Abstract
Database of published chromosome numbers and ploidy-level estimates of the tribe Alysseae is presented, together with the revised generic concept and the list of accepted names, to reflect the most recent taxonomic and phylogenetic studies in Alysseae. It is available on-line at www.alysseae.sav.sk. The tribe encompasses 24 genera and 277 species. Chromosome numbers and/or ploidy levels are known for 171 out of 297 recognized taxa. Of these, 95 (55.6 %) taxa are diploids, 43 (25.1 %) are polyploids, and 33 (19.3 %) involve both diploids and polyploids. The most common base chromosome number in the tribe is x = 8 and less frequent is x = 7. The highest variation in base chromosome numbers (x = 7, 8, 11, 15) is found in the genus Hormathophylla. A key to all genera and descriptions of the two new genera Cuprella and Resetnikia are presented. Many new nomenclatural combinations, mainly in the re-established Odontarrhena (77), are proposed.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Alysseae DC. is the third largest tribe of the family Brassicaceae (Cruciferae). Its native range is Eurasia and North Africa, and the center of its greatest diversity is the Mediterranean and Irano-Turanian regions. Members of the Alysseae are annual or perennial herbs or subshrubs morphologically characterized by having stellate trichomes, yellow or white (rarely pink) petals, appendaged filaments, and latiseptate or terete (rarely angustiseptate) few-seeded silicles (Al-Shehbaz 1987; Warwick et al. 2008).
Phylogenetic relationships in the Alysseae have been poorly explored until a recent series of molecular studies (Beilstein et al. 2008, ndhF, phyA; Warwick et al. 2008, 2010, ITS; Cecchi et al. 2010, ITS; German et al. 2009, ITS; Khosravi et al. 2009, ITS; Rešetnik et al. 2013, ITS, ndhF, trnL-F; Li et al. 2014, ITS) have demonstrated that the traditional morphology-based taxonomic concept of the tribe (Schulz 1936; Janchen 1942; Dorofeyev 2004) was artificial. In a treatment adopted in the most recent synopsis of the family Brassicaceae (Al-Shehbaz 2012), the tribe Alysseae comprised 262 species classified in 17 genera. This novel approach included several important changes in the delimitation of Alysseae based on molecular evidence, such as the transfer of Alyssum klimesii Al-Shehbaz [now Ladakiella klimesii (Al-Shehbaz) D.A.German & Al-Shehbaz] to Crucihimalayeae D.A.German & Al-Shehbaz (German et al. 2009; German and Al-Shehbaz 2010), transfer of Athysanus Greene and return of Botschantzevia Nabiev to Arabideae DC. (Warwick et al. 2008), the assignment of Farsetia Turra and Lobularia Desv. at first to Malcolmieae Al-Shehbaz & Warwick (Warwick et al. 2008) and later to Anastaticeae DC. (Warwick et al. 2010; Al-Shehbaz 2012), transfer of Leptoplax O.E.Schulz from Thlaspideae DC. to Alysseae (Cecchi et al. 2010), exclusion of Ptilotrichum C.A.Mey. (Warwick et al. 2008) and its placement in Stevenieae Al-Shehbaz et al. (Al-Shehbaz et al. 2011), the assignment of previously unplaced Physocardamum Hedge to Alysseae (Warwick et al. 2010), and the segregation of previously unrecognized genera Lepidotrichum Velen. & Bornm. and Phyllolepidum Trinajstić from Aurinia Desv. (Cecchi 2011). A recent comprehensive phylogenetic study by Rešetnik et al. (2013) supported the above-mentioned separation of Phyllolepidum and Lepidotrichum, suggested the placement of Leptoplax and Physocardamum in Bornmuellera Hausskn. (relevant combinations were published by Rešetnik et al. 2014), and demonstrated that several genera (Alyssoides Mill., Alyssum L., Fibigia Medik.) are para- or polyphyletic.
During the last decade, many genus- and species-level studies of the Alysseae dealt with (micro) evolution, origin of polyploids, phylogeography, population genetics, taxonomic diversity or nomenclature (Iljinska 2005; Ortiz and Rodríguez Oubiña 2005; Ančev and Goranova 2006; Orcan 2006; Wesche et al. 2006; Çelik et al. 2007; Iljinska et al. 2007; Avetisian 2009; Orcan and Binzet 2009; German 2010, 2011, 2012b, 2014a, b, c; German and Al-Shehbaz 2010; Cecchi 2011; Knjasev 2011; Marhold et al. 2011; Meyer 2011; Pakravan et al. 2011; Španiel et al. 2011a, b, 2012a, b; Çetin et al. 2012; Rusterholz et al. 2012; Yılmaz 2012; Al-Shehbaz 2013; Cecchi et al. 2013; Kavousi et al. 2014; Magauer et al. 2014; Ranjbar et al. 2014; Rešetnik et al. 2014; Zozomová-Lihová et al. 2014). Other studies in Alysseae focused on heavy-metals tolerance and uptake and potential for phytoremediation and phytomining applications (e.g., Cecchi et al. 2013; Adamidis et al. 2014; Bani et al. 2015; Ghaderian et al. 2015; Morais et al. 2015; and references therein).
The growing interest in the Alysseae and the increasing amount of molecular and other data contributed considerably to our understanding of the phylogenetic relationships in the tribe and emphasized the need to carry out necessary changes in circumscriptions and nomenclature of taxa. Although several phylogenetic incongruencies still remain to be addressed, the available data enable us to make substantial taxonomic re-arrangements among the most controversial genera of the Alysseae.
The Brassicaceae family is well known for the common occurrence of polyploids and considerable variation in chromosome numbers (Marhold and Lihová 2006). Three ancient whole-genome duplication events, named γ, β, α, were probably experienced by all core Brassicaceae, of which the most recent At-α event might have triggered extensive radiation and diversification of the family. In addition, several independent mesopolyploidizations have been assumed or revealed in specific lineages (reviewed by Franzke et al. 2011). It was estimated that approximately 37 % of the species may be polyploid (Warwick and Al-Shehbaz 2006) but this figure most likely represents an underestimation (Franzke et al. 2011). From the available data, it appears that chromosome and ploidy variation is substantial also in the Alysseae, and the most frequent base chromosome number is x = 8, but x = 7, 11, 15 were also reported (Warwick and Al-Shehbaz 2006). Although the majority of small genera are diploid, genera such as Alyssum, Hormathophylla Cullen & T.R.Dudley, and Clypeola L. (as circumscribed by Al-Shehbaz 2012) include diploid, polyploid, or diploid–polyploid species. The origin of polyploids and various base chromosome numbers in the Alysseae, as well as their evolutionary significance, have not yet been sufficiently understood. To address these questions, a detailed summary of the current knowledge on karyological variation of the tribe across all genera and the whole distribution area is an essential starting point. For this purpose, chromosome number and ploidy-level database is the most convenient information tool.
Our goals in this Alysseae paper are to: (1) provide an updated generic concept, an identification key to genera, and a list of accepted species and infraspecific names that should represent the taxonomic backbone for further studies; (2) present a research platform for Alysseae taxa consisting of the database of accepted species and infraspecific names and their synonyms, and published chromosome number/ploidy-level data; (3) present new names and combinations reflecting the revised generic concept and the current state of knowledge.
Materials and methods
Generic and species concepts
The generic and species concepts of the tribe Alysseae adopted in the present paper reflect the most recent phylogenetic, taxonomic, and nomenclatural studies at the levels of tribe and above (Warwick et al. 2008; Jaén-Molina et al. 2009; Al-Shehbaz 2012; Rešetnik et al. 2013), as well as at the levels of genera and species (German 2010, 2011, 2012a, b, 2014a, b, c; German and Al-Shehbaz 2010; Cecchi 2011; Marhold et al. 2011; Španiel et al. 2011a, b, 2012a, b; Cecchi et al. 2013; Magauer et al. 2014; Rešetnik et al. 2014; Zozomová-Lihová et al. 2014). Part of the decisions was also made based on our yet-to-be-published studies focusing on Hormathophylla (E. Salmerón Sánchez et al., unpublished data) and Alyssum montanum–A. repens Baumg. complex in the Balkan Peninsula (S. Španiel et al., unpublished data).
List of accepted species and infraspecific names and synonyms
An initial list of published species and infraspecific names of the Alysseae was assembled based on the existing nomenclatural databases of vascular plants (The International Plant Names Index, www.ipni.org; The Plant List, www.theplantlist.org; Tropicos, www.tropicos.org), and the Brassicaceae species checklist (Warwick et al. 2006). All names were classified into the following categories: accepted names, provisionally accepted names, synonyms, doubtful synonyms, and unresolved names. Additionally, basionyms, illegitimate, and replaced names were identified. The effort was made to consult original publications, protologues, and original material of all controversial names of taxa before classifying them into the above-mentioned categories. The final list of accepted and provisionally accepted species and infraspecific names is available in the printed version of this paper. Furthermore, an extended version containing also synonyms, doubtful synonyms, and unresolved names is available online at AlyBase web (www.alysseae.sav.sk). It also lists designations that were not validly published but have been registered in some of the above-mentioned databases. The online version will be continuously updated, including the less frequently cited synonyms that perhaps were overlooked in the present version.
Chromosome numbers and ploidy-level data
Published data on chromosome numbers and flow cytometry (FCM)-based ploidy-level estimates of the tribe Alysseae were gathered using the dataset by Warwick and Al-Shehbaz (2006), IPCN database by Goldblatt and Johnson (1979 onwards), printed volumes of Index of plant chromosome numbers (for references see Goldblatt and Lowry 2011), IAPT/IOPB Chromosome Data reports (Marhold 2006 onwards), Mediterranean chromosome number reports (Kamari et al. 1991 onwards), and several chromosome number atlases (e.g., Bolkhovskikh et al. 1969; Agapova et al. 1990; Dobeš and Vitek 2000). In addition, more recent papers with chromosome number records were searched primarily via portal Web of Science. References to all sources of the chromosome numbers and/or FCM ploidy-level estimates are displayed in the presented AlyBase database.
The primary sources of all chromosome number records and FCM ploidy-level estimates were checked, and additional relevant information was obtained and stored in the database. Taxonomic identity of the analyzed material was ascertained according to the localities of origin and/or voucher specimens (if available).
For each record, the following data were stored (some optionally, depending on their availability in the original source):
-
1.
Taxon name: name with authorship as published in the original source (including typographical errors), standardized name of the taxon (corrected spelling of the name and its authorship), and currently accepted name of taxon after taxonomic revision of the record, taking into account the voucher specimens (if available) and/or the collection place (following the checklist of accepted and provisionally accepted names presented in this article).
-
2.
Chromosome data: mitotic and/or meiotic chromosome number, ploidy level, name of the person who counted chromosomes, number of analyzed plants, presence or absence of karyotype, idiogram, and photograph of chromosomes or their drawing in the original publication.
-
3.
Genome-size data: ploidy-level estimate, chromosome number estimate, flow-cytometric method using fluorochromes 4′,6-diamidino-2-phenylindole (DAPI) or propidium iodide (PI), number of analyzed plants.
-
4.
Locality: details on locality from which material was collected, including geographical coordinates (if coordinates are lacking in the original publication, locality was geo-referenced, but the exactness of coordinates depends on the accuracy of locality description in the original source), and classification of the locality according to the World geographical scheme for recording plant distributions (Brummitt 2001).
-
5.
Voucher specimen: collector(s) name(s), collection number, date of collection, specimen number, and the herbarium in which the collection is deposited (herbarium acronyms follow Thiers 2014; when no acronym is available, full name of the institution, city, and country are provided).
-
6.
Place of publication: the authorship and title of the paper, name of the journal, volume and pages (including exact page(s) and year in which records were published); for books, the corresponding information is provided as well.
Local geographical names, titles of papers and their author names in Cyrillic were transliterated following British standard for transliteration of Cyrillic and Greek characters (BS 2979 1958).
Technical solution of the AlyBase database
The production version of entire project is deployed on a server that runs Debian Linux 2.6, “lenny” distribution. The database platform used is PostgreSQL 8.3. In the near future, it will be migrated to Ubuntu Linux 14.04 with PostgreSQL 9.3. The web server installation is Apache 2.2 HTTP with support of PHP. Client side uses CakePHP framework with HTML5, CSS, and JavaScript with the usage of jQuery 1.10 and jQueryUI 1.11 libraries. Visualization of geographic data is done by Google Maps API v3. Database is available at www.alysseae.sav.sk.
The final product contains 17 database tables. The detailed list of fields in tables is given in Online Resource 1. We have generally followed the model suggested by Berendsohn et al. (1997) in the construction of the database structure for chromosome number and ploidy-level data. Presentation interface is in the form of a website with pages offering to search accepted names and synonyms and chromosome and ploidy-level data (including references) (Figs. 1, 2). Each of these pages offers search fields that allow users to narrow down search results.
Presentation interface of AlyBase for chromosome and ploidy-level data search (including references). Search fields allow users to narrow down search results. Search results in a list of taxa that have specified properties. Clicking on the particular name brings a list of chromosome records associated with this taxon. At the top of the list, it is possible to click on a Map tab that shows respective records on a map. One can click the pin on the map to show chromosome count of the record and further obtain the detailed page of the record
Chromosome and ploidy-level data search results in a list of taxa that have properties specified by search terms. Then, a list of chromosome records associated with a given taxon appears, including chromosome count or FCM ploidy-level estimate and publication place of the record. Furthermore, it is possible to display respective records on a map. Single chromosome-record display provides detailed information about the particular record. Data are grouped into chromosome number and/or ploidy-level estimates according to the genome size, locality, material, and reference sections for clarity. This page also contains a map showing the locality, provided it has associated geographic coordinates (either published in original source or determined by the authors of the database). Taxon name of the record is linked to the nomenclature part of the website. The connected reference page offers a list of sources of the chromosome data.
Nomenclatural data are arranged in a list of names with their places of publication and status. For synonyms, column status contains a reference to its accepted name expressed by formulation “synonym of” or “doubtful synonym of”. The page of particular name contains details of the name, including its publication place, basionym (if available), and a list of other synonyms. For all synonyms, the accepted names are given.
The detailed pages of chromosome records and nomenclatural data offer possibility to add an annotation to specific records. Annotations are revised before their publication on the web.
In order to make the process of data insertion quick and distributable, a custom administration interface was created. It allows the logged user to insert all necessary data into respective fields. Moreover, it offers the possibility to update inserted data, as well as to manage checklist of names of taxa, person names, and literature references. This interface allows more project members to insert data simultaneously without accessing the database directly at its lowest level or without any knowledge of SQL language. The server side is written in PHP5 with usage of Medoo library. Client side does not implement any framework. We used PHP5, XHTML, CSS, and JavaScript.
Results and discussion
Revised generic concept
Although the treatment of the tribe Alysseae has recently undergone a number of changes in the circumscription and internal division into genera, as already reflected in the treatment by Al-Shehbaz (2012) and its adjustments by Rešetnik et al. (2014), polyphyly of the genera Alyssoides, Alyssum, and Fibigia and the relatedness of Clypeola to Alyssum (Rešetnik et al. 2013) are still major sources of controversy in the generic treatment of the tribe. Here we summarize the necessary changes in the generic concept of Alysseae, reflecting the current knowledge on its phylogeny (Fig. 3).
Polyphyly of the genus Alyssoides, which resulted from inclusion of the two unrelated species, A. cretica (L.) Medik. and A. utriculata (L.) Medik. (the type of the genus) (Warwick et al. 2008; Rešetnik et al. 2013), should be resolved by the transfer of A. cretica to Lutzia Gand., as suggested by Rešetnik et al. (2013). As a result, both Alyssoides and Lutzia become monotypic. Indeed, Rešetnik et al. (2013) argued that such treatment was already adopted by some authors (Greuter and Raus 1983; Kit Tan 2002; Cecchi 2011).
