium bromide (13 mg/ml) for fluorescence microscopy analysis (Olympus BX51 fluorescence microscope). The slides were triplicates per treatment, and the images were scored using a digital imaging system.
Statistical analysis
Each treatment was performed in triplicate. All values are presented as means ± standard deviation (SD). One-way analysis of variance (ANOVA) followed by a calculation of least significant differences (LSDs) at p < 0.05 was used to evaluate the statistical significance of the results between the exposed and control groups by using SPSS 18.0. A bi-factorial ANOVA was performed by SPSS 18.0 to analyze biochemical responses in terms of the concentration, time of exposure, and their interaction. And, a post hoc test by least significant difference (LSD) calculation at p < 0.05 was preceded for bi-factorial ANOVA results. If a significant interaction between both factors exists, one-way ANOVA followed by LSD was conducted to evaluate the statistical significance of the results among the treatments at the same sample times.
Results
ROS content
Figure 1 shows that in all the [omim]PF6 treatments, the ROS content underwent a monotonic increase compared to the controls with increasing concentration and time. The maximum ROS content (2.02 × 103/mg protein) was 51.9 % higher than the control (1.33 × 103/mg protein) at 40 mg/kg on day 28. Therefore, the generation of ROS was stimulated by all treatments.
Antioxidant enzymes activities and detoxifying enzyme activity
According to Fig. 2a, the SOD activity of the exposure groups was stimulated at first and then was gradually inhibited. All of the treatments on day 7 had distinct activating effects compared with the control groups, and the maximum SOD activity was 29.6 % higher than the control. On day 14, almost no obvious difference was visible between the treatments and the control group. However, on days 21 and 28, the SOD activity inhibition of all treatment groups was pronounced compared to the control group. The minimum SOD activities on days 21 and 28 were 15.2 and 21.8 % lower than the controls, respectively.
Effect of [omim]PF6 on the enzyme activities in earthworms: a SOD activity, b CAT activity, c POD activity, and d GST activity. Pr protein. Each bar is the mean of three replicates. The error bars represent standard deviation (SD). The different letters above the columns indicate significant differences at p < 0.05 level between treatments
As shown in Fig. 2b, CAT activities in all treatment groups were the same as in the control except on day 14. On that day, the contaminated groups underwent significant inhibition compared to the control group.
In Fig. 2c, the POD activity in the lowest concentration group (5 mg/kg) was significantly stimulated on day 7 and day 14 compared to the control groups, while it was restored to the control level on day 21 and day 28. In the other treatment groups (10, 20, and 40 mg/kg), POD activity was significantly higher than the control groups, which suggested significant stimulation of the POD enzyme.
Figure 2d shows that on day 7, the GST activities of all treatment groups were not distinct from the control. After this time point, the GST activities of the treatment groups were at least 10 % higher than the control, except for the 5 mg/kg treatment group on days 14 and 21.
Oxidative damage
MDA content
Figure 3 shows that, at the early [omim]PF6 exposure time (on days 7 and 14), the MDA contents were not obviously different from the control at the lower concentrations (5 and 10 mg/kg). However, the MDA contents were higher than the control in the 20 and 40 mg/kg treatments. With longer exposure times, the MDA content in only the 40 mg/kg treatment was higher than the control; the other treatments (5, 10, and 20 mg/kg) showed no obvious change compared to the control.
DNA damage
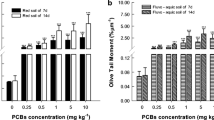
The images were obtained using the CASP software package and show the effect of [omim]PF6 on the olive tail moment (OTM) of coelomocyte DNA by [omim]PF6 as in Fig. 4. Representative pictures of the comet assay are shown in Fig. 5. Figure 4 shows that the OTM of all treatment groups was greatly elevated compared to the control groups. The elevation increased with increased concentration and extended time, which indicated that [omim]PF6 caused dose- and time-dependent DNA damage in earthworms. The maximum OTM was observed at 40 mg/kg on day 28 and was 5.97 times higher than the relative control.
