Abstract
Background and aims
Australian herbaceous native species have evolved in phosphorus (P) impoverished soils. Our objective was to explore shoot and root adaptations of two of these species with potential to be developed as pasture plants, at low, moderate and high P supply after 4 and 7 weeks of growth.
Methods
A glasshouse experiment examined the effect of 5, 20 and 80 mg P kg−1 air-dry soil on growth, rhizosphere carboxylate content, and mineral nutrition of two Australian native perennials, Kennedia nigricans (Fabaceae) and Ptilotus polystachyus (Amaranthaceae), and the exotic Medicago sativa (Fabaceae).
Key results
Leaf P concentrations at P80 were 6, 14 and 52 mg P g−1 leaf dry weight for M. sativa, K. nigricans and P. polystachyus, respectively. As soil P concentration increased, rhizosphere carboxylate content decreased for M. sativa, increased and then decreased for K. nigricans and was unchanged for P. polystachyus. For all species, the contribution of malic acid declined at the second harvest. For all species and P treatments, the amount of rhizosphere carboxylates per unit root length decreased as root length of a plant increased. Plant P content was determined more by P uptake rate per unit root length and time than by root length. Uptake of Mo for all species, and uptake of K, Mg and Mn for P. polystachyus, increased with soil P concentration. Uptake of Fe and S was higher when the content of carboxylates in the rhizosphere was higher.
Conclusion
Root physiological adaptations (i.e. rhizosphere carboxylate content and P-uptake rate) are more important than morphological adaptations (i.e. root length and diameter) to enhance the uptake of P and cations.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Phosphorus (P) in soil is relatively immobile and often poorly available to most plants (Vance et al. 2003). Therefore, morphological, physiological and biochemical mechanisms have evolved in plants to allow them to respond to P deficiency (Vance et al. 2003; Suriyagoda et al. 2010a). Typical morphological responses to low P availability include increased root development, higher root:shoot ratios, finer roots, longer root hairs and formation of mycorrhizas, all of which facilitate exploration of a greater soil volume (Wissuwa 2005; Smith and Read 2008).
In shoots, P deficiency can trigger a reduction of leaf expansion, photosynthetic rate and stomatal conductance (Jacob and Lawlor 1991; Ghannoum and Conroy 2007; Suriyagoda et al. 2010b), and thereby decrease plant growth. In roots, P availability can affect root morphological (e.g., architecture) and physiological (e.g., rhizosphere carboxylate dynamics) responses (Gahoonia and Nielsen 2004; Ho et al. 2005; Hammond et al. 2009; Liang et al. 2010; Suriyagoda et al. 2010a, 2011; Lynch 2011).
Plants can affect P acquisition by altering their root physiology (Neumann and Martinoia 2002; Wissuwa 2005), such as by an enhanced P-uptake rate (PUR- mg P cm−1 root day−1), and increased exudation of carboxylates (e.g., malate and citrate) and phosphohydrolases (Richardson et al. 2000; Wouterlood et al. 2005). Carboxylates can enhance the solubility of soil P and this is particularly important when the inherent soil P fertility is poor and/or most of the soil P is strongly sorbed. Carboxylates not only increase the uptake of P, but also contribute to the solubilisation of cations in the soil and, as a consequence, may lead to higher contents of these cations in plants (Page et al. 2006). When considering roots, little attention has been given to study of the relationship between the amount of carboxylates present in the rhizosphere of a plant and root length. In most instances the amount of carboxylates in the rhizosphere is reported per unit root dry weight (DW) (Pearse et al. 2007; Pang et al. 2010; Suriyagoda et al. 2010a; 2011). Even in the few instances where the relationships between carboxylate content and root length were established, only a small fraction of the root, near the root tip, was used (Peñaloza et al. 2002; Wouterlood et al. 2005). Due to the difficulty in assessing rhizosphere soil and roots from bulk soil, and because often only a single harvest was made, changes in rhizosphere carboxylate content (i.e. the balance between the amount exuded and that decomposed) with plant age are not well understood. Moreover, contrasting responses have been reported (Wouterlood et al. 2004; Marschner et al. 2011 and references therein). In summary, the dynamics of carboxylates in the rhizosphere (i.e. changes in content and composition) as affected by soil P supply, root length and plant growth (i.e. time) have rarely been explored and therefore warrant further investigation.
Australian native plant species have evolved in P-impoverished environments (Handreck 1997) and in some of these species, P uptake is little down-regulated at increased P supply (Shane et al. 2004; Ryan et al. 2009; Pang et al. 2010). Many native species, but by no means all, are therefore sensitive to P toxicity (Handreck 1997; Ryan et al. 2009; Pang et al. 2010). Because of inherently low fertility, large quantities of P fertilisers have been applied to agricultural soils in Australia to improve crop yields; this has resulted in substantial increases in soil P in many regions (Bolland et al. 1997). An understanding of the morphological and physiological traits that affect P acquisition of native plant species under high P availability is now important to facilitate the effective revegetation of native Australian agricultural landscapes with native species (Standish et al. 2007) and the development of native plants as pasture and crop species (Pang et al. 2010; Bell et al. 2011; Suriyagoda et al. 2011). However, knowledge to assist in the selection of species with enhanced P acquisition under low-P conditions is still required for low-input agroecosystems (Cornish 2009), and may be utilised to improve the productivity and sustainability of high-input agroecosystems (Lambers et al. 2011).
