Abstract
In this study, the effects of cadmium (Cd) stress on the activities of disaccharidases (sucrase, lactase, and maltase), amylase, trypsin, pepsase, superoxide dismutase (SOD), catalase (CAT), glutathione peroxidase (GPx), and malondialdehyde (MDA) content in the alimentary system of freshwater crabs Sinopotamon henanense were studied. Results showed that the enzyme activities in the stomach, intestine, and hepatopancreas changed with Cd concentration. In terms of digestive enzymes, Cd exposure had an inhibitory effect on the activities of the disaccharidases, amylase, and pepsase (only in the stomach). Significant induction of trypsin activity by Cd at a lower concentration was observed, but as Cd concentration increased, trypsin activity decreased. Maltase activity showed a slight recovery after inhibition by Cd. The activities of SOD and CAT increased initially and decreased subsequently. Cd significantly inhibited the activity of GPx. MDA content increased with increasing concentration of Cd. These results showed that acute Cd exposure led to harmful effects on the alimentary system of crabs, which are likely linked to Cd induced oxidative stress.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Cadmium (Cd), a widely used heavy metal in modern industry, is one of the most abundant, ubiquitously distributed toxic elements in aquatic ecosystems (Novelli et al. 2000). The effects of Cd on histology and physiology of aquatic animals have been relatively well studied (Chen et al. 2000; Tulasi et al. 1987; Paslor et al. 1988). Cd can result in acute and chronic intoxication in various organs, including the liver, kidney, testes, and intestine (Kuriwaki et al. 2005; Zhou et al. 1999; Xu et al. 2009).
Digestive enzymes can indicate the metabolic status of the animals and the degree of adaptation to the environment and can thus be used as biomarkers in environmental monitoring (Lai et al. 2011). In the present study, digestive enzymes have demonstrated post-assimilatory impacts on protease activity in grass shrimp collected along an impact gradient (Seebaugh et al. 2011). Results showed the separate and combined effects of Cd, temperature, and pH on digestive enzymes in three freshwater teleosts (Kuzmina et al. 2002), and a strong relationship between digestive enzymes activities, feeding rate, and metal contents (Dedourge-Geffard et al. 2009). The expression of amylase genes in Daphnia magna was upregulated after cadmium exposure (De Coen and Janssen 1997). So far, the toxic mechanism has remained unclear for the toxic effects that heavy metals have on the digestive systems of aquatic animals.
Studies have shown that Cd stimulates the production of reactive oxygen species (ROS), which can cause oxidative damage to tissues and organs, if the antioxidant defense system, consisting of superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GPx), was overwhelmed (Winston and Di Giulin 1991; Almeida et al. 2002). Alimentary tract mucosa is vulnerable to the injury of oxidative stress, free oxygen radicals, and lipid peroxidation (Wargle et al. 1996), and systemic oxidative stress can influence the activity of digestive enzymes (Lackeyram et al. 2007).
Freshwater crabs have the capability of accumulating heavy metals (Reinecke et al. 2003) and represent a suitable bioindicator for environmental contamination with these agents (Schuwerack et al. 2001). The freshwater crab Sinopotamon henanense is a species commonly found in China freshwater. Our earlier surveys on Cd accumulation in the S. henanense habitat in Qin River, the second longest river in Shanxi province, showed that Cd level could reach 12.46 ± 1.88 mg/kg in surface sediment, which poses a high ecological risk (Han et al. 2008). In some valleys near Cd-rich mines, the Cd level could reach 12.05 ± 1.47 mg/L in the water, where this crab species nearly vanished (Yuan et al. 2010). Considering the serious deterioration of the ecological environment, we chose a series of Cd concentrations of 7.25, 14.5, and 29.0 mg/L, which are 1/32, 1/16, and 1/8 of the 96 h LC50 for Cd to Sinopotamon yangtsekiense, respectively (Wang et al. 2008). These concentrations may be higher than general environmental Cd levels, but were similar to even lower than the Cd levels in a few serious pollution incidents.
Until now, most toxicity studies on crustaceans induced by Cd have been limited to hepatopancreas and gill injuries (Liu et al. 2011; Wu et al. 2008; Ma et al. 2009). The relevant studies on the heavy metal effects on enzymatic activities in alimentary system of S. henanense have not been reported yet. The current study investigated the effects of Cd stress on digestive enzymes and antioxidant enzymes of S. henanense’s alimentary system, and demonstrated that oxidative damage to the alimentary system could affect digestive function.
