Abstract
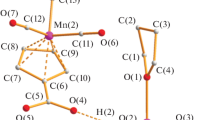
Joint thermolysis of the dinuclear pivalate complexes M2(μ-H2O)(μ-Piv)2(Piv)2(HPiv)4 (M = Co (1) and Ni (2), Piv- is the pivalate anion), in decane at 174 °C at the reactant ratio 1: 1 followed by treatment of the dry thermolysis product with methanol afforded crystals of a new cocrystallization product of the molecules containing the heterometallic cubane-like core M4(Co,Ni)O4. According to the X-ray diffraction data and the results of magnetic measurements, inductively coupled plasma atomic emission spectrometry (ICP-AES), and investigations of the solid-state thermal decomposition products, the isolated cocrystallization product has the general formula [Co1.6Ni2.4(μ3-OMe)4(μ2-Piv)2(pg2 -Piv)2(MeOH)4] ·4MeOH (3·4MeOH). Thermolysis of the crystals of the solvate 3·4MeOH is a destructive process accompanied by the intramolecular redox reaction. A mixture of metallic Ni and cobalt oxide (CoO) are the final solid decomposition products of 3 · 4MeOH in an argon atmosphere, whereas a mixture of the phases NiO, Co3O4, and NiCo2O4 is formed in air.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
J. A. Bertrand, A. P. Ginsberg, R. I. Kaplan, C. E. Kirkwood, R. L. Martin, R. C. Sherwood, Inorg. Chem., 1971, 10, 240.
M. Halcrow, J. S. Sun, J. C. Huffman, G. Christou, Inorg. Chem., 1995, 34, 4167.
V. Tangoulis, C. P. Raptopopoulu, S. Paschalidou, A. E. Tsohos, Inorg. Chem., 1997, 36, 5270.
T. O. Denisova, M. A. Golubnichaya, S. E. Nefedov, Izv. Akad. Nauk, Ser. Khim., 2003, 2612 [Russ. Chem. Bull., Int. Ed. (Engl. Transl.), 2003, 52, 2760].
K. Dimitrou, A. D. Brown, K. Folting, G. Christou, Inorg. Chem., 1999, 38,1834.
G. Chaboussant, R. Basler, H.-U. Gudel, S. T. Ochsenbein, A. Parkin, S. Parsons, G. Rajaraman, A. Sieber, A. A. Smith, G. A. Timco, R. E. P. Winpenny, Dalton Trans., 2004, 2758.
H. Zhao, J. Bacsa, K. R. Dunbar, Acta Crystallogr., Sect. E: Struct. Rep. Online, 2004, 60, m637.
K. Dimitrou, J.-S. Sun, K. Folting, G. Christou, Inorg. Chem., 1995, 34, 4160.
V. G. Kessler, S. G. Gohil, M. Kritikos, O. N. Korsak, E. E. Knyazeva, I. F. Moskovskaya, B. V. Romanovsky, Polyhedron, 2001, 20, 915.
J. K. Beattie, T. W. Hambley, J. A. Klepetko, A. F. Masters, P. Turner, Polyhedron, 1997, 16, 2109.
K. Dimitrou, A. D. Brown, T. E. Concolino, A. L. Rhein-gold, G. Christou, Chem. Commun., 2001, 1284.
G. Aromí, A. S. Batsanov, P. Christian, M. Helliwell, A. Parkin, S. Parsons, A. A. Smith, G. A. Timco, R. E. P. Winpenny, Chem. Eur. J., 2003, 9, 5142.
G. Aromí, A. S. Batsanov, P. Christian, M. Helliwell, O. Roubeau, G. A. Timco, R. E. P. Winpenny, Dalton Trans., 2003, 4466.
