Abstract
Background and aims
Iron plaque on roots has been hypothesized to be an effective restraint on the uptake of arsenic (As) by rice plants. Evaluating the formation of iron plaque and its effect on As uptake by various rice cultivars is valuable because selecting low As uptake rice cultivars results in reduced risks associated with rice consumption. This study examines iron plaque formation and its effect on As uptake by different genotypes of rice cultivars.
Methods
Hydroponic cultures were conducted in phytotron at day 25/night 20°C and the rice seedlings in fifth-leaf age were treated with Fe (II) at the levels of 0 and 100 mg L−1 in the Kimura B nutrient solutions for 14 days. The amount of iron plaque formation of 28 rice cultivars was determined by using the DCB extractable Fe of roots. Four cultivars representing high and low iron plaque formation capability, from indica and japonica respectively, were selected out of the 28 cultivars and processed for Fe and As treatments. After Fe treatments for 4 days, the seedlings were fed with As (III) at levels of 0, 0.5, and 1 mg L−1 for another 10 days. We were thus able to determine the amounts of iron plaque formation and the As content in iron plaque, roots, and shoots of the four tested cultivars.
Results
Iron plaque formation capability differed among tested twenty-eight rice cultivars. Feeding As to four tested cultivars enhanced iron plaque formation on roots; the As uptake by roots and shoots was decreased by the addition of Fe. Both the retention of As on iron plaque and the decrease of As uptake by the addition of Fe varied among tested cultivars and were not correlated with the iron plaque formation capability.
Conclusions
Iron plaque can sequestrate As on the roots and reduce rice’s As uptake. However, other factors also influence the As uptake, namely the differences in binding affinity of iron plaque to As, the existent As species in the rhizosphere, and the uptake capability of various As species by rice plants. These factors should also be considered when selecting low As uptake rice cultivars.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Arsenic (As) is a human carcinogenic and a ubiquitous metalloid. In an anaerobic soil environment, arsenite is the most prevalent species. Paddy rice is more efficient in accumulating As compared to other cereals (Su et al. 2010). Unfortunately, excessive As accumulation in rice grain poses a significant health risk to consumers (Meharg et al. 2009). Moreover, a high level of As may lead to increased phytotoxicity and decreases in grain yield in paddy fields (Panaullah et al. 2009).
Paddy rice is a staple foodstuff in Asia and is characterized by iron plaque on root surfaces. Ferrous ions form from the reduction of ferric ions under the reducing conditions such as paddy fields (Jien et al. 2010). Paddy rice roots can release oxygen and oxidants into the rhizosphere, thereby oxidize the ferrous ions transported to roots from paddy soils into ferric irons with the precipitation of iron oxides or hydroxides. Therefore, in paddy fields, the toxicity of iron to rice can be reduced through the formation of iron plaque on root surfaces (Chen et al. 1980; Green and Etherington 1977). Iron plaque is mainly composed of ferric hydroxides, goethite, and lepidocrocite (Chen et al. 1980); aside from iron, an element almost as important is manganese (Crowder and Coltman 1993). Iron plaque is characterized by having both amorphous and crystalline iron (hydr) oxides (Bacha and Hossner 1977; Chen et al. 1980). Under field conditions, iron plaque has also served as a restraint on the uptake of metal(loids) such as As by plants. This is probably due to its adsorption or co-precipitation processes (Liu et al. 2004; Otte et al. 1989; Ultra et al. 2009). At the same time, the sink-like characteristic of iron plaque lead to the concentrated metal(loid)s in the rhizosphere. In some cases, iron plaque may release these metal(loid)s and subsequently enhance uptake. For example, iron plaque can diminish the inhibition effect of phosphate on paddy rice’s arsenate uptake (Chen et al. 2005).