Rešetnik et al. (2013), confirming the findings of Warwick et al. (2008) and Khosravi et al. (2009), have further revealed that Fibigia, as delimited by Al-Shehbaz (2012), is polyphyletic. In order to achieve monophyly and more balanced taxonomic treatment, it should be either (1) split into genera Acuston Raf. [F. lunarioides (Willd.) Sweet], Brachypus Ledeb. [F. suffruticosa (Vent.) Sweet], Fibigia, Irania Hadač & Chrtek (comprising all the members of Fibigia sect. Edmondia Bunge ex Boiss.), Pterygostemon V.V.Botschantz. [F. spathulata (Kar. & Kir.) B.Fedtsch.], and here described monotypic genus Resetnikia [F. triquetra (DC.) Boiss. ex Prantl] (see “Taxonomic treatment” below), or (2) merged with Alyssoides, Clastopus Bunge ex Boiss., Degenia Hayek, and Physoptychis Boiss. to form a very broadly conceived, but still monophyletic genus Alyssoides. Because the second option would make Alyssoides extremely heterogeneous morphologically, we prefer to split from Fibigia the above-mentioned five genera. None of these were accepted in the most recent generic synopsis of Al-Shehbaz (2012), and we changed the broad concept of Fibigia following Rafinesque (1838), Dorofeyev (2012), Hadač and Chrtek (1973), and Boczantzeva (1976, 1977), respectively.
The polyphyletic Alyssum currently comprises three clades (Warwick et al. 2008; Rešetnik et al. 2013; E. Salmerón Sánchez et al., unpublished data): (1) “Alyssum s.str. clade” consisting of A. sect. Alyssum, A. sect. Gamosepalum (Hausskn.) T.R.Dudley, and A. sect. Psilonema (C.A.Mey.) Hook.f. (except “A. homalocarpum-A. antiatlanticum clade”), (2) “A. homalocarpum-A. antiatlanticum clade”, and (3) “Clypeola clade”, comprising A. sect. Odontarrhena (C.A.Mey. ex Ledeb.) W.D.J.Koch, A. sect. Meniocus (Desv.) Hook.f., and Clypeola.
While preparing a phylogenetic study of the genus Hormathophylla (E. Salmerón-Sánchez et al., unpublished data), we included in our analyses Alyssum antiatlanticum Emb. & Maire and A. homalocarpum (Fisch. & C.A.Mey.) Boiss. because of their uncertain generic affinities and their possible relationship to Hormathophylla. Our cpDNA and nuclear ITS sequence data showed that both species are neither related to Hormathophylla nor to most members of A. sect. Psilonema, into which they were previously classified. Our phylogenetic analyses revealed that these two Alyssum species form an isolated, well-supported lineage outside of the genus Alyssum (Fig. 4), from which it is also biogeographically distinct (contiguous vicariant). Treatment of this lineage as a separate taxonomic entity is also supported by morphological data. Alyssum sect. Psilonema, as circumscribed by Dudley (1964a), differed from the other five sections of the genus by having wingless, toothless, and unappendaged staminal filaments plus petals scarcely longer than sepals. In addition, Maire (1967) concluded that members of this section had elongated cylindrical nectaries. The section sensu Dudley (1964a) and Maire (1967) included A. antiatlanticum, A. homalocarpum, A. alyssoides (L.) L., A. damascenum Boiss. & Gaill., A. dasycarpum Stephan ex Willd., and A. granatense Boiss. & Reut. The inclusion of the first two species, however, was not unequivocal. Alyssum homalocarpum differs from the rest of the section by its broadly obovate, truncate, glabrous fruits with papillate margins, deciduous sepals, and leaves minutely denticulate at apex (unique in Alyssum; Dudley 1968; I. A. Al-Shehbaz personal observation). According to Maire (1967), A. antiatlanticum is the most isolated species in the genus, but he assigned it to A. sect. Psilonema due to its elongated cylindrical nectaries. On the other hand, the putative assignment of A. antiatlanticum to Hormathophylla was informally proposed by Philippe Küpfer on some annotated specimens (e.g., MA 121991), as well as by Maire who highlighted its morphological proximity to H. cochleata (Coss. & Durieu) P.Küpfer and Alyssum sect. Ptilotrichum (C.A.Mey.) Hook.f. (Maire 1967; Küpfer 1974). For the above-mentioned phylogenetic and morphological reasons, we propose here Cuprella Salmerón-Sánchez et al. as a new genus in the tribe Alysseae, comprising two species, C. antiatlantica (Emb. & Maire) Salmerón-Sánchez et al. and C. homalocarpa (Fisch. & C.A.Mey.) Salmerón-Sánchez et al. (see “Taxonomic treatment” below).
Due to incongruences among the results obtained from different molecular markers, the relationships within the Clypeola clade are not yet sufficiently resolved. While Alyssum sect. Odontarrhena appears to be monophyletic, Clypeola is not consistently separated from A. sect. Meniocus and is mostly paraphyletic in relation to A. sect. Odontarrhena (Rešetnik et al. 2013). To achieve monophyly of Alyssum, apart from the recognition of Cuprella, the Clypeola clade would have to be either split into three hitherto weakly phylogenetically supported genera (Odontarrhena C.A.Mey., Meniocus Desv., and Clypeola), or kept as one monophyletic, but morphologically heterogeneous genus Clypeola (the oldest available generic name for this group). The latter solution would require numerous new nomenclatural combinations that we presently want to avoid. Therefore, we prefer to treat these three genera separately, in agreement with their morphology, until their phylogenetic relationships are fully resolved by additional molecular markers.
For the genus Meniocus, we adopted here the traditional and morphology-based treatment despite the fact that its type, M. serpyllifolius (Desf.) Desv., the only species mentioned in the protologue of the genus (Desvaux 1815), unambiguously belongs to the genus Odontarrhena. While Odontarrhena encompasses 87 perennial species with uniovulate locules, Meniocus traditionally includes seven annual species with 2–4(−8)-ovuled locules (Dudley 1964a). The silicles with many seeds are also mentioned in the original description of the genus Meniocus. Because Odontarrhena serpyllifolia (Desf.) Jord. & Fourr. [≡ Alyssum serpyllifolium Desf.] is a perennial with uniovulate locules, as all other taxa of Odontarrhena, a conservation of the name Meniocus with a more appropriate type is necessary to resolve this inconsistency.
After the exclusion of Alyssum sect. Odontarrhena, A. sect. Meniocus, A. homalocarpum, and A. antiatlanticum, Alyssum s.str. consists of two clades (Rešetnik et al. 2013; E. Salmerón Sánchez et al., unpublished data): (1) most of the annual and perennial taxa of A. sect. Alyssum, (2) A. sect. Gamosepalum, few perennials of A. sect. Alyssum, and annual A. dasycarpum. In order to resolve the sectional classification of Alyssum, future phylogenetic studies should include more species that would also help in the identification of morphological synapomorphies for its infrageneric clades.
The name Alyssum montanum, which is the lectotype of the genus Alyssum (see Green 1925), was recently proposed to be conserved with a conserved type (Marhold et al. 2011). The original lectotype of A. montanum designated by Dudley (1964a) contradicted the widespread usage of the name, as it belongs to Odontarrhena obovata C.A.Mey. Therefore, the name A. montanum should be conserved with a conserved type, which is in agreement with the protologue and the traditional use of the name and which does not disrupt the well-established usage of the names A. sect. Alyssum and A. sect. Odontarrhena (Marhold et al. 2011).
The monotypic genus Takhtajaniella V.E.Avet. (Avetisian 1980, 2013), classified in synonymy of Alyssum by Al-Shehbaz (2012), was not sampled in the published phylogenetic studies of the Alysseae, and its status will be addressed in the future. In fact, Takhtajaniella with its white petals, 2–6-rayed trichomes, and globose fruits might be close to Bornmuellera (I. A. Al-Shehbaz and D. A. German, personal observation).
It seems that there are no morphological grounds for keeping Clastopus and Straussiella Hausskn. as separate genera, as was done by Al-Shehbaz (2012). The single available sequence for Straussiella purpurea (Bunge ex Boiss.) Hausskn. (Jaén-Molina et al. 2009), which is part of ITS2, is closer to that of Clastopus vestitus (Desv.) Boiss. (96.8 % identity) than to any other sampled Alysseae.
To summarize, compared with the generic treatment of the Alysseae adopted in the most recent synopsis of the family (Al-Shehbaz 2012), we suggest reassignment of Alyssoides cretica to Lutzia, exclusion of Alyssum sect. Odontarrhena and A. sect. Meniocus from Alyssum and resurrection of the genera Odontarrhena and Meniocus, transfer of Alyssum antiatlanticum and A. homalocarpum into the newly described genus Cuprella, reassignment of Alyssum globosum Grossh. to Takhtajaniella, placement of Physocardamum davisii Hedge and Leptoplax emarginata (Boiss.) O.E.Schulz into Bornmuellera, transfer of Fibigia suffruticosa into resurrected Brachypus, reassignment of Fibigia sect. Edmondia into Irania, reassignment of Fibigia lunarioides into Acuston, transfer of Fibigia spathulata into resurrected Pterygostemon, transfer of Fibigia triquetra into the newly described genus Resetnikia, and uniting Straussiella with Clastopus.
We recognize here the following 24 genera in the tribe Alysseae (the number of species is in parentheses, accepted and provisionally accepted species are included, unresolved cases are not taken into consideration; see also Fig. 5b): Acuston (1), Alyssoides (1), Alyssum (114), Aurinia (7), Berteroa DC. (5), Bornmuellera (9), Brachypus (1), Clastopus (2), Clypeola (9), Cuprella (2), Degenia (1), Fibigia (3), Galitzkya V.V.Botschantz. (3), Hormathophylla (11), Irania (5), Lepidotrichum (1), Lutzia (1), Meniocus (7), Odontarrhena (87), Phyllolepidum (2), Physoptychis (2), Pterygostemon (1), Resetnikia (1) and Takhtajaniella (1).
a Distribution of the chromosome number reports and ploidy-level estimates among the genera of the tribe Alysseae. b Number of accepted taxa (species, subspecies, varieties, autonyms excluded) and number of the taxa with the chromosome number records and/or ploidy-level estimates in the genera of the tribe Alysseae
An identification key to the genera of the tribe is in the Taxonomic treatment below. An online interactive identification key is available at the AlyBase web.
Accepted species and infraspecific names and synonyms
The nomenclature checklist of Alysseae currently available in the AlyBase database comprises a total of 281 accepted taxa, 16 provisionally accepted taxa, and 774 synonyms. In addition, there are 53 doubtful synonyms and 70 unresolved names that require further attention (Table 1). Full list of accepted and provisionally accepted taxa, including nomenclatural novelties (new combinations), is provided in the Taxonomic treatment below. The information presented in the AlyBase checklist is coordinated with the BrassiBase database (Koch et al. 2012; Kiefer et al. 2014), nevertheless, concentration on a single tribe gave us possibility to provide much more extensive synonymy compared with BrassiBase, apart from new generic concept presented here.
Chromosome numbers and ploidy-level data
Chromosome numbers and/or ploidy-level estimates presented in the database cover all genera currently placed in the tribe Alysseae (Fig. 5) except for Takhtajaniella. Out of the total number of currently accepted species, subspecies and varieties (297, excluding nominal taxa), chromosome number data and/or ploidy-level estimates are available for 171 taxa (57.6 %). Sole ploidy-level estimates are known for four taxa. In total, the database comprises 780 records of chromosome numbers and 209 of ploidy-level estimates based on genome-size studies that were published in 213 papers.
In terms of geographical coverage (according to the level 2 of the World geographical scheme for recording plant distributions; Brummitt 2001), the highest number of records is from southeastern Europe (386), followed by western Asia (155), southwestern Europe (143), and central Europe (105). Details are shown in Fig. 6 (unassigned records on this diagram are those based on material from botanical gardens, from old literature, or from experimental papers, where the localities of origin were not given).
Geographical distribution of the chromosome number and ploidy-level records for the tribe Alysseae. The geographical division of the world follows level 2 of the World geographical scheme for recording plant distributions (Brummitt 2001). Only areas gathering at least 1 % of the chromosome number records are marked
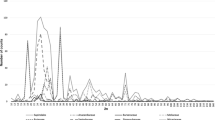
The most frequently registered ploidy level is diploid (593 records). Out of polyploids (395 records), the most often recorded ploidy level is tetraploid (341 records) and hexaploid (44 records) (Fig. 7; for the distribution details of ploidy levels in the genera Alyssum, Clypeola, Hormathophylla, and Odontarrhena see Online Resource 2).
Distribution of chromosome numbers and ploidy levels in the tribe Alysseae. For the construction of this histogram, haploid chromosome numbers were multiplied by two and ploidy-level estimates were assigned to the relevant chromosome numbers. Differences among diploids (2x) and polyploids are due to x = 7, 8, 11, 15
From the accepted taxa of the tribe for which we have chromosome number reports and/or ploidy-level estimates, 95 (55.6 %) taxa are diploid, 43 (25.1 %) polyploid, and 33 (19.3 %) with both diploids and polyploids. Based on Warwick and Al-Shehbaz (2006), the corresponding percentages for larger genera of other Brassicaceae tribes are as follows: Cardamine L.—32 % taxa diploid, 10 % both diploid and polyploid, and 58 % entirely polyploid (see also Kučera et al. 2005); Draba L.—25 % taxa diploid, 7 % both diploid and polyploid, and 68 % entirely polyploid; Lepidium L.—34 % taxa diploid, 14 % both diploid and polyploid, and 52 % entirely polyploid; Rorippa Scop.—48 % taxa diploid, 39 % both diploid and polyploid, and 13 % entirely polyploid. Such numbers underline the importance of polyploidy in the speciation and evolution of taxa of this family.
The most common base chromosome number reported for Alysseae is x = 8, less frequent is x = 7 (given for some Clypeola, Hormathophylla, and Meniocus species and for Alyssum umbellatum Desv.; for references see AlyBase). The highest variation in base chromosome numbers (x = 7, 8, 11, 15) is reported for Hormathophylla. Out of 11 species of this genus, chromosome or ploidy-level records are not available only for H. baetica P.Küpfer. Among the rest of Hormathophylla, x = 8 is reported for six species, five of them are tetraploid [H. ligustica (Breistr.) Španiel et al., H. purpurea (Lag. & Rodr.) P.Küpfer, H. pyrenaica (Lapeyr.) Cullen & T.R.Dudley, H. saxigena (Jord. & Fourn.) D.A.German & Govaerts] and one is diploid–tetraploid [H. spinosa (L.) P.Küpfer]. Base chromosome number x = 7 was recorded for three species, including the tetraploids H. cadevalliana (Pau) T.R.Dudley and H. reverchonii (Degen & Hervier) Cullen & T.R.Dudley and the octoploid H. longicaulis (Boiss.) Cullen & T.R.Dudley. Base chromosome number x = 11 is known only in H. cochleata (Coss. & Durieu) P. Küpfer and x = 15 in H. lapeyrouseana (Jord.) P. Küpfer. The taxa with 2n = 22 and 2n = 30 might be classified as secondarily diploid. The exact nature and origin of these chromosome numbers in Hormathophylla has not yet been explored. There are several taxa of Alysseae for which two different base chromosome numbers, x = 7 and x = 8, have been reported (references in AlyBase). In three such species [Alyssum szovitsianum Fisch. & C.A.Mey., Fibigia macrocarpa (Boiss.) Boiss., and Odontarrhena corsica (Duby) Španiel et al.] the chromosome counts corresponding to x = 8 are likely correct, while those corresponding to x = 7 are most probably erroneous. In Alyssum umbellatum, Meniocus linifolius (Stephan ex Willd.) DC., and M. meniocoides (Boiss.) Hadač & Chrtek, counts corresponding to x = 7 and x = 8 were reported with equal frequency, and none of them are treated here as likely incorrect. Further attention should be particularly paid to taxa of the genera Clypeola, Hormathophylla, and Meniocus to clarify the reported base chromosome number variation.