Materials and methods
Animals and treatments
S. henanense (hereinafter referred to as “crabs”) were purchased in October 2011 from the Wu Longkou Dong’an Aquatic Wholesale Market in Taiyuan and were acclimated in glass aquaria (130 cm × 50 cm × 60 cm) filled with tap water (aerated for 48 h, with a temperature of 16–20 °C, pH 6.8, and dissolved oxygen over 6 mg/L) for 2 weeks before experiments. Aquaria were shielded by a black plastic to reduce disturbance. All crabs were fed commercial feed (1 % of body weight) two times a week.
After acclimation, healthy, similar-sized adult crabs with a wet mass of 20.2 ± 2.5 g were randomly divided into four groups and exposed to a gradient of Cd concentrations (0, 7.25, 14.5, and 29.0 mg/L) in glass aquaria (20 cm × 20 cm × 20 cm) with 1 L of CdCl2 solution. During the experimental period, crabs were not fed. The exposure medium was changed every 24 h. All other conditions were kept the same as those used for acclimation. During the exposure period, there was no mortality of the experimental animals.
After 4 days, the crabs were sacrificed, and tissue samples of hepatopancreas, stomach, and intestine were immediately excised after opening the cephalothorax of each crab. These tissues were weighted and then added to phosphate buffer at a pH 7.2 in a proportion of 1:9 (w/v), and were homogenized at ice-bathing condition with an electronic homogenizer, and the homogenate then was frozen-centrifuged for 10 min (9,300×g). The supernatants were taken and stored at −80 °C for enzyme activity testing. All tests were performed in triplicate.
Biochemical assays
The analytical reagent CdCl2 was used. Disaccharidase (sucrase, lactase, and maltase), amylase, trypsin, pepsase, SOD, CAT, GPx, MDA, and total protein content were measured with kits (Nanjing Jiancheng Bioengineering Institute, Nanjing, China) following the manufacturer’s protocols. All measurements were carried out with a microplate reader (Spectramax M5; Molecular Devices, USA). All tests were performed in triplicate.
Disaccharidase (sucrase, lactase, and maltase) activities were determined as described by Dahlquist (1968), and activities of amylase, trypsin, and pepsase were determined as described by Pan and Wang (1997). SOD activity was measured by the xanthine/xanthine oxidase method (Nishikimi 1975); 1 U of SOD was defined as the amount of protein that inhibited the rate of NBT reduction by 50 %. CAT activity was determined by the ammonium molybdate colorimetric method (Góth 1991). GPx activity was quantified by the dithio-binitrobenzoic acid method (Rotruck et al. 1973), based on the reaction between remaining glutathione after the action of GPx and 5,59-dithio bis-(2-nitrobenzoic acid) to form a complex that absorbs maximally at 412 nm. Lipid peroxidation was measured using the thiobarbituric acid test for MDA according to Ohkawa et al. (1979). Protein content was determined according to Bradford (1976) using bovine serum albumin as the standard.
Statistical analysis
Statistical analyses were performed with SPSS 15.0 software. Data distributions and the homogeneity of variance were tested using Kolmogorov–Smirnov and Levene tests, respectively. When the data satisfy the prerequisites for parametric tests (analysis of variance, ANOVA), one-way ANOVA and the Dunnet’s test were used to evaluate the significance of differences between treated and control groups. The data were expressed as mean ± SD. The post hoc least significant difference (LSD) test was performed for inter-group comparisons. Probability values of P <0.05 were considered statistically significant.
Results
Effects of Cd on digestive enzymes activities in different groups
The results indicated that Cd significantly inhibited sucrase activity in the alimentary system as shown in Fig. 1a (P < 0.01). In the low concentration group (7.25 mg/L), Cd had a significant inhibitory effect on the activity of sucrase in the hepatopancreas and intestine, while its inhibitory effect towards the stomach was only obvious in the group containing middle concentrations (14.5 mg/L). In terms of the three tissues, sucrase activities reached the lowest point in the high concentration group (29.0 mg/L), and activities in the hepatopancreas, intestine, and stomach were reduced to 53 %, 31 %, and 17 % of those of the control group, respectively.