Bi-factorial ANOVA results of biochemical responses
Because a significant interaction between [omim]PF6 concentration and exposure time was found (Table 1), changes in the biomarkers should be the result of the combined effects of these two factors. Both [omim]PF6 concentration and exposure time were associated with significant changes for all the biomarkers except CAT activity. As shown in post hoc test results (Table 2), exposure dose and time have a different effect on biomarkers. Under different doses, only ROS and DNA damage were significantly different. As the exposure time lengthened, ROS, MDA, and DNA damage were different from controls.
Discussion
Effects of ROS and oxidative stress
ROS, comprising superoxide anion (O2·−), hydrogen peroxide (H2O2), and hydroxyl radicals (OH·), are always present under normal conditions in organisms. Various conditions can change this balance. In these experiments, the ROS level was elevated significantly in all the [omim]PF6 treatment groups. Our results demonstrate that the ROS balance was disrupted by external [omim]PF6. A similar result was found in the study of Du et al. (2012), who studied the toxic effect of ILs on zebrafish (Danio rerio). In conclusion, ROS levels in organisms could be elevated after exposure to [omim]PF6. When the ROS cannot be scavenged by antioxidative enzymes, the excess ROS may cause oxidative stress, lipid peroxidation, and DNA damage in earthworms.
SOD is the first defense line against O2 −, catalyzing the dismutase of O2 − into H2O2 and O2 (Bowler et al. 1992; Qu et al. 2010). In the present study, on day 7, the activity of SOD in the treatment groups was significantly stimulated. Therefore, the increase in SOD activity may be due to a protective strategy against a potential increase of ROS production and an adjustment in response to the oxidative conditions (Wu et al. 2013). On days 21 and 28, SOD activities were decreased in all the treatment groups. The decrease in SOD activity may be caused by the excess ROS, which exceeded the antioxidative capacity of the earthworms (Wang et al. 2006). Previous studies of the toxic effect of ILs in Lemna minor and Physa acuta have reported similar results (Zhang et al. 2013a, b; Ma et al. 2014). Then, H2O2 was taken over by CAT. CAT is a tetrameric heme-containing enzyme with the potential to directly transform H2O2 into H2O and O2 (Blokhina et al. 2003; Gill and Tuteja 2010). The CAT activities of the treatments were the same as the control on day 7. However, they became inhibited on day 14 and then recovered to the control level. The decrease in CAT activity may be due to the stress conditions, by reducing the rate of protein turnover (Liu et al. 2011). On days 21 and 28, the CAT activities recovered to the control level, perhaps caused by an adaptive mechanism. That trend was similar to the study of Li et al. (2010), who studied the toxic effects of [C8mim]Br on earthworms. Guaiacol peroxidase (Kronfuss et al. 1996; Liu et al. 2011) is important to catalyze the removal of H2O2, and POD activities were elevated significantly during the experiment in most treatment groups. That result may be due to the differences in their mechanisms of H2O2 removal. The CAT reaction is H2O2 dismutation, while the POD reaction is an oxidation with other substrates (Vidossich et al. 2012). A significant elevation in POD activity was also found by Zhang et al. (2013a, b), who studied the toxicity of [C8mim]Br to L. minor.
Therefore, [omim]PF6 caused oxidative damage in earthworms. Furthermore, the change in antioxidant enzyme activities, not only the increase in the rate of superoxide and/or hydrogen peroxide generation, but also the decrease in the antioxidant levels could result in oxidative damage (Escobar et al. 1996).
Effects of detoxifying enzyme
GSTs can detoxify xenobiotics and endogenous metabolic by-products via enzymatic glutathione conjugation, glutathione-dependent peroxidase activity, or isomerization reactions, to protect organisms from oxidative damage (Feng et al. 1999; LaCourse et al. 2009). In our study, on day 7, the GST activities of all treatment groups were not different compared to the control. That result may suggest that GSTs could handle the excess ROS with the help of antioxidant enzymes. From day 14, GST activities were stimulated, which may be related to the excess ROS. This result was similar to Yu et al. (2008), who studied [C8mim]Br toxicity to mouse liver and found an increase in GST activity. Therefore, our study showed that [omim]PF6 could induce oxidative stress in earthworms.