Aneuploidy was rarely reported in the Alysseae, and only eight cases are found in literature that might be considered as such (references in AlyBase). Three of them seem plausible [2n = 16, 18 in Alyssum contemptum Schott & Kotschy and A. erosulum Gennar. & Pestal., 2n = 17 in Galitzkya macrocarpa (Ikonn.-Gal.) V.V.Botschantz.], while two are likely erroneous (2n = 30 in Odontarrhena obovata C.A.Mey. and 2n = 36 in Alyssum simplex Rudolphi). Another three likely aneuploid chromosome numbers have been recorded repeatedly and thus appear plausible, but they deserve verification in future studies [2n = 46 in Alyssum hirsutum M.Bieb.; 2n = 24 and 2n = 26 in Clypeola aspera (Grauer) Turrill]. The highest polyploid recorded in Alysseae is the assumed hypododecaploid (2n = 92, in Alyssum harputicum T.R.Dudley), whose exact chromosome number needs verification. B-chromosomes were reported in Alysseae only three times [in Alyssum fastigiatum Heywood, Lutzia cretica (L.) Greuter & Burdet, and Odontarrhena tortuosa subsp. caliacrae (Nyár.) Španiel et al.; see AlyBase].
The currently available online resources on chromosome numbers mostly cover only some periods of time, certain areas, or taxonomic groups. The database of Index of Plant Chromosome Numbers (IPCN, Goldblatt and Johnson 1979 onwards), which is part of more complex Tropicos database by the Missouri Botanical Garden, is currently limited from years 1979 to 2006 (Goldblatt 2007; Goldblatt and Lowry 2011). However, final steps are being made to renew the collection of data for this database under auspices of the International Association for Plant Taxonomy (K. Marhold, unpublished data). Most recently, Chromosome Counts Database (CCDB, version 1.11) was made public (Rice et al. 2015) to represent a global and unlimited information source on chromosome numbers.
Other chromosome number databases are regionally or taxonomically restricted. These include CHROBASE—Chromosome Numbers for the Italian Flora (Bedini et al. 2010 onwards), Chromosome Number Survey of the Ferns and Flowering Plants of Slovakia (Marhold et al. 2007), Chromosome Number Database of Polish Plants (Góralski et al. 2009 onwards), BSBI Cytology Database (Botanical Society of the British Isles 2014), Chilean Plants Cytogenetic Database (Jara-Seguel and Urrutia 2011), and ChromoPar—Paraguay Chromosome Counts Database (Simon et al. 2000). Examples of taxonomically restricted databases include Index to Chromosome Numbers in Asteraceae (Watanabe 2002), Chromosome Counts in Hieracium L. (Schuhwerk 1996), BrassiBase (Koch et al. 2012; Kiefer et al. 2014), and Karyological Database of the Genus Cardamine (Kučera et al. 2005).
Unlike global sources, such as IPCN and CCDB, or widely conceived specialized BrassiBase database, which present very simple data (mostly just taxon names, chromosome numbers, and place of publication), AlyBase provides more complete information on the origin of analyzed material, voucher specimens and revision of the identification of plant material according to the collection place or voucher specimens (if available). Our aim is also to point to the records that are unequivocally erroneous or doubtful either with respect to the identity of analyzed material or chromosome count. In some cases, we suspected that meiotic counts were likely confused with mitotic ones (e.g., 2n = 8 in Alyssoides utriculata, Pogliani 1971 and Gagnidze 1983; 2n = 8 in Alyssum strictum Willd., Ghaffari 1987).
Another innovative feature of our database is that we register both the chromosome number counts and ploidy-level estimates based on flow-cytometric measurements. Flow cytometry has considerably changed the practice of studies of plant polyploids during the past two decades. Detailed studies at the population level enabled the discovery of cases of the occurrence of minority cytotypes that were not revealed using classical chromosome counting (Kron et al. 2007; Vrána et al. 2014). Such minority cytotypes are usually missing in standard-reference sources that refer solely to the chromosome number counts. The presentation of ploidy-level estimates based on flow-cytometric measurements is a practice applied recently also in the column IAPT/IOPB Chromosome Data published in Taxon (Marhold 2006 onwards).
Taxononomic treatment
Key to the genera of Alysseae
The number of ovules per ovary is an important character in distinguishing genera of the tribe. It can easily be obtained from the fruit by counting the number of seeds plus aborted ovules, or by counting the number of funicles or their scars from fruit that shed all of its seeds.
For an interactive key to the genera, see the AlyBase web.
-
1a.
Ovules 1 or 2 per ovary.
-
2a.
Annual herbs; septum absent or reduced to a rim; fruit indehiscent, samaroid, usually with setose, barbed, or hooked trichomes; ovules 1 per ovary … 9. Clypeola
-
2b.
Perennial herbs or subshrubs; septum complete; fruit dehiscent, rarely samaroid, without setose, barbed, or hooked trichomes; ovules 2 per ovary (rarely 1 in some Bornmuellera).
-
3a.
At least some trichomes malpighiaceous (2-rayed, medifixed), other trichomes 3–6-rayed; fruit inflated or rarely latiseptate … 6. Bornmuellera
-
3b.
All trichomes stellate, (6–)8–24-rayed; fruit latiseptate.
-
4a.
Filaments toothless, unappendaged; petals white; style to 0.7 mm … 16. Lepidotrichum
-
4b.
Filaments usually toothed, appendaged; petals yellow, rarely creamy white; style usually 1–3 mm … 19. Odontarrhena
-
4a.
-
3a.
-
2a.
-
1b.
Ovules 4–16 per ovary.
-
5a.
Sepals persistent well after fruit maturity, accrescent; fruit indehiscent or tardily dehiscent; seeds rugose or verrucose, wingless … 8. Clastopus
-
5b.
Sepals usually caducous well before fruit maturity, not accrescent; fruit dehiscent; seeds smooth or reticulate, winged, margined, or rarely wingless.
-
6a.
Petals deeply bifid to about middle.
-
7a.
Trichomes a mixture of simple and 2–8-rayed; lateral sepals not saccate at base; Mediterranean region, one cosmopolitan annual weed … 5. Berteroa
-
7b.
Trichomes exclusively 8–12-rayed; lateral sepals saccate at base; China, Mongolia … 13. Galitzkya
-
7a.
-
6b.
Petals obtuse, rounded, truncate, retuse, or emarginate.
-
8a.
Style (5–)6–10 mm long.
-
9a.
Petals purple or crimson red … 7. Brachypus
-
9b.
Petals yellow or white, if pinkish then plants subshrubs.
-
10a.
Filaments minutely appendaged at base; anthers apiculate; cotyledons oblique incumbent; at least some cauline leaves sinuate or dentate; lateral sepals not saccate at base … 4. Aurinia
-
10b.
Filaments unappendaged at base; anthers not apiculate; cotyledons accumbent; cauline leaves entire; lateral sepals saccate at base.
-
11a.
Fruit inflated.
-
12a.
Fruit valves thickened, leathery to subwoody, glabrous outside and inside; ovules 8–12 per ovary; at least some trichomes malpighiaceous, 2-rayed … 2. Alyssoides
-
12b.
Fruit valves papery, pubescent outside and inside; ovules 4 per ovary; all trichomes stellate, 12–20-rayed … 11. Degenia
-
12a.
-
11b.
Fruit strongly latiseptate.
-
13a.
Petals white or rarely pink; ovules 4–8 per ovary; fruit obovate, orbicular, elliptic, or cochleariform; style rarely to 5 mm, persistent … 14. Hormathophylla
-
13b.
Petals yellow; ovules 12–16 per ovary; fruit oblong to narrowly elliptic; style 7–10 mm, caducous … 23. Resetnikia
-
13a.
-
11a.
-
10a.
-
9a.
-
8b.
Style 0.1–3(–4) mm long.
-
14a.
Annual herbs.
-
15a.
Leaves distally denticulate; fruit margin papillate … 10. Cuprella
-
15b.
Leaves entire; fruit margin entire.
-
16a.
Ovules 4–8(–16) per ovary; fruit latiseptate, valves glabrous or with simple trichomes and/or papillae … 18. Meniocus
-
16b.
Ovules 4 per ovary; fruit fully or unilaterally inflated, valve glabrous or with stellate trichomes.
-
17a.
Petals yellow; infructescene distinctly elongated; fruit slightly inflated unilaterally, variously shaped … 3. Alyssum
-
17b.
Petals white; infructescene subcapitate; fruit fully inflated, globose … 24. Takhtajaniella
-
17a.
-
16a.
-
15a.
-
14b.
Perennial herbs, subshrubs, or shrubs.
-
18a.
Trichomes malpighiaceous or mixed with 3–6-rayed ones, simple trichomes absent … 6. Bornmuellera
-
18b.
Trichomes (6–)8–24-rayed, sessile or stalked, simple ones present or absent.
-
19a.
All filaments unappendaged, toothless, and wingless.
-
20a.
Fruit ovoid to globose-ovoid, strongly inflated; ovules 8–16 per ovary; cotyledons incumbent … 17. Lutzia
-
20b.
Fruit obovate, orbicular, elliptic, or cochleariform, strongly latiseptate; ovules 4–8 per ovary; cotyledons accumbent.
-
21a.
Petals yellow, shorter than or subequaling sepals, claw undifferentiated from blade … 10. Cuprella
-
21b.
Petals white or pink, distinctly longer than sepals, claw differentiated from blade.
-
22a.
Fruit replum concealed by valve margins; ovules 4 per ovary; nectar glands 1, confluent, median glands present … 20. Phyllolepidum
-
22b.
Fruit replum visible; ovules 4–8 per ovary; nectar glands 4, 1 on each side of lateral stamen, median glands absent … 14. Hormathophylla
-
22a.
-
21a.
-
20a.
-
19b.
At least median or lateral filaments appendaged, winged, or toothed.
-
23a.
Stigma strongly 2-lobed, lobes connivent, decurrent … 1. Acuston
-
23b.
Stigma entire, rarely slightly 2-lobed, lobes neither connivent nor decurrent.
-
24a.
Replum visible, not concealed by distinct valves margin; anthers apiculate (except for Alyssum).
-
25a.
Ovules 4–8(–16) per ovary; leaves dentate, sinuate, or pinnatifid, very rarely entire; cotyledons obliquely incumbent … 4. Aurinia
-
25b.
Ovules 4 per ovary; leaves entire; cotyledons accumbent.
-
26a.
Anthers not apiculate; fruit usually dehiscent, 2–4-seeded; seeds usually winged; valves not veined; style 0.5–3 mm long … 3. Alyssum
-
26b.
Anthers apiculate; fruit tardily dehiscent, 1-seeded; seeds wingless; valves reticulate veined; style 2.5–4 mm long … 15. Irania
-
26a.
-
25a.
-
24b.
Replum not visible, concealed by connate valves margin; anthers not apiculate.
-
27a.
Fruit indehiscent, globose or ovoid, inflated and bladderlike … 21. Physoptychis
-
27b.
Fruits dehiscent, oblong, orbicular to ovate, not inflated.
-
28a.
Ovules 12–16 per ovary; racemes elongated considerably in fruit, bracteate or ebracteate; style persistent; trichomes 6–8-rayed … 12. Fibigia
-
28b.
Ovules 4–8 per ovary; racemes slightly elongated in fruit, ebracteate; style caducous; trichomes 12–20-rayed … 22. Pterygostemon
-
28a.
-
27a.
-
24a.
-
23a.
-
19a.
-
18a.
-
14a.
-
8a.
-
6a.
-
5a.
List of names
The list contains the names of genera (numbered and in bold; with synonymy and distribution) and accepted and provisionally accepted (marked by #) names of species and infraspecific taxa in Alysseae, including nomenclatural novelties (in bold) and descriptions of new genera Cuprella and Resetnikia.
1. Acuston Raf., Sylva Tell. 131. 1838. [Oct–Dec 1838]–[1 sp.]. —TYPE: Acuston lunarioides (Willd.) Raf.
≡ Pevalekia Trinajstić
Distribution area: Greece (Crete, Cyclades, Dodecanese).
Acuston lunarioides (Willd.) Raf., Sylva Tellur. 131. 1838.
2. Alyssoides Mill., Gard. Dict. Abr., ed. 4. 1754. [28 Jan 1754]–[1 sp.]. —TYPE: Alyssum utriculatum L. [≡ Alyssoides utriculata (L.) Medik.].
≡ Cistocarpium Spach; = Vesicaria Adans.
Distribution area: Albania, Bosnia and Herzegovina, Bulgaria, Croatia, France, Greece, Gruziya, Italy, Macedonia, Montenegro, Romania, Russia, Serbia, Switzerland, Turkey.
Alyssoides utriculata (L.) Medik., Philos. Bot. 1: 189. 1789.
Alyssoides utriculata (L.) Medik. subsp. utriculata
Alyssoides utriculata subsp. bulgarica (Sagorski) Hartvig in Strid, Mount. Fl. Greece 1: 277. 1986.
3. Alyssum L., Sp. Pl.: 650. 1753. [1 Mai 1753]–[114 spp.]. —LECTOTYPE: Alyssum montanum L. (designated by Green 1925: 52).
= Gamosepalum Hausskn.; = Psilonema C.A.Mey.
Distribution area: throughout North Africa, Asia, and Europe; several species naturalized in North and South America, South Africa, and Australia.
Alyssum aizoides Boiss., Ann. Sci. Nat., Bot. sér. 2, 17: 153. 1842.
Alyssum alyssoides (L.) L., Amoen. Acad. 4 (Fl. Monsp.): 487. 1759; Reg. Veg. Syst. Nat., ed. 10, 2: 1130. 1759.
Alyssum andinum Rupr., Mém. Acad. Imp. Sci. Saint-Pétersbourg, sér. 7, 15(2) (Fl. Cauc.): 103. 1869.
Alyssum argyrophyllum Schott & Kotschy, Oesterr. Bot. Wochenbl 7: 229. 1857.
Alyssum armenum Boiss., Fl. Orient. 1: 278. 1867.
Alyssum artwinense N.Busch, Fl. Cauc. Crit. 3(4): 566. 1909.
Alyssum atlanticum Desf., Fl. Atlant. 2: 71, tab 149. 1798.
Alyssum aurantiacum Boiss., Fl. Orient. 1: 276. 1867.
Alyssum austrodalmaticum Trinajstić, Suppl. Fl. Anal. Jugosl. 8: 7. 1982.
#Alyssum bargalense Micevski, Fragm. Bot. Mus. Maced. Sci. Natur. 12(10): 105. 1985.
Alyssum baumgartnerianum Bornm. ex Jos.Baumgartner, Jahresber. Kais.-Franz-Jos. Oberrealschule Baden b. Wien 48: 16. 1911.
Alyssum blancheanum Gomb., Bull. Soc. Bot. France 112: 320. 1966.
Alyssum bornmuelleri Hausskn. ex Degen, Oesterr. Bot. Z. 48: 108. 1898.
Alyssum bosniacum Beck, Glasn. Zem. Mus. Bosn. i Hercegov. 28: 122. 1916, nom. alt.
Alyssum bulbotrichum Hausskn. & Bornm., Mitt. Thüring. Bot. Vereins 20: 3. 1905.
Alyssum cacuminum Španiel, Marhold & Lihová, Taxon 63: 585. 2014.
Alyssum caespitosum Jos.Baumgartner, Jahresber. Nieder-Österr. Landes-Lehrersem. Wiener-Neustadt 36: 26. 1909.
#Alyssum calycocarpum Rupr., Mém. Acad. Imp. Sci. St.-Pétersbourg, sér. 7, 15(2): 103. 1869.
Alyssum cephalotes Boiss., Diagn. Pl. Orient. Nov., ser. 2, 3(1): 34. 1854.
