Abstract
Microbial plant symbionts have been suggested to mediate plant-soil feedback and affect ecosystem functions. Systemic Epichloë fungal endophytes of grasses are found to mediate litter decomposition. These effects are often linked to alkaloids produced by Epichloë species, which are hypothesized to negatively affect decomposers. Although endophytes have been found to affect plant community and soil biota, direct (through litter quality) and indirect (through the environment) effects of fungal endophytes on litter decomposition have been scarcely scrutinized. We placed litterbags with endophyte-symbiotic (E+) and non-symbiotic (E−) Schedonorus pratensis plant litter in plots dominated by E+ or E− plants of the same species, and followed the dynamics of mass losses over time. We predicted the endophyte would hinder decomposition through changes in litter quality and that both types of litter would decompose faster in home environments. E+ litter decomposed faster in both environments. The mean difference between decomposition rate of E+ and E− litter tended to be higher in E− plots. Nitrogen and phosphorus, two elements usually associated with high decomposition rates, were significantly lower in E+ litter. We also detected a higher proportion of C in the cellulose form in E+ litter. Contrary to the general assumption, we found that symbiosis with Epichloë fungal endophytes can be associated with higher decomposition of plant litter. Since direct effects of Epichloë fungi were still stronger than indirect effects, it is suggested that besides the alkaloids, other changes in plant biomass would explain in a context-dependent manner, the endophyte effects on the litter decomposition.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Plant-soil feedback affects functional processes of ecosystems such as recycling of nutrients and primary productivity (Zhang et al. 2008; Hobbie 2015). Decomposition that implies the passage of dead plant material to organic matter and minerals can be affected by multiple biotic and abiotic factors (Melillo et al. 1982; Vivanco and Austin 2006; Austin et al. 2014; Cleveland et al. 2014; García-Palacios et al. 2016). Understanding the controls of litter decomposition is crucial for both productivity management in agroecosystems and for estimating carbon budgets in ecosystems (Zhang et al. 2008; Omacini et al. 2012; Austin et al. 2014; Crowther et al. 2015; Hobbie 2015).
Leaf fungal endophytes of the genus Epichloë have received certain attention with inconsistent results regarding their effects on host litter decomposition. Asexual Epichloë fungal endophytes (Clavicipitaceae) associate with species of cool-season grasses (subfamily Pooideae) growing systemically in the aboveground tissues and passing through generations by vertical transmission (Schardl et al. 2007; Gundel et al. 2017). Considering the 11 independent studies published until 2012, a meta-analysis reported an overall negative, although not significant, effect of endophytes on litter decomposition (Omacini et al. 2012). At the time the meta-analysis was performed, the preponderant hypothesis was that fungal alkaloids produced by endophyte symbionts which have shown deterrent effects on herbivores (see e.g., Clay 1988; Schardl et al. 2007), would have a direct inhibitory effect on litter decomposition. However, as observed for the effects of endophytes on plant–herbivore interactions (Saikkonen et al. 2010, 2013a; Ueno et al. 2016; Shukla et al. 2015), effects on decomposition have proven to be highly variable with negative, neutral, or positive results depending on host species, partners’ genotypes, and ecological conditions (Omacini et al. 2004; Lemons et al. 2005; Siegrist et al. 2010; Gundel et al. 2016; Mikola et al. 2016). Apart from the alkaloids, endophytes have been associated with changes in plant biomass quality that could be also linked to litter decomposition. These effects have been observed at the level of mineral content (e.g., Phosphorus (P), Carbon (C)/Nitrogen (N) ratio), metabolites (e.g., phenolic and antioxidant compounds, and sugar alcohols of fungal origin such as the mannitol), and structural parameters such as content of fibers and lignin (Zabalgogeazcoa et al. 2006; Rasmussen et al. 2008; Rogers et al. 2011; Hamilton et al. 2012; Vázquez-de-Aldana et al. 2013a; Soto-Barajas et al. 2016). However, it has been challenging to establish a direct association between the endophyte symbiosis, the changes in host plant biomass quality, and the decomposition rate of the litter (Siegrist et al. 2010; Gundel et al. 2016; Mikola et al. 2016).
