Abstract
The heavy metal and polycyclic aromatic hydrocarbons (PAHs) contents were evaluated in surface soil and plant samples of 18 wild species collected from 3 typical industrial sites in South Central China. The accumulative characteristics of the plant species for both heavy metal and PAHs were discussed. The simultaneous accumulation of heavy metal and PAHs in plant and soil was observed at all the investigated sites, although disparities in spatial distributions among sites occurred. Both plant and soil samples were characterized by high accumulation for heavy metal at smelting site, moderate enrichment at coke power and coal mining sites, whereas high level of PAHs (16 priority pollutants according to US Environmental Protection Agency) at coke power site, followed sequentially by coal mining and smelting sites. Based on the differences of heavy metal and PAH accumulation behaviors of the studied plant species, heavy metal and PAH accumulation strategies were suggested: Pteris vittata L. and Pteris cretica L. for As and PAHs, Boehmeria nivea (L.) Gaud for Pb, As, and PAHs, and Miscanthus floridulu (Labnll.) Warb for Cu and PAHs. These native plant species could be proposed as promising materials for heavy metal and PAHs combined pollution remediation.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Industrial contamination has long been recognized as priority environmental concern in the process of industrialization and urbanization. The European environmental protection agency in 2006 identified 3,000,000 potential industrial contaminated sites in Europe, 83 % of which would pose health threat to human beings (EEA 2007). In the USA, 1,536 industrial contaminated sites were classified as priority remediation sites during 1982–2005 (US EPA 2004). It is reported that Pb, As, Cr, Cd, Zn, polycyclic aromatic hydrocarbons (PAHs), etc. ranked the top 12 contaminants of concern at 568 Superfund sites in the USA (US EPA 2002). The simultaneous accumulation of heavy metals and PAHs have gained considerable attention since soils contaminated with PAHs were frequently reported to contain high amounts of heavy metal (Achten and Hofmann 2009; Wang et al. 2009; Sun et al. 2013). Several studies have demonstrated that the combination of these two types of contaminants could potentiate great environmental risk to vegetation, soil microbial, and human health (Maier et al. 2002).
Phytoremediation has been proposed as promising technology to treat a wide range of contaminants and applied well in many contaminated sites (US EPA 2005). Generally, collection of plant species with genetic ability to survive, reproduce in barren site, and tolerate excessive toxic substance plays a critical role in the success of phytotechnology application (Parizanganeha et al. 2010). So far, a number of metal-tolerant plant species have been identified to be efficient in heavy metal accumulation. Some plants have also been demonstrated for the phytoremediative potentials of PAHs (Sojinua et al. 2010). Recently, several researches have paid attention to screening effective plant species to remediate heavy metal and PAH combined pollution, most of which were based on the pot culture study. For a few field trials, the test subject was often the introduced plant species rather than the native or naturalized species (Vervaeke et al. 2003; Sébastien et al. 2005; Sun et al. 2011). Whether native plant species is of great significance in phytoremediation for heavy metal and PAH combined pollution was seldom reported. In fact, it is increasingly believed that plant species endemic to contaminated site would have excellent potential for phytoremediation with its adaptation to growth conditions (Parizanganeha et al. 2010). Thus, identifying and collecting plant species spontaneously grown in industrial areas, which are resistant to metals and PAHs co-exposure, and can accumulate considerable amounts of them, would facilitate the phytoremediation technology development.
China is one of the world’s largest coal and metallic mineral producer and consumer. Because of the rapid industrial growth in China over the past decades, the number of industry enterprises has reached 375,000 in 2008, and the pillar industry including coke power, coal mining, and metal smelting accounted for 12.5 % of the total (NBS 2010). The industries of coke power, coal mining, and metal smelting are generally recognized as the main heavy metal and PAH pollution sources (Cheng 2003; Achten and Hofmann 2009; Wang et al. 2009). However, limited information is available on the characterization of heavy metal and PAH pollution at these sites in China. In this study, heavy metal and PAHs of soil and plant samples collected from coke power, metal smelting, and coal mining sites were investigated. The main objectives were (1) to identify and characterize heavy metal and PAHs distribution and potential sources of three industrial sites and (2) to evaluate the phytoremediation potential of endurant plant species for heavy metal and PAH combined pollution.