#Alyssum clausonis Pomel, Nouv. Mat. Fl. Atl.: 236. 1874.
Alyssum contemptum Schott & Kotschy, Österr. Bot. Wochenbl. 4: 177. 1854.
Alyssum corningii T.R.Dudley, J. Arnold Arbor. 45: 72. 1964.
Alyssum cuneifolium Ten., Flora Napol. 1: XXXVII. 1812.
Alyssum dagestanicum Rupr., Mém. Acad. Imp. Sci. Saint-Pétersbourg, sér. 7, 15(2) (Fl. Cauc.): 104. 1869.
Alyssum damascenum Boiss. & Gaill., Diagn. Pl. Orient. Nov., ser. 2, 3(6): 18. 1859.
Alyssum dasycarpum Stephan ex Willd., Sp. Pl., ed. 4, 3(1): 469. 1800.
#Alyssum decoloratum Pomel, Nouv. Mat. Fl. Atl.: 236. 1874.
Alyssum diffusum Ten., Flora Napol. 1: XXXVII. 1812.
Alyssum diffusum Ten. subsp. diffusum
Alyssum diffusum subsp. calabricum Španiel, Marhold, N.G.Passal. & Lihová, Amer. J. Bot. 98: 1900. 2011.
Alyssum diffusum subsp. garganicum Španiel, Marhold, N.G.Passal. & Lihová, Amer. J. Bot. 98: 1900. 2011.
Alyssum djurdjurae Chabert, Bull. Soc. Bot. France 36: 19. 1889.
Alyssum doerfleri Degen, Denkschr. Kaiserl. Akad. Wiss., Wien. Math.-Naturwiss. Kl. 64: 708. 1897.
#Alyssum embergeri Quézel, Bull. Soc. Sci. Nat. Maroc 31: 254. 1953.
Alyssum erosulum Gennari & Pestal. ex Clementi, Mem. Reale Accad. Sci. Torino, ser. 2, 16: 248. 1857.
Alyssum fastigiatum Heywood, Bull. Brit. Mus. (Nat. Hist.) Bot. 1: 92. 1954.
#Alyssum flahaultianum Emb. ex Emb., Willdenowia 15: 62. 1985.
Alyssum flexicaule Jord., Observ. Pl. Nouv. 1: 12, pl. 1. 1846.
Alyssum foliosum Bory & Chaub., Expéd. Sci. Morée 3(2), Bot.: 185. 1832.
Alyssum fulvescens Sm., Fl. Graec. Prodr. 2(1): 13. 1813.
Alyssum fulvescens Sm. var. fulvescens
Alyssum fulvescens var. stellatocarpum Hub.-Mor., Feddes Repert. 48: 274. 1940.
Alyssum gallaecicum (S.Ortiz) Španiel, Marhold, Lihová, Taxon 63: 585. 2014.
Alyssum gmelinii Jord. & Fourr., Brev. Pl. Nov. 2: 8. 1868.
Alyssum granatense Boiss. & Reut., Pugillus Pl. Nov. Afr. Bor. Hispan.: 9. 1852 [Jan 1852].
Alyssum gustavssonii Hartvig, Mount. Fl. Greece 1: 293. 1986.
Alyssum hajastanum Avet., Izv. Biol. Nauki 14(11): 93. 1961.
Alyssum handelii Hayek, Beih. Bot. Centralbl. 45(2): 279. 1928.
Alyssum harputicum T.R.Dudley, J. Arnold Arbor. 45: 76. 1964.
Alyssum hezarmasjedense Kavousi & Nazary, Novon 23: 59. 2014.
Alyssum hirsutum M.Bieb., Fl. Taur.-Cauc. 2: 106. 1808.
Alyssum idaeum Boiss. & Heldr., Diagn. Pl. Orient. 8: 35. 1849.
Alyssum iljinskae V.I.Dorof., Fl. Russia 4: 486. 2001.
Alyssum iranicum Hausskn. ex Jos.Baumgartner, Jahresber. Landes-Gymn. Baden b. Wien 48: 9. 1911.
Alyssum kaynakiae Yılmaz, Nordic J. Bot. 30: 190. 2012.
#Alyssum kotovii A.P.Iljinsk., Ukrayins’k. Bot. Zhurn., n.s. 45(3): 82. 1988.
Alyssum lanceolatum Jos.Baumgartner, Jahresber. Landes-Gymn. Baden b. Wien 48: 11. 1911.
Alyssum lassiticum Halácsy, Consp. Fl. Graec., Suppl. 1: 10. 1908.
Alyssum lenense Adams, Mém. Soc. Imp. Naturalistes Moscou 5: 110. 1817.
Alyssum lepidoto-stellatum (Hausskn. & Bornm.) T.R.Dudley, J. Arnold Arbor. 45: 70. 1964.
Alyssum lepidotum Boiss., Diagn. Pl. Orient. Nov., ser. 1, 1(1): 73. 1843.
Alyssum loiseleurii P.Fourn., Quatre Fl. France: 425. 1936.
Alyssum lycaonicum (O.E.Schulz) T.R.Dudley, J. Arnold Arbor. 45: 79. 1964.
Alyssum macrocalyx Coss. & Durieu, Bull. Soc. Bot. France 4: 12. 1857.
Alyssum macropodum Boiss. & Balansa, Diagn. Pl. Orient. Nov., ser. 2, 3(6): 18. 1859.
Alyssum minutum Schltdl. ex DC., Reg. Veg. Syst. Nat. 2: 316. 1821.
Alyssum misirdalianum Orcan & Binzet, Novon 19: 494. 2009.
Alyssum moellendorfianum Asch. ex Beck, Ann. K. K. Naturhist. Hofmus. 2: 73, 174. 1887.
Alyssum montanum L., Sp. Pl.: 650. 1753, nom. cons. prop.
Alyssum mouradicum Boiss. & Balansa, Diagn. Pl. Orient. Nov., ser. 2, 6: 16. 1859.
Alyssum mozaffarianii Kavousi, Iran J. Bot. 9: 48. 2001.
Alyssum muelleri Boiss. & Buhse, Nouv. Mém. Soc. Imp. Naturalistes Moscou 12 (Aufzähl.): 16. 1860.
Alyssum neglectum Magauer, Frajman & Schönswetter, Bot. J. Linn. Soc. 176: 500. 2014.
#Alyssum nezaketiae Aytaç & H.Duman, Israel J. Bot. 48: 317. 2000 [2001].
#Alyssum niveum T.R.Dudley, J. Arnold Arbor. 45: 78. 1964.
#Alyssum numidium Pomel, Nouv. Mat. Fl. Atl. 2: 377. 1875.
Alyssum ochroleucum Boiss. & A.Huet, Diagn. Pl. Orient. Nov., ser. 2, 3(5): 36. 1856.
Alyssum orophilum Jord. & Fourr., Brev. Pl. Nov. 2: 11. 1868.
#Alyssum oschtenicum (N.Busch) Kharkev., Bot. Mater. Gerb. Bot. Inst. Komarova Akad. Nauk S.S.S.R. 15: 84. 1953.
Alyssum paphlagonicum (Hausskn.) T.R.Dudley, J. Arnold Arbor. 45: 71. 1964.
#Alyssum patulum Pomel, Nouv. Mat. Fl. Atl. 1: 238. 1874.
Alyssum persicum Boiss., Ann. Sci. Nat. Bot., sér. 2, 17: 152. 1842.
Alyssum pirinicum (Stoj. & Acht.) Ančev, Willdenowia 36: 195. 2006.
Alyssum pluscanescens (Raim. ex Jos.Baumgartner) Španiel, Lihová & Marhold, comb. et stat. nov. ≡ Alyssum montanum “proles” pluscanescens Raim. ex Jos.Baumgartner, Jahresber. Nieder-Österr. Landes-Lehrersem. Wiener-Neustadt 34: 21. 1907.
Alyssum pogonocarpum Carlström, Willdenowia 14: 15. 1985.
Alyssum praecox Boiss., Fl. Orient. 1: 275. 1867.
Alyssum propinquum Jos.Baumgartner, Jahresber. Nieder-Österr. Landes-Lehrersem. Wiener-Neustadt 36: 22. 1909.
Alyssum pseudomouradicum Hausskn. & Bornm. ex Jos.Baumgartner, Jahresber. Landes-Lehrers. Wiener-Neustadt 36: 7. 1909.
Alyssum pulvinare Velen., Sitzungsber. Königl. Böhm. Ges. Wiss., Math.-Naturwiss. Cl. 1889(2): 30. 1890.
Alyssum reiseri Velen., Sitzungsber. Königl. Böhm. Ges. Wiss., Math.-Naturwiss. Cl., 1893 (37): 9. 1894.
Alyssum repens Baumg., Enum. Stirp. Transsilv. 2: 237. 1816.
Alyssum rhodanense Jord. & Fourr., Brev. Pl. Nov. 2: 10. 1868.
Alyssum rostratum Steven, Mém. Acad. Imp. Sci. St. Pétersbourg Hist. Acad. 3: 295. 1810.
Alyssum scutigerum Durieu, Expl. Sci. Algérie, Atlas: tab. 72, Fig. 4. 1849.
Alyssum siculum Jord., Diagn. Esp. Nouv.: 202. 1864.
Alyssum simplex Rudolphi, J. Bot. (Schrader) 1799(2): 290. 1799.
Alyssum simulans Runemark ex Hartvig, Fl. Hellenica 2: 207. 2002.
Alyssum smyrnaeum C.A.Mey., Bull. Sci. Acad. Imp. Sci. Saint-Pétersbourg 7: 132. 1840.
#Alyssum speciosum Pomel, Nouv. Mat. Fl. Atl. 1: 237. 1874.
Alyssum sphacioticum Boiss. & Heldr., Diagn. Pl. Orient. Nov., ser. 1, 2(8): 35. 1849.
Alyssum stapfii Vierh., Verh. K. K. Zool.-Bot. Ges. Wien 64: 260, tab.7, Fig. 1. 1914.
#Alyssum stenostachyum Botsch. & Vved., Bot. Mater. Gerb. Bot. Inst. Uzbek. Fil. Akad. Nauk S.S.S.R. 3: 18. 1941.
Alyssum stribrnyi Velen., Fl. Bulg.: 640. 1891.
Alyssum strictum Willd., Sp. Pl., ed. 4, 3(1): 464. 1800.
Alyssum strigosum Banks & Sol. in Russell, Nat. Hist. Aleppo, ed. 2, 2: 257. 1794.
Alyssum strigosum Banks & Sol. subsp. strigosum
Alyssum strigosum subsp. cedrorum (Schott & Kotschy) T.R.Dudley, J. Arnold Arbor. 45: 67. 1964.
Alyssum sulphureum T.R.Dudley & Hub.-Mor., J. Arnold Arbor. 45: 74. 1964.
Alyssum szovitsianum Fisch. & C.A.Mey., Index Sem. Horti Petropol. 4: 31. 1838.
Alyssum taygeteum Heldr., Verh. K. K. Zool.-Bot. Ges. Wien 48: 703. 1897./Sched. Herb. Graec. Norm., ser. 2: No. 1405. 1897.
Alyssum tetrastemon Boiss., Ann. Sci. Nat. Bot., sér. 2, 17: 153. 1842.
Alyssum thymops (Hub.-Mor. & Reese) T.R.Dudley, J. Arnold Arbor. 45: 71. 1964.
Alyssum trichocarpum T.R.Dudley & Hub.-Mor., J. Arnold Arbor. 45: 68. 1964.
Alyssum trichostachyum Rupr., Mém. Acad. Imp. Sci. Saint-Pétersbourg, sér. 7, 15(2) (Fl. Cauc.): 101. 1869.
Alyssum turkestanicum Regel & Schmalh., Izv. Imp. Obshch. Lyubit. Estestv. Moskovsk. Univ. 34: 6. 1882.
Alyssum umbellatum Desv., J. Bot. Agric. 3: 173. 1814.
Alyssum vernale Kit. ex Hornem., Hort. Bot. Hafn. 2: 601. 1815.
Alyssum wierzbickii Heuff., Flora (Regensburg) 18: 242. 1835.
Alyssum wulfenianum Willd., Enum. Pl. Hort. Berol. Suppl.: 44. 1813 [publ. Jul.–Dec. 1814].
Alyssum wulfenianum Willd. subsp. wulfenianum
Alyssum wulfenianum subsp. ovirense (A.Kern.) Magauer, Schönswetter & Frajman, Bot. J. Linn. Soc. 176: 500. 2014.
Alyssum xanthocarpum Boiss., Ann. Sci. Nat., Bot., ser. 2, 17: 154. 1842.
4. Aurinia Desv., J. Bot. Agric. 3: 162. 1815.–[7 spp.]. —LECTOTYPE: Aurinia saxatilis (L.) Desv. (designated by Dudley 1964b: 391).
= Anodontea (DC.) Sweet
Distribution area: Albania, Austria, Bosnia and Herzegovina, Bulgaria, Croatia, Czech Republic, Germany, Greece, Gruziya, Hungary, Italy, Macedonia, Moldavia, Montenegro, Poland, Romania, Russia, Serbia, Slovakia, Slovenia, Turkey, Ukraine.
Aurinia corymbosa Griseb., Spicil. Fl. Rumel. 1: 271. 1843.
Aurinia gionae (Quézel & Contandr.) Greuter & Burdet, Willdenowia 13: 86. 1983.
Aurinia leucadea (Guss.) K.Koch, Hort. Dendrol.: 23. 1853.
Aurinia moreana Tzanoud. & Iatroú, Bot. Chronika 1: 22. 1981.
Aurinia petraea (Ard.) Schur, Enum. Pl. Transsilv.: 61. 1866.
Aurinia saxatilis (L.) Desv., J. Bot. Agric. 3: 162. 1815.
Aurinia saxatilis (L.) Desv. subsp. saxatilis
Aurinia saxatilis subsp. megalocarpa (Hausskn.) T.R.Dudley, J. Arnold Arbor. 45: 397. 1964.
Aurinia saxatilis subsp. orientalis (Ard.) T.R.Dudley, J. Arnold Arbor. 45: 394. 1964.
Aurinia sinuata (L.) Griseb., Spicil. Fl. Rumel. 1: 271. 1843.
5. Berteroa DC., Mém. Mus. Hist. Nat. 7: 232. 1821.–[5 spp.]. —LECTOTYPE: Berteroa incana (L.) DC. (designated by Pfeiffer 1871–1873: 397).
≡ Myopteron Spreng.
Distribution area: Albania, Armenia, Austria, Azerbaijan, Bosnia & Herzegovina, Bulgaria, China, Croatia, Czech Republic, Estonia, France, Greece, Gruziya, Hungary, Italy, Kazakhstan, Kyrgyzstan, Latvia, Lithuania, Macedonia, Moldavia, Mongolia, Montenegro, Poland, Romania, Russia, Serbia, Slovakia, Slovenia, Tajikistan, Turkey, Ukraine, Uzbekistan; B. incana is naturalized throughout much of Europe, Asia, and North America.
Berteroa gintlii Rohlena, Magyar Bot. Lapok 3: 232. 1904.
Berteroa incana (L.) DC., Reg. Veg. Syst. Nat. 2: 291. 1821.
Berteroa mutabilis (Vent.) DC., Reg. Veg. Syst. Nat. 2: 292. 1821.
Berteroa obliqua (Sm.) DC., Reg. Veg. Syst. Nat. 2: 292. 1821.
Berteroa orbiculata DC., Reg. Veg. Syst. Nat. 2: 293. 1821.
6. Bornmuellera Hausskn., Mitth. Thüring. Bot. Vereins, ser. 2, 11: 71. 1897.–[9 spp.]. —TYPE: Bornmuellera tymphaea (Hausskn.) Hausskn.
= Leptoplax O.E.Schulz; = Physocardamum Hedge
Distribution area: Albania, Greece, Serbia, Turkey.