Effects of oxidative damage
During lipid peroxidation (LPO), products can transform from polyunsaturated precursors including small hydrocarbon fragments such as ketones to MDA and related compounds (Garg and Manchanda 2009). The MDA content in organisms indirectly reflects the degree of intracellular injury (Bacanlı et al. 2014; Sharma et al. 2005). The MDA contents were not obviously different to the control in the lower-concentration treatments (5 and 10 mg/kg). The MDA contents in the 20 mg/kg treatment group were higher than the control on day 7 and 14 and then recovered to the control level. This pattern may be caused by the successful defense of antioxidant enzymes and detoxifying enzymes. Moreover, the MDA contents in the 40 mg/kg treatment were always higher than the control. The increase in MDA content may be attributed to the failure of antioxidant enzymes and detoxifying enzymes to defend against excess ROS. Our result was similar to Lin et al. (2012), who found that when earthworms were exposed to contamination, the content of MDA significantly increased at the higher doses but not at the lower doses. Previous studies and the present results all showed that ILs could cause LPO in earthworms.
In addition to LPO, the runaway ROS may induce many other negative effects, such as DNA strand breaks or removal of nucleotides during oxidative stress (Cooke et al. 2003). DNA containing breaks can form a comet-like image when viewed by fluorescence microscopy (Azqueta et al. 2009; Collins 2014). Single-cell gel electrophoresis (SCGE) is a useful tool to determine DNA damage (Reinecke and Reinecke 2004; Wada et al. 2014). Furthermore, the olive tail moment (OTM) of the test was chosen for evaluating DNA damage in earthworms (Olive et al. 1990; Hu et al. 2010). In Fig. 4, DNA damage showed a dose- and time-dependent effect. This result could be explained as the failure of antioxidant enzymes when defending against ROS. The results were similar to the outcomes of some former studies. Du and Dong (Dong et al. 2013; Du et al. 2012) discussed the genotoxicity of ILs to zebrafish (D. rerio). In addition, DNA damage was more sensitive than LPO.
Conclusion
The present study reveals the biochemical responses and DNA damage in earthworms exposed to [omim]PF6. At first, [omim]PF6 induced excess ROS. Then, antioxidant enzymes and detoxifying enzyme cooperated to scavenge excess ROS. In the variations of antioxidant enzymes and detoxifying enzyme, SOD, POD, and GST were more activated than CAT. The unscavenged excess ROS finally caused LPO and DNA damage in earthworms. LPO was observed at concentrations of 40 mg/kg from days 7 to 28, while DNA damage was obviously observed for all the treatment groups during the entire exposure period. In addition, SOD was more sensitive to ROS than the other enzymes studied, and the comet assay was more sensitive than MDA for the detection of oxidative damage.