The differences in iron plaque formation among rice genotypes have been noted (Geng et al. 2005; Liu et al. 2011). Specifically, the interactions between genotype, environment, and environment x genotype affect the uptake and accumulation of As (Ahmed et al. 2011; Dwivedi et al. 2010; Norton et al. 2009). Furthermore, Hu et al. (2005) reported that both As and phosphate concentrations in iron plaque had a strong positive correlation with the amounts of Fe in iron plaque (DCB-extractable) for three rice cultivars. The degree of radio oxygen loss (ROL) has been used to evaluate root aeration and has been found to have a strong correlation with As tolerance and accumulation in rice (Mei et al. 2009; Wu et al. 2011a) and various wetland plants (Li et al. 2011). Root aeration is a critical factor for different cultivars that affects the formation of iron plaque on root surfaces and subsequent As uptake by plants. The amount of iron plaque produced on root surfaces appears to be the main determinant of As mobility and uptake by rice. With that said, limited information is available on the relationship between plant As uptake and iron plaque formation on root for different rice cultivars. In order to select rice cultivars with low As uptake and thus reduce the risk from rice consumption, it is important to investigate the capability of iron plaque formation and its effect on As uptake by various rice cultivars. The objectives of this study are to evaluate the formation of iron plaque on the roots of commonly planted genotypes of rice cultivars in Taiwan and to investigate its overall effect on As uptake.
Material and methods
Rice cultivars
In this study, we used twenty-eight rice (Oryza sative L.) cultivars commonly planted in Taiwan, including fourteen japonica rice (TK 2, TK 4, TK 8, TK 9, TK 14, TK 16, TNG 16, TNG 67, TNG 71, TC 65, TC 192, TY 3, KS 139, and KS 145) and fourteen indica rice (TS 2, TCS 10, TCS 17, TCSW 1, TCSW 2, TCSY 112, TCSY 837, TCSY952031, TCSY 962021, TCSY 962024, TCSY 962037, TCSY 962045, and TCSY 962058) and evaluated their iron plaque forming capability.
Hydroponic cultures
The hydroponic cultures were cultivated in a phytotron glasshouse with a controlled condition (day 25/night 20°C; RH 70–95 %) with culture solutions refreshed every 2 days. Rice seeds were washed with distilled water for 10 min after being sterilized through being submerged in 1 % sodium hypochlorite solution with one drop of Tween 20 for 30 min. The seeds were then germinated under moist conditions at 37°C for 2–3 days. Fifty seeds of each rice cultivar were then transferred to and held by an iron mesh set on the surface of the culture solution in a beaker followed by raising seedlings in half-strength modified Kimura B nutrient solutions (pH 4.8–5) for 2 weeks. Afterward, the solutions were replaced by the full-strength nutrient solutions. After the fifth leaf of rice seedlings became visible, the treatments of Fe and As addition were administered. The solution cultures were stagnant without aeration or N2 flushing for rice growth to simulate a flooding condition of paddy rice cultivation. In addition, in order to avoid Fe(II) oxidation, the culture solutions were made just before use and refreshed every 2 days.
Since the Fe concentration in paddy soil solutions is usually around 100 mg L−1, a dose of additional 0 and 100 mg L−1 Fe(II) was added to the culture solutions as an Fe treatment for all cultivars for another 2 weeks. Finally, the seedlings were harvested and the iron plaque on root surfaces was extracted by using modified cold DCB solutions (Liu et al. 2004) to evaluate the differences in iron plaque formation capability between cultivars. The roots of cultivar TCN 1 before and after DCB extraction were fixed with agar and then the root cross-section was taken by cutting with a microtome at 5–7 cm below the base of a root (about half of root length). The cross-sections were examined using upright microscope.
To investigate the effect of iron plaque formation on As uptake by rice seedlings, two cultivars were selected respectively from indica and japonica based on iron plaque formation capability. These cultivars included TK 9 (high capability; japonica), TS 2 (high capability; indica), TY 3 (low capability; japonica), and TCSY 962021 (low capability; indica). After the seedlings of the four selected cultivars grew up to the fifth leaf age, additional Fe (II) was added into the culture solutions at the levels of 0 and 100 mg Fe (II) L−1 since the first day (+Fe0 and +Fe100). On the fourth day and after, sodium arsenite was added into the culture solutions at the levels of 0, 0.5, and 1.0 mg As (III) L−1 (As0, As0.5 and As1), respectively. The treatment of +Fe0/As0 was served as a control and the other treatments included +Fe0/As0, +Fe0/As0.5, +Fe0/As1, +Fe100/As0, +Fe100/As0.5, and +Fe100/As1. On the fourteenth day, seedlings were harvested for the analysis.