Bornmuellera angustifolia (Hausskn. ex Bornm.) Cullen & T.R.Dudley, Feddes Repert. 71: 228. 1965.
Bornmuellera baldaccii (Degen) Heywood, Feddes Repert. 69: 61. 1964.
Bornmuellera cappadocica (Willd.) Cullen & T.R.Dudley, Feddes Repert. 71: 228. 1965.
Bornmuellera davisii (Hedge) Rešetnik, Phytotaxa 159: 299. 2014 [18 Feb 2014].
Bornmuellera dieckii Degen, Oesterr. Bot. Z. 50: 313. 1900.
Bornmuellera emarginata (Boiss.) Rešetnik, Phytotaxa 159: 299. 2014 [18 Feb 2014].
Bornmuellera glabrescens (Boiss. & Balansa) Cullen & T.R.Dudley, Feddes Repert. 71: 228. 1965.
Bornmuellera kiyakii Aytaç & A.Aksoy, Bot. J. Linn. Soc. 134: 487. 2000.
Bornmuellera tymphaea (Hausskn.) Hausskn., Mitt. Thüring. Bot. Vereins 11: 72. 1897.
7. Brachypus Ledeb., Fl. Rossica 1: 133. 1841.–[1 sp.]. —TYPE: Brachypus asper Ledeb. [= Brachypus suffruticosus (Vent.) V.I.Dorof.].
Distribution area: Armenia, Azerbaijan, Iran, Iraq, Turkmenistan, Turkey.
Brachypus suffruticosus (Vent.) V.I.Dorof., Consp. Fl. Cauc. 3(2): 411. 2012.
8. Clastopus Bunge ex Boiss., Fl. Orient. 1: 261. 1867.–[2 spp.]. —LECTOTYPE: Clastopus vestitus (Desv.) Boiss. (designated by Al-Shehbaz 2012: 933).
= Straussiella Hauskn.
Distribution area: Iran, Iraq, Turkey.
Clastopus purpureus Bunge ex Boiss., Fl. Orient. 1: 261. 1867.
Clastopus vestitus (Desv.) Boiss., Fl. Orient. 1: 261. 1867.
9. Clypeola L., Sp. Pl.: 652. 1753.–[9 spp.]. —LECTOTYPE: Clypeola jonthlaspi L. (designated by Green 1925: 55).
≡ Fosselinia Scop.; = Bergeretia Desv. (1815), non Bubani (1901); = Orium Desv.; = Pseudoanastatica (Boiss.) Grossh.
Distribution area: Afghanistan, Albania, Algeria, Armenia, Azerbaijan, Bulgaria, Croatia, Cyprus, France, Greece, Gruziya, Iran, Iraq, Israel, Italy, Jordan, Kazakhstan, Kyrgyzstan, Lebanon, Libya, Macedonia, Morocco, Oman, Pakistan, Romania, Russia, Serbia, Saudi Arabia, Spain, Switzerland, Syria, Ukraine, Tajikistan, Tunisia, Turkmenistan, Turkey, Uzbekistan.
Clypeola aspera (Grauer) Turrill, J. Bot. (London) 60: 269. 1922.
Clypeola ciliata Boiss., Fl. Orient. 1: 309. 1867.
Clypeola cyclodontea Delile, Bull. Soc. Centr. Agric. Dép. Hérault 23: 258. 1830.
Clypeola dichotoma Boiss., Ann. Sci. Nat. Bot., sér. 2, 17: 175. 1842.
Clypeola elegans Boiss. & A.Huet, Diagn. Pl. Orient. Nov., ser. 2, 3(5): 38. 1856.
Clypeola eriocarpa Cav., Descr. Pl.: 401. 1802.
Clypeola jonthlaspi L., Sp. Pl.: 652. 1753.
Clypeola lappacea Boiss., Ann. Sci. Nat. Bot., sér. 2, 17: 174. 1842.
Clypeola raddeana Albov, Bull. Herb. Boissier, sér. 1, 2: 448. 1894.
10. Cuprella Salmerón-Sánchez, Mota & Fuertes, gen. nov.–[2 spp.]. —TYPE: Cuprella antiatlantica (Emb. & Maire) Salmerón-Sánchez, Mota & Fuertes
Description: Herbs annual or perennial with woody base. Trichomes sessile, 10–18-rayed stellate, without webbing between slender rays, those on leaves not umbonate, simple trichomes present or absent. Multicellular glands absent. Stems erect to ascending, sometimes prostrate, simple, leafy, not spiny. Basal and lowermost stem leaves short petiolate, not rosulate, simple, entire or distally denticulate; cauline leaves subsessile, cuneate to attenuate, not auriculate at base, entire or denticulate. Racemes many-flowered, ebracteate, corymbose, elongated in fruit; rachis straight; fruiting pedicels divaricate or recurved, sometimes straight, pubescent, persistent. Sepals ovate, free, deciduous, erect, pubescent, equal, base of lateral pair not saccate; petals yellow, erect, shorter than or subequaling sepals; blade obovate to oblanceolate, glabrous, apex retuse; claw hardly differentiated from blade, glabrous, unappendaged, entire; stamens 6, included, erect, subequal; filaments wingless, toothless, unappendaged, glabrous, free; anthers ovate or oblong, not apiculate; nectar glands 4, lateral, cylindrical, 1 on each side of lateral stamen; ovules 4 per ovary; placentation subapical. Fruit dehiscent, capsular silicles, obovate, latiseptate, not inflated, unsegmented; valves papery, veinless or with an obscure midvein, pubescent outside or glabrous with papillate margin, not keeled, smooth, wingless, unappendaged; gynophore absent; replum rounded, visible; septum complete, membranous, veinless; style 0.5–1 mm long, cylindric, persistent; stigma capitate, entire, unappendaged. Seeds aseriate, usually 1 per locule, narrowly winged, ovate, flattened; seed coat smooth, not mucilaginous when wetted; cotyledons accumbent.
Diagnosis: Cuprella is readily distinguished from Alyssum sect. Psilonema, within which its two species were formerly placed, by having deciduous sepals longer than or subequaling the petals and by the broadly obovate silicles truncate at apex. One of its species (C. antiatlantica) is woody at base, and the other (C. homalocarpa) has denticulate leaves and papillate fruit margin. The four species of A. sect. Psilonema are annuals with orbicular or ovate fruits emarginate or rarely subobtuse at apex and have persistent sepals slightly shorter than petals.
Cuprella is defined by forming a well-supported phylogenetic lineage within the tribe Alysseae quite isolated from other lineages of Alyssum and Clypeola. The GenBank accession numbers for Cuprella homalocarpa and C. antiatlantica are KR269770, KM033824 for ITS, KR269768, KR269769 for trnL, trnL-F, and KP276174, KP276175 for ndhF, respectively.
Distribution area: Cuprella antiatlantica is a narrow endemic to Morocco, whereas C. homalocarpa is widely distributed from Egypt eastward into Israel, Jordan, Syria, Saudi Arabia, Iraq, Kuwait, Iran, and Pakistan.
Habitats: The species of Cuprella occupy xeric environments on limestone (C. antiatlantica) or basaltic and siliceous substrates (C. homalocarpa).
Etymology: The name of the genus honors Phillipe Küpfer (1924-), a Swiss professor emeritus at the University of Neuchâtel, where he focused on the study of the tribe Alysseae. The root is formed by late Latin cuprum, after his family name, which means copper trader or artisan in German, and the suffix “–ella” indicating the minute habit of the two species included in the genus.
Key to the species of Cuprella:
-
1a.
Perennials with woody base; fruit stellate pubescent, not papillate; leaves entire … C. antiatlantica
-
1b.
Annuals; fruit glabrous, papillate at margin; leaves denticulate … C. homalocarpa
Cuprella antiatlantica (Emb. & Maire) Salmerón-Sánchez, Mota & Fuertes, comb. nov. ≡ Alyssum antiatlanticum Emb. & Maire, Bull. Soc. Hist. Nat. Afrique N. 23: 165. 1932. Described from: “in rupestribus siliceis editis Anti-Atlasis: prope castellum Igherm, et in monte Fidoust, ad alt. 1700–2200 m, april florens (Emberger, Jahandiez, Maire et Weiller).” —LECTOTYPE (here designated): In Anti-Atlante: in rupestris arenaceis prope castellum Igherm, 1700–1800 m, 21 Apr 1931, R. Maire s.n. (lectotype: MPU! MPU002814, http://plants.jstor.org/stable/10.5555/al.ap.specimen.MPU002814); isolectotype: MA49591).
Cuprella homalocarpa (Fisch. & C.A.Mey.) Salmerón-Sánchez, Mota & Fuertes, comb. nov. ≡ Psilonema homalocarpum Fisch. & C.A.Mey., Index Seminum [St.Petersburg (Petropolitanus)] 6: 63. 1840. Described from: “Semina in Arabia petraea legit Dr. Schimper [cultivated in Hortus Botanicus Imperialis Petropolitanus].” —LECTOTYPE vel HOLOTYPE (designated as “holotype” by Dudley in: 1965: 210): Semina in Arabia petraea legit Dr Schimper, Cult[a] in hb. [horto botanico] Petropolit. 1839, Meyer s.n. (LE!).
11. Degenia Hayek, Oesterr. Bot. Z. 60: 93. 1910.–[1 sp.]. —TYPE: Degenia velebitica (Degen) Hayek
Distribution area: Croatia (Velebit and Velika Kapela Mts).
Degenia velebitica (Degen) Hayek, Oesterr. Bot. Z. 60: 93. 1910.
12. Fibigia Medik., Pfl.-Gatt.: 90. 1792.–[3 spp.]. —TYPE: Fibigia clypeata (L.) Medik.
Distribution area: Afghanistan, Albania, Armenia, Azerbaijan, Bulgaria, Cyprus, Egypt, Greece, Gruziya, Iran, Iraq, Israel, Italy, Jordan, Lebanon, Macedonia, Serbia, Syria, Turkey, Ukraine.
Fibigia clypeata (L.) Medik., Pfl.-Gatt. 1: 90. 1792.
Fibigia clypeata (L.) Medik. subsp. clypeata
Fibigia clypeata subsp. anatolica A.Duran & Tuştaş, Afr. J. Biotech. 11: 112. 2012.
Fibigia clypeata subsp. eriocarpa (DC.) Greuter, Bocconea 25: 50. 2012.
Fibigia heterophylla Rech.f., Ark. Bot., ser. 2, 5: 166. 1960.
Fibigia macrocarpa (Boiss.) Boiss., Fl. Orient. 1: 258. 1867.
13. Galitzkya V.V.Botschantz., Bot. Zhurn. (Moscow & Leningrad) 64: 1440. 1979.–[3 spp.]. —TYPE: Galitzkya spathulata (Stephan ex Willd.) V.V.Botschantz.
Distribution area: NW China, Kazakhstan, SW Mongolia, Russia (SE of European part, SW Siberia).
Galitzkya macrocarpa (Ikonn.-Gal.) V.V.Botschantz., Bot. Zhurn. (Moscow & Leningrad) 64: 1442. 1979.
Galitzkya potaninii (Maxim) V.V.Botschantz., Bot. Zhurn. (Moscow & Leningrad) 64: 1442. 1979.
Galitzkya spathulata (Stephan ex Willd.) V.V.Botschantz., Bot. Zhurn. (Moscow & Leningrad) 64: 1442. 1979.
14. Hormathophylla Cullen & T.R.Dudley, Feddes Repert. 71: 225. 1965.–[11 spp.]. —TYPE: Hormathophylla reverchonii (Degen & Hervier) Cullen & T.R.Dudley.
= Nevadensia Rivas Mart.
Distribution area: Algeria, France, Italy, Morocco, Spain.
#Hormathophylla baetica P.Küpfer, Anales Inst. Bot. Cavanilles 35: 123. 1978.
Hormathophylla cadevalliana (Pau) T.R.Dudley, Feddes Repert. 71: 226. 1965.
Hormathophylla cochleata (Coss. & Durieu) P. Küpfer, Boissiera 23: 215. 1974.
Hormathophylla lapeyrouseana (Jord.) P. Küpfer, Boissiera 23: 213. 1974.
Hormathophylla ligustica (Breistr.) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum ligusticum Breistr., Bull. Soc. Sci. Dauph. 61: 616. 1947.
Hormathophylla longicaulis (Boiss.) Cullen & T.R.Dudley, Feddes Repert. 71: 226. 1965.
Hormathophylla purpurea (Lag. & Rodr.) P. Küpfer, Fl. Iberica 4: 193. 1993.
Hormathophylla pyrenaica (Lapeyr.) Cullen & T.R.Dudley, Feddes Repert. 71: 226. 1965.
Hormathophylla reverchonii (Degen & Hervier) Cullen & T.R.Dudley, Feddes Repert. 71: 225. 1965.
Hormathophylla saxigena (Jord. & Fourn.) D.A.German & Govaerts, comb. nov. ≡ Ptilotrichum saxigenum Jord. & Fourr., Brev. Pl. Nov. 2: 13. 1868.
Hormathophylla spinosa (L.) P. Küpfer, Boissiera 23: 208. 1974.
15. Irania Hadač & Chrtek, Acta Univ. Carol., Biol. 1971(4): 248. 1973 [Sep 1973].–[5 spp.]. —TYPE: Irania umbellata (Boiss.) Hadač & Chrtek
Distribution area: Afghanistan, Iran, Iraq.
Irania compacta (Rech.f.) Hadač & Chrtek, Acta Univ. Carol., Biol. 1971 (4): 248. 1973.
Irania membranacea (Rech.f.) Hadač & Chrtek, Acta Univ. Carol., Biol. 1971 (4): 248. 1973.
Irania multicaulis (Boiss. & Hohen.) Hadač & Chrtek, Acta Univ. Carol., Biol. 1971 (4): 248. 1973.
Irania pendula (Boiss.) Hadač & Chrtek, Acta Univ. Carol., Biol. 1971 (4): 248. 1973.
Irania umbellata (Boiss.) Hadač & Chrtek, Acta Univ. Carol., Biol. 1971 (4): 248. 1973.
16. Lepidotrichum Velen. & Bornm., Oesterr. Bot. Z. 39: 323. 1889.–[1 sp.]. —TYPE: Lepidotrichum uechtritzianum (Bornm.) Velen. & Bornm.
Distribution area: Black Sea coast of Bulgaria and Turkey.
Lepidotrichum uechtritzianum (Bornm.) Velen. & Bornm., Oesterr. Bot. Z. 39: 324. 1889.
17. Lutzia Gand., Bull. Soc. Bot. France 67 Sess. Extraord.: viii. 1923.–[1 sp.]. —LECTOTYPE (designated here): Lutzia fruticosa Gand., Bull. Soc. Bot. France 67, Sess. Extr.: viii (1920) [1923], nom. illeg. (Art. 52) [≡ Lutzia cretica (L.) Greuter & Burdet].
Distribution area: Greece (Astipalea, Crete, Karpathos, Kasos).
Lutzia cretica (L.) Greuter & Burdet, Willdenowia 13: 94. 1983.
18. Meniocus Desv., J. Bot. Agric. 3: 173. 1815.–[7 spp.]. —TYPE: Meniocus serpyllifolius (Desf.) Desv. [= Odontarrhena serpyllifolia (Desf.) Jord. & Fourr.].
Distribution area: Afghanistan, Algeria, Armenia, Azerbaijan, Bulgaria, China, Gruziya, Iran, Iraq, Israel, Jordan, Kazakhstan, Kuwait, Kyrgyzstan, Lebanon, Macedonia, Moldavia, Mongolia, Morocco, Pakistan, Romania, Russia, Saudi Arabia, Spain, Syria, Tajikistan, Turkmenistan, Turkey, Ukraine, Uzbekistan. Introduced in Australia.
