Abstract
Many of the plants found in serpentine areas are endemics and they may accumulate Ni at high concentration. High accumulation of Cr is rare, or in some views, never properly demonstrated. Generally, a very small proportion of any serpentine flora shows high accumulation of Ni, in some serpentine areas Ni accumulators are completely absent. There are approximately 570 hyperaccumulator plant species found on earth, 450 of them are Ni hyperaccumulators. A few of the Ni accumulators have potential for phytoremediation or phytomining. In the present study, 19 different Brassicaceae members growing in serpentine habitats in the district of Yahyalı, Kayseri province, Turkey viz., members of genera Aethionema, Alyssum, Arabis, Heldreichia, Hesperis, Iberis, Isatis, Microthlaspi, Odontarrhena, Pseudosempervivum and Thlaspi were investigated. Nickel concentrations in the soil and underground and aboveground parts of plants were determined by using ICP-OES. It was observed that Ni concentrations of seven taxa (Odontarrhena muralis, O. oxycarpa, Isatis cappadocica subsp. cappadocica, Microthlaspi perfoliatum, Pseudosempervivum sempervivum, Thlaspi triangulare, Thlaspi rosulare) reach the threshold criterion of 1,000 mg kg−1 for Ni hyperaccumulation. In this study Ni concentrations in aboveground tissues of I. cappadocica subsp. cappadocica are determined as 5,587 mg kg−1 (in dry weight). Accordingly, it is suggested that this taxon be added to the list of Ni hyperaccumulator plants.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The soils derived from ultramafic rocks often have a unique biogeochemistry. Serpentine soils are found in many parts of the world e.g. California, New Caledonia, Cuba, central Brazil, parts of Mediterranean Europe and Turkey. These soils have some characteristic properties: (1) deficient in essential plant nutrients such as Ca, K, and P, (2) rich in trace metal concentrations like Ni, Cr and Fe, (3) a high magnesium/calcium (Mg/Ca) quotient (Proctor and Woodell 1975; Brooks 1987; Roberts and Proctor 1992; Reeves and Adıgüzel 2008). Therefore, the serpentine vegetation is different from the vegetation of the surrounding rocks and corresponding soils. In addition, the some ultramafic areas are very rich in terms of endemic (Proctor and Woodell 1971).
Serpentinophytes are model organisms that can be used in phytoremediation technology and in the resolution of environmental problems by removing toxic doses of trace metals (Brooks 1987; Chaney et al. 2005). These plants can accumulate trace metals in their roots and shoots, at a level greater than that in the soil (Raskin et al. 1994; Sheoran et al. 2013). In other words, plants that can tolerate high metal concentration by detoxifying metal collected in organs such as stem and leaf are known as hyperaccumulators. Some of the earlier dry matter concentration thresholds for hyperaccumulation of trace elements in the terrestrial plants have been revised more recently. Recommendations by Krämer (2010) and van der Ent et al. (2013) include: Cd > 100 mg kg−1; Co, Cu 300 mg kg−1; Ni 1,000 mg kg−1; Zn 3,000 mg kg−1; Mn 10,000 mg kg−1.
More than 570 plant species have been recorded as trace metal or metalloid hyperaccumulators worldwide. Of these, at least 450 are Ni hyperaccumulators (van der Ent et al. 2013). Brassicaceae is the one of the most important families in which Ni hyperaccumulators have been found (Reeves and Adıgüzel 2008). The Brassicaceae family is of particular interest, because it includes the largest numbers of species capable of accumulating exceptionally high content of compounds of Ni, Zn and Cd (Krämer 2010).
According to Flora of Turkey records, it is reported that there are 248 plants specific to serpentine, of which 119 are serpentinophytes and 129 are serpentinophages (Özdeniz et al. 2017). 55 serpentine areas in Turkey have been examined and 60 Ni-accumulators and more than 40 serpentine-endemics have been reported from these areas (Adıgüzel and Reeves 2012). The majority of these species belong to the genera Alyssum, Bornmuellera, Cochlearia and Thlaspi under the Brassicaceae family (Brooks et al. 1979; Reeves and Brooks 1983; Reeves et al. 1983; Reeves 1988: Reeves and Adıgüzel 2004, 2008). Also the Turkish species of Cochlearia that are Ni hyperaccumulators are now placed in the genus Pseudosempervivum as P. aucheri (Boiss.) Pobed. and P. sempervivum (Boiss. & Bal.) Pobed., and Thlaspi s.l. has been broken up into several genera, several of which (including Noccaea, Microthlaspi and some species still classified under Thlaspi) contain Ni hyperaccumulators (Reeves and Adıgüzel 2004, 2008). Isatis pinnatiloba and Noccaea camlikensis are the most recent recorded species of Ni hyperaccumulators from Turkey serpentine soils (Altınözlü et al. 2012; Aksoy et al. 2015).