References
Azqueta A, Shaposhnikov S, Collins AR (2009) DNA oxidation: investigating its key role in environmental mutagenesis with the comet assay. Mutat Res 67:101–108
Bacanlı M, Aydın S, Taner G, Göktas HG, Sahin T, Basaran AA, Basaran N (2014) The protective role of ferulic acid on sepsis-induced oxidative damage in Wistar albino rats. Environ Toxicol Pharmacol 38:774–782
Blokhina O, Virolainen E, Fagerstedt KV (2003) Antioxidants, oxidative damage and oxygen deprivation stress: a review. Ann Bot-London 91:179–194
Bowler C, Montagu MV, Inze D (1992) Superoxide dismutase and stress tolerance. Annu Rev Plant Physiol Plant Mol Biol 43:83–116
Bradford MM (1976) A rapid sensitive method for the quantification of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Bubalo MC, Radošević K, Redovniković IR, Halambek J, Srček VG (2014) A brief overview of the potential environmental hazards of ionic liquids. Ecotoxicol Environ Saf 99:1–12
Collins AR (2014) Measuring oxidative damage to DNA and its repair with the comet assay. Biochim Biophys Acta 1840:794–800
Cooke MS, Evans MD, Dizdaroglu M, Lunec J (2003) Oxidative DNA damage: mechanisms, mutation, and disease. FASEB J 17:1195–1214
Deng Y, Beadham I, Wu J, Chen XD, Hu L, Gu J (2015) Chronic effects of the ionic liquid [C4mim][Cl] towards the microalga Scenedesmus quadricauda. Environ Pollut 204:248–255
Dong M, Zhu LS, Zhu SY, Wang JH, Wang J, Xie H, Du ZK (2013) Toxic effects of 1-decyl-3-methylimidazolium bromide ionic liquid on the antioxidant enzyme system and DNA in zebrafish (Danio rerio) livers. Chemosphere 91:1107–1112
Du ZK, Zhu LS, Dong M, Wang JH, Wang J, Xie H, Zhu SY (2012) Effects of the ionic liquid [Omim]PF6 on antioxidant enzyme systems, ROS and DNA damage in zebrafish (Danio rerio). Aquat Toxicol 124–125:91–93
Earle MJ, Seddon KR (2000) Ionic liquids. Green solvents for the future. Pure Appl Chem 72:1391–1398
Escobar JA, Rubio MA, Lissi EA (1996) SOD and catalase inactivation by singlet oxygen and peroxyl radicals. Free Radical Biol Med 20:285–290
Feng QL, Davey KG, Pang ASD, Primavera M, Ladd TR, Zheng SC, Sohi SS, Retnakaran A, Palli SR (1999) Glutathione S-transferase from the spruce budworm, Choristoneura fumiferana: identification, characterization, localization, cDNA cloning, and expression. Insect Biochem Mol Biol 29:779–793
Garg N, Manchanda G (2009) ROS generation in plants: boon or bane? Plant Biosyst 143:81–96
Gill SS, Tuteja N (2010) Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants. Plant Physiol Biochem 48:909–930
Guo YY, Liu T, Zhang J, Wang JH, Wang J, Zhu LS, Yang JH (2015). Biochemical and genetic toxicity of the ionic liquid 1-octyl-3-methylimidazolium chloride on earthworms (Eisenia fetida). Eviron Toxicol Chem.
Habig WH, Pabst MJ, Jacoby WB (1974) Glutathione-S-transferases: the first enzymatic step in mercapturic acid formation. J Biol Chem 249:1730–1739
Han YN, Zhu LS, Wang JH, Wang J, Xie H, Zhang SM (2014) Integrated assessment of oxidative stress and DNA damage in earthworms (Eisenia fetida) exposed to azoxystrobin. Ecotoxicol Environ Saf 107:214–219
Hu CW, Li M, Cui YB, Li DS, Chen J, Yang LY (2010) Toxicological effects of TiO2 and ZnO nanoparticles in soil on earthworm Eisenia fetida. Soil Biol Biochem 42:586–591
ISO 11268–1 (1993) Soil quality; Biological method; Effects of pollutants on soil fauna; Part 1: determination of acute toxicity to earthworms (Eisenia fetida) using artificial soil substrate
Kronfuss G, Wieser G, Havranek WM, Polle A (1996) Effects of ozone and mild drought stress on total and apoplastic guaiacol peroxidase and lipid peroxidation in current-year needles of young Norway spruce (Picea abies L., Karst.). J Plant Physiol 148:203–206