Meniocus aureus Fenzl, Pugillus Pl. Nov. Syr.: 13. 1842.
Meniocus blepharocarpus (T.R.Dudley & Hub.-Mor.) Hadač & Chrtek, Acta Univ. Carol., Biol. 1971 (4): 260. 1973.
Meniocus heterotrichus (Boiss.) Hadač & Chrtek, Acta Univ. Carol., Biol. 1971 (4): 260. 1973.
Meniocus huetii (Boiss.) Hadač & Chrtek, Acta Univ. Carol., Biol. 1971 (4): 260. 1973.
Meniocus linifolius (Stephan ex Willd.) DC., Reg. Veg. Syst. Nat. 2: 325. 1821.
Meniocus meniocoides (Boiss.) Hadač & Chrtek, Acta Univ. Carol., Biol. 1971 (4): 260. 1973.
Meniocus stylaris Boiss. & Balansa, Diagn. Pl. Orient. Nov., ser. 2, 1(6): 16. 1859.
19. Odontarrhena C.A.Mey. ex Ledeb., Icon. Pl. Nov. 2: 15. 1830.–[87 spp.]. —TYPE: Odontarrhena microphylla Ledeb. (= Odontarrhena obovata C.A.Mey.).
= Triplopetalum Nyár.
Distribution area: Predominantly mountainous areas of Palaeoarctics (one native species in north-west N America) with the center of diversity in Mediterranean region, Balkans, and SW Asia.
Odontarrhena akamasica (B.L.Burtt) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum akamasicum B.L.Burtt, Kew Bulletin 4: 100. 1949.
Odontarrhena albiflora (F.K.Mey.) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum albiflorum F.K.Mey., Haussknechtia Beih. 15: 63, Fig. 9. 2011.
Odontarrhena alpestris (L.) Ledeb., Fl. Ross. 1: 142. 1841.
Odontarrhena anatolica (Hausskn. ex Nyár.) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum anatolicum Hausskn. ex Nyár., Bul. Grăd. Bot. Univ. Cluj 9: 40. 1929.
Odontarrhena argentea (All.) Ledeb., Fl. Ross. 1: 141, 751. 1841.
Odontarrhena bertolonii (Desv.) Jord. & Fourr., Brev. Pl. Nov. 2: 6. 1868.
Odontarrhena bertolonii (Desv.) Jord. & Fourr. subsp. bertolonii
Odontarrhena bertolonii subsp. scutarina (Nyár.) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum bertolonii subsp. scutarinum Nyár., Bul. Grăd. Bot. Univ. Cluj 7: 101. 1927 [1928].
Odontarrhena borzaeana (Nyár.) D.A.German, Turczaninowia 17(4): 30. 2014.
Odontarrhena bracteata (Boiss. & Buhse) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum bracteatum Boiss. & Buhse, Nouv. Mém. Soc. Imp. Naturalistes Moscou 12 (Aufzähl.): 18. 1860.
Odontarrhena callichroa (Boiss. & Balansa) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum callichroum Boiss. & Balansa, Diagn. Pl. Orient. Nov., ser. 2, 3(5): 34. 1856.
Odontarrhena carica (T.R.Dudley & Hub.-Mor.) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum caricum T.R.Dudley & Hub.-Mor., J. Arnold Arbor. 45: 89. 1964.
Odontarrhena cassia (Boiss.) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum cassium Boiss., Diagn. Pl. Orient. Nov., ser. 1, 2(8): 34. 1849.
Odontarrhena chalcidica (Janka) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum chalcidicum Janka, Oesterr. Bot. Z. 22: 175. 1872.
Odontarrhena chondrogyna (B.L.Burtt) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum chondrogynum B.L.Burtt, Kew Bulletin 4: 101. 1949.
Odontarrhena cilicica (Boiss. & Balansa) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum cilicicum Boiss. & Balansa, Diagn. Pl. Orient. Nov., ser. 2, 3(5): 34. 1856.
Odontarrhena condensata (Boiss. & Hausskn.) Jord. & Fourr., Brev. Pl. Nov. 2: 4. 1868.
Odontarrhena condensata (Boiss. & Hausskn.) Jord. & Fourr. subsp. condensata
Odontarrhena condensata subsp. flexibilis (Nyár.) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum flexibile Nyár., Bul. Grăd. Bot. Univ. Cluj 7: 157. 1927 [1928].
Odontarrhena constellata (Boiss.) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum constellatum Boiss., Ann. Sci. Nat. Bot., sér. 4, 2: 244. 1854.
Odontarrhena corsica (Duby) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum corsicum Duby, Bot. Gall. 1: 34. 1828.
Odontarrhena corymbosoidea (Formánek) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum corymbosoides Formánek, Verh. Naturf. Vereins Brünn 34: 329. 1896.
Odontarrhena crenulata (Boiss. & Heldr.) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum crenulatum Boiss. & Heldr., Diagn. Pl. Orient. Nov., ser. 1, 2(8): 33. 1849.
Odontarrhena cyprica (Nyár.) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum cypricum Nyár., Bul. Grăd. Bot. Univ. Cluj 7: 156. 1927 [1928].
Odontarrhena davisiana (T.R.Dudley) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum davisianum T.R.Dudley, J. Arnold Arbor. 45: 81. 1964.
Odontarrhena debarensis (Micevski) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum debarense Micevski, Prilozi Oddel. Biol. Med. Nauki Makedonska Akad. Nauk. Umet. 15: 50. 1994.
Odontarrhena diffusa Jord. & Fourr., Brev. Pl. Nov. 2: 5. 1868.
Odontarrhena discolor (T.R.Dudley & Hub.-Mor.) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum discolor T.R.Dudley & Hub.-Mor., J. Arnold Arbor. 45: 80. 1964.
Odontarrhena dubertretii (Gomb.) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum dubertretii Gomb., Mém. Soc. Bot. France 1952: 4. 1952.
Odontarrhena dudleyi (Adıgüzel & R.D.Reeves) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum dudleyi Adıgüzel & R.D.Reeves, Edinburgh J. Bot. 59: 216. 2002.
Odontarrhena elatius (F.K.Mey.) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum elatius F.K.Mey., Haussknechtia Beih., 15: 64. 2011.
Odontarrhena eriophylla (Boiss. & Hausskn.) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum eriophyllum Boiss. & Hausskn., Fl. Orient. 1: 273. 1867.
Odontarrhena euboea (Halácsy) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum euboeum Halácsy, Consp. Fl. Graec. 1: 93. 1900.
Odontarrhena fallacina (Hausskn.) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum fallacinum Hausskn., Mitth. Thüring. Bot. Vereins, n.f., 3–4: 114. 1893.
Odontarrhena fedtschenkoana (N.Busch) D.A.German, Komarovia 6(2): 85. 2008 [19 May 2010].
Odontarrhena filiformis (Nyár.) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum filiforme Nyár., Bul. Grăd. Bot. Univ. Cluj 9: 35. 1929.
Odontarrhena floribunda (Boiss. & Balansa) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum floribundum Boiss. & Balansa, Diagn. Pl. Orient. Nov., ser. 2, 3(5): 33. 1856.
Odontarrhena fragillima (Bald.) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum alpestre var. fragillimum Bald., Malpighia 9: 58. 1895.
Odontarrhena gehamensis (Fed.) D.A.German, Turczaninowia 17(4): 30. 2014.
Odontarrhena gevgelicensis (Micevski) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum gevgelicense Micevski, Prilozi Oddel. Biol. Med. Nauki Makedonska Akad. Nauk. Umet. 15: 55. 1994.
Odontarrhena giosnana (Nyár.) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum giosnanum Nyár., Bul. Grăd. Bot. Univ. Cluj 7: 127. 1927 [1928].
Odontarrhena haradjianii (Rech.f.) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum haradjianii Rech.f., Ark. Bot., ser. 2, 5: 172. 1960.
Odontarrhena haussknechtii (Boiss.) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum haussknechtii Boiss., Fl. Orient. 1: 269. 1867.
Odontarrhena heldreichii (Hausskn.) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum heldreichii Hausskn., Mitt. Thüring. Bot. Vereins 3-4: 113. 1893.
Odontarrhena huber - morathii (T.R.Dudley) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum huber-morathii T.R.Dudley, J. Arnold Arbor. 45: 83. 1964.
Odontarrhena inflata (Nyár.) D.A.German, Turczaninowia 17(4): 31. 2014.
Odontarrhena kavadarcensis (Micevski) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum kavadarcense Micevski, Prilozi Oddel. Biol. Med. Nauki Makedonska Akad. Nauk. Umet. 15: 52. 1994.
Odontarrhena kurdica (Boiss.) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum oxycarpum var. kurdicum Boiss., Fl. Orient. 1: 269. 1867.
Odontarrhena lanigera (DC.) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum lanigerum DC., Reg. Veg. Syst. Nat. 2: 308. 1821.
Odontarrhena lesbiaca P.Candargy, Bull. Soc. Bot. France 44: 151. 1897.
Odontarrhena libanotica (Nyár.) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum libanoticum Nyár., Bul. Grăd. Bot. Univ. Cluj 18: 83. 1938 [1939].
Odontarrhena litvinovii (Knjaz.) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum litvinovii Knjaz., Novosti Sist. Vyssh. Rast. 42: 143 (-145; Fig. 3). 2011 [13 Jul 2011].
Odontarrhena lurensis (F.K.Mey.) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum lurense F.K.Mey., Haussknechtia Beih., 15: 64. 2011.
Odontarrhena markgrafii (O.E.Schulz) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum markgrafii O.E.Schulz, Ber. Deutsch. Bot. Ges. 44: 422. 1926.
Odontarrhena masmenaea (Boiss.) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum masmenaeum Boiss., Diagn. Pl. Orient. Nov., ser. 2, 3(5): 36. 1856.
Odontarrhena metajnae (Plazibat) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. et stat. nov. ≡ Alyssum serpyllifolium var. metajnae Plazibat, Nat. Croatica 18: 413. 2009.
Odontarrhena mughlaei (Orcan) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum mughlaei Orcan, Nordic J. Bot. 23: 703. 2006.
Odontarrhena muralis (Waldst. & Kit.) Endl., Cat. Horti Vindob. 2: 245. 1841 [Nov–Dec 1842].
Odontarrhena nebrodensis (Tineo) L.Cecchi & Selvi, Inform. Bot. Ital. 45: 308. 2013 [11 Dec 2013].
Odontarrhena nebrodensis (Tineo) L.Cecchi & Selvi subsp. nebrodensis
Odontarrhena nebrodensis subsp. tenuicaulis (Hartvig) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum nebrodense subsp. tenuicaule Hartvig, Mount. Fl. Greece 1: 300. 1986.
Odontarrhena obovata C.A.Mey. in Ledebour, Fl. Alt. 1: 61. 1831 [Jul.-Dec. 1831].
Odontarrhena obtusifolia (Steven ex DC.) C.A.Mey., Verz. Pfl. Casp. Meer (C.A. von Meyer). 181. 1831 [Nov-Dec 1831].
Odontarrhena orbelica (Ančev & Uzunov) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum orbelicum Ančev & Uzunov, Phytol. Balcan. 8: 26. 2002.
Odontarrhena oxycarpa (Boiss. & Balansa) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum oxycarpum Boiss. & Balansa, Diagn. Pl. Orient. Nov., ser. 2, 3(5): 35. 1856.
Odontarrhena pateri (Nyár.) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum pateri Nyár., Bul. Grăd. Bot. Univ. Cluj 9: 33. 1929.
Odontarrhena pateri (Nyár.) Španiel, Al-Shehbaz, D.A.German & Marhold subsp. pateri
Odontarrhena pateri subsp. prostrata (Boiss. & A.Huet ex Nyár.) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum prostratum Boiss. & A.Huet ex Nyár., Bul. Grăd. Bot. Univ. Cluj 18: 98. 1939.
Odontarrhena peltarioidea (Boiss.) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum peltarioides Boiss., Ann. Sci. Nat. Bot., sér. 2, 17: 158. 1842.
Odontarrhena peltarioidea (Boiss.) Španiel, Al-Shehbaz, D.A.German & Marhold subsp. peltarioidea
Odontarrhena peltarioidea subsp. virgatiformis (Nyár.) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum peltarioides var. virgatiforme Nyár., Anal. Acad. Rep. Pop. Române, Biol. ser. A, mem. 3, 1: 84. 1949.
Odontarrhena penjwinensis (T.R.Dudley) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum penjwinense T.R.Dudley, Notes Roy. Bot. Gard. Edinburgh 24: 162. 1962.
Odontarrhena pinifolia (Nyár.) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Triplopetalum pinifolium Nyár., Magyar Bot. Lapok 24: 97. 1925 [1926].
Odontarrhena polyclada (Rech.f.) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum polycladum Rech.f., Phyton (Horn, Austria) 3: 55. 1951.
Odontarrhena pterocarpa (T.R.Dudley) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum pterocarpum T.R.Dudley, J. Arnold Arbor. 45: 370. 1964.
Odontarrhena pugiostyla (Nyár.) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum pugiostylum Nyár., Bul. Grăd. Bot. Univ. Cluj 18: 89. 1938 [1939].
Odontarrhena robertiana (Bernard ex Gren. & Godr.) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum robertianum Bernard ex Gren. & Godr., Fl. France 1: 117. 1847.
Odontarrhena samarifera (Boiss. & Hausskn.) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum samariferum Boiss. & Hausskn., Fl. Orient. 1: 272. 1867.
Odontarrhena samia (T.R.Dudley & Christod.) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum samium T.R.Dudley & Christod., Notes Roy. Bot. Gard. Edinburgh 45: 433. 1988.
Odontarrhena serpentina (Micevski) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum serpentinum Micevski, Prilozi Oddel. Biol. Med. Nauki Makedonska Akad. Nauk. Umet. 15: 46. 1994.
Odontarrhena serpentinicola (F.K.Mey.) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum serpentinicola F.K.Mey., Haussknechtia Beih. 15: 65. 2011.
Odontarrhena serpyllifolia (Desf.) Jord. & Fourr., Brev. Pl. Nov. 2: 1. 1868.
Odontarrhena sibirica (Willd.) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum sibiricum Willd., Sp. Pl., ed. 4, 3(1): 465. 1800.
Odontarrhena singarensis (Boiss. & Hausskn.) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum singarense Boiss. & Hausskn., Fl. Orient. Suppl.: 49. 1888.
Odontarrhena skopjensis (Micevski) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum skopjense Micevski, Prilozi Oddel. Biol. Med. Nauki Makedonska Akad. Nauk. Umet. 15: 43. 1994.
Odontarrhena smolikana (Nyár.) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum smolikanum Nyár., Bul. Grăd. Bot. Univ. Cluj 9: 43. 1929.
Odontarrhena stipitata (Kavousi & T.R.Dudley) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum stipitatum Kavousi & T.R.Dudley, Iran J. Bot. 9: 48. 2001.
Odontarrhena subspinosa (T.R.Dudley) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum subspinosum T.R.Dudley, Notes Roy. Bot. Gard. Edinburgh 24: 160. 1962.
Odontarrhena syriaca (Nyár.) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum syriacum Nyár., Bul. Grăd. Bot. Univ. Cluj 18: 84. 1938 [1939].
Odontarrhena szarabiaca (Nyár.) D.A.German, Komarovia 6(2): 85. 2008 [19 May 2010].
#Odontarrhena subalpina (M.Bieb.) D.A.German, Turczaninowia 17(4): 30. 2014.
Odontarrhena tavolarae (Briq.) L.Cecchi & Selvi, Inform. Bot. Ital. 45: 308. 2013 [31 Dec 2013].
Odontarrhena tortuosa (Waldst. & Kit. ex Willd.) C.A.Mey. in Ledebour, Fl. Alt. 1: 60. 1831 [Jul.-Dec. 1831].