Effects of cadmium on the activities of digestive enzymes in S. henanense (a sucrase activity; b lactase activity; c maltase activity; d amylase activity; e trypsin activity; f pepsase activity). The values are means ± SD from three individual samples. Using one-way ANOVA, compared with the control, significance is indicated by * P <0.05, ** P <0.01
Cd had a significant inhibitory effect on lactase activity in the alimentary system (Fig.1b). When compared to the control, lactase activity significantly decreased to its lowest level (39 % of control) both in the low concentration group (7.25 mg/L) in the hepatopancreas and in the high concentration group (29.0 mg/L) in intestine and stomach, a decrease of 51 % and 58 %, respectively.
As seen in Fig. 1c, after being inhibited by Cd, maltase activity in crab alimentary system showed a slight recovery. But when compared with the control group, maltase activity of the alimentary system in the Cd-treated group was significantly (P<0.05) or highly significantly (P<0.01) decreased. Maltase activity reached its lowest value at 7.25 mg/L Cd, with enzymatic activities in the hepatopancreas, intestine, and stomach being 68 %, 56 %, and 53 % that of the control, respectively.
Cd had an inhibitory effect on amylase activity in alimentary system of crabs (Fig.1d). Amylase activity in the hepatopancreas, intestine, and stomach were 46 %, 49 %, and 42 % that of the respective control group, respectively. When compared with the control, amylase activities of the hepatopancreas and stomach showed a significant decrease in the middle concentration group (14.5 mg/L) and decreased to its lowest in the high concentration group (29.0 mg/L). Amylase activity of intestine at 29.0 mg/L of Cd concentration showed a significant difference (P<0.05) from the control.
Initially, exposure concentrations up to 7.25 mg/L Cd resulted in a concentration-dependent increase in the activity of trypsin, and a maximum increase of 51 %, 41 %, and 26 % in the hepatopancreas, intestine, and stomach, respectively, were observed as compared to control (Fig. 1e). With increasing Cd concentration, trypsin activity in the hepatopancreas, intestine, and stomach dropped by 86 %, 62 %, and 93 %, respectively, in the 29.0 mg/L Cd group compared to the control. But in the stomach, the inhibition was not significant (P > 0.1).
Pepsase activity was only detected in the stomach of crabs (Fig. 1f). Cd had an obvious inhibitory effect on pepsase activity. Compared with the control group, at 14.5 mg/L and 29.0 mg/L of Cd concentration, pepsase activity showed significant decreases (P<0.01), while in the high concentration group (29.0 mg/L), it reached its lowest value: 49 % of control.
Effects of Cd on antioxidant enzymes activities and lipid peroxidation in different groups
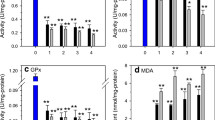
During the 4-day acute treatment with Cd, SOD activities in three tissues of crabs all increased at the least exposure concentration and then decreased at greater concentrations (Fig. 2a). In intestine and stomach, when Cd concentration was low (7.25 mg/L), SOD activity showed highly significant induction (P<0.01). With increasing Cd concentration, SOD activities in these three tissues were significantly inhibited at 29.0 mg/L Cd.
Effects of cadmium on the activities of antioxidant enzymes and the membrane lipid peroxidation in S. henanense (a SOD activity; b CAT activity; c GPx activity; d MDA content). The values are means ± SD from three individual samples. Using one-way ANOVA, compared with the control, significance is indicated by * P <0.05, ** P <0.01
As seen in Fig. 2b, CAT activities in the crab’s alimentary system (stomach and intestine) were stimulated by Cd and reached maximum activities when Cd concentration was low (7.25 mg/L); CAT activities in hepatopancreas, intestine, and stomach were 17 %, 89 %, and 138 % greater than the control, respectively. Enzymatic activities decreased slightly in the 14.5 mg/L exposure group, but in the intestine and stomach the activities were still significantly higher than the control. In crabs from the high concentration (29.0 mg/L) group, CAT activities in these two tissues were significantly inhibited. In the hepatopancreas, the activity of CAT was significantly decreased in the high Cd concentration (29.0 mg/L) group.
The results showed that in the high concentration (29.0 mg/L) group, GPx activities in the three tissues were lower than the control group (P<0.01) (Fig. 2c). Cd had a significant inhibitory effect on the activity of GPx. In intestine, GPx activity for crabs treated with 14.5 mg/L Cd was significantly lower than in the control.
In each Cd-treated group, the content of MDA rose with increasing Cd concentration (Fig. 2d). In the middle concentration (14.5 mg/L) group, the MDA content in the hepatopancreas and intestine, compared with the control group, increased significantly and reached its peak at the highest Cd concentration (29.0 mg/L). The maximal increases in MDA of three tissues after Cd treatment were 34 %, 40 %, and 28 % greater than the control, respectively.