LaCourse EJ, Viadel MH, Jefferies JR, Svendsen C, Spurgeon DJ, Barrett J, Morgan AJ, Kille P, Brophy PM (2009) Glutathione transferase (GST) as a candidate molecular-based biomarker for soil toxin exposure in the earthworm Lumbricus rubellus. Environ Pollut 157:2459–2469
Lawes SDA, Hainsworth SV, Blake P, Ryder KS, Abbott AP (2010) Lubrication of steel/steel contacts by choline chloride ionic liquids. Tribol Lett 37(2):103–110
Lawler JM, Song W, Demaree SR (2003) Hindlimb unloading increases oxidative stress and disrupts antioxidant capacity in skeletal muscle. Free Radical Biol Med 35:9–16
Li XY, Luo YR, Yun MX, Wang J, Wang JJ (2010) Effects of 1-methyl-3-octylimidazolium bromide on the antioxidant system of earthworm. Chemosphere 78:853–858
Li H, Imai Y, Takiue T, Matsubara H, Aratono M (2013) Effect and mixing of counter anions at the surface of aqueous solution of imidazolium-based ionic liquids. Colloids and Surfaces A: Physicochem Eng Aspects 427:26–32
Lin DS, Zhou QX, Xu YM, Chen C, Li Y (2012) Physiological and molecular responses of the earthworm (Eisenia fetida) to soil chlortetracycline contamination. Environ Pollut 171:46–51
Liu S, Zhou QX, Wang YY (2011) Ecotoxicological responses of the earthworm Eisenia fetida exposed to soil contaminated with HHCB. Chemosphere 83:1080–1086
Liu HJ, Zhang XQ, Chen CD, Du ST, Dong Y (2015) Effects of imidazolium chloride ionic liquids and their toxicity to Scenedesmus obliquus. Ecotoxicol Environ Saf 122:83–90
Liu T, Zhu LS, Xie H, Wang JH, Wang J, Sun FX, Wang FH (2014) Effects of the ionic liquid 1-octyl-3-methylimidazolium hexafluorophosphate on the growth of wheat seedlings. Environ Sci Pollut Res 21:3936–3945
Liu XL, Sun ZJ, Chong W, Sun ZT, He CK (2009) Growth and stress responses of the earthworm Eisenia fetida to Escherichia coli O157:H7 in an artificial soil. Microb Pathogenesis 46:266–272
Luo YR, Wang SH, Yun MX, Li XY, Wang JJ, Sun ZJ (2009) The toxic effects of ionic liquids on the activities of acetylcholinesterase and cellulase in earthworms. Chemosphere 77:313–318
Luo YR, Wang SH, Li XY, Yun MX, Wang JJ, Sun ZJ (2010) Toxicity of ionic liquids on the growth, reproductive ability, and ATPase activity of earthworm. Ecotoxicol Environ Saf 73:1046–1050
Ma JG, Dong XY, Fang Q, Li XY, Wang JJ (2014) Toxicity of imidazolium-based ionic liquids on Physa acuta and the snail antioxidant stress response. J Biochem Mol Toxicol 28:69–75
MacFarlane DR, Pringle JM, Howletta PC, Forsytha M (2010) Ionic liquids and reactions at the electrochemical interface. Phys Chem Chem Phys 12:1659–1669
Moosavi M, Daneshvar A (2014) Investigation of the rheological properties of two imidazolium-based ionic liquids. J Mol Liq 190:59–67
OECD (1984) The current Organization of Economic and Cooperative Development acute earthworm toxicity test (OECD). Guidelines for the testing of chemicals, no. 207., Earthworm acute toxicity tests
Olive PL, Banath JP, Durand RE (1990) Heterogeneity in radiation induced DNA damage and repair in tumor and normal cells measured using the Comet assay. Radiat Res 122:86–94
Qu BC, Zhao HX, Zhou JT (2010) Toxic effects of perfluorooctane sulfonate (PFOS) on wheat (Triticum aestivum L.) plant. Chemosphere 79:555–560
Reinecke SA, Reinecke AJ (2004) The comet assay as biomarker of heavy metal genotoxicity in earthworms. Arch Environ Contam Toxicol 46:208–215
Sharma Y, Bashir S, Irshad M, Nag TC, Dogra TD (2005) Dimethoate-induced effects on antioxidant status of liver and brain of rats following subchronic exposure. Toxicology 215:173–181