Materials and methods
Site description and sampling
This study was conducted in industrial region in Chenzhou city, Hunan province in South Central China, which has long been recognized as the base of nonferrous metals and energy. The climate of the study area is typically subtropical with a mean annual rainfall around 1,500 mm and temperature of 17.4 °C. The mean altitude above the sea level is 400 m. Two large coke power stations, three smelting factories, and two coal mining workings are located (Fig. 1). The coke power plants are important producers of electricity and coal tar in Chenzhou. The main products of the smeltery are Pb, Zn, Cu, Au, and Ag. The surface coal mining is on the basis of Bituminous coal. Two areas in the proximity of the coke battery for coke power site, three areas nearby the smelting furnace of smelting site, and two areas in the vicinity of coal washing poor and coal picker for coal mining site, each of about 25 × 25 m2, were selected for soil and plant sampling. Soil samples of references were collected from an unaffected area far from the influence of the anthropogenic activities. Forty-nine surface (0–20 cm) soil samples were collected, and each soil sample was gathered and mixed properly by five subsamples collected randomly from the surroundings following different directions. Among 49 soil samples, 12 were collected from coke power site, 18 from smelting site, 13 from coal mining site, and 6 from reference area.
Plant species sampling was performed in the vicinity of soil sampling points where the plants were grown. At least three or five individual plants of each plant species were collected randomly within the sampling area, and then they were mixed to give a composite plant sample for each species. The corresponding soil samples were also a mixture of single samples taken from three to five locations where the plants were sampled. The identification of species was realized with the help from the Institute of Botany, Chinese Academy of Sciences. Plant samples were cleaned with tap water and distilled water and then prepared for heavy metal and PAH analysis. Soil was dried and sieved through a 2-mm screen for analysis.
Chemical analysis
For heavy metal analysis, the dried and ground plant samples were digested with HNO3–HClO4, while soil samples were digested with HNO3–H2O2 (EPA 3050B). The elements of Cu, Pb, Zn, and Cd were determined by inductively coupled plasma optical emission spectrometry (5300DV, PerkinElmer U. S.). As was determined using atomic fluorescence spectroscopy (9130, Beijing Titan Instruments Co., China). Accuracy of the elemental analysis was verified by standard reference material. The results showed a recovery of Cu, Pb, Zn, Cd, and As in the range of 90.6–103.3 %, 93.4–106.8 %, 89.2–98.5 %, 95.7–112.4 %, and 95.6–109.6 %.
For PAH analysis, certain amounts of freeze-dried soil or plant fragment were extracted by ultrasonication with dichloromethane or acetone–dichloromethane mixture. The extracts were concentrated and then transferred to silica gel column for cleanup by washing with hexane and dichloromethane mixture. The eluate was concentrated and analyzed by Agilent GC-MS [Agilent 7890A gas chromatography (GC) coupled with a 5975C mass spectroscopy (MS) system]. A mixture of PAH containing 16 compounds were determined: naphthalene, acenaphthylene, acenaphthene, fluorine, phenanthrene, anthracene, fluoranthene, pyrene, benz[a]anthracene, chrysene, benzo[b]fluoranthene, benzo[k]fluoranthene, benzo[a]pyrene, indeno[1,2,3-cd]pyrene, dibenz[a,h]anthracene, and benzo[ghi]perylene.
Data analysis
Statistical analysis was performed using SPSS 13.0. The difference of heavy metal and ∑16 PAHs concentrations in soil and plant samples among different sampling sites was determined by the Kruskal–Wallis test. Spearman’s correlation was conducted to examine the relationships among heavy metal and PAHs in soil and plant samples. A probability of 0.05 or lower was considered as significant. Principal component analysis (PCA) was performed to investigate the relationship between plant species and contaminant concentrations.
Results and discussion
The concentrations of As, Cd, Cu, Pb, Zn, and ∑16 PAHs in soil samples are listed in Table 1. According to the heavy metal contents determined in the reference area, which were in line with the corresponding natural background values in Hunan province (As, Cd, Cu, Pb, and Zn at the levels of 13.6, 0.079, 25.4, 27.3, and 88.6 mg/kg, respectively) (MEP 1990), the total metal concentrations in the soil samples of industrial sites were higher than the background values by approximately 1 to several hundred times based on the mean concentration. Compared with ∑16 PAHs levels determined in the reference area, which were below the documented standard in Dutch (1 mg/kg), the contents of ∑16 PAHs in the soil samples of industrial sites exceeded the background level by approximately 78 times at coke power site, about 32.5 times at coal mining site, and more than 20 times at smelting site. The concentrations of heavy metals and ∑16 PAHs in soils of the study area were among the high level compared to the reported values for other contaminated soils at industrial sites in the literature, which clearly highlighted the strong anthropogenic input from industrial processing activities on these spots.