In this study of some of the Brassicaceae of serpentine areas in the Aladağlar district of Yahyalı (Kayseri) we aimed to investigate trace metal concentrations in underground and aboveground tissues and soil of 19 taxa growing on ultramafic soils.
Materials and methods
Materials
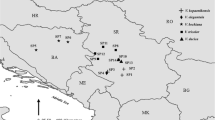
The study site is quite extensive and not uniform in its topography and vegetation. 70% of the study area is mountainous and varies in terms of vegetation cover. The large areas to the south of the area are covered with forest, while in the north the steppe vegetation is dominant. The ultramafic area of Yahyalı in Kayseri province in Turkey has a large number of limestone–serpentine contacts. The most important ultramafic rocks in this area are located in the east and south side of Aladağ. The study area is lies between 37°46–55′N and 35°19–30′E and at altitude 700–2000 m (Fig. 1).

The map is adopted from Reeves and Adıgüzel (2004)
Map of Turkey, showing areas of Ultramafic geology (in black) and the study area.
Soils and specimens of the following plant taxa were collected from this serpentine habitat in 2012 and 2013: Aethionema armenum Boiss. (J. Çelik 1060), Aethionema speciosum Boiss & A. Huet subsp. speciosum (J. Çelik 1063), Alyssum pseudomouradicum Hausskn. & Bornm. ex Baumg (J. Çelik 1078), Alyssum strigosum Banks & Sol. subsp. strigosum (J. Çelik 1018), Arabis alpina L. subsp. alpina (J. Çelik 1005), Arabis aucheri Boiss. (J. Çelik 1017), Arabis nova Vill. (J. Çelik 1042), Heldreichia bupleurifolia Boiss. subsp. rotundifolia (Boiss.) Parolly, Nordt & Mumm var. rotundifolia (J. Çelik 1114), Hesperis anatolica A. Duran (J. Çelik 1052), Iberix simplex DC. (J. Çelik 1029), Isatis cappadocica Desv. subsp. cappadocica (J. Çelik 1044), Microthlaspi perfoliatum (L.) F. K. Mey (J. Çelik 1006), Odontarrhena muralis (Waldst. & Kit) Endl. (≡ Alyssum murale) (J. Çelik 1093), Odontarrhena oxycarpa (Boiss. & Balansa) Španiel, Al-Shehbaz, D. A. German & Marhold (≡ Alyssum oxycarpum) (J. Çelik 1068), Pseudosempervivum aucheri (Boiss.) Pobed. (J. Çelik 1045), Pseudosempervivum sempervivum (Boiss. & Balansa) Pobed. (J. Çelik 1036), Thlaspi triangulare (F. K. Meyer) Greuter & Burdet. (J. Çelik 1034), Thlaspi oxyceras (Boiss.) Hedge (J. Çelik 1043) and Thlaspi rosulare Boiss. & Balansa (J. Çelik 1112) (Table 1). The specimens collected were deposited in the Herbarium of Akdeniz University.
Sampling
Soils were taken from the 0–5 cm zone, returned to the laboratory, sieved with a standard 4 mm sieve and air-dried. At least 15–30 adult plants were randomly selected and collected from each site. Five plants were retained as herbarium specimens. Other plants from each site were divided into above- and below-ground parts. These were transferred to the laboratory in plastic bags. Plant samples were washed with tap water, followed by deionized water. They were dried in an oven (80 °C) until constant weight and brittleness. The samples were subsequently ground with a pestle and mortar. Homogenized plant materials and soil samples were then stored in clean paper bags before heavy-metal analysis.
Chemical and statistical analyses
Soil samples (0.5 g dry weight) were digested with 10 mL of pure HNO3 (65%), using a CEM–MARS 5 (CEM Corporation, Matthews, NC, USA) microwave digestion system (digestion conditions were the following: maximum power 1200 W; power 100%; ramp time 20 min, pressure 180 psi; temperature 180 °C; and hold time 10 min). After digestion, the volume of each sample was adjusted to 25 mL by using double-deionized water. Homogenized plant samples (0.5 g dry weight) were also prepared using the same procedure as for heavy-metal analysis. The soil and plant samples were analyzed for Ni, Cr, Cu, Zn and Fe by inductively coupled plasma–optical emission spectroscopy (ICP–OES; Varian-Liberty II Varian Australia Pty Ltd, Mulgrave, Vic., Australia). All chemicals were of analytical reagent grade. Standard peach leaves (NIST, SRM-1547) were used as a reference material. All analytical procedures were also performed using this reference material. Soil and plant samples were digested in triplicate and analyzed. The means and standard deviation (SD) of the data were calculated by using the SPSS 15.00 version package (SPSS Inc., Chicago, IL, USA).