Odontarrhena tortuosa (Waldst. & Kit. ex Willd.) C.A.Mey. subsp. tortuosa
Odontarrhena tortuosa subsp. caliacrae (Nyár.) Španiel, Al-Shehbaz & Marhold, comb. nov. ≡ Alyssum caliacrae Nyár., Bul. Grăd. Bot. Univ. Cluj 6: 92. 1926.
Odontarrhena tortuosa subsp. cretacea (Kotov) Španiel, Al-Shehbaz & Marhold, comb. nov. ≡ Alyssum tortuosum subsp. cretaceum Kotov, Zhurn. Inst. Bot. Vseukraïns’k. Akad. Nauk 21–22: 238. 1939.
Odontarrhena tortuosa subsp. heterophylla (Nyár.) Španiel, Al-Shehbaz & Marhold, comb. nov. ≡ Alyssum tortuosum subsp. heterophyllum Nyár., Bul. Grăd. Bot. Univ. Cluj 7: 132. 1928 [sep. ed. Vorst. Alyssum: 118. 1929.].
Odontarrhena tortuosa subsp. savranica (Andrz.) Španiel, Al-Shehbaz & Marhold, comb. nov. ≡ Alyssum savranicum Andrz., in Besser, Enum. Pl. Volhyn., ed. 2: 82. 1822.
Odontarrhena trapeziformis (Bornm. ex Nyár.) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum trapeziforme Bornm. ex Nyár., Anal. Acad. Rep. Pop. Române, Biol. ser. A, mem. 3, 1: 83. 1949.
Odontarrhena troodi (Boiss.) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum troodi Boiss., Fl. Orient. Suppl.: 49. 1888.
Odontarrhena turgida (T.R.Dudley) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum turgidum T.R.Dudley, Great Basin Naturalist 24: 7. 1964.
Odontarrhena virgata (Nyár.) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Alyssum virgatum Nyár., Bul. Grăd. Bot. Univ. Cluj 7: 115. 1927 [1928].
20. Phyllolepidum Trinajstić, Razpr. Slov. Akad. Znan. Umetn. 31: 362. 1990.–[2 spp.]. —TYPE: Phyllolepidum rupestre (Sweet) Trinajstić.
≡ Lepidophyllum Trinajstić (1980), non Cass. (1816), nec Brongn. (1828).
Distribution area: Albania, Greece, Italy, Macedonia, Montenegro, Serbia, Turkey.
Phyllolepidum cyclocarpum (Boiss.) L.Cecchi, Pl. Biosystems 145: 828. 2011.
Phyllolepidum cyclocarpum (Boiss.) L.Cecchi subsp. cyclocarpum
Phyllolepidum cyclocarpum subsp. pindicum (Hartvig) L.Cecchi, Pl. Biosystems 145: 828. 2011.
Phyllolepidum rupestre (Sweet) Trinajstić, Razpr. Slov. Akad. Znan. Umet. 31: 363. 1990.
21. Physoptychis Boiss., Fl. Orient. 1: 260. 1867.–[2 spp.]. —TYPE: P. gnaphalodes (DC.) Boiss. (= Physoptychis caspica (Hablitz) V.V.Botschantz.).
Distribution area: Armenia, Azerbaijan, Iran, Iraq, Turkey.
Physoptychis caspica (Hablitz) V.V.Botschantz., Bot. Zhurn. (Moscow & Leningrad) 61: 1440. 1976.
Physoptychis haussknechtii Bornm., Mitt. Thüring. Bot. Vereins 13–14: 1. 1899.
22. Pterygostemon V.V.Botschantz., Bot. Zhurn. (Moscow & Leningrad) 62: 1504. 1977 [6–31 Oct 1977].–[1 sp.]. —TYPE: Pterygostemon spathulatus (Kar. & Kir.) V.V.Botschantz.
≡ Asterotricha V.V.Botschantz. (1976), non Kuntze (1903), nec Astrotricha DC. (1829).
Distribution area: NW China, E Kazakhstan.
Pterygostemon spathulatus (Kar. & Kir.) V.V.Botschantz., Bot. Zhurn. (Moscow & Leningrad) 62: 1504. 1977.
23. Resetnikia Španiel, Al-Shehbaz, D.A.German & Marhold, gen. nov.–[1 sp.]. —TYPE: Resetnikia triquetra (DC.) Španiel, Al-Shehbaz, D.A.German & Marhold.
Description: Herbs perennial, canescent throughout, with simple caudex terminated in a central rosette and covered with leaf or petiole remains of previous seasons. Trichomes sessile, 12–20-rayed, stellate, without webbing between rigid rays, at least those on leaves often umbonate, simple trichomes absent. Multicellular glands absent. Stems erect to ascending, originating laterally from below central rosette, leafy, not spiny. Basal leaves petiolate, rosulate, simple, entire, with a distinct midvein abaxially; cauline leaves petiolate, not auriculate at base, entire. Racemes several- to many-flowered, ebracteate, corymbose, slightly elongated in fruit; rachis straight; fruiting pedicels ascending to divaricate, straight, densely pubescent, persistent. Sepals oblong, free, deciduous, erect, pubescent, unequal, base of lateral pair saccate; petals yellow, erect at base with flaring blade, much longer than sepals; blade obovate, glabrous, apex obtuse; claw strongly differentiated from blade, longer than sepals, glabrous, unappendaged, entire; stamens 6, distinctly exserted, erect, tetradynamous; filaments narrowly winged, unappendaged or lateral pair sometimes minutely toothed, glabrous, free; anthers oblong, not apiculate; nectar glands 2, lateral, ring-like, surrounding base of lateral stamen, or U-shaped and subtending it, median glands absent; ovules 12–16 per ovary; placentation parietal. Fruit dehiscent, capsular broad siliques or narrow silicles, oblong to elliptic or narrowly so, strongly latiseptate, not inflated, unsegmented; valves thin leathery, veinless, densely pubescent outside, very sparsely pubescent inside, not keeled, smooth, wingless, unappendaged; gynophore absent; replum rounded concealed by connate valve margins; septum complete, membranous, veinless; style 7–10 mm long, filiform, pubescent along proximal half, caducous; stigma capitate entire, unappendaged. Seeds biseriate, winged all around, ovate or broadly so, strongly flattened; seed coat not mucilaginous when wetted; cotyledons accumbent.
Diagnosis: Resetnikia is readily distinguished from Fibigia, in which its single species was formerly placed, by having 12–20-rayed (vs. 6–8-rayed) stellate trichomes, fruiting style 7–10 (vs. 1–2) mm long, unequal (vs. equal) sepals the lateral pair of which saccate (vs. not saccate) at base, fruit valves sparsely pubescent (vs. glabrous) inside, and ebracteate racemes slightly elongated in fruit (vs. at least basally bracteate or rarely ebracteate racemes elongated considerably in fruit).
Distribution area: Resetnikia triquetra is a narrow endemic to Croatian Adriatic coast.
Habitats: vertical fissures of calcareous rocks.
Etymology: the name of the genus honors Ivana Rešetnik (University of Zagreb, Croatia) in recognition for her work (and other collaborators) on the phylogenetic relationships of the tribe Alysseae.
Resetnikia triquetra (DC.) Španiel, Al-Shehbaz, D.A.German & Marhold, comb. nov. ≡ Farsetia triquetra DC. Reg. Veg. Syst. Nat. 2: 290. 1821. Described from: “in Dalmatiâ ad rupes arcis Clissa”. —HOLOTYPE: [Croatia] In Dalmat: ad rupes arcis Clissa, misit 1820, Portenschlag s.n. (G-DC! G00205396, http://plants.jstor.org/stable/viewer/10.5555/al.ap.specimen.g00205396).
24. Takhtajaniella V.E.Avet., Sist. Evol. Vyssh. Rast.: 54. 1980.–[1 sp.]. —TYPE: Takhtajaniella globosa V.E.Avet.
Distribution area: Azerbaijan (Karabakh).
Takhtajaniella globosa V.E.Avet., Sist. Evol. Vyssh. Rast.: 54. 1980.
References
Adamidis GC, Dimitrakopoulos PG, Manolis A, Papageorgiou AC (2014) Genetic diversity and population structure of the serpentine endemic Ni hyperaccumulator Alyssum lesbiacum. Pl Syst Evol 300:2051–2060. doi:10.1007/s00606-014-1047-3
Agapova ND, Arkharova KB, Vakhtina LI, Zemskova EA, Tarvis LV (1990) Chisla khromosom tsvetkovykh rastenii flory SSSR: Semeistva Aceraceae—Menyanthaceae. Nauka, Leningrad
Al-Shehbaz IA (1987) The genera of Alysseae (Cruciferae; Brassicaceae) in the Southeastern United States. J Arnold Arbor 68:185–240
Al-Shehbaz IA (2012) A generic and tribal synopsis of the Brassicaceae (Cruciferae). Taxon 61:931–954
Al-Shehbaz IA (2013) Clypeola is united with Alyssum (Brassicaceae). Harvard Pap Bot 18:125–128. doi:10.3100/025.018.0204
Al-Shehbaz IA, German DA, Karl R, Jordon-Thaden I, Koch MA (2011) Nomenclatural adjustments in the tribe Arabideae. Pl Diversity Evol 129:71–76. doi:10.1127/1869-6155/2011/0129-0044
Ančev M, Goranova V (2006) Trichome morphology of eleven genera of the tribe Alysseae (Brassicaceae) occurring in Bulgaria. Willdenowia 36:193–204
Avetisyan VE (1980) Takhtajaniella V. Avet. – novyi rod krestotsvetnykh (Brassicaceae) s Kavkaza. In: Zhilin SG (ed) Sistematika i evolyutsiya vysshikh rastenii, Leningrad, Nauka, pp 54–56
Avetisian VE (2009) New data for the flora of Armenia and Caucasia (Brassicaceae: Alyssum, Erysimum). Fl Rastit Rast Res Armenii 17:20–22
Avetisian VE (2013) On the distribution of genus Takhtajaniella (Brassicaceae). Takhtajania 2:114
Bani A, Echevarria G, Zhang X, Benizri E, Laubie B, Morel JL, Simonnot M-O (2015) The effect of plant density in nickel-phytomining field experiments with Alyssum murale in Albania. Austral J Bot 63:72–77. doi:10.1071/BT14285
Bedini G, Garbari F, Peruzzi L (2010 onwards). Chrobase.it—Chromosome numbers for the Italian flora. Available at: http://www.biologia.unipi.it/chrobase/. Accessed 6 Dec 2014
Beilstein MA, Al-Shehbaz IA, Mathews S, Kellogg EA (2008) Brassicaceae phylogeny inferred from phytochrome A and ndhF sequence data: tribes and trichomes revisited. Amer J Bot 95:1307–1327. doi:10.3732/ajb.0800065
Berendsohn WG, Greilhuber J, Anagnostopoulos A, Bedini G, Jakupovic J, Nimis PL, Valdés B (1997) A comprehensive datamodel for karyological databases. Pl Syst Evol 205:85–98. doi:10.1007/BF00982799
Boczantzeva VV (1976) New genus Asterotricha V. Bocz. (Cruciferae) from Kazakhstan. Bot Zhurn (Moscow & Leningrad) 61:930–931
Boczantzeva VV (1977) Chromosome numbers of two species from the family Cruciferae. Bot Zhurn (Moscow & Leningrad) 62:1504–1505
Bolkhovskikh ZV, Grif VG, Zakharyeva OI, Matveva TS (1969) Chromosome numbers of flowering plants. Nauka, Leningrad
Botanical Society of the British Isles (2014) BSBI database—Cytology. Available at: http://rbg-web2.rbge.org.uk/BSBI/cytsearch.php. Accessed 6 Dec 2014
British Standard BS 2979 (1958) Transliteration of Cyrillic and Greek characters. BSI, London
Brummitt RK (2001) World geographical scheme for recording plant distributions, 2nd edn. Hunt Institute for Botanical Documentation Carnegie Mellon University, Pittsburgh
Cecchi L (2011) A reappraisal of Phyllolepidum (Brassicaceae), a neglected genus of the European flora, and its relationships in tribe Alysseae. Pl Biosyst 145:818–831. doi:10.1080/11263504.2011.580789
Cecchi L, Gabbrielli R, Arnetoli M, Gonnelli C, Hasko A, Selvi F (2010) Evolutionary lineages of nickel hyperaccumulation and systematics in European Alysseae (Brassicaceae): evidence from nrDNA sequence data. Ann Bot (Oxford) 106:751–767. doi:10.1093/aob/mcq162
Cecchi L, Colzi I, Coppi A, Gonnelli C, Selvi F (2013) Diversity and biogeography of Ni-hyperaccumulators of Alyssum section Odontarrhena (Brassicaceae) in the central western Mediterranean: evidence from karyology, morphology and DNA sequence data. Bot J Linn Soc 173:269–289. doi:10.1111/boj.12084
Çelik N, Akpulat HA, Dömnez E (2007) A new species of Physoptychis (Brassicaceae) from central Anatolia, Turkey. Bot J Linn Soc 154:393–396. doi:10.1111/j.1095-8339.2007.00662.x
Çetin Ö, Duran A, Martin A, Tuştaş S (2012) A taxonomic study of the genus Fibigia Medik. (Brassicaceae). African J Biotechnol 11:109–119. doi:10.5897/AJB11.302
Desvaux NA (1815) Sur la famille des plantes Crucifères. J Bot Agric 3:145–187
Dobeš C, Vitek E (2000) Documented chromosome number checklist of Austrian vascular plants. Verlag des Naturhistorischen Museums Wien, Wien
Dorofeyev VI (2004) System of family Cruciferae B. Juss. (Brassicaceae Burnett). Turczaninowia 7:43–52
Dorofeyev VI (2012) Brassicaceae Burnett, nom. cons., nom. alt. (Cruciferae Juss., nom. cons.). In: Takhtajan AL (ed) Caucasian Flora Conspectus 3(2). KMK Scientific Press, Saint Petersburg, Moscow, pp 371–469
Dudley TR (1964a) Synopsis of the genus Alyssum. J Arnold Arbor 45:358–373
Dudley TR (1964b) Synopsis of the genus Aurinia in Turkey. J Arnold Arbor 45:390–400
Dudley TR (1965) Studies in Alyssum: near Eastern representatives and their allies, II. Section Meniocus and section Psilonema. J Arnold Arbor 46:181–217
Dudley TR (1968) [Alyssum] Sect. Psilonema (C.A.Mey.) Hook. In: Hedge J, Rechinger KH (eds), Flora Iranica 57. Akademische Druck- u. Verlagsanstalt, Graz, pp 153–155
Franzke A, Lysák M, Al-Shehbaz IA, Koch MA, Mummenhoff K (2011) Cabbage family affairs: the evolutionary history of Brassicaceae. Trends Pl Sci 16:108–116. doi:10.1016/j.tplants.2010.11.005
Gagnidze R (1983) Investigatio caryologica complexuum floristicorum calcareorum Transcaucasiae occidentalis [in Georgian]. Zametki Sist Geogr Rast 39:21–38
German DA (2010) (1979) Proposal to conserve the name Odontarrhena obovata (Alyssum obovatum) against O. microphylla (A. microphyllum) (Cruciferae). Taxon 59:1897–1898
German DA (2011) Taxonomical confusions in the Cruciferae of North and Central Asia I. Alyssum fischerianum and Alyssum canescens. Turczaninowia 14(4):18–28
German DA (2012a) Plant genera and species new to China recently found in northwest Xinjiang. Nordic J Bot 30:61–69. doi:10.1111/j.1756-1051.2011.01341.x
German DA (2012b) (2058) Proposal to conserve Odontarrhena obovata (Alyssum obovatum), nom. cons. prop., against Alyssum fischerianum (Cruciferae. Taxon 61:470
German DA (2014a) New synonyms and combinations in Eurasian Brassicaceae (Cruciferae). Phytotaxa 173:31–40. doi:10.11646/phytotaxa.173.1.2
German DA (2014b) Revised typifications and nomenclatural notes in N Eurasian Cruciferae. Willdenowia 44:351–361. doi:10.3372/wi.44.44305