Two perennial herbaceous species native to Australia, Kennedia nigricans Lindl. (Fabaceae) and Ptilotus polystachyus (Gaudich.) F. Muell. (Amaranthaceae) were used in the current experiment. K. nigricans is considered to have potential for use as a grain crop due to its large seed size and favourable seed chemical composition (Rivett et al. 1983; Bell et al. 2011). Kennedia nigricans also has high amounts of rhizosphere carboxylates (i.e. >100 μmol g−1 root DW) (Ryan et al. 2012). P. polystachyus exhibits a remarkable P physiology, being very tolerant of low-P conditions, for reasons that are not yet known, but can accumulate very high leaf [P] (40 mg g−1) at high P supply, without any P-toxicity symptoms (Ryan et al. 2009). In P. polystachyus, high concentrations of Mg and K (greater than 23 and 136 mM, respectively) were found in the vacuoles of leaf cells which also contained high [P] (Ryan et al. 2009). Indeed, for a wide range of plant species, uptake of various nutrients is correlated (Norvell and Welch 1993; Conn and Gilliham 2010), possibly due to their solubilisation in soil through release of carboxylates into the rhizosphere by the plant (Page et al. 2006). Thus it appears that K. nigricans and P. polystachyus are both well adapted to grow in low-P soils and probably possess a range of adaptations to low soil [P] and, for P. polystachyus, to high soil [P].
The objective of the present study was to investigate the growth of K. nigricans and P. polystachyus at low, moderate and high P supply, with a focus on root morphological (i.e. root length, diameter) and root physiological (i.e. nutrient-uptake rate, rhizosphere carboxylates) characteristics after 4 and 7 weeks of growth. Medicago sativa L. (lucerne), a widely grown perennial pasture legume, was also included. We investigated six hypotheses: (i) an increase in soil [P] and thereby tissue [P] would decrease the amount of carboxylates in the rhizosphere; (ii) the composition of carboxylates would not change across P treatments and plant age/harvests; (iii) the total amount of carboxylates in the rhizosphere would increase as root length increased; (iv) higher tissue [P] at increased soil P supply would be due to both an increased root length and an increased PUR (mg P cm−1 root day−1); (v) plants would accumulate high concentrations of mineral elements (i.e. both cations and anions) at high soil [P]; and, (vi) the accumulation of high amounts of mineral elements per plant would also be correlated with the amount of carboxylates in the rhizosphere.
Materials and methods
Growth conditions
The experiment was a fully crossed factorial design of two factors: plant species (K. nigricans, M. sativa, P. polystachyus) and P (P5, 5 mg P kg−1 air-dry soil; P20, 20 mg P kg−1 air-dry soil; P80, 80 mg P kg−1 air-dry soil). In previous experiments, growth of most of the Australian native perennial pasture species was maximum at a soil P supply of ~20 mg P kg−1, and growth was reduced at a P supply over 50 mg P kg−1 (Pang et al. 2010; Suriyagoda et al. 2010a). Therefore, we expected P5 and P80 to be at the sub- and supra-optimal range of P supply. There were two harvests, at 4 and 7 weeks after seedling emergence, with four replicates at each harvest. K. nigricans accession NS19026, M. sativa cv. SARDI-10 and P. polystachyus accession MGP001 were used in the experiment. A sandy loam soil was collected from the top 10-cm layer of a native, virgin forest near Perth (32°2′S, 115°8′E). Soil was air dried and sieved through a 3-mm diameter mesh to remove debris such as plant roots and other organic matter. Initial soil nutrient status as determined by CSBP FutureFarm analytical laboratories, Bibra Lake, Australia, is given in Table 1. As the bicarbonate-extractable soil [P] (Colwell-P) in its native state was 5 mg P kg−1 air-dry soil, the three P treatments received 0, 15 and 75 mg P kg−1 air-dry soil as KH2PO4, in order to obtain P5, P20 and P80, respectively. Additional potassium was supplied in the P5 and P20 treatments as KCl to balance the level of potassium. NH4NO3 (40 mg kg−1 dry soil) was also added to all pots. To prepare the soil for each P treatment, soil was thoroughly mixed using a mechanical mixer until homogeneous. Mixing was done for P5 soil first and then for the P20 and P80 treatments. Pots of 10-cm diameter and 17-cm length were filled with 1.8 kg of soil to a bulk density of 1.4 g cm−3. De-ionised water was added to each pot to reach the field capacity and left for 3 days. The experiment was carried out in a glasshouse at the University of Western Australia, Perth (31°59′S, 115°53′E) as a randomised complete block design. Three seedlings, which were raised on a sterilised sand bed, were planted in each pot and thinned to one plant at 3–4 days. From week 4, 50 mL of 2 mM NH4NO3 was added twice a week to ensure adequate nitrogen supply. Each pot was weighed and watered to reach the field capacity daily. The glasshouse was unheated under natural light and had an average day and night time temperatures of 23 and 13 °C, respectively during the experiment, which was conducted from August to September 2010.