Results
The concentrations of trace metals (Ni, Co, Cr, Cu, Fe, Pb, Zn) in soil, aboveground and underground parts of the plants are given in Tables 2 and 3.
Ni concentration in the studied plants ranged from 8 to 8,100 mg kg−1 in the above ground (AG) parts and 10–17,000 mg kg−1 in the underground (UG) parts. The Ni concentrations in the dry weight of the aboveground parts of plants were found as follows: 2,497 mg kg−1 in Odontarrhena muralis, 1,873 mg kg−1 in O. oxycarpa, 8,085 mg kg−1 in Thlaspi triangulare, 5,587 mg kg−1 in Isatis cappadocica subsp. cappadocica, 2,270 mg kg−1 in Microthlaspi perfoliatum (= Thlaspi perfoliatum), 8,100 mg kg−1 in Pseudosempervivum sempervivum (= Cochlearia sempervivum) and 4,563 mg kg−1 in Thlaspi rosulare. Ni concentrations in the aboveground parts of these seven taxa are higher than both normal values and other taxa (Table 2). Mean concentration of Ni in the dry weight of the underground (roots) parts of plants was up to 6,390 mg kg−1 in Odontarrhena muralis, 3,408 mg kg−1 in O. oxycarpa, 17,000 mg kg−1 in Thlaspi triangulare, 1,010 mg kg−1 in Microthlaspi perfoliatum, 8,173 mg kg−1 in Pseudosempervivum aucheri, 12,600 mg kg−1 in P. sempervivum, 12,200 mg kg−1 in Thlaspi oxyceras and 5,813 mg kg−1 in T. rosulare (Table 2).
The concentrations of Ni in the aboveground and underground parts of T. rosulare and T. oxyceras collected from Yahyalı are 4,563–5,813 mg kg−1 and 33–12,000 mg kg−1 respectively (Table 2). The highest Ni concentration in a Thlaspi species was 8,085 mg kg−1 in T. triangulare from Yahyalı. Also in dry of matter aboveground parts of Microthlaspi perfoliatum (= Thlaspi perfoliatum) a concentration of 2,270 mg kg−1 was detected (Table 2). Ni concentrations in two Pseudosempervivum species collected from Yahyalı district are found to be 8,100 mg kg−1 in P. sempervivum and 101 mg kg−1 in P. aucheri (Table 2).
In our study, concentrations of Zn, Cu and Pb for three different Arabis species (A. alpina subsp. alpina, A. aucheri and A. nova) were determined to be normal, as serpentine soils are not normally enriched in these elements (Table 2). The highest Ni concentration in aboveground parts of I. cappadocica subsp. cappadocica is found to be 5,587 mg kg−1 in our study (Table 2).
Discussion
In this study Ni, Co, Cr, Cu, Fe, Pb and Zn concentrations of the Yahyalı Aladağlar serpentine soils and above-ground and underground parts of 19 species of Brassicaceae were studied to find possible relationships between soil and plant concentrations, and to seek potential accumulator plants. The concentrations of Ni, Cr and Fe in the soil of the study area exceeded the limit values stated by the Ministry of Environment and Urban Planning of Republic of Turkey (2017). Such high concentrations are one of the most prominent features of ultramafic rocks (Brooks 1987; Roberts and Proctor 1992). The following comments can be made on the accumulation of Ni and other elements in the species studied here.