Plant community can have strong effects on abiotic conditions (e.g., humidity and soil temperature), as well as on soil microbial and faunal communities through plant-soil feedbacks, and ultimately affect litter decomposition (Van der Putten et al. 2013; Gong et al. 2015). Some of these changes may be exerted by the endophytes through their impact on the diversity of grassland community (Rudgers and Clay 2008; Saikkonen et al. 2013b; Vazquez-de-Aldana et al. 2013b) and thereby, on the community of decomposers (Lemons et al. 2005; Rudgers and Clay 2008). Thus endophytes may have indirect effects on litter decomposition through effects on plant community and on soil biota. The endophyte Epichloë coenophiala of tall fescue (Schedonorus arundinaceus) had a negative effect on host litter decomposition through the environment (patches dominated by endophyte-symbiotic plants vs endophyte-free counterparts) although it was of smaller magnitude compared to the direct effect through litter source (Siegrist et al. 2010). With the same grass species, Lemons et al. (2005) found that the exclusion of meso-invertebrates shifted the endophyte effect from decreasing to increasing the rate of litter decomposition. However, endophyte influence on invertebrates (although not necessarily involved in decomposition) associated to host plants has been variable depending on grass species and genotype, and the ecological conditions (see e.g., Rudgers and Clay 2008; Vesterlund et al. 2011; Popay and Jensen 2005; Shukla et al. 2015). Studies have revealed that symbiosis with leaf fungal endophytes affects abundance and structure of the soil microbial community associated with the rhizosphere of tall fescue plants (Buyer et al. 2011; Rojas et al. 2016). Assuming a local adaptation of the soil biota through plant-soil feedback processes, decomposition would be faster if plant litter is similar to the locally produced biomass (home-field advantage hypothesis (HFA); Ayres et al. 2006; Austin et al. 2014; Veen et al. 2015; but see Freschet et al. 2012).
In this paper, we tested direct (through changes in plant litter quality) and indirect (by means of changes in the abiotic and biotic environment) effects of the endophyte Epichloë uncinatum on litter decomposition of its host, the perennial grass Schedonorus pratensis (common name: meadow fescue). Although the majority of studies have found inhibitory effects of endophytes on litter decomposition (see the meta-analysis by Omacini et al. 2012), we might expect positive or neutral effects of endophytes on litter decomposition based on previous results. A positive effect of the endophyte E. uncinatum on litter decomposition of S. pratensis was observed in a common garden experiment (Gundel et al. 2016). Using the same symbiotic interaction (S. pratensis–E. uncinatum), a short-time experiment (it covered 70 days during summer) failed to find either positive or negative effects of the endophyte on litter decomposition (Mikola et al. 2016). Therefore, the relative importance of direct and indirect effects of fungal endophytes on plant litter decomposition is yet uncertain in the long-term. To address this, we used a reciprocal experimental design by crossing litterbags with endophyte-symbiotic (E+) and non-symbiotic (E−) S. pratensis plant material in patches dominated by E+ or E− plants of the same species. Since the fungus E. uncinatum has been shown to affect plant growth, stand stability, and soil invertebrates (Malinowski et al. 1997; Saikkonen et al. 2013b; Bylin et al. 2014; Shukla et al. 2015), we predicted an interaction between symbiotic status of the plant litter (Litter type, E+ and E−) and symbiont influence through the environment (Environment type, E+ and E−). In accordance with the home-field advantage hypothesis, we also predicted that both types of litter (E+ and E−) will decompose faster in environments dominated by the same plant type (i.e., E+ and E−, respectively).
Materials and methods
Origin of plant litter
Endophyte-symbiotic (E+) and non-symbiotic (E−) litter was collected from S. pratensis plants growing in a common garden (Ruissalo Botanical Garden, University of Turku, Finland). Epichloë uncinatum is the loline-producing endophyte hosted by S. pratensis (Craven et al. 2001; Schardl et al. 2007). Individual plants of S. pratensis of the Finnish cultivar ‘Kasper’, symbiotic (E+) and non-symbiotic (E−) with endophyte (ten plants each) were placed at random in a grid with 1 m2 per plant in 2008 (Saikkonen et al. 2013b). Symbiotic status of the established plants was checked and confirmed several times since the establishment of the common garden (see Saikkonen et al. 2013b). At the end of the growing season of the third year (2011), aboveground biomass from the 10 E+ and 10 E− plants was harvested and mixed to E+ and E− batches. The harvest of plant material at the end of the growing season simulates plant senescence before winter in northern latitudes. Air-dried and chopped leaf and pseudostem biomass (E+ or E−) were enclosed in litterbags (100 × 140 mm size, 1 mm−2 mesh, 4 ± 0.05 g biomass/litterbag).