Plant analyses
Harvests were conducted 4 and 7 weeks after establishment. At each harvest, the shoot was removed from the base (i.e. collar area) and used for leaf area measurements with a LiCor LI-3000 Portable Area Meter, which was equipped with a LI-3050A Transparent Belt Conveyer Accessory (LiCor, Lincoln, NE, USA). Soil was taken out of the pot and roots were shaken lightly to remove the excess soil; remaining soil was defined as the rhizosphere soil (Veneklaas et al. 2003). Roots were transferred to a 200-mL vial and washed in a measured amount of 0.2 mM CaCl2 solution ranging from 20 to 100 mL depending on the root volume. The root system was gently dunked in the solution until as much rhizosphere soil as possible was removed. Care was taken to minimise root damage. A 1-mL subsample of the rhizosphere extract was then filtered using a 0.2-μm syringe filter into a 1-mL HPLC vial. The vial was acidified with one drop of concentrated phosphoric acid, placed in dry ice, and transferred to a −20 °C freezer until HPLC analysis. Detailed methodology for the carboxylate analysis is given in Suriyagoda et al. (2010a). The root system was then washed more thoroughly to remove any residual soil. These roots were used to measure the root length, diameter and the distribution of the root length according to diameter classes (i.e. <0.5, 0.5–1, 1–2, 2–3, 3–4, and >4 mm) using the commercial software package WinRHIZO 4.1 (Regent Instruments Inc., Quebec, Canada, 2000). Calculated root length and the soil volume in a pot were used to calculate the root density (i.e. half distance between roots) as given in Bhadoria et al. (2004).
Root, stem and leaf dry weights (DW) were determined after drying at 60 °C for a week. Root mass ratio (RMR) was calculated as the ratio between root dry weight and total plant dry weight. Roots, stems and leaves of each plant were ground separately in a stainless steel ball mill. For analysis of leaf nutrient concentrations, approximately 100 mg of leaf was taken and digested in nitric/perchloric acid and analysed using inductively coupled plasma (ICP) atomic absorption with a Perkin Elmer Optima 5300 DV optical emission spectrometer (OES; Shelton, CT, USA). For stem and root [P] determination, an approximately 100-mg subsample was taken and digested in nitric/perchloric acid, and analysed using the molybdate and malachite green method (Motomizu et al. 1983). Leaf, stem and root [P] and DWs were used to calculate the amount of P taken up by each plant at both harvests. The ΔP uptake and Δroot length for each treatment combination were calculated by taking the difference of mean P content and mean root length, respectively between the second (7 wk) and first (4 week) harvest. The mean PUR (mg P cm−1 root day−1) for each treatment combination was calculated by taking the difference of total amount of P present in plant tissues (i.e. leaves, stems and roots) between the second and first harvest and dividing that value by the difference of average root length at the two harvests.
Soil analyses
Ammonium-N and nitrate-N concentrations were determined as described by Searle (1984), “plant-available” phosphorus (P) and potassium (K) were measured using the Colwell test (Colwell 1965; Rayment and Higginson 1992), available sulfur (S) according to Blair et al. (1991), organic carbon according to Walkley and Black (1934), with the remainder of soil analyses using protocols outlined in Rayment and Higginson (1992). Cation-exchange capacity (CEC) was calculated from exchangeable soil cations (Black 1968).
Statistical analyses
Data were subjected to 2-way analyses of variance in SAS/STAT software Version 9.1 (SAS Institute Inc., Cary, NC, USA) to examine the impact of species, P treatments and their interaction on response variables at each harvest separately. No transformations were needed to meet ANOVA assumptions. Comparisons between means were made using Tukey’s Honest Significant Difference procedure. Means are presented with standard errors of the mean (std.err) and significance is expressed at p < 0.05. Shoot and root DW, root length, root diameter and tissue nutrient concentrations are presented for the second harvest only. When estimating net PUR, ΔP uptake and Δroot length from measured variables, means of the measured variables within each treatment combination at each harvest were used. When establishing the relationship between root length and rhizosphere carboxylate content, heteroscedasticity was present. Therefore, log-transformed root carboxylate data were used to correct this heteroscedasticity.
Results
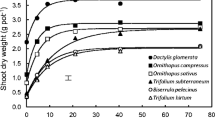
For both shoot and root DW there was an interaction between P treatment and species (Table 2). Shoot and root DW of K. nigricans and M. sativa increased from P5 to P20, while P. polystachyus was less responsive (Fig. 1). At P80, shoot DW of K. nigricans and P. polystachyus was reduced compared with that at P20. Root DW of P. polystachyus was reduced at P80 compared with that at P20. At the first harvest, the impact of P treatment on growth of all species was similar to that at the second harvest (data not shown). Leaf area followed a similar trend to shoot DW (data not shown). Root mass ratio (i.e. root to plant DW ratio) of K. nigricans (0.17) and P. polystachyus (0.17) was much lower than that of M. sativa (0.40) and was unaffected by P treatment (data not shown).