Alyssum and Odontarrhena
The genus Alyssum contains the largest number of Ni hyperaccumulators known worldwide at the present time. All are in sect. Odontarrhena which has been regarded as a separate genus at various times since 1830. From about 200 currently recognised taxa in Alyssum s.l., 110 are found in Turkey, including 38 from sect. Odontarrhena. Of these, 32 have been listed as Ni hyperaccumulators by Reeves and Adıgüzel (2008); some appear to be serpentine-obligate while others are found both on serpentine and on other substrates. In a recent taxonomic and phylogenetic study genus of the Alyssum are separated 3 genera (Alyssum L., Odontarrhena C. A. Mey. ex Ledeb. and Meniocus Desv. by Španiel et al. (2015). After separation of the genus Alyssum in Turkey, there are 42 taxa in Odontarrhena, 7 species in Meniocus and remain taxa in Alyssum. All hyperaccumulator species of former genus Alyssum are now included in genus Odontarrhena. O. muralis is a polymorphic species that has been much studied over the whole of its very wide range in southeastern Europe, Turkey and further east. It is consistently found with high Ni concentrations when occurring on ultramafic soils (Doksopulo 1961; Brooks and Radford 1978). However, it is a facultative hyperaccumulator in the sense described by Pollard et al. (2014). Several subspecific taxa from non-metalliferous soils have been found to have low Ni concentrations (Reeves et al. 1983), whereas in some regions such as central and northern Greece the species is a reliable indicator of ultramafics and always has high Ni concentrations (Kelepertsis et al. 1990). In Turkey two of the three varieties of A. murale subsp. murale recognised by Dudley (1965), var. murale (now Odontarrhena muralis) and var. haradjianii (Rech.) Dudley (now Odontarrhena haradjianii) have been collected from serpentine soils and are Ni hyperaccumulators; the highest values for specimens collected in Turkey are given as 21,340 mg kg−1 for O. muralis and 16,220 mg kg−1 for O. haradjianii (Reeves and Adıgüzel 2008). In the present work the mean Ni concentration in above ground parts of O. muralis was 2,497 mg kg−1. Odontarrhena muralis specimens with up to 34,700 mg kg−1 Ni have been found in the Balkans (Bani et al. 2010), leading to extensive studies on the use of this species for phytoremediation and phytomining (Chaney et al. 2005; Bani et al. 2015).
Of the other Odontarrhena species collected, O. oxycarpa is a known Ni hyperaccumulator with up to 7,290 mg kg−1 being recorded for the specimen Kühne 1418 from the Amanus Mts. near Erzin (Hatay province) (Brooks et al. 1979; Reeves 2017, unpublished data). It is a widely scattered Turkish endemic. Other collections with 7,286–16,250 mg kg−1 Ni (Reeves and Adıgüzel 2008; Reeves 2017, unpublished data) were made from (1) the valley of Ecemiş Çayı SE of Hamidiye (Adana province); (2) 2 km SSW of Yelatan (Niğde province, on the W side of the Aladağ massif), and (3) 25 km E of Refahiye (Erzincan province). Because Brooks et al. (1979) also recorded two Ni values below 10 mg kg−1 it seems likely that this species should also be regarded as a facultative hyperaccumulator. In the present study, O. oxycarpa was found at the location given in Table 1 with a mean Ni concentration of 1,873 mg kg−1.
The two Alyssum species collected in this study are A. pseudomouradicum Hausskn. & Bornm. and A. strigosum Banks & Sol. subsp. strigosum, both in sect. Alyssum (Dudley 1965). These have the expected low Ni concentrations (no more than 12 mg kg−1), as recorded by Brooks et al. (1979). Underground parts of the plants collected in this study were: O. muralis, 6,390 mg kg−1; O. oxycarpa 3,408 mg kg−1; A. pseudomouradicum 69 mg kg−1; A. strigosum 10 mg kg−1. Again, the distinction between the hyperaccumulators and the non-accumulators is clearly seen. Close to the present study area, near Büyükcakır village (Kayseri province), Altınözlü et al. (2012) found Odontarrhena peltarioidea (Boiss.) Španiel, Al-Shehbaz, D. A. German & Marhold subsp. peltarioidea (formerly A. peltarioides Boiss. subsp. peltarioides) with 4,411 mg kg−1 Ni.