Litter decomposition field experiment
In order to examine direct and indirect endophyte effects, we assigned the litterbags into an experimental field established at MTT Agrifood Research Finland (Jokioinen) in May 2006 (Saikkonen et al. 2013b). The experimental field consisted of ten blocks with two paired plots (25 × 39 m) sown with either E+ (79% frequency of endophyte-symbiotic plants) or E− (0% endophyte frequency) seeds of S. pratensis cultivar ‘Kasper’ (sowing rate: 20 kg ha−1). The symbiotic status remained high (80–90%) and low (0–3%) in E+ and E− plots, respectively (Saikkonen et al. 2013b). Because the cover of S. pratensis diminished by approximately 23% in E− plots after four years (Saikkonen et al. 2013b), we identified plant stands within each plot that were dominated by S. pratensis in order to avoid the effect of other plant species. After 6 years, total C and N soil contents were not different between E+ and E− plant stands (Mikola et al. 2016).
On October 20, 2011, we placed 12 E+ and 12 E− litterbags on each of the 20 plots (10 E+ and 10 E− environments). Three E+ and three E− litterbags were randomly removed from each plot at four dates: June and September 2012, and May and September 2013. The decomposition was determined by weighing the remaining mass of litter in each bag, and expressed as percentage of litter mass loss (initial mass − final mass) on a dry weight basis.
Litter quality parameters
At the beginning of the experiment, three litterbags per symbiotic status were randomly selected and analyzed for mineral and chemical characterization. Mineral concentration (P, K, Ca, Mg, S, Fe, Mn, Zn, and Cu) was analyzed by the ICP-OES method (inductively coupled plasma optical emission spectrometry). For this, samples were digested in concentrated nitric acid (10 ml) and evaporated to about 1–2 ml. The sample was then transferred into a 50 ml volumetric flask, diluted with MILLI-Q purified water, and filtered before the ICP-OES measurement. Concentration of trace elements was evaluated by high resolution ICP-OES (Perkin Elmer Optima 8300) (Kumpulainen and Paakki 1987). Carbon and nitrogen were determined with an automated dry combustion method (Dumas method) by a Leco TruMac CN-analyzer, Leco Corporation, USA. Acid detergent fiber [ADF: cellulose + lignin + ash (minerals and silica)] and acid detergent lignin (ADL: lignin) were determined using the filter bag technique, with an Ankom Automated Fiber Analyzer A2000, based on the analytical method of Goering and Van Soest (1970).
Data analysis
For analyzing the direct (symbiotic status of the litter: E+ or E−) and indirect (through the environment: E+ or E−) effects of endophyte symbiosis, time (four levels: days of decomposition), and all their interactions on litter mass loss (g), we estimated a general linear mixed-effects model (lme function, nlme package, Pinheiro et al. 2014). Random effects accounted for the fact that litterbags were nested within 20 plots, and plots were nested within 10 blocks to avoid pseudo-replication (i.e., random intercept models). As a complementary analysis, decomposition rates were estimated by calculating the slope (k) of the exponential decay curves for each combination of endophyte-symbiotic status and environment. In the formula: ln (M t /M 0) = y − k t , M 0 and M t represent the initial litter mass and the mass remaining at time t (in years), respectively, y is the intercept and k is the decomposition constant (Wieder and Lang 1982). Least-square regression analyses were used to estimate the k values considering all the replicates for all treatments (combination of symbiotic status and environment) and decomposition times, including litterbag mass in time 0 (i.e., 4 g). Endophyte effects on each litter quality parameter were analyzed by means of t-student test. The analyses were performed in R, version 3.0.2 (R Development Core Team 2013).