The effect of P treatment on root length varied with species, with root length of all species increasing from P5 to P20 and that of P. polystachyus then declining greatly to P80 (Fig. 2a, Table 2). K. nigricans had a smaller root system (i.e. total root length and root DW were less) than that of M. sativa and P. polystachyus in most instances (Figs 1 and 2a). Root surface area showed a similar trend to root length (data not shown).
a Root length and b root diameter of Kennedia nigricans, Medicago sativa and Ptilotus polystachyus grown at 5, 20 or 80 mg P kg−1 air-dry soil, and c root length distribution based on diameter classes (in mm) for Kennedia nigricans, Medicago sativa and Ptilotus polystachyus after 7 weeks of growth (harvest 2). Note: P treatment had no effect on root length distribution
For root diameter, there was an interaction between species and P treatment (Fig. 2b, Table 2). For M. sativa only, root diameter increased from P5 to P80 (Fig. 2b). Due to this change, specific root length (SRL; root length per unit root DW) of M. sativa changed among P treatments from 121 ± 11 m g−1 at P5 to 58 ± 6 m g−1 and 59 ± 9 m g−1 at P20 and P80, respectively. For K. nigricans and P. polystachyus, SRL remained at 115 ± 12 m g−1 and 157 ± 11 m g−1, respectively, across all treatments.
Root length distribution of a plant based on root diameter classes differed only with species (Fig. 2c, Table 2), with the distribution for K. nigricans and P. polystachyus being very similar and including a higher proportion of fine roots than for M. sativa. For instance, the proportion of root length with a diameter of <1 mm was 21 % for K. nigricans, 27 % for P. polystachyus and 10 % for M. sativa.
Rhizosphere carboxylate content measured at 4 and 7 weeks after seedling emergence showed a species and P treatment interaction (Fig. 3, Table 2). At both harvests, carboxylate content (μmol g−1 root DW) of K. nigricans increased from P5 to P20 and then decreased at P80. For M. sativa, rhizosphere carboxylate content decreased as soil [P] increased, while rhizosphere carboxylate content of P. polystachyus was unchanged with P treatment. For M. sativa and P. polystachyus only, there was a reduction in rhizosphere carboxylates at the second harvest compared with that at the first harvest across P treatments (F 1, 55 = 18.7, P < 0.001). Carboxylates in the rhizosphere mainly comprised citric, malic, malonic and oxalic acid, with the contribution from other organic acids (i.e. acetic, cis-aconitic, fumaric, lactic, succinic and trans-aconitic) less than 8 % (Fig. 3). Composition differed among harvests and species. Contribution of malic acid was reduced from the first to the second harvest for all species. At the second harvest, malonic, citric and oxalic acids were the main rhizosphere carboxylates for K. nigricans, M. sativa and P. polystachyus, respectively (Fig. 3).
Heteroscedasticity observed in root carboxylate content with increase in root length could be corrected for by using the log-transformed root carboxylate data (Fig. 4a). Root carboxylate content (or log-transformed root carboxylate content) in the rhizosphere of a plant increased at a decreasing rate when root length increased, irrespective of species and P treatment (Fig. 4a). Moreover, when higher root lengths (see Fig. 2a) caused roots to be in closer proximity, rhizosphere carboxylates increased (Fig. 4b).
a Relationship between log-transformed rhizosphere carboxylate content and root length of Kennedia nigricans, Medicago sativa and Ptilotus polystachyus grown at 5, 20 or 80 mg P kg−1 air-dry soil, and b log-transformed rhizosphere carboxylate content and half the distance between roots across P treatments and species . Note: only for b P treatment and species had no effect
For leaf [P] there was an interaction between species and P treatment (Fig. 5a, Table 2). For all species, leaf [P] increased with increasing soil [P]. However, the increase from P20 to P80 was much greater for P. polystachyus than for the other two species. The total amount of P taken up by each plant showed a similar response to P treatment as leaf tissue [P] (Table 2, data not shown). Therefore, P. polystachyus achieved a leaf [P] and total P content that was 6.7 and 3.5 times higher, respectively, than those of M. sativa, and 3.1 and 3.4 times higher, respectively, than those of K. nigricans.
Net P-uptake rate (PUR- mg P cm−1 root day−1) increased with P supply for all species (Fig. 5b). However, M. sativa had the lowest PUR at all levels of P supply, while P. polystachyus had the highest. The relationship between ΔP taken up (i.e. amount of P taken up by a plant between two harvests) and PUR was stronger (r = 0.98) than the relationship between ΔP taken up and Δ root length (i.e. difference in root length between two harvests) (r = −0.50) (Fig. 6).
Leaf tissue Fe and S contents per plant were correlated with rhizosphere carboxylate content (Fig. 7). However, there was no relationship with leaf tissue concentrations of K, Mg, Al, Co, Na, Ca, Mn, Cu, Zn or Mo.
Leaf tissue concentrations of most elements were affected by species and P treatment (Tables 3 and 4, Fig. 8). For P. polystachyus at P80, [K], [Mg], [Mo] and [Mn] were higher, and, irrespective of the P treatment, [Na] and [Zn] were higher than those of K. nigricans and M. sativa (Fig. 8). Therefore, the uptake of both cations (i.e. K, Mg, Mn and Na) and anions (i.e. Mo) of P. polystachyus was highly correlated with P uptake, irrespective of P treatment (r = 0.82, 0.51, 0.33, −0.32, and 0.72 for K, Mg, Mn, Na and Mo, respectively, p < 0.05). For K. nigricans, only K and Mo uptake were correlated with P uptake (r = 0.65 and 0.62, respectively, p < 0.05) and for M. sativa only Mo uptake was correlated with with P uptake (r = 0.36, p < 0.02). Moreover, in a narrow range of leaf [P] (i.e. within each soil P treatment) there were numerous correlations between leaf [P] and other mineral elements (e.g., Mg, Na and Zn) for all the species (Fig. 8).