Variation of industrial pollution feature was observed at three types of sites. In general, the soil heavy metal concentrations were found to decrease in the order smelting site > coal mining site > coke power site; while in contrast, ∑16 PAHs concentrations were highest at coke power site, and followed sequentially by coal mining and smelting sites. The polymetallic pollution was observed due to As, Cd, Cu, Zn, and Pb at smelting site with the concentration median values of 251, 34.9, 851, 2,315, and 4,610 mg/kg, amounting several orders of magnitude higher than those at coal mining and coke power sites (P < 0.01). The primary metal-bearing minerals for smelting in the study area are galena (PbS), sphalerite (ZnS), chalcopyrite (CuFeS2), and arsenopyrite (FeAsS). The high concentrations of As, Cd, Cu, Zn, and Pb in the soil are probably due to airborne emissions, transportation, and deposition of volatile particles and aerosols from stack emission or the fugitive emissions. High As level was recorded at coal mining site, with the highest concentration of 127 mg/kg. He et al. (2002) reported that coal mines undergone mineralization could result in enrichment of toxic trace elements, e.g., arsenic and fluorine. It was speculated that the local coal as potential pollution source would result in emission of unburnt coal particles containing As in the process of coal mining, storage, and transportation. The heavy metal concentrations were relatively lower at coke power site; however, compared to the background, the enrichment of Pb was obvious (P < 0.05).The enrichment of heavy metal in soil was also observed at several coal-based electricity production sites where heavy metals are released into the atmosphere during the combustion of fossil fuels, as well as from high temperature industrial processes and ultimately accumulate in the surface soil (Cheng 2003; Petaloti et al. 2006; Shukurov et al. 2009). Therefore, the combustion for power generation can be interpreted as main heavy metal source at coke power site.
The ∑16 PAHs concentrations at industrial sites ranged from 215 μg/kg (smelting site) to 32,474 μg/kg (coke power site). Soils collected from the coke power and coal mining sites showed higher ∑16 PAHs concentrations than those from smelting site (P < 0.05). This is in accordance with previous findings that coal combustion for power generation and coal mining activities are likely the main sources of PAHs (Musa et al. 2010). The contribution of the two- and three-ring PAHs to ∑16 PAHs were <50 % (9.6–46.2 %) at coke power site, whereas they were >50 % at smelting (50.8–86.9 %) and coal mining sites (50.8–79.8 %) (Fig. 2). The mixtures of ∑16 PAHs in soil were dominated by fluoranthene (13.8–23.2 %) and pyrene (9.3–16.8 %) at coke power site. The predominant PAH species were naphthalene (12.1–28.1 %), acenaphthene (8.0–26.7 %), and phenanthrene (7.8–18.8 %) at smelting site, and phenanthrene (12.5–36.5 %) and acenaphthene (9.2–38.2 %) at coal mining site. It is suggested that higher molecular weight PAHs are deposited near coal combustion emission sources for power generation as a consequence of lower vapor pressure (Bakker et al. 2000). The local Bituminous coal dust with distinct low molecular weight aromatic compounds emanating from the coal mining activities would contribute to higher contents of two- and three-ring PAHs in coal mining site.
Heavy metal in plant
Eighteen plant species from 16 families were found in the studied area (Table 2). These plant species are perennials belonging to herb, shrub, and tree. The ferns of Pteris vitatta, Pteris cretica, and Cyclosorus parasiticus, and the grass of Boehmeria nivea dominate the sampling areas at three industrial sites. Non-endemic tree species of Cunninghamia lanceolata, and Broussonetia papyrifera are able to grow on coke power and coal mining sites. However, they all show a leafy crown and thick trunks with clear signs of poor growth.