Results
The endophyte status of the litter (E+ and E−), the endophyte status of the environment, and decomposition time, interactively affected the litter mass loss of S. pratensis (F 4 = 2.806, P = 0.025; Table 2) (Fig. 1). E+ litter lost more mass than E− litter, and the average difference between E+ and E− litter was significantly greater in environments growing E− grass (Figs. 1, 2). Averaged over the four retrieval times, the mean difference between E+ and E− litter biomass was 0.11 g in environments growing E+ and 0.21 g in environments growing E− grasses. The difference between the environments (E+ and E− plots) was most evident at the first litterbag removal time (summer 2012) when decomposition was 7% higher in endophyte-symbiotic than endophyte-free environments (Fig. 1).
Temporal dynamics of litter mass loss (%) of Schedonorus pratensis cv. Kasper plants symbiotic (E+ dark bars) and non-symbiotic (E− white bars) with fungal endophyte during the experimental time (days). Retrieval times are indicated together with the corresponding season of each year. The litterbags were placed in an environment where endophyte-symbiotic (E+ patterned bars) or non-symbiotic (E− non-patterned bars) plants of the same grass species were growing. Values are means ±95% CI (n = 10)
Mean values (±95% CI) of decomposition constants (k, year−1) for endophyte-symbiotic (E+ dark bars) and non-symbiotic (E− white bars) litter from Schedonorus pratensis plants (cultivar ‘Kasper’) in environments dominated by endophyte-symbiotic (E+ patterned bars) and non-symbiotic (E− non-patterned bars) plants of the same grass species
The pattern of biomass loss from the litterbags along experimental time was apparent through decomposition constants for each type of litter in each environment (Fig. 2). The reaction norm shows graphically the interaction between factors in which E+ litter decomposed faster than E− litter but with a tendency to show more decomposition in stands dominated by E− plants than in stands dominated by E+ plants (notice that 95% confidence intervals of E+ and E− litter do not overlap in the E− environment). Thus, E+ litter tended to decompose faster in E− environment than in E+ environment, and E− litter tended to decompose slower in E− environment than in E+ environment (Fig. 2).
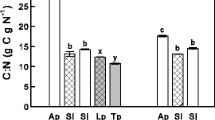
Several parameters related to the quality of litter and decomposition rate at the beginning of the experiment differed between E+ and E− litter (Table 1). The endophyte was associated with a significant decrease in litter nitrogen content (≈26% lower) but not in carbon (C mean = 42.75%). E+ litter was significantly higher in C:N ratio (E+ = 62.20) than E− litter (E− = 45.69) (t 4 = 4.244, P = 0.013). Mg and Mn contents were higher in E+ litter, while E− litter was higher in P, Cu, Fe, K, S, and Zn contents (Table 1). There was a marginal positive effect of endophyte on ADF (cellulose + lignin + ash). Together with dry matter, ash, lignin, and Ca contents, the N:P ratio (t 4 = 1.726, P = 0.159) was not significantly affected by the endophyte symbiosis (N:P ≈ 0.41) (Table 1).
Discussion
In contrast to the hypothesis that Epichloë fungal endophytes decelerate litter decomposition of host plants, we found a direct positive effect of the endophyte symbiosis on litter decomposition of the host grass, S. pratensis. These results are in accordance with our previous experiment in which the E. uncinatum fungal endophyte increased the decomposition rate of the S. pratensis litter when incubated in a common garden without vegetation (see Gundel et al. 2016). As we predicted, however, the endophyte modulated decomposition appeared to depend on the frequency of Epichloë endophyte-symbiotic grasses in the plant community and its consequences on the biotic and abiotic environment (i.e., indirect effects). Litter from E+ plants tended to decompose faster in both E+ and E− environments compared to litter from E− plants. In contrast to home-field advantage hypothesis (HFA), the decomposition rates of E+ were higher than that of E− litter in the study plots dominated by endophyte-free S. pratensis plants. Overall, direct effects of the endophyte status of litter seemed to be more important than indirect effects mediated through the community of decomposers in the soil. Similar direct versus indirect effects were observed for the endophyte E. coenophiala on the litter decomposition of its regular host S. arundinaceus (Siegrist et al. 2010).