Relationship between leaf tissue concentrations of K, Mg, Mo, Mn, Na and Zn and P for Kennedia nigricans (diamonds), Medicago sativa (squares) and Ptilotus polystachyus (circles) grown at 5, 20 or 80 mg P kg−1 air-dry soil (black, white and grey symbols, respectively). Correlation coefficients (r) for K. nigricans, M. sativa and P. polystachyus are given as r K , r M and r P , respectively, where, ns no significant difference; * p < 0.05; ** p < 0.01; *** p < 0.001
Discussion
Only K. nigricans and M. sativa increased growth from P5 to P20, and only K. nigricans and P. polystachyus decreased growth from P20 to P80. Therefore, for K. nigricans P5 and P80 were in the sub- and supra-optimal range of soil [P], respectively. For M. sativa, P5 was a sub-optimal soil [P], but plants grew equally well at P20 and P80. For P. polystachyus P5 and P20 can be considered as optimal, while P80 was supra-optimal. Thus the growth of both Australian native species was negatively affected by the higher soil [P], while M. sativa grew well even at the higher soil [P] of P80. The contrasting response of K. nigricans and M. sativa to increasing P supply is consistent with that reported for K. prostrata, K. prorepens and M. sativa by Pang et al. (2010). P toxicity at high P supply is relatively common for Australian native plant species that have evolved on P-impoverished soils (Shane et al. 2004; Pang et al. 2010).
Soil [P] and rhizosphere carboxylate dynamics
Hypothesis (i) that an increase in soil [P] and thereby tissue [P] will decrease the amount of carboxylates in the rhizosphere, was only partly supported. Only for K. nigricans (only from P20 to P80) and M. sativa did the amount of carboxylates in the rhizosphere decrease when soil [P] increased. Recently, Pang et al. (2010) and Suriyagoda et al. (2010a, 2011) reported that carboxylate composition did not change with soil [P] for Kennedia spp. and M. sativa. However, using a range of plant species, Lambers et al. (2002) and Pearse et al. (2007) showed that the composition of carboxylates did change with the source of P. Moreover, Veneklaas et al. (2003) found that composition of Cicer arietinum L. carboxylates changed when grown in different soils with varying P availability. When considering the composition of carboxylates along the root length of Lupinus albus L., malate was predominantly exuded from apices of both seedling taproots and proteoid roots, whereas citrate exudation was restricted to proteoid root clusters (Peñaloza et al. 2002). Most of these experiments have tested the amount of carboxylates in the rhizosphere only once during the growth period (i.e. at final harvest) and contrasting results are reported. Moreover, changes in rhizosphere carboxylate composition with time/plant age have been paid little attention. Therefore, we tested hypothesis (ii) that composition of carboxylates would not change across P treatments and plant age/harvests. When considering soil [P] only for K. nigricans, carboxylate composition did not change with soil [P] while for all species, especially K. nigricans, carboxylate composition changed with harvest. Overall, the present findings support previous findings (Lambers et al. 2002; Veneklaas et al. 2003; Pearse et al. 2007) that the amount and composition of carboxylates in the rhizosphere is variable and depend on the plant species, soil [P] and growth stage of the plant. However, the impact of changes in amount and composition of rhizosphere carboxylates on P uptake is not yet clear and needs further attention as not all carboxylates may have a similar effect on plant P uptake. For instance, Ryan et al. (2012) found that plant P content in a range of Kennedia species was strongly correlated with citric acid, but not malic or malonic acids.
Root length and rhizosphere carboxylate content
Roots exude carboxylates into the rhizosphere to enhance uptake of P for plant growth (Lambers et al. 2002). Therefore, our third hypothesis was that the total amount of carboxylates in the rhizosphere of a plant would increase as the root length increased. However, the hypothesis was only partially supported as the rate of increase of rhizosphere carboxylate content decreased and varied (i.e. heteroscedasticity) as the root length increased (Fig. 4a). This might be due to several reasons. First, mature and senescing roots may have had reduced carboxylate exudation compared with active root tips (Marschner et al. 1997; Marschner et al. 2011 and references there in) as we examined plants that grew for 7 weeks and had root lengths up to over 150 m. A decrease in the rhizosphere malate content along the root length was found in L. albus while the response of citrate changed depending on the [P] in soil (Peñaloza et al. 2002). However, Wouterlood et al. (2004) found that carboxylate content of C. arietinum increased with increasing distance from the root apex only up to 2.5 cm from the root tip. Decomposition of carboxylates also can reduce the amount of carboxylates present in the rhizosphere (Jones 1998; Van Hees et al. 2002; Marschner et al. 2011). Second, plants might decrease carboxylate exudation from root regions where soil P has already been depleted. Third, the reduced rate of increase of rhizosphere carboxylate content as the root length increases can also be affected by root morphology, soil characteristics and rhizosphere microbial properties (Suriyagoda et al. 2010a; Marschner et al. 2011 and references there in). As far as we are aware this is the first attempt to establish relationships between total amount of rhizosphere carboxylate content of a plant and root length and this concept warrants further testing.