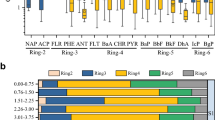
Heavy metal contents in aerial part of plant samples showed significant differences among industrial sites except As (P < 0.01) (Table 2). Chaney (1989) reported that heavy metal concentrations in plant growing on uncontaminated sites were generally in the range of 0.01–1.0 mg/kg for As, 0.05–0.2 mg/kg for Cd, 0.1–10 mg/kg for Pb, 27–150 mg/kg for Zn, and 5.0–30 mg/kg for Cu, respectively. A comparison of these known background values with determined metal levels in plant samples of industrial sites indicates that plant samples collected from the coke power and coal mining sites had the normal concentrations for Cd, Cu, Pb, and Zn, while As (0.89–381 mg/kg) was relatively higher in current study. Plants collected from the smelting site were characterized by high levels of As, Cd, Cu, Pb, and Zn with the maximum concentrations reaching 1,276, 34.8, 1,187, 4,430, and 6,565 mg/kg, respectively. Analytical results for comparing heavy metal composition of both plant and corresponding soil samples of three industrial sites are given in Fig. 3. It was shown that the order of metal concentration in plant samples followed a similar pattern to that in soil samples, reflecting the accumulative indication potential of these wild plants, which was also reported in the corresponding studies (Guo et al. 2009; Franco-Hernández et al. 2010). However, no significant correlation between plant and soil heavy metal content was found (P > 0.05).
Box plot of log transformation value of heavy metal in plant and corresponding soil samples at three industrial sites. The whiskers from the upper and lower edge of the box define the range of all the data; the central line inside the box represents the median; data outliers are marked by circles (N1.5 × hinge spread) and extreme outliers by stars (N3 × hinge spread)
Plant As content varied between species, but independently of the soil As concentration, the species P. vitatta had the highest capacity to accumulate As with the maximum content of 1,276 mg/kg in shoots. Furthermore, high As accumulation levels were also measured in P. cretica, B. nivea, and Selaginella davidii, with their shoots As concentrations up to 216, 360, and 169 mg/kg, respectively. Regarding the plant accumulation of Cd, Equisetum hyemale and P. vitatta had significantly higher concentrations with the values of 34.8 and 14.0 mg/kg, respectively. Efficient accumulation of Cu was found in Miscanthus floridulu (1,187 mg/kg), E. hyemale (265 mg/kg), Polygonum pubescens (113 mg/kg), and P. vitatta (107 mg/kg). Specially, it is worth noting that Cu concentration in aerial part of M. floridulu reached the criterion for considering a hyperaccumulator plant (1,000 mg/kg) (Brooks et al. 1998). Considerable amounts of Zn and Pb were accumulated in more species, exactly in plant species from the smelting industrial site, where maximum of Pb (4,430 mg/kg) and Zn (6,565 mg/kg) were found concurrently in E. hyemale. The calculated bioconcentration factor (BCFs), defined as the concentration ratio of heavy metal in plant aerial part to that in soil, demonstrated that most of the collected plants behaved as endurant excluder with low BCFs (≤1), except that P. vitatta (1.61–6.53), P. cretica (1.27–4.60), and B. nivea (1.99–4.59) had relatively high BCFs for As, and B. nivea had high BCFs for Pb (2.07–3.10).
The excellent As-accumulating ability of P. vitatta, P. cretica, and B. nivea, and Pb-accumulating ability of B. nivea have been well recognized (Wei and Chen 2002; Zhao et al. 2002; Yang et al. 2010), which was also demonstrated in the present study. Although soil with high level of combined pollution would restrict the plant accumulation ability, it seemed that several plant species possess a certain degree of constitutional metal tolerance and accumulating potential for various heavy metals. For instance, P. vitatta was able to accumulate considerable amounts of heavy metals such as As, Cd, Cu, Pb, and Zn. B. nivea was efficient in accumulating As, Pb, and Zn; P. cretica for As and Pb; and M. floridulu for Cu, Pb, and Zn.
PAHs in plant
It is generally believed that the uptake of PAHs by plant is primarily from atmosphere through gas and particle-bound depositions (Slaski et al. 2000).
Thus, ∑16 PAHs concentrations in plant aerial part samples are shown in Table 2. The aerial part of wild plants showed similar PAHs spacial distribution pattern to soil, recurring in high ∑16 PAHs amount in plant samples of the coke power and coal mining sites relative to those of smelting site. Furthermore, there was a significant variation of PAHs profiles in plant samples of three sites (Fig. 4). The levels of four-, five-, and six-ring PAHs in plant samples were much higher at coke power site (50.4–72.0 %), whereas two- and three-ring PAHs were much higher at smelting (50.4–97.0 %) and coal mining (68.6–89.1 %) sites, which was consistent with PAHs distribution in soil samples. However, no significant correlation between plant and soil ∑16 PAHs content was found (P > 0.05). Furthermore, the PAH profiles were not much similar for the plant and soil samples. The concentrations of two- and three-ring PAHs were relatively higher, and four- and six-ring PAHs were lower in plant samples than those of soils. The phenomenon has also previously been described by Bakker et al. (2000) and Sojinua et al. (2010), who proposed that this could be attributed to wind drift and wash-off of the high molecular compounds from the leaves or the low absorptivity of these classes of compounds.