The harvested seedlings were washed thoroughly with tap water and then with deionized water; they were later separated into roots and shoots. Iron plaque on roots was determined by extraction using DCB solutions from a portion of root samples of each treatment. Briefly, 1 g of roots was extracted for 60 min at room temperature (20–25°C) in a 40 mL solution containing 0.03 M sodium citrate and 0.125 M sodium bicarbonate, with the addition of 0.6 g sodium dithionite. The roots were then rinsed three times with deionized water and the eluates were also added to the DCB extracts.
The roots without removing iron plaque and the shoots were digested separately with concentrated HNO3/H2O2 following the procedure adapted by Sun et al. (2009). The concentrations of Fe and P in the DCB extracts and plant sample digested solutions were measured by ICP-OES (Perkin-Elmer, Optima 2000); Arsenic concentrations were measured by ICP-MS (Agilent, 7700x). The concentrations of As in roots were calculated from the difference between the amounts of As in roots without removing iron plaque and those in iron plaque.
Data analyses
The statistical analysis was carried out using ANOVA (analysis of variance) to test the effect of cultivar genotypes and Fe addition treatments on iron plaque formation and As distribution in iron plaque, roots, and shoots. The General Linear Model of SAS was used to test the difference in the measurements between treatments with least significant difference test (LSD test) at a level of P = 0.05 (SAS Institute). Regression analyses were conducted using SigmaPlot 10.
Results
Observation of iron plaque on cross-section of roots
The fresh cross-sections of the root of one tested rice cultivar TCN 1 (shown as an example) observed by upright microscope indicated that iron plaque visibly formed around the epidermis cells when treated with an additional 100 mg Fe L−1 in the culture solutions (Fig. 1a). Iron plaque was reddish brown and covered the cell wall of epidermis. Fe precipitation was distributed around epidermis cells and predominantly extended inward to the cortex or aerenchyma and rarely to the endodermis (Green and Etherington 1977). DCB extraction rendered iron plaque invisible and partially damaged the epidermis cells; neither exodermis nor any other tissue inside was found damaged (Fig. 1b). Cold DCB extraction procedure has been widely used for the removal of iron plaque to determine the constituents of iron plaque and As uptake by roots. It should be noted that small numbers of the epidermis cells were damaged by the DCB extraction procedure, possibly resulting in overestimation of the amounts of As sequestrated in iron plaque because As might be leaked out from damaged epidermis cells.
Iron-plaque formation capability of 28 genotypes of rice cultivars
Figure 2 shows the amounts of iron plaque (expressed by DCB extractable Fe) formed on root surfaces, the shoot height, root length, and total biomass of the 28 rice cultivars tested in this study. The amounts of DCB extractable Fe of tested rice cultivars grown in culture solutions with additional Fe ranged from 6.03 to 13.3 μmol Fe plant−1 (Fig. 2a). However, they were relatively small grown in culture solutions without additional Fe (data not shown). In addition, they were not significantly different (P > 0.05) between the indica and japonica rice cultivars. Nevertheless, ANOVA revealed a significant effect (P < 0.001) of rice cultivar genotypes on the amounts of DCB extractable Fe of roots in both with and without additional Fe treatments. The genotypes of TS 2 and TCSY 962021 were selected to represent high and low capability of iron plaque formation respectively from indica cultivars. Another two genotypes representing high and low capability of iron plaque formation, TK 9 and TY 3, were also selected from japonica cultivars for As uptake experiments. We also found significant effects of cultivar genotypes on shoot height, root length, and total biomass (P < 0.001). The relative percentages of shoot height with additional Fe treatments compared to control ranged from 86.9 % to 105.2 % in all tested cultivars while the extent of inhibition by Fe on indica cultivars was higher than that of Japonica (Fig. 2b). The relative percentages of root length ranged from 69.3 % to 100 % while the extent of inhibition effect by Fe on japonica was higher than indica (Fig. 2c). For the total biomass, japonica was larger than indica and the extent of inhibition effect by Fe on japonica was higher than indica (Fig. 2d). We observed no significant negative correlation (P < 0.05) between the extent of plant growth (shoot height, root length, and total biomass) inhibition by the additional Fe treatment and the amounts of iron plaque formation of all the cultivars.