Root growth and morphology
Hypothesis (iv) that increased tissue [P] at higher soil [P] would be due to both an increased PUR (mg P cm−1 root day−1) and an increased root length was only partly supported. Even though there was an increase in root length from P5 to P20 for all the species, supporting the hypothesis, root length of K. nigricans and P. polystachyus was reduced at P80 compared with that at P20 which did not support the hypothesis. Reduced root length of K. nigricans and P. polystachyus at P80 was correlated with reduced shoot and root growth (Figs 1 and 2). Reduced root length at P80 might be an adaptive response to avoid P toxicity at a higher P supply (Shane et al. 2004). Increased root growth and relative allocation to root growth (higher RMR) is beneficial for P acquisition, since inorganic P is relatively immobile in soil, but may slow overall plant growth because of the increased respiratory burden of root tissue (Lynch and Ho 2005, and references there in). At low- to optimal soil P supply, root length increase of Oryza sativa L. was better correlated with the amount of P taken up than with PUR (Wissuwa 2005). However, in the present experiment, as hypothesised, the PUR increased from P5 to P80 for all species (Fig. 5). Therefore, the amount of P taken up between the two harvests (ΔP) was more strongly correlated with PUR (r = 0.98) than with root length (r = −0.5) (Fig. 6). Moreover, when comparing among species at P80, the PUR of the two Australian native legumes, K. nigricans and P. polystachyus was 2–6 times higher than that of M. sativa. The higher PUR of K. nigricans and P. polystachyus may reflect a higher proportion of fine roots than for M. sativa (Fig. 2), and/or reduced root length as a morphological adaptation coupled with an inability to down-regulate P uptake proportionately. However, Shane and Lambers (2006) reported that the net-P uptake of Grevillea crithmifolia R. Br. was reduced at higher P supply which was considered an adaptation to decrease the rate of P influx at increased P supply.
Nutrient accumulation
Accumulation of one nutrient often has consequences for the accumulation of others, with changes seemingly driven largely by a need to maintain charge balance, although it may also serve as a detoxification mechanism (Zaharieva and Römheld 1991; Ryan et al. 2003; Conn and Gilliham 2010). P. polystachyus has previously been found to accumulate high [P], [K] and [Mg] in its leaf cell vacuoles (Ryan et al. 2009). Therefore, we tested hypothesis (v) plants would accumulate high concentrations of ions (i.e. both cations and anions) at high soil P supply. As hypothesised, P. polystachyus accumulated high concentrations of K, Mg and Mo at P80 (Fig. 8). Furthermore, irrespective of the P treatment, P. polystachyus accumulated very high concentrations of Na, Mn and Zn. Therefore, correlations of ion uptake (i.e. K, Mg, Mo, Mn and Na) of P. polystachyus with P uptake, irrespective of the P treatment, were significant (i.e. r = 0.82, 0.51, 0.72, 0.33 and -0.32 for K, Mg, Mo, Mn and Na, respectively) (Fig. 7, Table 4). For all the species, numerous strong positive correlations were present between a narrow range of leaf [P] (i.e. within each P treatment) and mineral elements. However, to test hypothesis (v) we considered correlations across all P treatments (i.e. wide range of leaf [P]) (Fig. 8) in order to study broad, general trends. Brennan et al. (2000) found that at the seedling stage of P. polystachyus, the critical concentration for shoot growth to avoid deficiency is about 15 mg g−1 DW for K, and 9 mg g−1 DW for P. Adequate concentrations in shoots were 17–27 mg g−1 DW for K and 10–16 mg g−1 DW for P. In the present experiment, at P20, similar results were found, while at P80 concentrations of K and P were higher than those reported by Brennan et al. (2000). Therefore, the P80 treatment was in the supra-optimal range of P for P. polystachyus. However, P. polystachyus did not show any visual P toxicity symptoms, apart from the reduced DW at P80.
Hypothesis (vi) that the accumulation of high amounts of mineral elements would be correlated with the amount of carboxylates in the rhizosphere was only supported for Fe and S (Fig. 8). Argyriadis et al. (1976) found similar results for S. Even though rhizosphere carboxylate dynamics and plant nutritional status were measured in many previous studies, their correlation was not tested (Shen et al. 2004; Suriyagoda et al. 2011). This merits further detailed study.
Concluding remarks
The response of rhizosphere carboxylate content and composition varied with soil P supply, plant species and plant age. For all species and P treatments, the amount of rhizosphere carboxylates per unit root length decreased as root length of a plant increased. Higher rhizosphere carboxylates corresponded with enhanced uptake of Fe and S from soil. The amount of P taken up by plants was determined more by the PUR (i.e. physiological adaptation) than by the root length (i.e. morphological adaptation). When P (i.e. an anion) was taken up in greater quantities, cation uptake was also increased.