Thlaspi
Many species of Thlaspi have been well established as zinc and/or nickel hyperaccumulators, as discussed in publications involving comprehensive surveys of Thlaspi s.l. (Reeves and Brooks 1983; Reeves 1988). Nomenclature in this genus has been controversial. The revision of Meyer (1973) which recognized more than 100 species subdivided into 12 smaller genera, was rejected in the publication of Flora of Turkey (Davis et al. 1988), but has received some support from more recent molecular work (e.g. Mummenhoff and Koch 1994; Mummenhoff et al. 1997; Koch and Mummenhoff 2001; Al-Shehbaz et al. 2007; Al-Shehbaz 2014). Many Thlaspi species (including many of the Ni hyperaccumulators) have been transferred to the resurrected genus Noccaea, but hyperaccumulation is also seen in species classified in Meyer’s genera Masmenia, Microthlaspi and Thlaspiceras. Using the nomenclature of Thlaspi s.l. Reeves (1988) recorded Ni hyperaccumulation in Thlaspi cyprium, T. elegans, T. jaubertii, T. ochroleucum, T. oxyceras and T. rosulare. The listing under T. oxyceras included 11 specimens of taxa described by Meyer as species of Thlaspiceras, using small samples sent by Meyer (e.g. T. bovis, T. crassifolium, T. eigii, T. rechingeri, T. triangulare), finding very high Ni in each one. Because of the uncertainty about the status of these taxa, the results were summarized under Thlaspi oxyceras, showing 14 samples with 3,080–35,600 mg kg−1 Ni. Extreme values included 35,600 mg kg−1 in Meyer’s Thlaspiceras crassifolium, 34,700 mg kg−1 in T. dolichocarpum, 30,000 mg kg−1 in T. rechingeri and 25,000 mg kg−1 in T. triangulare. Full details of the Ni analyses and specimens of Meyer’s Thlaspiceras taxa are given in Reeves and Adıgüzel (2008). Their publication also included Ni concentrations as high as 56,020 mg kg−1 for Thlaspi oxyceras in specimens collected near Yarpuz (Osmaniye province) in the Amanus Mts.; this is one of the highest Ni concentrations recorded in plant leaves.
Analyses of 11 specimens of Thlaspi perfoliatum L. (= Microthlaspi perfoliatum (L.) F. K. Meyer) were also reported by Reeves and Adıgüzel (2008); five were found with high Ni, extreme values being 8,120 mg kg−1 for a sample from Hazar (Elazığ province) and 7,370 mg kg−1 for a sample from Kabaktepe, SW of Elbistan (Maraş province) (Reeves 2017, unpublished data).
In the present work, specimens identified as Thlaspi oxyceras, T. rosulare, T. triangulare and Microthlaspi perfoliatum have been obtained and analyzed (Table 2). The values found here for samples of above-ground material of T. rosulare (4,563 mg kg−1), T. triangulare (8,085 mg kg−1) and Microthlaspi perfoliatum (2,270 mg kg−1) confirm the earlier observations of Ni hyperaccumulation by these species, noted above. Underground parts of T. rosulare and T. triangulare had even higher Ni concentrations than the above-ground parts. The low value (33 mg kg−1) recorded here for T. oxyceras is very surprising, in the light of the consistently high values, always > 500 mg kg−1 and often > 10,000 mg kg−1, recorded for leaves of this species in Reeves and Adıgüzel (2008). In the present case, the high Ni seems confined to the underground parts.
Pseudosempervivum
Two Turkish endemic species of interest here were formerly included in Cochlearia, but were transferred to the genus Pseudosempervivum by Pobedimova as P. aucheri (Boiss.) Pobed. and P. sempervivum (Boiss. & Bal.) Pobed. These species were established as Ni hyperaccumulators under their Cochlearia names (Reeves 1988); further specimens were obtained and analyzed in the work reported by Reeves and Adıgüzel (2008) who recorded 3,130–21,550 mg kg−1 Ni in 11 specimens of P. aucheri and 2,210–34,130 mg kg−1 Ni in 10 specimens of P. sempervivum. In the present work, both species have been found in the study area; high Ni (8,100 mg kg−1) was measured in above-ground parts of P. sempervivum, but surprisingly, there was a much lower concentration (101 mg kg−1) in P. aucheri, although both species had very high Ni in underground parts.
Isatis
It has been stated (Reeves 2017, unpublished data) that analysis of 24 specimens of 17 different Isatis taxa from Turkey showed that all but two had Ni concentrations below 20 mg kg−1. Some of these were definitely from serpentine soils, some were possibly from serpentine (because of their collection locations) and others were from other substrates. The samples were from both herbarium and field collections. The two exceptions were serpentine specimens: I. huber-morathii Davis with 34 mg kg−1 from Sopan dağ, Kayseri, 15 km S of Pınarbaşı (Hüber–Morath 11017) and I. pinnatiloba Davis with 33 mg kg−1 from Marmaris–Enecik, Muğla (Davis 25334). A specimen of I. glauca ssp. exauriculata collected in 2001 from serpentine soil on the road from Kurutilek–Karataş, Erzincan was found to have 14.7 mg kg−1 Ni (Reeves 2017, unpublished data). All these values are normal for plants on serpentine.