Discussion
Digestive enzymes activities of crustaceans are important physiological parameters that reflect digestive physiology of a crustacean and are closely related to heredity, environment, and food habit. Digestive enzymes of crabs are mainly provided and excreted by the digestive tract (consisting of stomach and intestine) and digestive gland (hepatopancreas) (Li et al. 1994). The stomach grates and preliminarily digests the food (Fang et al. 2002); midgut and hindgut digest the food that is not completely digested by the stomach (Barker and Gibson 1978); hepatopancreas is made of one left lobe and one right lobe, which is the most developed digestive gland and detoxification organ of crabs (Lou et al. 2010). There are different kinds of digestive enzymes that exist in different tissues (Jiang et al. 2010). In our experiment, trypsin activity of the hepatopancreas was the highest. In the intestine, the maltase activity was higher than the other two tissues. Pepsase activity was only detected in the stomach as indicated by Sastry and Gupta (1979). Through reacting with digestive enzymes, heavy metal ions influence the growth or even survival of the organisms. The induction or inhibitory effects vary between different digestive enzymes. In addition, metal ions have different action mechanisms towards different digestive enzymes (Sastry and Gupta 1978).
There are two ways for heavy metals to influence enzymes: one is to stimulate enzymatic activity and the second is to inhibit enzymatic activity (Zhao 2009). Our present research showed that the effects of Cd on three tissues and six kinds of digestive enzymes of the digestive system of crabs were not consistent. At a low Cd concentration, the activities of trypsin in alimentary system of crabs increased and fell back to a high Cd concentration, and maltase activity showed a slight recovery after inhibition by Cd. Concerning the induction mechanism of exogenous compounds on enzymes, it is generally assumed that exogenous compounds induce the production of enzymes mainly by means of manipulating genes’ repression. Exogenous compounds and repressors manipulate genes form a complex, which in return can disable the repressor and thus result in an increasing synthesis of proteinaceous enzymes (Kong 2000).
The influence of Cd on crab digestive enzymes was also reflected by the inhibitory effects on the activities of sucrose, lactase, amylase, and pepsase. With rising concentration of Cd, the activities of these four enzymes were all inhibited to a certain extent. Cd is a kind of soft acid that can connect with mercapto groups of cysteine residue at the activity center of digestive enzyme. This connection can inhibit the activity of the enzyme. Cd can also connect with the non-active center of the enzyme, thus changing the composition of the enzyme to reduce its activity. This is assumed to be the main mechanism of Cd in inhibiting digestive enzymes activities in the alimentary system of crabs.
There have been a number of studies about digestive enzymes being used as biomarker for pollutant stress (Douhri and Sayah 2009; Boldina-Cosqueric et al. 2010; Palais et al. 2012). Barfield et al. (2001) found that after 28 days of exposure to Cd, the activity of Corbicula fluminea’s digestive enzyme (cellulose enzyme) decreased with the increase of the concentration gradient, so they concluded that digestive enzymes could be used as a biomarker for Cd stress towards heavy metals. In the present paper, the crabs were exposed for 4 days, and the activities of sucrase, amylase, and pepsase exhibited a linear decrease with the rise of Cd concentration, so there was a significant negative correlation between enzyme and concentration. This phenomenon testified that the sucrase, amylase, and pepsase enzyme activity in the digestive system of crabs can also be used as a biomarker for monitoring heavy metal pollution.
When aquatic invertebrates are exposed to heavy metals, animals will respond by activating the mechanism of detoxification. For example, metallothionein (MT) is abundantly expressed, which is closely related with excretion and detoxification of Cd. Cd is then accumulated and detoxified by chelation (Ma et al. 2008). But with increasing exposure concentration, Cd may exceed the body’s limits of detoxification. Then, Cd will combine with large molecule proteins in the body (such as a variety of enzymes) and cause toxic effects (Zhou et al. 2010).
The present study showed that Cd effects on antioxidant enzymes activities and MDA content of three tissues in alimentary system of crabs are a dynamic process. Induction and inhibitory effects vary with biochemical parameters tested and treatment concentration of Cd (Lei et al. 2011).