(a) The amounts of DCB extractable Fe, (b) shoot height, (c) root length, and (d) total plant biomass of 28 rice cultivars grown in culture solutions with additional 100 mg Fe (II) L−1. The percentages in the bars are the values of additional Fe treatments relative to those without additional Fe treatments (control). NS indicates the difference in the values between Fe treatments was not significant based on the LSD test (P > 0.05)
Iron plaque formation as effected by As
Figure 3 shows the amounts of iron plaque formed on the roots of four tested cultivars grown in the culture solutions with additional 100 mg L−1 Fe and with/without the addition of As. The amounts of DCB extractable Fe of roots of four tested cultivars followed the order of TK 9 >TY 3 and TS 2 > TCSY 962021 for japonica and indica, respectively under the treatment without As addition (As0). The amounts of DCB extractable Fe were increased as increase of amounts of As addition except the TCSY 96201 cultivar under the addition of 0.5 mg As L−1 treatment (Fig. 3). The extent of amounts of DCB extractable Fe were increased 51.7, 77.2, 94.6, and 57.9 % for TY 3, TK 9, TCSY 962021, and TS 2, respectively under the addition of 1.0 mg As L−1 treatment. Among the four tested cultivars, TK 9 had the highest amounts of extractable Fe (0.88 mmol g−1 root DW) without As addition and also the extent of increase of amounts of extractable Fe (0.68 mmol g−1 root DW) by addition of 1 mg L−1 of As.
The effect of As addition (0, 0.5, and 1 mg L−1) on the amounts of DCB extractable Fe on the roots of four rice cultivars grown in solutions with additional 100 mg L−1 Fe. Different letters above the bars indicate significant difference in the values among As treatments based on the LSD test (P < 0.05)
Growth and As uptake by rice plants as effected by Fe and As
Figure 4 shows the shoot height and root length of the four rice cultivars under different Fe and As treatments. It can be seen that the addition of 100 mg Fe L−1 into the culture solutions significantly inhibited (P < 0.05) the growth of rice seedlings for all As treatments and tested cultivars with the exception of shoot height of TY 3 treated with 0.5 mg As L−1 (Fig. 4a). Under the treatments without Fe, the shoot height of all cultivars were significantly decreased (P < 0.05) by As at 1 mg L−1. For those with the treatment of an additional 100 mg L−1 Fe, the growth of rice seedling was also inhibited by the addition of 1 mg L−1As. The shoot height of TY 3, TK 9, and TCSY 962021 and the root length of TCSY 962021 were significantly decreased (P < 0.05).
The effects of additional Fe (0 and 100 mg L−1) and As (0, 0.5, and 1 mg L−1) on (a) shoot height and (b) root length of four rice cultivars grown in culture solutions. Different letters above the bars indicate the differences among the treatments on each cultivar were significant based on the LSD test (P < 0.05)
Figure 5 shows the As contents in roots and shoots of rice seedlings of four tested cultivars and indicates that the As contents in the roots ranged from 3.5 to 23.8 μmol g−1 DW and those of the shoots were much lower and ranged from 0.12 to 0.53 μmol g−1 DW. It can be seen that the As content in both roots and shoots were increased through the addition of As. The extent of As increase in roots through the addition of As was higher compared to those of shoots. The levels of As accumulation in roots were 18.6–75.4 times higher than those of shoots, thereby implicating the low translocation of As from roots to shoots and that As had accumulated mainly in roots. Among the four tested cultivars, japonica cultivars accumulated higher amounts of As than indica in shoots for all Fe and As addition treatments. Figure 5 also shows that at the same levels of As, the addition of 100 mg L−1 Fe decreased the As contents in roots and shoots of tested cultivars with the exception of roots of TK 9 and TS 2 and shoots of TS 2 under the treatment of As 0.5 mg L−1. Under 1 mg As L−1 treatment, the As contents in the root of TCSY 962021 and the shoot of TY 3 were the highest in these cultivars.