Epichloë endophytes can directly promote decomposition by modulating the leaf chemistry or the microbial community, which may act as saprotrophic in abscised plant parts (Zabalgogeazcoa et al. 2013; Saikkonen et al. 2015). The majority of the chemical ecology literature on Epichloë endophytes has focused on endophyte-origin alkaloids, which are lolines in the case of E. uncinatum (Lehtonen et al. 2005; Schardl et al. 2007; Bylin et al. 2014). Conventionally, Epichloë endophytes that produce alkaloids are treated as plant defensive mutualists providing protection to the host plant against herbivores and pathogens (Saikkonen et al. 2010, 2013a; Huitu et al. 2014; Ueno et al. 2016). Thus, these alkaloids are often hypothesized to also negatively affect decomposer organisms in the soil (Omacini et al. 2012; Saikkonen et al. 2015; Mikola et al. 2016). In contrast to the hypothesis, in our previous study with tall fescue (S. phoenix) we detected a positive association between alkaloid level (ergovaline) at the beginning of the trial, and litter decomposition (Gundel et al. 2016). These results do not, however, rule out alkaloid mediated effects on decomposers. Positive effects on decomposition rate might partly be explained by nitrogen burst from quickly degrading alkaloids (Siegrist et al. 2010) or changes in the decomposer community (see Saikkonen et al. 2015; Rojas et al. 2016). Alternatively, Epichloë species may differently modify the plant metabolomic profile favoring the stock of labile carbon (e.g., non-structural carbohydrates, phenolic compounds; Rasmussen et al. 2008; Hamilton et al. 2012) and thus differently modulating decomposition processes (García-Palacios et al. 2016).
In order to understand Epichloë endophyte-mediated litter decomposition via altered plant quality, we looked at differences in chemistry of leaf litter between E+ and E− plants. Several chemical parameters known to positively affect litter decomposition, such as high N and P contents (Melillo et al. 1982; Vivanco and Austin 2006; Güsewell and Gessner 2009; Gong et al. 2015; Hobbie 2015), were lower in E+ litter compared to those of E− litter. In addition, endophyte presence had no effect on lignin which is known to decelerate decomposition (Melillo et al. 1982; Vivanco and Austin 2006). However, ecological roles of individual chemical parameters may be context dependent. For example, N has sometimes detected to be associated with lower decomposition rates (Hobbie 2015). Contrarian results from studies focusing on individual elements can be partly explained by interactive effects of elements. E+ litter had higher C:N ratio, and lignin:N ratio (Lignin:NE+ = 5.39, and Lignin:NE− = 4.41); acid detergent fiber (ADF) contents tended to be also higher in E+ litter. The latter indicates a greater content of C in the form of cellulose because the proportion of other components of ADF, i.e., ash and lignin, remained the same in E+ and E− litter. Consequently, the cellulose:lignin ratio {[ADF − (ADL + Ash)]/ADL} was 9.11 and 6.70 for E+ and E− litters, respectively.
Differences in litter quality often reflect the relative amount of fungi and bacteria colonizing the litter. A proper balance of C:N or N:P may be required for the optimal activity of decomposer microorganisms in the litter (Vivanco and Austin 2006; Güsewell and Gessner 2009). Fungi typically dominate microbial communities in nutrient-poor organic matter, as they may have lower nutrient requirements and a lower metabolic activity than bacteria (Güsewell and Gessner 2009). Accordingly, the relative P requirements of fungi should be lower than those of bacteria (Smith 2002), so that fungi are expected to dominate in litter with high N:P ratios. Similarly, fungi dominate and drive decomposition on substrates with high C:N ratios, due to the lower N requirement, while bacteria will dominate on substrates with low C:N ratios. The question to be solved in future studies is how these Epichloë endophyte-mediated changes in litter chemistry affect microbial communities that take part in decomposition (Vázquez-de-Aldana et al. 2013c; Zabalgogeazcoa et al. 2013; Saikkonen et al. 2015). Our results suggest that cellulose-rich E+ litter with high C:N ratio may favor fungi, especially cellulolytic fungi (Vázquez-de-Aldana et al. 2013a), in decomposing microbial community. Similarly, the greater Mn found in E+ litter, suggests that endophyte symbiosis may promote fungi that use Mn peroxidases to break down lignin and thus decomposition rate (Hobbie 2015). By contrast, N:P ratio remained the same in E+ and E− litters and thus appears to play a less significant role in determining microbial decomposition in this study.