However, Altınözlü et al. (2012) collected I. pinnatiloba from various serpentine localities in SW Turkey, and state that 7 specimens showed 23–1,441 mg kg−1 Ni, two of them having more than 1,000 mg kg−1 (1,441; 1,288). These authors show a strange and inexplicable variation of plant Ni with DTPA-extractable Ni in the soil (the highest foliar Ni was from soil with the lowest extractable Ni). The exact localities of the high-Ni specimens were not stated. They also recorded 275 mg kg−1 in this species in greenhouse experiments, and indicated that I. pinnatiloba should be considered as a new Ni hyperaccumulator. This species is endemic to SW Turkey (Muğla province) and is recorded as occurring on both limestone and serpentine (Davis 1965); it has been noted as a species at Kersele bay, L. Köyceğiz (Reeves 2017, unpublished data), possibly on serpentine. It may be a facultative hyperaccumulator of Ni in the sense of Pollard et al. (2014). However, the wide range of Ni concentrations for specimens all collected from serpentine probably puts this species in the category of ‘erratic’ hyperaccumulation as described by Reeves et al. (2015) for Pimelea leptospermoides in Queensland, Australia, where it was shown that pH variation, even for a serpentine endemic, could produce a wide variation in Ni uptake. Possibly this is also the explanation for the variable Ni concentrations in serpentine specimens of I. pinnatiloba.
Isatis cappadocica (with its several subspecific taxa) is a much more widespread species, occurring not only Turkey but in Armenia, Azerbaijan, Georgia, Iraq and Iran, on a wide variety of substrates. A specimen of subsp. alyssifolia (Davis 31671) collected from Keşiş dağ above Cimin (Üzümlü) may well have been from an ultramafic substrate, but was found to have only 10 mg kg−1 Ni (Reeves 2017, unpublished data). The present observation of 5,587 mg kg−1 in I. cappadocica should stimulate further investigation of this genus in Turkey. This should include analyses of specimens from known ultramafic substrates and comparison with those from other soils, together with detailed analysis of the soils themselves (including pH measurements and total and extractable metal concentrations). This would help to ascertain the conditions under which Ni hyperaccumulation in this species can be expected. It is also of interest that this species has also been recorded as a hyyperaccumulator of arsenic, with up to 3,000 mg kg−1 (Karimi et al. 2010).
Other Isatis species in Turkey may be suitable subjects for further work on possible Ni accumulation: (1) I. spectabilis may be a serpentine endemic, being found on ‘igneous slopes’ of the Tunceli–Pülümür region (Davis 1965); (2) several other species may be serpentine endemic or may occur sometimes on serpentine: I. amani Davis; I. takhtajanii (from Erzincan-Keşiş dağ); I. huber-morathii (but as noted above, the Type, from serpentine has only 34 mg kg−1 Ni); I. davisiana H. Misirdali.
Other genera
Species analyzed from the sites studied in the present work in Aethionema, Arabis, Heldreichia, Hesperis and Iberis have not exhibited Ni hyperaccumulation, although Aethionema speciosum had 593 mg kg−1 Ni. It is noted that Ae. spicatum has been found with 1,110 mg kg−1 (Reeves et al. 2001).
Elements other than Ni. Strongly elevated Cu concentrations are only occasionally found in ultramafic soils: examples from Malaysia and Brazil (with soil Cu sometimes exceeding 5,000 mg kg−1) have been discussed by van der Ent and Reeves (2015). Even here, however, the foliar Cu concentrations are generally tightly controlled so that concentrations above 75 mg kg−1 are rarely found. In the present work, soil Cu concentrations are not abnormal and all foliar Cu concentrations lie in the range 1–8 mg kg−1.
Where high Ni concentrations in plants from ultramafic soils are accompanied by unusually high concentrations of both Cr (> 15 mg kg−1) and Fe (> 1,500 mg kg−1), this suggests the possibility of soil contamination. However, in this study (Table 2) there is no evidence of such a problem in the analyses of the above-ground plant parts. High Fe concentrations in the underground parts, as seen in some analyses here, are not unusual, especially as insoluble coatings rich in Fe and some other elements can form on root surfaces and the Fe concentrations in ultramafic soils are naturally high. There is nothing abnormal about the Zn concentrations in either the soils or the plants analyzed in the present work.