Singh NP, McCoy MT, Tice RR, Schneider EL (1988) A simple technique for quantitation of low levels of DNA damage in individual cells. Exp Cell Res 175:184–191
Song Y, Zhu LS, Xie H, Wang J, Wang JH, Liu W, Xie H (2009) DNA damage and effects on antioxidative enzymes in earthworm (Eisenia fetida) induced by atrazine. Soil Biol Biochem 28:1059–1062
Stolte S, Stephanie S, Amaya I, Piotr S (2011) The Biodegradation of ionic liquids—the view from a chemical structure perspective. Curr Org Chem 15:1946–1973
Sun Y, Oberley LW, Li Y (1988) A simple method for clinical assay of superoxide dismutase. Clin Chem 34:497–500
Tsarpali V, Belavgeni A, Dailianis S (2015) Investigation of toxic effects of imidazolium ionic liquids, [bmim][BF4] and [omim][BF4], on marine mussel Mytilus galloprovincialis with or without the presence of conventional solvents, such as acetone. Aqua Toxicol 164:72–80
Tsarpali V, Dailianis S (2015) Toxicity of two imidazolium ionic liquids, [bmim][BF4] and [omim][BF4], to standard aquatic test organisms: role of acetone in the induced toxicity. Ecotoxicol Environ Saf 117:62–71
Ueno K, Hata K, Katakabe T, Kondoh M, Watanabe M (2008) Nanocomposite ion gels based on silica nanoparticles and an ionic liquid: ionic transport, viscoelastic properties, and microstructure. J Phys Chem B 112(30):9013–9019
Ueno K, Watanabe M (2011) From colloidal stability in ionic liquids to advanced soft materials using unique media. Langmuir 27:9105–9115
Vidossich P, Alfonso-Prieto M, Rovira C (2012) Catalases versus peroxidases: DFT investigation of H2O2 oxidation in models systems and implications for heme protein engineering. J Inorg Biochem 117:292–297
Wada K, Yoshida T, Takahashi N, Matsumoto K (2014) Effects of seven chemicals on DNA damage in the rat urinary bladder: a comet assay study. Mutat Res 769:1–6
Wang XF, Xing ML, Shen Y, Zhu X, Xu LH (2006) Oral administration of Cr(VI) induced oxidative stress, DNA damage and apoptotic cell death in mice. Toxicology 228:16–23
Wu X, Tong ZH, Li LL, Yu HQ (2013) Toxic effects of imidazolium-based ionic liquids on Caenorhabditis elegans: the role of reactive oxygen species. Chemosphere 93:2399–2404
Yu M, Li SM, Li XY, Zhang BJ, Wang JJ (2008) Acute effects of 1-octyl-3- methylimidazolium bromide ionic liquid on the antioxidant enzyme system of mouse liver. Ecotoxicol Environ Saf 71:903–908
Zhang QM, Zhu LS, Wang J, Xie H, Wang JH, Han YN, Yang JH (2013a) Oxidative stress and lipid peroxidation in the earthworm Eisenia fetida induced by low doses of fomesafen. Environ Sci Pollut Res 20:201–208
Zhang BJ, Li XY, Chen DD, Wang JJ (2013b) Effects of 1-octyl-3-methylimidazolium bromide on the antioxidant system of Lemna minor. Protoplasma 250:103–110
Zhu LS, Dong XL, Xie H, Wang J, Wang JH, Su J, Yu CW (2011) DNA damage and effects on glutathione-S-transferase activity induced by atrazine exposure in zebrafish (Danio rerio). Environ Toxicol 26:480–488
Acknowledgments
The present study was supported by grants from the National Natural Science Foundation of China (No. 21277083 and 21377075).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Responsible editor: Cinta Porte
Rights and permissions
About this article
Cite this article
Liu, X., Zhang, S., Wang, J. et al. Biochemical responses and DNA damage in earthworms (Eisenia fetida) induced by ionic liquid [omim]PF6 . Environ Sci Pollut Res 23, 6836–6844 (2016). https://doi.org/10.1007/s11356-015-5827-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-015-5827-2