The effects of additional Fe (0 and 100 mg L−1) and As (0.5, and 1 mg L−1) on As contents in (a) shoots and (b) roots of four rice cultivars grown in culture solutions. Different letters above the bars indicate the differences among the treatments on each cultivar were significant based on the LSD test (P < 0.05)
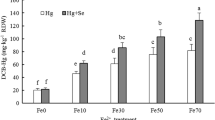
Table 1 shows the As uptake and distribution in iron plaque and plant tissues of rice seedlings grown in the culture solutions containing 0.5 and 1 mg L−1 of As as effected by Fe treatment. It indicates that As sorbed on iron plaque and total uptake (including adsorption on iron plaque and uptake by roots and shoots) by plants increased through the addition of 100 mg Fe L−1 except the total uptake by TK 9 under 0.5 mg L−1 As treatment. However, the As uptake in roots and shoots decreased through the addition of Fe with the exception of shoots of TY 3 and TS 2 under 0.5 mg L−1 As treatment and roots of TY 3 under 1 mg L−1 As treatment. The results also showed that there was an As distribution of 5–50 %, 40–90 %, and 2–13 % in iron plaques, roots and shoots respectively. Although TK 9 had the highest iron plaque formation capability, the amount of As sorbed on iron plaque and the extent of decrease of As uptake by roots and shoots as affected by addition of 100 mg Fe L−1 were not the largest under 1 mg L−1 As treatment. The correlation between the amount of DCB extractable Fe and As of roots of the tested cultivars shown in Fig. 6(a) was much lower compared to that between DCB extractable Fe and P shown in Fig. 6(b).
Discussion
Iron plaque formation capability of the twenty-eight genotypes of rice cultivars including one half as japonica and one half as indica was found to be different among cultivars. The various amounts of iron plaque formed on different rice cultivars implicated different root oxidation ability of various rice genotypes. The effect of genotypes on iron plaque formation on rice was also found in previous studies (Geng et al. 2005; Liu et al. 2004). Although iron plaque formation can play a role in preventing iron toxicity (Chen et al. 1980; Green and Etherington 1977), the iron plaque formation capability was not significantly correlated with the tolerance of rice cultivars to Fe as demonstrated by that no significantly negative correlation (P < 0.05) between the extent of plant growth (shoot height, root length, and total biomass) inhibition by the additional Fe treatment and the amounts of iron plaque formation of all the cultivars in this study was observed.
We observed enhanced iron plaque formation caused by the addition of As to the cultivars (Fig. 3). The ratios of the increase of iron plaque (moles DCB extractable Fe) formation to the moles of Fe or As added into the culture solutions ranged from 0.2 to 0.47 for Fe; respective ratios for As ranged from 0.7 to 45. Seemingly, this suggests that enhanced formation of iron plaque in response to As stress was much stronger than to Fe stress; reasons why have yet to be determined. In additional, the mole ratios increase of Fe to As in iron plaque of tested cultivars were higher than 8 and up to 57. Although no direct mineralogical measurements were made, the higher Fe/As ratios in iron plaque than those of common Fe-As minerals [1, 3, and 0.5 for arsenopyrite (FeAsS), tennantite((Cu,Fe)12As4S13) and oregonite (Ni2FeAs2), respectively] might suggest that the enhancement of iron plaque formation by As was not mainly caused by the precipitation of Fe-As minerals. Mishra et al. (2011) reported that feeding arsenite caused oxidative stress (increase in free radicals) in rice seedlings, an increase in the level of many enzymatic and non-enzymatic antioxidants, and the synthesis of phytochelatin, which mitigates arsenite-induced oxidative damage. They also demonstrated that the addition of As increases O .−2 and H2O2 in both root and shoot tissue. This plant response as one of mechanisms for As detoxification by rice might happen and these free radicals as well as O2 might be released outward from the roots and caused oxidation of Fe (II) to Fe (III) on the root surfaces and subsequent formation of iron plaque. In addition, there was a significant effect of rice genotype on the amount of iron plaque induced by As, thereby implying that the As-induced oxidation ability of roots differed among rice cultivars. It was shown that in synthetic systems with ferrihydrite (which is the predominant Fe (hydr)oxide in iron plaque), the Fe-Fe correlations in ferrihydrite are progressively disrupted which slows the transformation from poorly-crystalline ferrihydrite to a more crystalline Fe phase like goethite or magnetite as the As/Fe ratio in solution is increased (Waychunas et al. 1993). Hence, the possibility that the enhancement of DCB-extractable Fe with increasing As is due to changes in the surface chemistry of the Fe oxide in iron plaque. In addition, a synergistic effect of As(III) and Fe(II) sorption onto goethite was observed at a total As(III) concentration of 1000 μM (Dixit and Hering 2006). Moreover, it is possible that the addition of As promoted the oxidation of Fe(II) to form goethite, as has been seen for other anions like molybdate (Cornell and Schwertmann 2003). The above mentioned Fe-As chemico-physical interactions are also possible reasons for explaining the enhancement of iron plaque formation by addition of As.
