Abstract
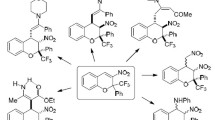
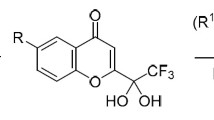
The reactions of 3-nitro-2-trifluoromethyl-and 3-nitro-2-trichloromethyl-2H-chromenes with thiols and aromatic amines proceed via the nucleophilic addition type to the activated double bond to form 2,3,4-trisubstituted chromanes in high yields. The stereoisomeric compositions and structures of the diastereomers were determined by 1H, 19F NMR and 2D NOESY spectroscopies and X-ray diffraction analysis.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
G. P. Ellis, in The Chemistry of Heterocyclic Compounds, Ed. G. P. Ellis, Wiley, New York, 1977, 31.
W. S. Bowers, in Comprehensive Insect Physiology, Biochemistry and Pharmacology, Eds L. I. Gilbert and G. A. Kerkut, Pergamon Press, Oxford, 1985, 8, 551.
W. S. Bowers, T. Ohta, J. S. Cleere, and P. A. Marsella, Science, 1976, 193, 542.
R. Bergmann and R. Gericke, J. Med. Chem., 1990, 33, 492.
G. Burrell, F. Cassidy, J. M. Evans, D. Lightowler, and G. Stemp, J. Med. Chem., 1990, 33, 3023.
R. Gericke, J. Harting, I. Lues, and C. Schittenhelm, J. Med. Chem., 1991, 34, 3074.
A. E. Fenwick, Tetrahedron Lett., 1993, 34, 1815.
H. Koga, H. Sato, T. Ishizawa, N. Taka, and T. Takahashi, Tetrahedron Lett., 1995, 36, 87.
T. Takahashi, H. Koga, H. Sato, T. Ishizawa, N. Taka, and J. Imagawa, Bioorg. Med. Chem., 1998, 6, 323.
T. A. Engler, J. P. Reddy, K. D. Combrink, and D. V. Velde, J. Org. Chem., 1990, 55, 1248.
M. G. Murugesh, K. Subburaj, and G. K. Trivedi, Tetrahedron, 1996, 52, 2217.
J. D. Hepworth and R. Livingstone, J. Chem. Soc. C, 1966, 2013.
G. Descotes and D. Missos, Synthesis, 1971, 149.
R. C. Jennings and A. P. Ottridge, J. Chem. Soc., Perkin Trans. 1, 1984, 1733.
P. Anastasis and P. E. Brown, J. Chem. Soc., Perkin Trans. 1, 1982, 2013.
P. Anastasis and P. E. Brown, J. Chem. Soc., Perkin Trans. 1, 1983, 197.
J. Bujons, F. Camps, and A. Messeguer, Tetrahedron Lett., 1990, 31, 5235.
T. S. Rao, A. K. Singh, and G. K. Trivedi, Heterocycles, 1984, 22, 1377.
R. P. K. Kodukulla, S. Hariharan, and G. K. Trivedi, Tetrahedron, 1994, 50, 4623.
S. Hariharan, H. H. Mathur, and G. K. Trivedi, Indian J. Chem., 1988, 27B, 994.
M. Nyerges, A. Virányi, G. Marth, A. Dancsó, G. Blaskó, and L. Töke, Synlett, 2004, 2761.
N. Ono, N. Banshou, S. Ito, T. Murashima, and T. Ogawa, J. Heterocycl. Chem., 1997, 34, 1243.
T. Shimizu, Y. Hayashi, K. Yamada, T. Nishio, and K. Teramura, Bull. Chem. Soc. Jpn, 1981, 54, 217.
S. Biswas, P. R. Maulik, R. C. Gupta, M. Seth, and A. P. Bhaduri, Acta Crystallogr., Sect. C, 1996, C52, 1036.
V. Yu. Korotaev, I. B. Kutyashev, and V. Ya. Sosnovskikh, Heteroatom. Chem., 2005, 16, 492.
V. Yu. Korotaev, V. Ya. Sosnovskikh, I. B. Kutyashev, and M. I. Kodess, Lett. Org. Chem., 2005, 2, 616.
H. Hofmann and G. Salbeck, Chem. Ber., 1970, 103, 2768.
A. Arduini, A. Bosi, A. Pochini, and R. Ungaro, Tetrahedron, 1985, 41, 3095.
N. Ishizuka, K. Matsumura, K. Sakai, M. Fujimoto, S. Mihara, and T. Yamamori, J. Med. Chem., 2002, 45, 2041.
A. Arduini, A. Pochini, and R. Ungaro, J. Chem. Soc., Perkin Trans. 1, 1986, 1391.
F. H. Allen, O. Kennard, D. G. Watson, L. Brammer, A. G. Orpen, and R. Taylor, J. Chem. Soc., Perkin Trans 2, 1987, S1.
F. H. Allen and O. Kennard, Chemical Design Automation News, 1993, 8, 31 (Version 5.23).
R. S. Rowland and R. Taylor, J. Phys. Chem., 1996, 100, 7384.
M.-C. Yan, Y.-J. Jang, W.-Y. Kuo, Z. Tu, K.-H. Shen, T.-S. Cuo, C.-H. Ueng, and C.-F. Yao, Heterocycles, 2002, 57, 1033.
G. M. Sheldrick, SHELX-97, Programs for Crystal Structure Determination and Refinement, University of Göttingen, Göttingen (Germany), 1997.
Author information
Authors and Affiliations
Additional information
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 2, pp. 309–321, February, 2006.
Rights and permissions
About this article
Cite this article
Korotaev, V.Y., Sosnovskikh, V.Y., Kutyashev, I.B. et al. Reactions of 3-nitro-2-trihalomethyl-2H-chromenes with S-and N-nucleophiles. Synthesis and stereochemistry of 2,3,4-trisubstituted chromanes. Russ Chem Bull 55, 317–330 (2006). https://doi.org/10.1007/s11172-006-0255-6
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/s11172-006-0255-6