The species B. nivea, P. vitatta, and P. cretica with rough or hairy leaf surfaces, larger exposed foliage surface area, or the presence of trichome were more efficient in accumulating and retaining high-molecular PAHs. S. bungeana and M. floridulu with the characteristics of thin layer of cutile or smooth and sharp leaf were observed to have higher concentrations of low-molecular PAHs (Table 2). B. nivea showed the strongest accumulation of PAHs with the value of 2,136 μg/kg at coke power site, especially four-, five-, and six-ring PAHs (71.9 %). Researchers defined that plants with hairy foliage would be more efficient in accumulating PAHs especially the high molecular weight PAHs than the hairless foliage (Howsam et al. 2000; Mikefryer and Agrosso 2006). Consequently, the rough leaf surface and the large leaf area might contribute to the high PAHs accumulation in B. nivea. Brighigna et al. (2002) and Pereira et al. (2007) demonstrated that the trichomes, which increase the plant–air interfaces, play an important role in retaining the particles such as the particle-bound PAHs. It is well known that P. vitatta and P. cretica are abundant of trichomes (Xie et al. 2009), which could be one reason for the excellent capacity of the two ferns in accumulating PAHs. Mclachlan et al. (1995) and Sojinua et al. (2010) considered that the plant species belonging to Gramineae with the characteristics of succulent, porous stems or smooth and sharp leaf were potential accumulating material for low molecular PAHs, which was also observed in the current study that the concentrations of PAHs were relatively higher in S. bungeana (980 μg/kg) and M. floridulu (795 μg/kg) at smelting site.
Plants can reduce the amount of PAHs by gaseous diffusion and intercepting particles on their leaf and bark surfaces. Simonich and Hites (1995) remarked that, on average, 40 % of emitted PAHs were removed by plants with higher PAH burdens in areas adjacent to local sources. Sojinua et al. (2010) also demonstrated the bioconcentration potentials of plant aerial parts for PAHs at polluted sites. In the present study, ∑16 PAHs concentrations in the species B. nivea, P. vitatta, P. cretica, and S. bungeana were relatively higher than the values determined in the soils from the same location, indicating the bioaccumulation and phytoremediation potentials of these species under investigation.
Heavy metal and PAH combined accumulation
Correlation analysis was applied to investigate the relationship among heavy metal and ∑16 PAHs in soil and plant samples (Table 3). Generally, there was significant positive correlation between Pb and ∑16 PAHs concentrations (P < 0.05) in soil samples regardless of industrial sites. In addition, ∑16 PAHs was positively correlated with As and Zn (P < 0.05) at coke power site, while positively correlated with Cu and Zn (P < 0.05) at smelting site. The significant positive correlations between ∑16 PAHs and various heavy metals produced from industrial emissions indicate the common source of PAHs and these metals in soil (Musa et al. 2010; Fabietti et al. 2010). Significant positive correlations among As, Pb, Zn, and ∑16 PAHs (P < 0.05) in plant samples were evident at coke power site, which was in accordance with those in soil. At coal mining site, ∑16 PAHs closely positively related to the element of As. No significant correlation between ∑16 PAHs and heavy metals was observed for plants at smelting site.
PCA based on heavy metal and ∑16 PAH concentrations in plant samples was employed for the plant accumulation characteristics evaluation. For coke power and coal mining sites, two factors explaining 92.5 and 87.4 % of the total variance were extracted, respectively. Three components were extracted covering 90.9 % of the data variance for smelting site. The results obtained further showed a positive association among the contents of As, Pb, Zn, and ∑16 PAHs in plants samples at coke power site. The contents of ∑16 PAHs in plants were associated with Cu at smelting site while they were positively correlated with As at coal mining site (Table 4).