The addition of Fe caused an increase in As distribution in iron plaque (from <13 % to 25–52 %). The As contents in roots and shoots of rice decreased through addition of Fe resulting iron plaque formation for four tested cultivars (Figs. 3 and 5b). These two observations suggest that iron plaque appears to be able to sequestrate As; this has been reported in previous studies (e.g. Hu et al. 2005; Wu et al. 2012). It was hypothesized that higher amounts of iron plaque formed would increase As retention by iron plaque and therefore decrease the As uptake by rice. However, the cultivar TK 9, with the highest amounts of iron plaque formation among the four tested cultivars in this study, actually did not have the largest amounts of As retention by iron plaque and the corresponding decrease in As content and uptake by roots and shoots under all treatments. This suggests that the disparities in iron plaque formation capability do not reflect the difference in As retention capability among rice cultivars. The extent of As decrease in roots and shoots by iron plaque was not solely dependent on the amounts of iron plaque formation of rice cultivars. Furthermore, it was observed that a correlation between amounts of DCB extractable Fe and As of roots of tested cultivars was much lower than that between DCB extractable Fe and P in this study, suggesting that As sorption behavior by iron plaque was different from that of P and the binding affinity of iron plaque for As varied among different cultivars. Our results differed from those of Hu et al. (2005) who found that the correlation of both amounts of As and P with Fe in the iron plaque was extremely high for the rice cultivars they tested.
From this study, we found that As can induce iron plaque formation and iron plaque can sequestrate As for tested cultivars. In addition, As retention on iron plaque and decreased As uptake by rice plants varied among rice cultivars. We also found that reducing As uptake by rice cultivars was determined by other factors in addition to iron plaque formation capability. Seyfferth et al. (2010; 2011) also discovered that iron plaque is not a direct interceptor of As uptake by rice through various optical observation methods. Thus, simply evaluating iron plaque formation capability of rice cultivars may be insufficient to ultimately predict their ability to reduce As uptake. The differences in the binding affinity of iron plaque to As (Raven et al. 1998), As species existing in rhizosphere (Wu et al. 2011a), pathways of As uptake (Abedin et al. 2002; Ma et al. 2008; Wu et al. 2011b), uptake capability of various As species (Arao et al. 2011; Deng et al. 2010) by various rice cultivars may complicate the effect of iron plaque formation on As uptake. This requires further investigation in order to ultimately select rice cultivars with the lowest As uptake and thus reduce the risk associated with rice consumption.