The current experiment showed that the changes of antioxidant enzymes and MDA content differed from organ to organ. CAT, GPx activities, and MDA content of hepatopancreas were higher than those of the other two tissues. This may be related to the fact that hepatopancreas is the key site of Cd accumulation in Crustacea (Wang et al. 2001) and one of the most important organs that play important roles in metal detoxification (Li et al. 2008).
SOD can instantly convert the harmful oxygen-free radicals into H2O2, and maintain metabolic balance of free radicals in the body, thereby protecting cells from damage. The ability of SOD to remove the oxygen-free radicals is related to its content and activity (Cao et al. 2010). In the experiment presented here, the lower concentration of Cd increased SOD activity of crabs, resulting in "hormesis", but when Cd concentration exceeded a certain range, the activity of SOD was reduced, which indicated that SOD could not remove O2 − in the alimentary system of crabs in a timely manner, and thus Cd had a toxic effect on the crabs. With the accumulation of O2 −, oxidative damage for cells of the alimentary system can become more severe which could finally result in the death of cells (Yan et al. 2007).
Both CAT and GPx can transform H2O2 into H2O and O2 (Yao et al. 2006). Therefore, CAT and GPx could reduce the tissue injury by removing the H2O2. The activity of CAT is relevant to the concentration of H2O2 (Fomazier et al. 2002). Results of the present study revealed first an activation and then an inhibition of CAT activity at 7.25 mg/L Cd, indicating that when a small amount of Cd entered the organism, the activity of SOD was stimulated to increase the concentration of H2O2 consequently. The higher concentration of H2O2 induces the activity of CAT (Chen et al. 1994). With increasing concentration of Cd, a large amount of extra ROS were induced, and the scavenging capability of CAT was reduced, leading to a decrease in CAT activity of three tissues. GPx is an enzyme requiring selenium for its activity. The combination of Cd and GPx’s active site (Se-Cys) can reduce the toxic effect of Cd on the organism and, on the other hand, can lead to changes at the active site and cause GPx to lose its activity (Iszard et al. 1995), hurting the organism through the accumulation of H2O2 and ·OH (Hultberg et al. 1998). In the current experiment, by increasing the concentration of Cd, ROS kept accumulating, and the activities of GPx in three tissues changed and its synthesis was inhibited, leading to a significant decrease in GPx activity.
Lipid peroxidation is one of the main indicators of oxidative damage (Anane and Creppy 2001). MDA is not only an important product of lipid peroxidation but also can react with a protein’s free amino acid and cause cross linking within and between protein molecules, which consequently damage the cell (Papadimitriou and Loumbourdis 2002). Our data confirmed that acute intoxication with Cd after 4 days of exposure caused a statistically significant increase in MDA concentration. The content of MDA is representative of oxidative damages.
Currently, a large number of studies have shown that heavy metals and other stressors may lead to oxidative stress, and can cause gastrointestinal damage (Chen 2008). Cd had a significant inhibitory effect on the activities of SOD, CAT, and GPx at 29.0 mg/L, and MDA content increased with increasing Cd concentration, suggesting that the antioxidant defense system of the alimentary system of crabs was damaged under high Cd stress, which resulted in structural damages of the alimentary system, directly affecting the activities of a variety of digestive enzymes (Zhao 2011).
Conclusions
In summary, with increasing concentration of Cd, the activities of four kinds of digestive enzymes were inhibited to a certain degree, and the activity of trypsin in alimentary system of crabs was increased in the low Cd concentration group and decreased in the high Cd concentration group. Maltase activity showed a slight recovery after inhibition by Cd. At a low concentration of Cd, antioxidant enzymes activities were stimulated, but inhibited at higher Cd concentrations. This indicated that the damage to antioxidant defense system may be the factor affecting the activities of digestive enzymes. All these may contribute to the establishment of predictive models for the toxicity of Cd. In the future, it will be interesting to investigate histological damages of Cd to the alimentary system of S. henanense.