Conclusions
Hyperaccumulation of Ni has been observed in seven of the species from the ultramafic soils of the Aladağlar area near Yahyalı (Kayseri province), Turkey. Six of these were previously recorded as hyperaccumulators from studies in other ultramafic areas of Turkey. However, the record of 5,587 mg kg−1 in Isatis cappadocica adds another species to the list of Turkish hyperaccumulators. As it occurs on other substrates with low Ni in both the soils and plants, it may be regarded as a facultative hyperaccumulator. Further studies are needed of about species of this genus on ultramafic soils. Of the species studied here Odontarrhena muralis is the one with the greatest potential for phytoremediation and phytomining.
References
Adıgüzel N, Reeves RD (2012) Important serpentine areas of Turkey and distribution patterns of serpentine endemics and nickel accumulators. Bocconea 24:7–17
Aksoy A, Leblebici Z, Prasad MNV (2015) Metal–accumulating plants from serpentine habitats of Kızıldağ, Konya Province, Turkey. Aust J Bot 63:372–378
Al-Shehbaz IA (2014) A synopsis of the genus Noccaea (Coluteocarpeae, Brassicaceae). Harv Papers Bot 19:25–51
Al-Shehbaz IA, Mutlu B, Donmez AA (2007) The Brassicaceae (Cruciferae) of Turkey, updated. Turk J Bot 31:327–336
Altınözlü H, Karagöz A, Polat T, Ünver İ (2012) Nickel hyperaccumulation by natural plants in Turkish serpentine soils. Turk J Bot 36:269–280
Bani A, Pavlova D, Echevarria G, Mullaj A, Reeves RD, Morel JL, Sulçe S (2010) Nickel hyperaccumulation by species of Alyssum and Thlaspi (Brassicaceae) from the ultramafics of the Balkans. Botanica Serbica 34:3–14
Bani A, Echevarria G, Sulçe S, Morel JL (2015) improving the agronomy of Alyssum murale for extensive phytomining: a 5-year field study. Int J Phytoremed 17:117–127
Brooks RR (1987) Serpentine and its vegetation: a multidisciplinary approach. Dioscorides Press, Portland
Brooks RR, Radford CC (1978) Nickel accumulation by European species of the genus Alyssum. Proc R Soc Lond B Biol Sci 200:217–224
Brooks RR, Morrison RS, Reeves RD, Dudley TR, Akman Y (1979) Hyperaccumulation of nickel by Alyssum Linnaeus (Cruciferae). Proc R Soc Lond B Biol Sci 203:387–403
Chaney RL, Angle JS, McIntosh MS, Reeves RD, Li YM, Brewer EP, Chen KY, Roseberg RJ, Perner H, Synkowski EC, Broadhurst CL, Wang S, Baker AJM (2005) Using hyperaccumulator plants to phytoextract soil Ni and Cd. Z Naturforsch C 60:190–198
Davis PH (1965) Isatis. In: Davis PH (ed) Flora of Turkey, vol 1. Edinburgh Univ Press, Edinburgh, pp 287–307
Davis PH, Mill RR, Tan K (1988) Cruciferae. Flora of Turkey and the East Aegean Islands, vol 10. University of Edinburgh Press, Edinburgh, pp 29–58
Doksopulo EP (1961) Nickel in rocks, soils, water and plants adjacent to the talc deposits of the Chorchanskaya group. Izdat Tbilisk Univ, Tbilisi
Dudley TR (1965) Alyssum L. In: Davis PH (ed) Flora of Turkey and the East Aegean Islands, vol 1. University of Edinburgh Press, Edinburgh, pp 362–409
Karimi N, Ghaderian SM, Maroofi H, Schat H (2010) Analysis of arsenic in soil and vegetation of a contaminated area in Zarshuran. Iran Int J Phytoremediat 12:159–173
Kelepertsis AE, Reeves RD, Baker AJM (1990) The use of the genus Alyssum as a safe geobotanical–biogeochemical indicator for geological mapping of ultrabasic rocks in Greece. Praktika tis Akademias Athinon 65:170–176
Koch M, Mummenhoff K (2001) Thlaspi s.str. (Brassicaceae) versus Thlaspi s.l. morphological and anatomical characters in the light of ITS and nrDNA sequence data. Plant Syst Evol 227:209–225
Krämer U (2010) Metal hyperaccumulation in plants. Annu Rev Plant Biol 61:517–534
Meyer FK (1973) Conspectus der “Thlaspi”-Arten Europas, Afrikas und Vorderasiens. Feddes Rep 84:449–470
Ministry of Environment and Urban Planning of Republic of Turkey (2017) Heavy metal limit values in soil. http://sgb.csb.gov.tr/mevzuat/dosyalar/r_20131001003814507_7542c1f8-141f-4e9d-bc2f-3c94f17f0c62.pdf. Accessed 29 May 2017