References
Abedin MJ, Feldmann J, Meharg AA (2002) Uptake kinetics of arsenic species in rice plants. Plant Physiol 128:1120–1128
Ahmed ZU, Panaullah GM, Gauch H, McCouch SR, Tyagi W, Kabir MS, Duxbury JM (2011) Genotype and environment effects on rice (Oryza sativa L.) grain arsenic concentration in Bangladesh. Plant Soil 338:367–382
Arao T, Kawasaki A, Baba K, Matsumoto S (2011) Effects of arsenic compound amendment on arsenic speciation in rice grain. Environ Sci Technol 45:1291–1297
Bacha RE, Hossner LR (1977) Characteristics of coatings formed on rice roots as affected by iron and manganese additions. Soil Sci Soc Am J 41:931–935
Chen CC, Dixon JB, Turner FT (1980) Iron coating on rice roots: Morphology and models of development. Soil Sci Soc Am J 44:1113–1119
Chen Z, Zhu YG, Liu WJ, Meharg AA (2005) Direct evidence showing the effect of root surface iron plaque on arsenite and arsenate uptake into rice (Oryza sativa) roots. New Phytol 165:91–97
Cornell RM, Schwertmann U (2003) The iron oxides- structure, properties, reactions occurrences and uses. Wiley-VCH, Weinheim
Crowder AA, Coltman DW (1993) Formation of manganese oxide plaque on rice roots in solution culture under varying pH and manganese (Mn2+) concentration conditions. J Plant Nutr 16:589–559
Deng D, Wu SC, Wu FY, Deng H, Wong MH (2010) Effects of root anatomy and Fe plaque on arsenic uptake by rice seedlings grown in solution culture. Environ Pollut 158:2589–2595
Dixit S, Hering J (2006) Sorption of Fe(II) and As(III) on goethite in single- and dual-sorbate systems. Chem Geol 228:6–15
Dwivedi S, Tripathi RD, Srivastava S, Singh R, Kumar A, Tripathi P, Dave R, Rai UN, Chakrabarty D, Trivedi PK, Tuli R, Adhikari B, Bag MK (2010) Arsenic affects mineral nutrients in grains of various Indian rice (Oryza sativa L.) genotypes grown on arsenic-contaminated soils of West Bengal. Protoplasma 245:113–124
Geng CN, Zhu YG, Liu WJ, Smith SE (2005) Arsenate uptake and translocation in seedlings of two genotypes of rice is affected by external phosphate concentrations. Aquat Bot 83:321–331
Green MS, Etherington JR (1977) Oxidation of ferrous iron by rice (Oryza-sativa-L.) roots: Mechanism for waterlogging tolerance. J Exp Bot 28:678–690
Hu Y, Li JH, Zhu YG, Huang YZ, Hu HQ, Christie P (2005) Sequestration of As by iron plaque on the roots of three rice (Oryza sativa L.) cultivars in a low-P soil with or without P fertilizer. Environ Geochem Health 27:169–176
Jien SH, Hseu ZY, Chen ZS (2010) Hydropedological implications of ferromanganiferous nodules in rice-growing plinthitic Ultisols under different moisture regimes. Soil Sci Soc Am J 74:880–891
Li H, Ye ZH, Wei ZJ, Wong MH (2011) Root porosity and radial oxygen loss related to arsenic tolerance and uptake in wetland plants. Environ Pollut 159:30–37
Liu JG, Leng XM, Wang MX, Zhu ZQ, Dai QH (2011) Iron plaque formation on roots of different rice cultivars and the relation with lead uptake. Ecotox Environ Safe 74:1304–1309
Liu WJ, Zhu YG, Smith FA, Smith SE (2004) Do iron plaque and genotypes affect arsenate uptake and translocation by rice seedlings (Oryza sativa L.) grown in solution culture? J Exp Bot 55:1707–1713
Ma JF, Yamaji N, Mitani N, Xu XY, Su YH, McGrath SP, Zhao FJ (2008) Transporters of arsenite in rice and their role in arsenic accumulation in rice grain. Proc Natl Acad Sci USA 105:9931–9935
Meharg AA, Williams PN, Adomako E, Lawgali YY, Deacon C, Villada A, Cambell RCJ, Sun G, Zhu YG, Feldmann J, Raab A, Zhao FJ, Islam R, Hossain S, Yanai J (2009) Geographical variation in total and inorganic arsenic content of polished (wite) rice. Environ Sci Technol 43:1612–1617