References
Almeida JA, Diniz YS, Marques SF, Faine LA, Ribas BO, Burneiko RC, Novelli EL (2002) The use of oxidative stress response as biomarkers in Nile tilapia (Oreochromis niloticus) exposed to in vivo cadmium contamination. Environ Int 27:673–679
Anane R, Creppy EE (2001) Lipid peroxidation as pathway of aluminium cytotoxicity in human skin fibroblast cultures: prevention by superoxide dismutase and catalase and vitamins E and C. Hum Exp Toxicol 20:477–48
Barfield ML, Farris JL, Black MC (2001) Biomarker and bioaccumulation responses of Asian clams exposed to aqueous cadmium. J Toxicol Environ Health A 63:495–510
Barker PL, Gibson RJ (1978) Observations on the structure of the mouthparts, histology of the alimentary tract, and digestive physiology of the mud crab Scylla serrata (Decapoda: Portunidae). J Exp Mar Biol Ecol 58:175–191
Boldina-Cosqueric I, Amiard JC, Amiard-Triquet C, Dedourge-Geffard O, Métais I, Mouneyrac C, Moutel B, Berthet B (2010) Biochemical, physiological and behavioral markers in the endobenthic bivalve Scrobicularia plana as tools for the assessment of estuarine sediment quality. Ecotoxicol Environ Saf 73(7):1733–1741
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein using the principle of protein–dye binding. Anal Biochem 72:248–254
Cao L, Huang W, Liu J, Yin X, Dou S (2010) Accumulation and oxidative stress biomarkers in Japanese flounder larvae and juveniles under chronic cadmium exposure. Comp Biochem Physiol C Toxicol Pharmacol 151(3):386–392
Chen Q (2008) The studies of the effect of oxidative stress on animal’s gastrointestinal tract structure and function. Thesis for Doctor of Science, Jiangnan University, Wuxi (in Chinese)
Chen T, Furst A, Chien PK (1994) The effects of cadmium and iron on catalase activities in Tubifex. J Am Coll Toxicol 13:112
Chen M, Xie JM, Gao XY, Jing JJ (2000) Effects of lead on lipid peroxidation in organs of mice. Chin Pub Health 16:1107–1108 (in Chinese)
Dahlquist A (1968) Assay of intestinal disaccharidases. Anal Biochem 22:99–105
De Coen WM, Janssen CR (1997) The use of biomarkers in Daphnia magna toxicity testing II. Digestive enzyme activity in Daphnia magna exposed to sublethal concentrations of cadmium, chromium and mercury. Chemosphere 35(5):1053–1067
Dedourge-Geffard O, Palais F, Biagianti-Risbourg S, Geffard O, Geffard A (2009) Effects of metals on feeding rate and digestive enzymes in Gammarus fossarum: an in situ experiment. Chemosphere 77:1569–1576
Douhri H, Sayah F (2009) The use of enzymatic biomarkers in two marine invertebrates Nereis diversicolor and Patella vulgate for the biomonitoring of Tangier’s bay (Morocco). Ecotoxicol Environ Saf 72(2):394–399
Fang ZP, Pan QS, Huang FJ, Luo LT, He J (2002) Studies on histology and scanning electron microscope of digestive tract of Chinese mitten-handed crab (Eriocheir sinensis). Acta Hydrobiol Sin 26(2):136–141 (in Chinese)
Fomazier RF, Ferreira RR, Viteria AP, Molina SMG, Lea PJ, Azevedo RA (2002) Effects of cadmium on antioxidant enzyme activities in sugar cane. Biol Plant 45:91–97
Góth L (1991) A simple method for determination of serum catalase activity and revision of reference range. Clin Chim Acta 196:143–151
Han T, Wang Q, Wang L (2008) Ecological investigation of freshwater crab and river pollution in basin of Qinhe River. Sichuan J Zool 27:804–806 (in Chinese)
Hultberg B, Andersson A, Isaksson A (1998) Alterations of thiol metabolism in human cell lines induced by low amounts of copper, mercury of cadmium ions. Toxicology 126:203–212
Iszard MB, Liu J, Klaassen CD (1995) Effect of several metallothionein inducers on oxidative stress defense mechanisms in rats. Toxicology 104:25–33
Jiang HX, Kong XH, Zhou XY, Guo HY (2010) Effect of Cu2+ on digestive enzymes activity in the digestive organs of Cyprinus carpio. J Hydroecol 3(4):108–112 (in Chinese)
Kong FX (2000) Environmental biology. Higher Education Press, Beijing, in Chinese