References
Argyriadis GA, Dijkshoorn W, Lampe JEM (1976) Level and origin of carboxylate in buckwheat. Plant Soil 44:669–681
Bell LW, Bennett RG, Ryan MH, Clarke H (2011) The potential of herbaceous native Australian legumes as grain crops: a review. Renew Agr Food Syst 26:72–91
Bhadoria PS, El Dessougi H, Liebersbach H, Claassen N (2004) Phosphorus uptake kinetics, size of root system and growth of maize and groundnut in solution culture. Plant Soil 262:327–336
Black CA (1968) Soil-plant relationships. 2nd edn. Wiley, New York. Department of Agronomy, Iowa
Blair GJ, Chinoim N, Lefroy RDB, Anderson GC, Crocker GJ (1991) A soil sulphur test for pastures and crops. Aust J Soil Res 29:619–626
Bolland MDA, Lewis DC, Gilkes RJ, Hamilton LJ (1997) Review of Australian phosphate rock research. Aust J Agr Res 37:845–859
Brennan RF, Webb MG, Crowhurst AM (2000) Yield responses of mulla mulla (Ptilotus exaltatus Nees.) seedlings to additions of nitrogen, potassium and phosphorus fertiliser. Aust J Exp Agr 40:867–871
Colwell JD (1965) An automatic procedure for the determination of phosphorus in sodium hydrogen carbonate extract of soil. Chem Ind 10:893–895
Conn S, Gilliham M (2010) Comparative physiology of elemental distributions in plants. Ann Bot 105:1081–1102
Cornish PS (2009) Phosphorus management on extensive organic and low-input farms. Crop Past Sci 60:105–115
Gahoonia TS, Nielsen NE (2004) Root traits as tools for creating phosphorus efficient crop varieties. Plant Soil 260:47–57
Ghannoum O, Conroy JP (2007) Phosphorus deficiency inhibits growth in parallel with photosynthesis in a C3 (Panicum laxum) but not two C4 (P. coloratum and Cenchrus ciliaris) grasses. Funct Plant Biol 34:72–81
Handreck KA (1997) Phosphorus requirements of Australian native plants. Aust J Soil Res 35:241–289
Hammond JP, Broadley MR, White PJ, King GJ, Bowen HC, Hayden R, Meacham MC, Mead A, Overs T, Spracklen WP, Greenwood DJ (2009) Shoot yield drives phosphorus use efficiency in Brassica oleracea and correlates with root architecture traits. J Exp Bot 60:1953–1968
Ho MD, Rosas JC, Brown KM, Lynch JP (2005) Root architectural tradeoffs for water and phosphorus acquisition. Funct Plant Biol 32:737–748
Jacob J, Lawlor DW (1991) Stomatal and mesophyll limitations of photosynthesis in phosphate deficient sunflower, maize and wheat plants. J Exp Bot 42:1003–1011
Jones DL (1998) Organic acids in the rhizosphere - a critical review. Plant Soil 205:25–44
Lambers H, Finnegan PM, Laliberté E, Pearse SJ, Ryan MH, Shane MW, Veneklaas EJ (2011) Phosphorus nutrition of Proteaceae in severely phosphorus-impoverished soils: are there lessons to be learned for future crops? Plant Physiol 156:1058–1066
Lambers H, Juniper D, Cawthray GR, Veneklaas EJ, Martínez-Ferri E (2002) The pattern of carboxylate exudation in Banksia grandis (Proteaceae) is affected by the form of phosphate added to the soil. Plant Soil 238:111–122
Liang Q, Cheng X, Mei M, Yan X, Liao H (2010) QTL analysis of root traits as related to phosphorus efficiency in soybean. Ann Bot 106:223–234
Lynch JP (2011) Root phenes for enhanced soil exploration and phosphorus acquisition: tools for future crops. Plant Physiol 156:1041–1049
Lynch JP, Ho MD (2005) Rhizoeconomics: carbon costs of phosphorus acquisition. Plant Soil 269:45–56
Marschner P, Crowley DE, Higashi RM (1997) Root exudation and physiological status of a root-colonizing fluorescent pseudomonas in mycorrhizal and nonmycorrhizal pepper (Capsicum annuum L.). Plant Soil 189:11–20
Marschner P, Crowley D, Rengel Z (2011) Rhizosphere interactions between microorganisms and plants govern iron and phosphorus acquisition along the root axis – model and research methods. Soil Biol Biochem 43:883–894
Motomizu S, Wakimoto T, Toei S (1983) Spectrophotometric determination of phosphate in river waters with molybdate and malachite green. Analyst 118:361–367
Neumann G, Martinoia E (2002) Cluster roots: an underground adaptation for survival in extreme environments. Trends Plant Sci 7:162–167
Norvell WA, Welch RM (1993) Growth and nutrient uptake by barley (Hordeum vulgare L. cv Herta): Studies using an N-(2-Hydroxyethyl) ethylenedinitrilotriacetic acid-buffered nutrient solution technique (I. Zinc Ion Requirements). Plant Physiol 101:619–625
Page V, Weisskopf L, Feller U (2006) Heavy metals in white lupin: uptake, root-to-shoot transfer and redistribution within the plant. New Phytol 171:329–341
Pang J, Tibbett M, Denton MD, Lambers H, Siddique KHM, Bolland MDA, Revell CK, Ryan MH (2010) Variation in seedling growth of 11 perennial legumes in response to phosphorus supply. Plant Soil 328:133–143
Pearse SJ, Veneklaas EJ, Cawthray G, Bolland MDA, Lambers H (2007) Carboxylate composition of root exudates does not relate consistently to a crop species’ ability to use phosphorus from aluminium, iron or calcium phosphate sources. New Phytol 173:181–190
Peñaloza E, Corcuera LJ, Martinez J (2002) Spatial and temporal variation in citrate and malate exudation and tissue concentration as affected by P stress in roots of white lupin. Plant Soil 241:209–221
Rayment GE, Higginson FR (1992) Australian laboratory handbook of soil and water chemical methods. Inkata Press, Melbourne
Richardson AE, Hadobas PA, Hayes JE (2000) Acid phosphomonoesterase and phytase activities of wheat (Triticum aestivum L.) roots and utilization of organic phosphorus substrates by seedlings grown in sterile culture. Plant Cell Environ 23:397–405
Rivett D, Tucker D, Jones G (1983) The chemical composition of seeds from some Australian plants. Aust J Agr Res 34:427–432
Ryan MH, Ehrenberg S, Bennett RG, Tibbett M (2009) Putting the P in Ptilotus: a phosphorus-accumulating herb native to Australia. Ann Bot 103:901–911
Ryan MH, McCully ME, Huang CX (2003) Location and quantification of phosphorus and other elements in fully hydrated, soil-grown arbuscular mycorrhizas: a cryo-analytical scanning electron microscopy study. New Phytol 160:429–441
Ryan MH, Tibbett M, Edmonds-Tibbett T, Suriyagoda LDB, Lambers H, Cawthray GR, Pang J (2012) Carbon trading for phosphorus gain: the balance between rhizosphere carboxylates and arbuscular mycorrhizal symbiosis in plant phosphorus acquisition. Plant, Cell Environ. In press.