Mei XQ, Ye ZH, Wong MH (2009) The relationship of root porosity and radial oxygen loss on arsenic tolerance and uptake in rice grains and straw. Environ Pollut 157:2550–2557
Mishra S, Jha AB, Dubey RS (2011) Arsenite treatment induces oxidative stress, upregulates antioxidant system, and causes phytochelatin synthesis in rice seedlings. Protoplasma 248:565–577
Norton GJ, Duan GL, Dasgupta T, Islam MR, Lei M, Zhu YG, Deacon CM, Moran AC, Islam S, Zhao FJ, Stroud JL, McGrath SP, Feldmann J, Price AH, Meharg AA (2009) Environmental and genetic control of arsenic accumulation and speciation in rice grain: Comparing a range of common cultivars grown in contaminated sites across Bangladesh, China, and India. Environ Sci Technol 43:8381–8386
Otte ML, Rozema J, Koster L, Haarsma MS, Broekman RA (1989) Iron plaque on roots of Aster tripolium L.: interaction with zinc uptake. New Phytol 111:309–317
Panaullah GM, Alam T, Hossain MB, Loeppert RH, Lauren JG, Meisner CA, Ahmed ZU, Duxbury JM (2009) Arsenic toxicity to rice (Oryza sativa L.) in Bangladesh. Plant Soil 317:31–39
Raven KP, Jain A, Loeppert RH (1998) Arsenite and arsenate adsorption on ferrihydrite: Kinetics, equilibrium, and adsorption envelopes. Environ Sci Technol 32:344–349
Seyfferth AL, Webb SM, Andrews JC, Fendorf S (2010) Arsenic localization, speciation, and co-occurrence with iron on rice (Oryza sativa L.) roots having variable Fe coatings. Environ Sci Technol 44:8108–8113
Seyfferth AL, Webb SM, Andrews JC, Fendorf S (2011) Defining the distribution of arsenic species and plant nutrients in rice (Oryza sativa L.) from the root to the grain. Geochim Cosmochim Acta 75:6655–6671
Su YH, McGrath SP, Zhao FJ (2010) Rice is more efficient in arsenite uptake and translocation than wheat and barley. Plant Soil 328:27–34
Sun G, Williams PN, Zhu Y, Deacon C, Carey A, Raab A, Feldmann J, Meharg AA (2009) Survey of arsenic and its speciation in rice products such as breakfast cereals, rice crackers and Japanese rice condiments. Environ Int 35:473–475
Ultra VU, Nakayama A, Tanaka S, Kang YM, Sakurai K, Iwasaki K (2009) Potential for the alleviation of arsenic toxicity in paddy rice using amorphous iron-(hydr)oxide amendments. Soil Sci Plant Nutri 55:160–169
Waychunas GA, Rea BA, Fuller CC, Davis JA (1993) Surface chemistry of ferrihydrite: Part 1. EXAFS studies of the geometry of coprecipitated and adsorbed arsenate. Geochim Cosmochim Acta 57:2251–2269
Wu C, Ye ZH, Shu WS, Zhu YG, Wong MH (2011a) Arsenic accumulation and speciation in rice are affected by root aeration and variation of genotypes. J Exp Bot 62:2889–2898
Wu C, Ye ZH, Li H, Wu SC, Deng D, Zhu YG, Wong MH (2012) Do radial oxygen loss and external aeration affect iron plaque formation and arsenic accumulation and speciation in rice? J Exp Bot. doi:10.1093/jxb/ers017
Wu ZC, Ren HY, McGrath SP, Wu P, Zhao FJ (2011b) Investigating the contribution of the phosphate transport pathway to arsenic accumulation in rice. Plant Physiol 157:498–508
Acknowledgments
The financial support from the National Science Council, Executive Yuan, Taiwan (grant no. NSC-98-2313-B-002-028-MY3) is sincerely appreciated.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Henk Schat.
Dr. Chia-Hsing Lee and Mr. Yi-Cheng Hsieh are equal contributors to this paper.
Rights and permissions
About this article
Cite this article
Lee, CH., Hsieh, YC., Lin, TH. et al. Iron plaque formation and its effect on arsenic uptake by different genotypes of paddy rice. Plant Soil 363, 231–241 (2013). https://doi.org/10.1007/s11104-012-1308-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-012-1308-2