Kuriwaki J, Nishijo M, Honda R, Tawara K, Nakagawa H, Hori E, Nishijo H (2005) Effects of cadmium exposure during pregnancy on trace elements in fetal rat liver and kidney. Toxicol Lett 156:369–376
Kuzmina VV, Golovanova IL, Kovalenko E (2002) Separate and combined effects of cadmium, temperature and pH on digestive enzymes in three freshwater teleosts. Bull Environ Contam Toxicol 69:302–308
Lackeyram D, Archbold T, Shoveller AK, Mine Y, Fan MZ (2007) Gut carbohydrate digestive enzymes respond differentially to oxidative stress induced by in vivo infusion of hydrogen peroxide (H2O2) in formula-fed piglets. FASEB J 21:839–913
Lai TH, He BY, Fan HQ, Zhou RQ, Yang Y (2011) Effects of cadmium stress on the activities of antioxidant enzymes, digestive enzymes and the membrane lipid peroxidation of the mangrove mud clam Geloina coaxans (Gmelin). Acta Ecol Sin 31:3044–3053 (in Chinese)
Lei WW, Wang L, Liu DM, Xu T, Luo JX (2011) Histopathological and biochemical alternations of the heart induced by acute cadmium exposure in the freshwater crab Sinopotamon yangtsekiense. Chemosphere 84(5):689–694
Li TW, Su XR, Zhang F (1994) Histology, histochemistry of the digestive system in blue crab Portunus trituberculatus (Decapoda, Portunidae). J Liaoning Normal Univ (Nat Sci) 17(3):230–237 (in Chinese)
Li YQ, Wang L, Liu N, Wang Q, He YJ, Meng F (2008) Effects of cadmium on enzyme activity and lipid peroxidation in freshwater crab Sinopotamon yangtsekiense. Acta Hydrobiol Sin 32(3):373–379 (in Chinese)
Liu DM, Yan B, Yang J, Lei WW, Wang L (2011) Mitochondrial pathway of apoptosis in the hepatopancreas of the freshwater crab Sinopotamon yangtsekiense exposed to cadmium. Aquat Toxicol 105:394–402
Lou D, Yang JF, Xie H, Chen JG (2010) A preliminary study of comparative histology of major organs from Portunus trituberculatus and Sinopotamon. J Marine Sci 28(3):72–78 (in Chinese)
Ma WL, Wang L, He YJ, Yan Y (2008) Tissue-specific cadmium and metallothionein levels in freshwater crab Sinopotamon henanense during acute exposure to waterborne cadmium. Environ Toxicol 23(3):393–400
Ma WL, Yan T, He YJ, Wang L (2009) Purification and cDNA cloning of a cadmium-binding metallothionein from the freshwater crab Sinopotamon henanense. Arch Environ Contam Toxicol 56:747–753
Nishikimi M (1975) Oxidation of ascorbic acid with superoxide anion generated by the xanthine–xanthine oxidase system. Biochem Biophys Res 63:463–468
Novelli F, Novelli E, Manzano MA, Lopes AM, Cataneo AC, Barbosa LL, Ribas BO (2000) Effect of alpha-tocopherol on superoxide radical and toxicity of cadmium exposure. Int J Environ Health Res 10:125–134
Ohkawa H, Ohishi N, Yagi K (1979) Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem 95:351–358
Palais F, Dedourge-Geffard O, Beaudon A, Pain-Devin S, Trapp J, Geffard O, Noury P, Gourlay-Francé C, Uher E, Mouneyrac C, Biagianti-Risbourg S, Geffard A (2012) One-year monitoring of core biomarker and digestive enzyme responses in transplanted zebra mussels (Dreissena polymorpha). Ecotoxicology 21(3):888–905
Pan LQ, Wang KX (1997) Studies on digestive enzyme activities and amino acid in the larvae of Eriocheir sinensis. J Fish Sci Chin 4(2):13–20 (in Chinese)
Papadimitriou E, Loumbourdis NS (2002) Exposure of the frog Rana ridibunda to copper impact on two biomarkers, lipid peroxidation, and glutathione. Bull Environ Contam Toxicol 69:885–891
Paslor A, Medina J, Rarno JD, Torreblanca A, Díaz-Mayans J, Hernandez F (1988) Determination of lead in treated crayfish Procambarus clarkii: accumulation in different tissues. Bull Environ Contam Toxicol 41:412–418
Reinecke AJ, Snyman RG, Nel JAJ (2003) Uptake and distribution of lead (Pb) and cadmium (Cd) in the freshwater crab, Potamonautes perlatus (Crustacea) in the Eerste River, South Africa. Water Air Soil Pollut 145:395–408
Rotruck JT, Pope AL, Ganther HE, Swanson AB, Hafeman DG, Hoekstra WG (1973) Selenium: biochemical role as a component of glutathione peroxidase. Science 179:588–590