Mummenhoff K, Koch M (1994) Chloroplast DNA restriction site variation and phylogenetic relationships in the genus Thlaspi sensu lato (Brassicaceae). Syst Bot 19:73–88
Mummenhoff K, Franzke A, Koch M (1997) Molecular phylogenetics of Thlaspi s.l. (Brassicaceae) based on chloroplast DNA restriction site variation and sequences of internal transcribed spacers of nuclear ribosomal DNA. Can J Bot 75:469–482
Özdeniz E, Özbey BG, Kurt L, Bölükbaşı A (2017) Serpantin ekolojisi ve Türkiye serpantin florası’na katkılar. Toprak Bilimi ve Bitki Besleme Dergisi 5(1):22–33
Pollard AJ, Reeves RD, Baker AJM (2014) Facultative hyperaccumulation of heavy metals and metalloids. Plant Sci 217–218:8–17
Proctor J, Woodell SRJ (1971) The plant ecology of serpentine: i. Serpentine vegetation of England and Scotland. J Ecol 59(2):375–395
Proctor J, Woodell SRJ (1975) The ecology of serpentine soils. Adv Ecol Res 9:255–366
Raskin I, Nanda Kumar PBA, Dushenkov S, Salt DE (1994) Bioconcentration of heavy metals by plants. Curr Opin Biotech 5(3):285–290
Reeves RD (1988) Nickel and zinc accumulation by species of Thlaspi L., Cochlearia L. and other genera of the Brassicaceae. Taxon 37:309–318
Reeves RD, Adıgüzel N (2004) Rare plants and nickel accumulators fromTurkish serpentine soils, with special reference to Centaurea species. Turk J Bot 28:147–153
Reeves RD, Adıgüzel N (2008) The nickel hyperaccumulating plants of the serpentines of Turkey and adjacent areas: a review with new data. Turk J Biol 32:143–153
Reeves RD, Brooks RR (1983) European species of Thlaspi L. (Cruciferae) as indicators of nickel and zinc. J Geochem Explor 18:275–283
Reeves RD, Brooks RR, Dudley TR (1983) Uptake of nickel by species of Alyssum, Bornmuellera, and other genera of Old World Tribus Alysseae. Taxon 32:184–192
Reeves RD, Kruckeberg AR, Adıgüzel N, Krämer U (2001) Studies on the flora of serpentine and other metalliferous areas of western Turkey. S Afr J Sci 97:513–517
Reeves RD, Laidlaw WS, Doronila A, Baker AJM, Batianoff GN (2015) Erratic hyperaccumulation of nickel, with particular reference to the Queensland serpentine endemic Pimelea leptospermoides F. Mueller. Aust J Bot 63:119–127
Roberts BA, Proctor J (eds) (1992) The ecology of areas with serpentinized rocks: a world view. Kluwer Academic Publishers, Dordrecht
Sheoran V, Sheoran AS, Poonia P (2013) Phytomining of gold: a review. J Geochem Explor 128:42–50
Španiel S, Kempa M, Salmerón-Sánchez E, Fuertes-Aguilar J, Mota JF, Al-Shehbaz IA, German DA, Olšavská K, Šingliarová B, Zozomová-Lihová J, Marhold K (2015) AlyBase: database of names, chromosome numbers, and ploidy levels of Alyssae (Brassicaceae), with a generic concept of the tribe. Plant Syst Evol 301:2463–2491
van der Ent A, Reeves RD (2015) Foliar metal accumulation in plants from copper-rich ultramafic outcrops: case studies from Malaysia and Brazil. Plant Soil 389:401–418
van der Ent A, Baker AJM, Reeves RD, Pollard AJ, Schat H (2013) Hyperaccumulators of metal and metalloid trace elements: facts and fiction. Plant Soil 362:319–334
Acknowledgements
This study was funded by Nevşehir Hacıbektaş Veli University Scientific Research Projects Coordination Unit as Project No. NEUBAP-13F28. The authors thanks Professor Roger D. Reeves for critical reading, improving quality of this manuscript and improving the English of this manuscript. Also, we would like to thanks Prof. Dr. Şinasi Yıldırımlı for identification of Isatis cappadocica subsp. cappadocica.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Çelik, J., Aksoy, A. & Leblebici, Z. Metal hyperaccumulating Brassicaceae from the ultramafic area of Yahyalı in Kayseri province, Turkey. Ecol Res 33, 705–713 (2018). https://doi.org/10.1007/s11284-018-1606-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11284-018-1606-0