The scatter plot of the component scores of plant species is depicted in Fig. 5. At coke power site, the PC1-positive loadings for P. vitatta, P. cretica, and B. nivea indicated main accumulation of As, Pb, Zn, and ∑16 PAHs, with average contents of As, Pb, Zn, and ∑16 PAHs relatively higher than other species. At coal mining site, the distribution in PC2 showed that P. vitatta, P. cretica, B. nivea, and P. asiatica accumulated statistically higher As and PAHs. In particular, B. nivea was characterized as high accumulation for As, Pb, Cu, Zn, and ∑16 PAHs as all the data were in the positive PC sectors both at coke power and coal mining sites. At smelting site, M. floridulu showed high positive loading in PC3 indicative of the high accumulation for Cu and PAHs. P. cretica and S. bungeana showed accumulation of As, Cu, and PAHs due to the positive loading in PC2 and PC3. P. vitatta was characterized as accumulator for As, Cd, Pb, and Zn with positive loading in PC1 and PC2.
Based on the results obtained from PCA, together with heavy metal and PAHs contents in plant samples, the accumulation potential of plant species was compared. Generally, the species of P. vitatta, P. cretica, and B. nivea with significantly higher contents of As and PAHs wherever these species were located showed excellent As accumulating capacity and positive potential for PAHs uptake. In addition, P. vitatta was capable of simultaneously accumulating high levels of As, Pb, Zn, and PAHs. The species B. nivea was characterized by high accumulation ability for Pb, As, and PAHs. M. floridulu showed the maximum and significant Cu accumulation and a relative accumulation capacity for PAHs. It is well recognized that the promising plant material for phytoremediation would be characterized by the applicability to extensive geographical areas in abundant communities, the resistance to combined pollution, and great phytoaccumulating potential for contaminants (Jørgensen et al. 2005). The species P. vitatta, P. cretica, B. nivea, and M. floridulu are generally identified as growing widely and colonizing contaminated sites in South Central China. The applications of these plant species for phytoremediation of heavy metal contamination have also been documented (Wei and Chen 2002; Yang et al. 2010). Recently, a variable of researches have demonstrated that, even at low doze, the combination of heavy metal and PAHs could strongly cause oxidative stress and cellular organelle deformation in plant, resulting in the suppression of heavy metal uptake (Maier et al. 2002; Li et al. 2010). In the present study, it seemed that, at the levels of heavy metal and PAHs determined in plant species, these indigenous species were tolerant to high heavy metal and PAH co-exposure with good growth conditions and showed high accumulation potential for various heavy metals and PAHs and, therefore, would serve as good phytoremediators for heavy metal and PAH combined pollution.
Conclusion
The present study clearly demonstrates that endemic plant species growing at the industrial sites can colonize and survive in soils with multiple heavy metals and PAHs. Although spatial distributions of the heavy metal and PAHs varied greatly among industrial sites, it is possible to conclude that the species P. vitatta, P. cretica, B. nivea, and M. floridulu have potential in co-exposure tolerating and accumulating heavy metals and PAHs. Based on the examination of plant species for possible application of phytoremediation technologies, the plant strategy indicated that P. vitatta and P. cretica would be the promising remediation material for As and PAH combined pollution. B. nivea would be a candidate for Pb, As, and PAH combined pollution remediation while M. floridulu for Cu and PAH combined pollution remediation. It should also be noted that plants usually have significant metabolic activities in the rhizosphere for degrading PAHs. Thus, field-scale studies of heavy metal and PAH removal efficiency in soil should also be undertaken to further evaluate the phytoremediation capability of these plant species.