Sastry KV, Gupta PK (1978) In vitro inhibition of digestive enzymes by heavy metals and their reversal by chelating agent: part I. Mercuric chloride intoxication. Bull Environ Contam Toxicol 20:729–735
Sastry KV, Gupta PK (1979) The effect of cadmium on the digestive system of the teleost fish, Heteropneustes fossilis. Environ Res 19:221–230
Schuwerack PM, Lewis JW, Jones P (2001) The potential use of the South African river crab, Potamonautes warreni, as a bioindicator species for heavy metal contamination. Ecotoxicology 10:159–166
Seebaugh DR, L’Amoreaux WJ, Wallacea WG (2011) Digestive toxicity in grass shrimp collected along an impact gradient. Aquat Toxicol 105:609–617
Tulasi SJ, Yasmeen R, Reddy CP, Rao VJR (1987) Lead uptake and lead loss in the freshwater field crab, Barytelphusa guerini, on exposure to organic and inorganic lead. Bull Environ Contam Toxicol 39:63–68
Wang L, Yang XQ, Wang Q, Wang DX (2001) The accumulation of Cd and the effect of EST in five tissues and organs of Eriocheir sinensis. Acta Zool Sin 47(Suppl):96–100 (in Chinese)
Wang L, Yan B, Liu N, Li YQ, Wang Q (2008) Effects of cadmium on glutathione synthesis in hepatopancreas of freshwater crab, Sinopotamon yangtsekiense. Chemosphere 74:51–56
Wargle TD, Hall L, Turnberg LA (1996) Platelet activating factor: release from colonic mucosa on patients with ulcerative colitis and its effect on colinic secretion. Gut 38(3):355–361
Winston GW, Di Giulin RT (1991) Prooxidant and antioxidant mechanism in aquatic organism. Aquat Toxicol 24:143–152
Wu JP, Chen HC, Huang DJ (2008) Histopathological and biochemical evidence of hepatopancreatic toxicity caused by cadmium and zinc in the white shrimp, Litopenaeus vannamei. Chemosphere 73:1019–1026
Xu XQ, Weng XL, Zhao YL, Qin F (2009) Effects of water-borne cadmium on development, reproduction and ultrastructure of Daphnia carinata. J Fudan Univ (Nat Sci) 48(3):375–380 (in Chinese)
Yan B, Wang L, LI YQ, Liu N, Wang Q (2007) Effects of cadmium on hepatopancreatic antioxidant enzyme activity in a freshwater crab Sinopotamon yangtsekiense. Acta Zool Sin 53:1121–1128 (in Chinese)
Yao CL, Wang ZY, Xiang JH (2006) Crustacean haemocytes and their function in immune responses. Zoo Res 27:549–557 (in Chinese)
Yuan QH, Ye YL, He ZS (2010) Research on heavy metal pollution of water in Lanping mining area. Occup Health 26:2235–2236 (in Chinese)
Zhao YM (2009) Studies on the toxic effects of water-borne Hg2+ on Eriocheir sinensis. Thesis for Doctor of Science, Nankai University, Tianjin (in Chinese)
Zhao KL (2011) Effects of composite antioxidants on oxidative stress and intestine in animals. Thesis for Master of Science, Shanghai Jiaotong University, Shanghai (in Chinese)
Zhou T, Zhou G, Song W, Eguchi N, Lu W, Lundin E, Jin T, Nordberg G (1999) Cadmium-induced apoptosis and changes in expression of p53, c-jun and MT-I genes in testes and ventral prostate of rats. Toxicology 142:1–13
Zhou YF, Wu W, You Y, Chen JC (2010) Dynamics of metallothionein in organs of Carassius auratus under combined stresses of Cd and Zn. J Ecol Rural Environ 26(1):63–67 (in Chinese)
Acknowledgments
This work was supported by the National Nature Science Foundation (no. 30970361, to Lan Wang). The authors are thankful to Professor Enmin Zou and Professor Hans-Uwe Dahms for language editing of the manuscript.
Conflict of interest statement
The authors declare that there is no conflict of interest
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Markus Hecker
Rights and permissions
About this article
Cite this article
Wu, H., Xuan, R., Li, Y. et al. Effects of cadmium exposure on digestive enzymes, antioxidant enzymes, and lipid peroxidation in the freshwater crab Sinopotamon henanense . Environ Sci Pollut Res 20, 4085–4092 (2013). https://doi.org/10.1007/s11356-012-1362-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-012-1362-6