To conclude, Epichloë endophytes can affect plant litter decomposition through multiple ecological pathways. In previous studies we have shown that E. uncinatum increases plant vigor and thereby promotes high frequencies of E+ S. pratensis individuals in the plant community (Saikkonen et al. 2013b). Thus, endophyte symbiosis can indirectly affect environmental conditions for both degradation of litter by chemical and physical processes or by decomposers via the structure of the plant community and microclimate (e.g., temperature, humidity, etc.). In this study we demonstrated Epichloë endophyte-mediated changes in litter quality and that, although influenced by the frequency of infected grasses in the plant community, decomposition rate of E+ litter was higher compared to E− litter, lending no support for home-field advantage hypothesis. Accumulating conflicting literature evidence suggests, however, the effects of endophytes on litter quality and decomposition are context dependent, and are not necessarily translated into changes in nutrient cycling (Mikola et al. 2016).
References
Austin AT, Vivanco L, González-Arzac A, Pérez LI (2014) There’s no place like home? An exploration of the mechanisms behind plant litter-decomposer affinity in terrestrial ecosystems. New Phytol 204:307–314
Ayres E, Dromph KM, Bardgett RD (2006) Do plant species encourage soil biota that specialise in the rapid decomposition of their litter? Soil Biol Biochem 38:183–186
Buyer JS, Zuberer DA, Nichols KA, Franzluebbers AJ (2011) Soil microbial community function, structure, and glomalin in response to tall fescue endophyte infection. Plant Soil 339:401–412
Bylin AG, Hume DE, Card SD, Mace WJ, Lloyd-West CM, Huss-Danell K (2014) Influence of nitrogen fertilization on growth and loline alkaloid production of meadow fescue (Festuca pratensis) associated with the fungal symbiont Neotyphodium uncinatum. Botany 92:370–376
Clay K (1988) Fungal endophytes of grasses: a defensive mutualism between plants and fungi. Ecology 69:10–16
Cleveland CC, Reed SC, Keller AB, Nemergut DR, O’Neill SP, Ostertag R, Vitousek PM (2014) Litter quality versus soil microbial community controls over decomposition: a quantitative analysis. Oecologia 174:283–294
Craven KD, Blankenship JD, Leuchtmann A, Hignight K, Schardl CL (2001) Hybrid fungal endophytes symbiotic with the grass Lolium pratense. Sydowia 53:44–73
Crowther TW, Thomas SM, Maynard DS, Baldrian P, Covey K, Frey SD, van Diepen LTA, Bradford MA (2015) Biotic interactions mediate soil microbial feedbacks to climate change. Proc Natl Acad Sci USA 112:7033–7038
Freschet GT, Aerts R, Cornelissen JHC (2012) Multiple mechanisms for trait effects on litter decomposition: moving beyond home-field advantage with a new hypothesis. J Ecol 100:619–630
García-Palacios P, Shaw AE, Wall DH, Hättenschwiler S (2016) Temporal dynamics of biotic and abiotic drivers of litter decomposition. Ecol Lett 19:554–563
Goering HK, Van Soest PJ (1970) Forage fiber analyses (apparatus, reagents, procedures, and some applications). U.S. Agricultural Research Service, Ames
Gong S, Guo R, Zhang T, Guo J (2015) Warming and nitrogen addition increase litter decomposition in a temperate meadow ecosystem. PLoS ONE 10(3):e0116013
Gundel PE, Helander M, Garibaldi LA, Vázquez-de-Aldana BR, Zabalgogeazcoa I, Saikkonen K (2016) Role of foliar fungal endophytes in litter decomposition among species and population origins. Fungal Ecol 21:50–56
Gundel PE, Rudgers JA, Whitney KD (2017) Vertically transmitted symbionts as mechanisms of transgenerational effects. Am J Bot 104:787–792
Güsewell S, Gessner MO (2009) N:P ratios influence litter decomposition and colonization by fungi and bacteria in microcosms. Funct Ecol 23:211–219