German DA (2014c) Some new and revised typifications in North Eurasian Cruciferae. Turczaninowia 17:29–41
German DA, Al-Shehbaz IA (2010) Nomenclatural novelties in miscellaneous Asian Brassicaceae (Cruciferae). Nordic J Bot 28:646–651. doi:10.1111/j.1756-1051.2010.00983.x
German DA, Friesen N, Neuffer B, Al-Shehbaz IA, Hurka H (2009) Contribution to ITS phylogeny of the Brassicaceae, with special reference to some Asian taxa. Pl Syst Evol 283:33–56. doi:10.1007/s00606-009-0213-5
Ghaderian SM, Ghasemi R, Heidari H, Vazirifar S (2015) Effects of Ni on superoxide dismutase and glutathione reductase activities and thiol groups: a comparative study between Alyssum hyperaccumulator and non-accumulator species. Austral J Bot 63:65–71. doi:10.1071/BT14282
Ghaffari SM (1987) Chromosome studies in some flowering plants of Iran. Rev Cytol Biol Vég, Botaniste 10:3–8
Goldblatt P (2007) The index to plant chromosome numbers—past and future. Taxon 56:984–986
Goldblatt P, Johnson DE (eds) (1979) Index to plant chromosome numbers. Missouri Botanical Garden, St. Louis
Goldblatt P, Lowry PP II (2011) The index to plant chromosome numbers (IPCN): three decades of publication by the Missouri Botanical Garden come to an end. Ann Missouri Bot Gard 98:226–227. doi:10.3417/2011027
Góralski G, Lubczynska P, Joachimiak A (2009 onwards). Chromosome number database. Available at: http://chromosomes.binoz.uj.edu.pl/chromosomes/. Accessed 6 Dec 2014
Green ML (1925) Standard species of the Linnean genera of Tetradynamia. Bull Misc Inform Kew 1925:49–58
Greuter W, Raus T (1983) Med-checklist Notulae 7. Willdenowia 13:79–99
Hadač E, Chrtek J (1973) A contribution to the Brassicaceae of Iraq. Acta Univ Carol, Biol 1971:231–265
Iljinska AP (2005) Species of the genus Alyssum L. (sect. Alyssum) in the flora of Ukraine. Ukrainian Bot J 62:223–234
Iljinska A, Didukh Y, Burda R, Korotchenko I (2007) Brassicaceae Burnett (Cruciferae Juss.). In: Didukh Y (ed) Ecoflora of Ukraine 5. Phytosociocentre, Kyiv, pp 7–538
Jaén-Molina R, Caujapé-Castells J, Reyes-Betancort JA, Akhani H, Fernández-Palacios O, Pérez de Paz J, Febles-Hernández R, Marrero-Rodríguez Á (2009) The molecular phylogeny of Matthiola R. Br. (Brassicaceae) inferred from ITS sequences, with special emphasis on the Macaronesian endemics. Molec Phylogenet Evol 53:972–981. doi:10.1016/j.ympev.2009.08.031
Janchen E (1942) Das system der Cruciferen. Oesterr Bot Z 91:1–28
Jara-Seguel P, Urrutia J (2011) Chilean plants cytogenetic database. Jardin Botanico Nacional, Chile. Available at: http://www.chileanpcd.com/. Accessed 6 Dec 2014
Kamari G, Felber F, Garbari F (1991) Mediterranean chromosome number reports—1. Fl Medit 1:223–245
Kavousi K, Nazary Z, Nejad FG (2014) A new species of Alyssum (Brassicaceae) from Northeastern Iran. Novon 23:59–61. doi:10.3417/2010116
Khosravi AR, Mohsenzadeh S, Mummenhoff K (2009) Phylogenetic relationships of Old World Brassicaceae from Iran based on nuclear ribosomal DNA sequences. Biochem Syst Ecol 37:106–115. doi:10.1016/j.bse.2009.01.010
Kiefer M, Schmickl R, German DA, Mandáková T, Lysak MA, Al-Shehbaz IA, Franzke A, Mummenhoff K, Stamatakis A, Koch MA (2014) BrassiBase: introduction to a novel knowledge database on Brassicaceae evolution. Pl Cell Physiol 55:e3. doi:10.1093/pcp/pct158
Knjasev M (2011) Notes on some species of Brassicaceae in Urals and adjacent territories. Novosti Sist Vyssh Rast 42:136–146
Koch MA, Kiefer M, German DA, Al-Shehbaz IA, Franzke A, Mummenhoff K, Schmickl R (2012) BrassiBase: tools and biological resources to study characters and traits in the Brassicaceae—version 1.1. Taxon 61:1001–1009
Kron P, Suda J, Husband BC (2007) Applications of flow cytometry to evolutionary and population biology. Annual Rev Ecol Syst 38:847–876. doi:10.1146/annurev.ecolsys.38.091206.095504
Kučera J, Valko I, Marhold K (2005) On-line database of the chromosome numbers of the genus Cardamine (Brassicaceae). Biologia 60:473–476. Available at: http://www.cardamine.sav.sk/www/index.php. Accessed 6 Dec 2014
Küpfer Ph (1974) Recherches sur les liens de parenté entre la flore orophile des Alpes et celle des Pyrénées. Boissiera 23:1–322
Li Y, Kong Y, Zhang Z, Yin YQ, Liu B, Lv GH, Wang XY (2014) Phylogeny and biogeography of Alyssum (Brassicaceae) based on nuclear ribosomal ITS DNA sequences. J Genet 93:313–323. doi:10.1007/s12041-014-0362-3
Magauer M, Schönswetter P, Jang T-S, Frajman B (2014) Disentangling relationships within disjunctly distributed Alyssum ovirense/A. wulfenianum group (Brassicaceae), including description of a novel species from the north-eastern Alps. Bot J Linn Soc 176:486–505. doi:10.1111/boj.12214
Maire R (1967) Flore d’Afrique du Nord 13. Lechevalier, Paris
Marhold K (2006) IAPT/IOPB chromosome data 1. Taxon 55:443–445
Marhold K, Lihová J (2006) Polyploidy, hybridization and reticulate evolution: lessons from the Brassicaceae. Pl Syst Evol 259:143–174. doi:10.1007/s00606-006-0417-x
Marhold K, Mártonfi P, Mereďa P, Mráz P, Hodálová I, Kolník M, Kučera J, Lihová J, Mrázová V, Perný M, Valko I (2007) Chromosome number survey of the ferns and flowering plants of Slovakia VEDA, Bratislava. Available at: http://www.chromosomes.sav.sk/. Accessed 6 Dec 2014
Marhold K, Zozomová-Lihová J, Španiel S (2011) (1993) Proposal to conserve the name Alyssum montanum (Cruciferae) with a conserved type. Taxon 60:237–238
Meyer FK (2011) Beiträge zur flora von Albanien. Haussknechtia 15:1–220
Morais I, Campos JS, Favas PJC, Pratas J, Pita F, Prasad MNV (2015) Nickel accumulation by Alyssum serpyllifolium subsp. lusitanicum (Brassicaceae) from serpentine soils of Bragança and Morais (Portugal) ultramafic massifs: plant–soil relationships and prospects for phytomining. Austral J Bot 63:17–30. doi:10.1071/BT14245
Orcan N (2006) Alyssum mughlaei (Brassicaceae), a new species from Southwest Anatolia. Nordic J Bot 23:703–705. doi:10.1111/j.1756-1051.2003.tb00446.x
Orcan N, Binzet R (2009) Alyssum misirdalianum (Brassicaceae), a new species from Southern Turkey. Novon 19:494–496. doi:10.3417/2007142
Ortiz S, Rodríguez Oubiña J (2005) Caracterización taxonómica das poboacións galegas de Alyssum loiseleurii P.Fourn. (Brassicaceae). Nova Acta Ci Compostelana, Biol 14:51–56
Pakravan M, Bokaee ZN, Bolourian S (2011) A biosystematic study of the four varieties of Alyssum minus (Brassicaceae) in Iran. Iran J Bot 17:55–62
Pfeiffer L (1871–1873) Nomenclator botanicus 1. Sumptibus Theodori Fischeri, Casselis [Kassel]
Pogliani M (1971) Numeri cromosomici per la flora Italiana: 86–91. Inform Bot Ital 3:155–157
Rafinesque CS (1838) Sylva telluriana. Philadelphia. doi:10.5962/bhl.title.2757
Ranjbar M, Karami S, Rostami M (2014) A revision of Fibigia sect. Purpureae (Brassicaceae, Alysseae) in Iran, and the description of three new species. Biol Div Conserv 7:32–43
Rešetnik I, Satovic Z, Schneeweiss GM, Liber Z (2013) Phylogenetic relationships in Brassicaceae tribe Alysseae inferred from nuclear ribosomal and chloroplast DNA sequence data. Molec Phylogenet Evol 69:772–785. doi:10.1016/j.ympev.2013.06.026
Rešetnik I, Schneeweiss GM, Liber Z (2014) Two new combinations in the genus Bornmuellera (Brassicaceae). Phytotaxa 159:298–300. doi:10.11646/phytotaxa.159.4.8
Rice A, Glick L, Abadi S, Einhorn M, Kopelman NM, Salman-Minkov A, Mayzel J, Chay O, Mayrose I (2015) The Chromosome Counts Database (CCDB)—a community resource of plant chromosome numbers. New Phytol 206:19–26. doi:10.1111/nph.13191
Rusterholz HP, Aydin D, Baur B (2012) Population structure and genetic diversity of relict populations of Alyssum montanum on limestone cliffs in the Northern Swiss Jura mountains. Alpine Bot 122:109–117. doi:10.1007/s00035-012-0105-0
Schuhwerk F (1996) Published chromosome-counts in Hieracium. Available at: http://www.botanischestaatssammlung.de/projects/chrzlit.html. Accessed 6 Dec 2014
Schulz OE (1936) Cruciferae. In: Engler A, Prantl K (eds) Die natürlichen Pflanzenfamilien 17B, 2 edn, Wilhelm Engelmann, Leipzig, pp 227–658
Simon J, Franco D, Rivarola N, Daviña J, Duré R, Molero J (2000) SIIDIGE: una perspectiva de futuro para CromoPar. Available at: http://www.ub.edu/botanica/cromopar/news.htm, Database available at: http://www.ub.edu/botanica/cromopar/. Accessed 6 Dec 2014
Španiel S, Marhold K, Passalacqua NG, Zozomová-Lihová J (2011a) Intricate variation patterns in the diploid-polyploid complex of Alyssum montanum-A. repens (Brassicaceae) in the Apennine peninsula: evidence for long-term persistence and diversification. Amer J Bot 98:1887–1904. doi:10.3732/ajb.1100147
Španiel S, Marhold K, Filová B, Zozomová-Lihová J (2011b) Genetic and morphological variation in the diploid-polyploid Alyssum montanum in Central Europe: taxonomic and evolutionary considerations. Pl Syst Evol 294:1–25. doi:10.1007/s00606-011-0438-y
Španiel S, Zozomová-Lihová J, Passalacqua NG, Marhold K (2012a) Infraspecific classification of Alyssum diffusum (Brassicaceae) in Italy. Willdenowia 42:37–56. doi:10.3372/wi.42.42104
Španiel S, Marhold K, Thiv M, Zozomová-Lihová J (2012b) A new circumscription of Alyssum montanum ssp. montanum and A. montanum ssp. gmelinii (Brassicaceae) in Central Europe: molecular and morphological evidence. Bot J Linn Soc 169:378–402. doi:10.1111/j.1095-8339.2012.01225.x
Tan K (2002) Lutzia Gand. In: Strid A, Tan K (eds) Flora Hellenica 2. A.R.G. Gantner, Ruggell, p 199
Thiers B (2014) Index Herbariorum: A global directory of public herbaria and associated staff. New York: New York Botanical Garden’s Virtual Herbarium. Available at: http://sciweb.nybg.org/science2/IndexHerbariorum.asp. Accessed 6 Dec 2014
Vrána J, Cápal P, Bednářová M, Doležel J (2014) Flow cytometry in plant research: a success story. In: Nick P, Opatrný Z (eds) Applied plant cell biology, plant cell monographs 22, Springer Verlag, Berlin, pp 395–430. doi: 10.1007/978-3-642-41787-0_13
Warwick SI, Al-Shehbaz IA (2006) Brassicaceae: chromosome number index and database on CD-Rom. Pl Syst Evol 259:237–248. doi:10.1007/s00606-006-0421-1
Warwick SI, Francis A, Al-Shehbaz IA (2006) Brassicaceae: species checklist and database on CD-Rom. Pl Syst Evol 259:249–258. doi:10.1007/s00606-006-0422-0
Warwick SI, Sauder CA, Al-Shehbaz IA (2008) Phylogenetic relationships in the tribe Alysseae (Brassicaceae) based on nuclear ribosomal ITS DNA sequences. Botany 86:315–336. doi:10.1139/B08-013
Warwick SI, Mummenhoff K, Sauder CA, Koch MA, Al-Shehbaz IA (2010) Closing the gaps: phylogenetic relationships in the Brassicaceae based on DNA sequence data of nuclear ribosomal ITS region. Pl Syst Evol 285:209–232. doi:10.1007/s00606-010-0271-8
Watanabe K (2002) Index to chromosome numbers in Asteraceae. Available at: http://www.lib.kobe-u.ac.jp/infolib/meta_pub/G0000003asteraceae_e. Accessed 6 Dec 2014
Wesche K, Hensen I, Undrakh R (2006) Genetic structure of Galitzkya macrocarpa and G. potaninii, two closely related endemics of Central Asian mountain ranges. Ann Bot (Oxford) 98:1025–1034. doi:10.1093/aob/mcl182
Yılmaz Ö (2012) Alyssum kaynakiae sp. nov. (sect. Gamosepalum, Brassicaceae) from southwest Anatolia, Turkey. Nordic J Bot 30:190–194. doi:10.1111/j.1756-1051.2011.01402.x
Zozomová-Lihová J, Marhold K, Španiel S (2014) Taxonomy and evolutionary history of Alyssum montanum (Brassicaceae) and related taxa in southwestern Europe and Morocco: diversification driven by polyploidy, geographic and ecological isolation. Taxon 63:562–591. doi:10.12705/633.18
Acknowledgments
This study was financially supported by the Grant Agency VEGA, Bratislava, Slovak Republic (grant no. 2/0004/13 to K. Marhold) and by the European Social Fund and the state budget of the Czech Republic (CZ.1.07/2.3.00/30.0022) to S. Španiel, for which we are profoundly grateful. Participation of Dmitry A. German was supported by the German Research Foundation (Deutsche Forschungsgemeinschaft, DFG, grant KO2302-13/1 within the framework of the priority research program “Adaptomics”, DFG-SSP 1529).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Handling editor: Martin Lysak.
S. Španiel and M. Kempa contributed equally to this paper.
Electronic supplementary material
Below is the link to the electronic supplementary material.
606_2015_1257_MOESM2_ESM.pdf
Supplementary material 2 Online Resource 2. Distribution of chromosome numbers and ploidy levels in the genera Alyssum, Clypeola, Hormathophylla and Odontarrhena. For the construction of this histogram, haploid chromosome numbers were multiplied by two and ploidy-level estimates were assigned to the relevant chromosome numbers. (EPS 68 kb)
Rights and permissions
About this article
Cite this article
Španiel, S., Kempa, M., Salmerón-Sánchez, E. et al. AlyBase: database of names, chromosome numbers, and ploidy levels of Alysseae (Brassicaceae), with a new generic concept of the tribe. Plant Syst Evol 301, 2463–2491 (2015). https://doi.org/10.1007/s00606-015-1257-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00606-015-1257-3