Searle PL (1984) Berthelot or indophenol reaction and its use in the analytical chemistry of nitrogen. A review. Analyst 109:549–568
Shane MW, Lambers H (2006) Systemic suppression of cluster-root formation and net P-uptake rates in Grevillea crithmifolia at elevated P supply: a proteacean with resistance for developing symptoms of ‘P toxicity’. J Exp Bot 57:413–423
Shane MW, McCully ME, Lambers H (2004) Tissue and cellular phosphorus storage during development of phosphorus toxicity in Hakea prostrate (Proteaceae). J Exp Bot 55:1033–1044
Shen J, Tang C, Rengel Z, Zhang F (2004) Root-induced acidification and excess cation uptake by N2-fixing Lupinus albus grown in phosphorus-deficient soil. Plant Soil 260:69–77
Smith SE, Read DJ (2008) Mycorrhizal symbiosis, 3rd edn. Academic, London
Standish RJ, Cramer VA, Wild SL, Hobbs RJ (2007) Seed dispersal and recruitment limitation are barriers to native recolonization of old-fields in Western Australia. J Appl Ecol 44:435–445
Suriyagoda LDB, Lambers H, Ryan MH, Renton M (2010a) Effects of leaf development and phosphorus supply on the photosynthetic characteristics of perennial legume species with pasture potential: modelling photosynthesis with leaf development. Funct Plant Biol 37:713–725
Suriyagoda LDB, Ryan MH, Renton M, Lambers H (2010b) Multiple adaptive responses of Australian native perennial legumes with pasture potential to grow in phosphorus- and moisture-limited environments. Ann Bot 105:755–767
Suriyagoda LDB, Ryan MH, Renton M, Lambers H (2011) Above- and below-ground interactions of grass and pasture legume species when grown together under drought and low phosphorus availability. Plant Soil 348:281–297
Van Hees PAW, Jones DL, Godbold DL (2002) Biodegradation of low molecular weight organic acids in coniferous forest podzolic soils. Soil Biol Biochem 34:1261–1272
Vance CP, Uhde-Stone C, Allan DL (2003) Phosphorus acquisition and use: critical adaptations by plants for securing a non renewable resource. New Phytol 157:423–447
Veneklaas EJ, Stevens J, Cawthray GR, Turner S, Grigg AM, Lambers H (2003) Chickpea and white lupin rhizosphere carboxylates vary with soil properties and enhance phosphorus uptake. Plant Soil 248:187–197
Walkley A, Black IA (1934) Estimation of soil organic carbon by the chromic acid titration method. Soil Sci 37:29–38
Wissuwa M (2005) Combining a modelling with a genetic approach in establishing associations between genetic and physiological effects in relation to phosphorus uptake. Plant Soil 269:57–68
Wouterlood M, Cawthray GR, Scanlon TT, Lambers H, Veneklaas EJ (2004) Carboxylate concentrations in the rhizosphere of lateral roots of chickpea (Cicer arietinum) increase during plant development, but are not correlated with phosphorus status of soil or plants. New Phytol 162:745–753
Wouterlood M, Lambers H, Veneklaas EJ (2005) Plant phosphorus status has a limited influence on the concentration of phosphorus-mobilising carboxylates in the rhizosphere of chickpea. Funct Plant Biol 32:153–159
Zaharieva T, Römheld V (1991) Factors affecting cation-anion uptake balance and iron acquisition in peanut plants grown on calcareous soils. Plant Soil 130:81–86
Acknowledgments
We thank Kevin Foster for his valuable suggestions, which greatly improved the manuscript. This study was supported by the School of Plant Biology, and the Future Farm Industries Cooperative Research Centre and The University of Western Australia. LDB Suriyagoda also appreciates the SIRF/UIS Scholarship awarded by the University of Western Australia and further scholarship support from the late Frank Ford. We thank Tammy Edmonds-Tibbett and Greg Cawthray for technical help.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Tim Simon George.
Rights and permissions
About this article
Cite this article
Suriyagoda, L.D.B., Lambers, H., Renton, M. et al. Growth, carboxylate exudates and nutrient dynamics in three herbaceous perennial plant species under low, moderate and high phosphorus supply. Plant Soil 358, 105–117 (2012). https://doi.org/10.1007/s11104-012-1311-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-012-1311-7