Hamilton CE, Gundel PE, Helander M, Saikkonen K (2012) Endophytic mediation of reactive oxygen species and antioxidant activity in plants: a review. Fungal Diversity 54:1–10
Hobbie SE (2015) Plant species effects on nutrient cycling: revisiting litter feedbacks. Trends Ecol Evol 30:357–363
Huitu O, Forbes KM, Helander M, Julkunen-Tiitto R, Lambin X, Saikkonen K, Stuart P, Sulkama S, Hartley S (2014) Silicon, endophytes and secondary metabolites as grass defenses against mammalian herbivores. Front Plant Sci 5:478
Kumpulainen J, Paakki M (1987) Analytical quality control program used by the trace elements in foods and diets sub-network of the FAO European cooperative network on trace elements. Fresenius J Anal Chem 326:684–689
Lehtonen P, Helander M, Wink M, Sporer F, Saikkonen K (2005) Transfer of endophyte-origin defensive alkaloids from a grass to a hemiparasitic plant. Ecol Lett 8:1256–1263
Lemons A, Clay K, Rudgers JA (2005) Connecting plant–microbial interactions above and belowground: a fungal endophyte affects decomposition. Oecologia 145:595–604
Malinowski D, Leuchtmann A, Schmidt D, Nösberger J (1997) Growth and water status in meadow fescue is affected by Neotyphodium and Phialophora species endophytes. Agron J 89:673–678
Melillo JM, Aber JD, Steudler PA, Schimel JP (1982) Nitrogen and lignin control of hardwood leaf litter decomposition dynamics. Ecology 63:621–626
Mikola J, Helander M, Saikkonen K (2016) No effects of Epichloë endophyte infection on nitrogen cycling in meadow fescue (Schedonorus pratensis) grassland. Plant Soil 405:257–264
Omacini M, Chaneton E, Ghersa CM, Otero P (2004) Do foliar endophytes affect grass litter decomposition? A microcosm approach using Lolium multiflorum. Oikos 104:581–590
Omacini M, Semmartin MG, Perez LI, Gundel PE (2012) Grass-endophyte symbiosis: a neglected aboveground interaction with multiple belowground consequences. Appl Soil Ecol 325(61):273–279
Pinheiro J, Bates D, DebRoy S, Sarkar D, Team RDC (2014) Nlme: linear and nonlinear mixed effects models. R Package Version 3.1-117. http://cran.r-project.org/package¼nlme
Popay AJ, Jensen JG (2005) Soil biota associated with endophyte-infected tall fescue in the field. N Zeal Plant Prot 58:117–121
R Development Core Team (2013) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Rasmussen S, Parsons AJ, Fraser K, Xue H, Newman JA (2008) Metabolic profiles of Lolium perenne are differentially affected by nitrogen supply, carbohydrate content, and fungal endophyte infection. Plant Physiol 146:1440–1453
Rogers JK, Morton BC, Mosali J (2011) Plant and endophyte effect on fiber, N, and P concentrations in tall fescue. Int J Agron 2011:1–7
Rojas X, Guo J, Leff JW, McNear DH Jr, Fierer N, McCulley RL (2016) Infection with a shoot specific fungal endophyte (Epichloë) alters tall fescue soil microbial communities. Microb Ecol 72:197–206
Rudgers JA, Clay K (2008) An invasive plant-fungal mutualism reduces arthropod diversity. Ecol Lett 11:1–10
Saikkonen K, Saari S, Helander M (2010) Defensive mutualism between plants and endophytic fungi? Fungal Diversity 41:101–113
Saikkonen K, Gundel PE, Helander M (2013a) Chemical ecology mediated by fungal endophytes in grasses. J Chem Ecol 39:962–968
Saikkonen K, Ruokolainen K, Huitu O, Gundel PE, Piltti T, Hamilton CE, Helander M (2013b) Fungal endophytes help prevent weed invasions. Agr Ecosyst Environ 165:1–5
Saikkonen K, Mikola J, Helander M (2015) Endophytic phyllosphere fungi and nutrient cycling in terrestrial ecosystems. Curr Sci 109:121–126
Schardl CL, Grossman RB, Nagabhyru P, Faulkner JR, Mallik UP (2007) Loline alkaloids: currencies of mutualism. Phytochemistry 68:980–996
Shukla K, Hager HA, Yurkonis KA, Newman JA (2015) Effects of the Epichloë fungal endophyte symbiosis with Schedonorus pratensis on host grass invasiveness. Ecol Evol 5:2596–2607