References
Achten C, Hofmann T (2009) Native polycyclic aromatic hydrocarbons (PAH) in coals—a hardly recognized source of environmental contamination. Sci Total Environ 407:2461–2473
Bakker MI, Casado B, Koerselman JW, Tolls J, Kollöffel C (2000) Polycyclic aromatic hydrocarbons in soil and plant samples from the vicinity of an oil refinery. Sci Total Environ 263:91–100
Brighigna L, Papini A, Mosti S, Cornia A, Bocchini P, Galletti G (2002) The use of tropical bromeliads (Tillandsia spp.) for monitoring atmospheric pollution in the town of Florence, Italy. Rev Biol Trop 50:577–584
Brooks RR, Chambers MF, Nicks LJ, Robinson BH (1998) Phytomining. Trends Plant Sci 3:359–362
Chaney RL (1989) Toxic element accumulation in soils and crops: protecting soil fertility and agricultural food chains. In: Bar-Yosef B, Barrow NJ, Goldshmid J (eds) Inorganic contaminants in the vadose zone. Springer, Berlin, pp 140–158
Cheng SP (2003) Heavy metal pollution in China: origin, pattern and control. Environ Sci Pollut R 10:192–198
EEA (2007) Progress in management of contaminated sites (CSI 015). European Environment Agency, Copenhagen, Denmark
Fabietti G, Biasioli M, Barberis R, Ajmone-Marsan F (2010) Soil contamination by organic and inorganic pollutants at the regional scale: the case of Piedmont, Italy. J Soil Sed 10:290–300
Franco-Hernández MO, Vásquez-Murrieta MS, Patiño-Siciliano A, Dendooven L (2010) Heavy metals concentration in plants growing on mine tailings in Central Mexico. Bioresour Technol 101:3864–3869
Guo ZH, Megharaj M, Beer M, Ming H, Rahman MM, Wu WH, Naidu R (2009) Heavy metal impact on bacterial biomass based on DNA analyses and uptake by wild plants in the abandoned copper mine soils. Bioresour Technol 100:3831–3836
He B, Liang LN, Jiang GB (2002) Distributions of arsenic and selenium in selected Chinese coal mines. Sci Total Environ 296:19–26
Howsam M, Jones KC, Meson P (2000) PAHs associated with the leaves of three deciduous tree species. I—concentrations and profiles. Environ Pollut 108:413–424
Jørgensen SE, Costanza R, Xu FL (2005) Handbook ecological indicators for assessment of ecosystem health. CRC Press/Taylor & Francis, Boca Raton
Li Q, Lauer FT, Liu KJ, Hudson LG, Burchiel SW (2010) Low-dose synergistic immunosuppression of T-dependent antibody responses by polycyclic aromatic hydrocarbons and arsenic in C57BL/6J murine spleen cells. Toxicol Appl Pharmacol 245:344–351
Maier A, Schumann BL, Chang XQ, Talaska G, Puga A (2002) Arsenic co-exposure potentiates benzo[a]pyrene genotoxicity. Mutat Res 517:101–111
Malawska M, Wiłkomirski B (2001) An analysis of soil and plant (Taraxacum Officinale) contamination with heavy metals and polycyclic aromatic hydrocarbons (PAHs) in the area of the railway junction iława główna, Poland. Water Air Soil Pollut 127:339–349
McLachlan MS, Welsch-Pausch K, Tolls J (1995) Field validation of a model of the uptake of gaseous SOC in Lolium multrum (ryegrass). Environ Sci Technol 29:1998–2004
Mikefryer C, Agrosso A (2006) Plant uptake of non-ionic organic chemicals. Environ Sci Technol 40:45–52
Ministry of Environmental Protection of the People’s Republic of China (1990) Background value of soil elements in China. China Environmental Science Press, Beijing
Musa BBA, Benjamin A, Musa B, Nosir S, Michael K, Wolfgang W (2010) Polycyclic aromatic hydrocarbons (PAHs) and their oxygen-containing derivatives (OPAHs) in soils from the Angren industrial area, Uzbekistan. Environ Pollut 158:2888–2899
Nadal M, Schuhmacher M, Domingo JL (2004a) Levels of PAHs in soil and vegetation samples from Tarragona County, Spain. Environ Pollut 132:1–11
Nadal M, Schuhmacher M, Domingo JL (2004b) Metal pollution of soils and vegetation in an area with petrochemical industry. Sci Total Environ 321:59–69
National Bureau of Statistics of the People’s Republic of China (2010) China Environment Yearbook (2002–2009). China Statistic Press, Beijing
Odabasi M, Bayram A, Elbir T, Seyfioglu R, Dumanoglu Y, Ornektekin S (2010) Investigation of soil concentrations of persistent organic pollutants, heavy metals, and anions due to iron–steel plant emissions in an industrial region in Turkey. Water Air Soil Pollut 213:375–388
Parizanganeha A, Hajisoltanib P, Zamani A (2010) Concentration, distribution and comparison of total and bioavailable metals in top soils and plants accumulation in Zanjan zinc industrial town-Iran. Procedia Environ Sci 2:167–174