J. K. Beattie, T. W. Hambley, J. A. Klepetko, A. F. Masters, P. Turner, Polyhedron, 1998, 17, 1343.
K. Dimitrou, K. Folting, W. E. Streib, G. Christou, J. Am. Chem. Soc., 1993, 115, 6432.
I. G. Fomina, Zh. V. Dobrokhotova, G. G. Aleksandrov, O. Yu. Proshenkina, M. L. Kovba, A. S. Bogomyakov, V. N. Ikorskii, V. M. Novotortsev, I. L. Eremenko, Izv. Akad. Nauk, Ser. Khim., 2009, 11 [Russ. Chem. Bull., Int. Ed., 2009, 58, 11].
W. Bidell, V. Shklover, H. Berke, Inorg. Chem., 1992, 31, 5561.
M. A. Yampoĺskaya, G. S. Matuzenko, N. V. Gerbeleu, Yu. A. Simonov, A. A. Dvorkin, Zh. Neorg. Khim., 1989, 34, 681 [J. Inorg. Chem. USSR(Engl. Transl.), 1989, 34].
T. M. Abdel-Fattah, G. Davies, B. V. Romanovsky, O. L. Shakhnovskaya,A. M. Larin, S. A. Jansen, M. J. Palmieri, Catal. Today, 1996, 89, 670.
M. Hamid, A. A. Tahir, M. Mazhar, M. Zeller, K. C. Mol-loy, A. D. Hunter, Inorg. Chem., 2006, 26, 10457.
I. G. Fomina, G. G. Aleksandrov, Zh. V. Dobrokhotova, O. Yu. Proshenkina, M. A. Kiskin, Yu. A. Velikodnyi, V. N. Ikorskii, V. M. Novotortsev, I. L. Eremenko, Izv. Akad. Nauk, Ser. Khim., 2006, 1841 [Russ. Chem. Bull., Int. Ed., 2006, 55, 1909].
A. Albinati, V. I. Bakhmutov, N. V. Belkova, Eur. J. Inorg. Chem., 2002, 1530.
Zh. V. Dobrokhotova, I. G. Fomina, M. A. Kiskin, A. A. Sidorov, I. L. Eremenko, Izv. Akad. Nauk, Ser. Khim., 2006, 250 [Russ. Chem. Bull., Int. Ed., 2006, 55, 256].
I. G. Fomina, Zh. V. Dobrokhotova, M. A. Kiskin, G. G. Aleksandrov, O. Yu. Proshenkina, A. L. Emelina, V. N. Ikor-skii, V. M. Novotortsev, I. L. Eremenko, Izv. Akad. Nauk, Ser. Khim., 2007, 1650 [Russ. Chem. Bull., Int. Ed., 2007, 56, 1712].
I. G. Fomina, Zh. V. Dobrokhotova, G. G. Aleksandrov, M. A. Kiskin, M. A. Bykov, V. N. Ikorskii, V. M. Novotort-sev, I. L. Eremenko, Izv. Akad. Nauk, Ser. Khim., 2007, 1660 [Russ. Chem. Bull., Int. Ed., 2007, 56, 1722].
T. B. Mikhailova, A. A. Sidorov, I. F. Golovaneva, G. G. Aleksandrov, V. N. Ikorskii, V. M. Novotortsev, I. L. Ere-menko, Zh. Neorg. Khim., 2003, 48, 1648 [Russ. J. Inorg. Chem. (Engl. Transl.), 2003, 48, 1505].
SMART (Control) and SAINT (Integration) Software, Version 5.0, Bruker AXS Inc., Madison, WI, 1997.
G. M. Sheldrick, SADABS, Program for Scanning and Correction of Area Detector Data, Göttingen University, Göttingen, Germany, 1997.
G. M. Sheldrick, SHELX97, Program for the Solution of Crystal Structures, Göttingen University, Göttingen, Germany, 1997.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 4, pp. 685–691, April, 2010.
Rights and permissions
About this article
Cite this article
Fomina, I.G., Dobrokhotova, Z.V., Aleksandrov, G.G. et al. Carboxylate clusters with the M4O4 cubane-like core: pivalate cocrystal containing CoII and NiII . Russ Chem Bull 59, 699–705 (2010). https://doi.org/10.1007/s11172-010-0149-5
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-010-0149-5