Siegrist J, McCulley R, Bush L, Phillips T (2010) Alkaloids may not be responsible for endophyte associated reductions in tall fescue decomposition rates. Funct Ecol 24:460–468
Smith VH (2002) Effects of resource supply ratios on the structure and function of microbial communities. Antonie Van Leeuwenhoek 81:99–106
Soto-Barajas MC, Zabalgogeazcoa I, Gómez-Fuertes J, González-Blanco V, Vázquez-de-Aldana BR (2016) Epichloë endophytes affect the nutrient and fiber content of Lolium perenne regardless of plant genotype. Plant Soil 405:265–277
Ueno A, Gundel PE, Omacini M, Ghersa CM, Bush LP, Martínez-Ghersa MA (2016) Mutualism effectiveness of fungal endophyte in grasses is reduced by ozone. Funct Ecol 30:226–234
van der Putten WH, Bardgett RD, Bever JD, Bezemer TM, Casper BB, Fukami T, Kardol P, Klironomos JN, Kulmatiski A, Schweitzer JA, Suding KN, Van de Voorde TFJ, Wardle DA (2013) Plant–soil feedbacks: the past, the present and future challenges. J Ecol 101:265–276
Vázquez-de-Aldana BR, García-Ciudad A, García-Criado B, Vicente-Tavera S, Zabalgogeazcoa I (2013a) Fungal endophyte (Epichloë festucae) alters the nutrient content of Festuca rubra regardless of water availability. PLoS ONE 8(12):e84539
Vázquez-de-Aldana BR, Zabalgogeazcoa I, García Ciudad A, García Criado B (2013b) An Epichloë endophyte affects the competitive ability of Festuca rubra against other grassland species. Plant Soil 362:201–213
Vázquez-de-Aldana BR, Bills G, Zabalgogeazcoa I (2013c) Are endophytes an important link between airborne spores and allergen exposure? Fungal Diversity 60:33–42
Veen GF, Freschet GT, Ordonez A, Wardle DA (2015) Litter quality and environmental controls of home-field advantage effects on litter decomposition. Oikos 124:187–195
Vesterlund S-R, Helander M, Faeth SH, Hyvönen T, Saikkonen K (2011) Environmental conditions and host plant origin override endophyte effects on invertebrate communities. Fungal Diversity 47:109–118
Vivanco L, Austin AT (2006) Intrinsic effects of species on leaf litter and root decomposition: a comparison of temperate grasses from North and South America. Oecologia 150:97–107
Wieder RK, Lang GE (1982) A critique of the analytical methods used in examining decomposition data obtained from litter bags. Ecology 63:1636–1642
Zabalgogeazcoa I, García-Ciudad A, Vázquez-de-Aldana BR, García-Criado B (2006) Effects of the infection by the fungal endophyte Epichloë festucae in the growth and nutrient content of Festuca rubra. Eur J Agron 24:374–384
Zabalgogeazcoa I, Gundel PE, Helander M, Saikkonen K (2013) Non-systemic fungal endophytes in Festuca rubra plants infected by Epichloë festucae in subarctic habitats. Fungal Diversity 60:25–32
Zhang D, Hui D, Luo Y, Zhou G (2008) Rates of litter decomposition in terrestrial ecosystems: global patterns and controlling factors. J Plant Ecol 1(2):85–93
Acknowledgements
We thank Serdar Dirihan for his help in the field. This work was supported by the Academy of Finland Grants 137909, 281354, and 292732, Turku University Foundation, and Spanish Ministry of Economy and Competitiveness (Grant AGL2011-22783). We also thank the three anonymous reviewers and the subject editor for their constructive comments which significantly improved our article.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by Christina Birnbaum.
Appendix
Appendix
See Table 2.
Rights and permissions
About this article
Cite this article
Gundel, P.E., Helander, M., Garibaldi, L.A. et al. Direct and indirect effects of the fungal endophyte Epichloë uncinatum on litter decomposition of the host grass, Schedonorus pratensis . Plant Ecol 218, 1107–1115 (2017). https://doi.org/10.1007/s11258-017-0755-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11258-017-0755-5