Pereira MDS, Heitmann D, Reifenhäuser W, Rodrigo Ornellas Meire Santos LS, Torres JPM, Malm O, Körner W (2007) Persistent organic pollutants in atmospheric deposition and biomonitoring with Tillandsia usneoides (L.) in an industrialized area in Rio de Janeiro state, southeast Brazil—Part II: PCB and PAH. Chemosphere 67:1736–1745
Petaloti C, Triantafyllou A, Kouimtzis T, Samara C (2006) Heavy metals in atmospheric particulate matter over a coal burning power production area of western Macedonia, Greece. Chemosphere 65:2233–2243
Sébastien R, Suzanne L, Punita M, Anca M, Nathalie F, Claude M, René L, Guy C, Christine S, Christine G, Caroline O, Serge D, Michel L, Charles G (2005) Phytoremediation of heavy metal and PAH-contaminated brownfield sites. Plant Soil 272:277–290
Shukurov N, Pen-Mouratov S, Steinberger Y, Kersten M (2009) Soil biogeochemical properties of Angren industrial area, Uzbekistan. J Soil Sed 9:206–215
Simonich SL, Hites RA (1995) Organic pollutant accumulation in vegetation. Environ Sci Technol 29:2905–2914
Slaski JJ, Archambault DJ, Li X (2000) Evaluation of polycyclic aromatic hydrocarbon (PAH) accumulation in plants. The potential use of PAH accumulation as a marker of exposure to air emissions from oil and gas flares. ISBN 0-7785-1228-2. Report prepared for the Air Research Users Group, Alberta Environment, Edmonton, Alberta
Sojinua OS, Sonibare OO, Ekundayo O, Zeng EY (2010) Biomonitoring potentials of polycyclic aromatic hydrocarbons (PAHs) by higher plants from an oil exploration site, Nigeria. J Hazard Mater 184:759–764
Stalikas CD, Chaidou CI, Pilidisb GA (1997) Enrichment of PAHs and heavy metals in soils in the vicinity of the lignite-fired power plants of West Macedonia (Greece). Sci Total Environ 204:135–146
Sun YB, Zhou QX, Xu YM, Wang L, Liang XF (2011) Phytoremediation for co-contaminated soils of benzo[a]pyrene (B[a]P) and heavy metals using ornamental plant Tagetes patula. J Hazard Mater 186:2075–2082
Sun M, Luo Y, Teng Y, Jia Z, Li Z, Deng S (2013) Remediation of polycyclic aromatic hydrocarbon and metalcontaminated soil by successive methyl-β-cyclodextrin-enhanced soil washing–microbial augmentation: a laboratory evaluation. Environ Sci Pollut Res 20:976–986
Tang X, Shen C, Shi D, Cheema SA, Khan MI, Zhang C, Chen Y (2010) Heavy metal and persistent organic compound contamination in soil from Wenling: an emerging e-waste recycling city in Taizhou area, China. J Hazard Mater 17:653–660
US EPA (2002) Arsenic treatment technologies for soil, waste, and water. EPA-542-R-02-004. U.S. Environmental Protection Agency, Washington, DC
US EPA (2004) Cleaning up the nation’s waste sites: markets and technology trends. EPA 542-R-04-015. U.S. Environmental Protection Agency, Washington, DC
US EPA (2005) Use of field-scale phytotechnology for chlorinated solvents, metals, explosives and propellants, and pesticides. EPA 542-R-05-002. U.S. Environmental Protection Agency, Washington, DC
Vervaeke P, Luyssaert S, Mertens J, Meers E, Tack FMG, Lust N (2003) Phytoremediation prospects of willow stands on contaminated sediment: a field trial. Environ Pollut 126:275–282
Wang YH, Lin C, Lai YC, Chang-Chien GP (2009) Characterization of PCDD/Fs, PAHs, and heavy metals in a secondary aluminum smelter. J Environ Sci Health A 44:1335–1342
Wei CY, Chen TB (2002) The ecological and chemical characteristics of plants in the areas of high arsenic levels. Acta Phytoecol Sin 26:695–700
Xie QE, Yan XL, Liao XY, Li X (2009) The arsenic hyperaccumulator fern Pteris vittata L. Sci Total Environ 43:8488–8495
Yang B, Zhou M, Shu WS (2010) Constitutional tolerance to heavy metals of a fiber crop, ramie (Boehmeria nivea), and its potential usage. Environ Pollut 158:551–558
Zhao FJ, Dunham SJ, McGrath SP (2002) Arsenic hyperaccumulation by different fern species. New Phytol 156:27–31
Acknowledgements
This work was supported by the National Natural Science Foundation of China (grant no. 41271339).
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Philippe Garrigues
Rights and permissions
About this article
Cite this article
Sun, L., Liao, X., Yan, X. et al. Evaluation of heavy metal and polycyclic aromatic hydrocarbons accumulation in plants from typical industrial sites: potential candidate in phytoremediation for co-contamination. Environ Sci Pollut Res 21, 12494–12504 (2014). https://doi.org/10.1007/s11356-014-3171-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-014-3171-6