Abstract
Cereals are considered to be a major source of carbohydrates and proteins as well as minor micronutrients such as minerals, vitamins and antioxidants, including carotenoids. Carotenoids are natural lipophilic pigments biosynthesized mainly by plants, and certain bacteria and fungi. Cereals, although having a low carotenoid content when compared with the majority of fruits and vegetables, may have an important impact in the nutritional status of consumers. The daily consumption of cereals, and products derived from them, by a large part of the population, especially in under-developed and developing countries, makes cereals a contributor of carotenoids which should not be overlooked and must be taken into consideration in biofortification strategies. In the present manuscript, we revise the existing information about the composition and distribution of carotenoids in cereals, highlighting factors which alter their profile, such as domestication of wild varieties, genotype, storage, milling and processing techniques. Strategies for stimulating the carotenoid content in cereals, either by means of traditional breeding methods or by genetic manipulation, are also discussed.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Carotenoid pigments are molecules of lipophilic nature and are responsible for some of the yellow, orange and red colour of a wide range of fruits and some animal teguments (Britton and Hornero-Méndez 1997). Animals are not able to synthesize them de novo and so they must be acquired through the diet. Carotenoids are located in oil droplets, crystalloids and membrane structures within specialized vegetable organelles, the so-called plastids, being the most common the chloroplasts (in green tissues) and the chromoplasts (yellow, orange and red tissues), and are found in all parts of the plant: roots, leaves, flowers, fruits and seeds (Howitt and Pogson 2006). Carotenoids carry out their most basic functions in vegetables as pigment collectors of light in the photosynthetic apparatus, and preventing oxidative damage acting as antioxidants.
When carotenoids are ingested, they exert important biological activities; antioxidant, inhibition of carcinogenesis, enhancement of the immune response and cell defence against reactive oxygen species (ROS) and free radicals, and the reduction on the risk of developing cardiovascular and other degenerative diseases (Nishino 1997; recently reviewed by Britton et al. 2009). In addition some carotenoids (β-carotene, α-carotene, β-cryptoxanthin, etc.) have provitamin A activity (Baker and Günter 2004; Olson 1989). Of particular interest are the epidemiological studies showing an inverse correlation between the progression of age-related macular degeneration (AMD) and cataracts and the high intake of lutein and zeaxanthin rich-vegetables, being both pigments present in high concentration at the macula in the retina humans and primates (Ahmed et al. 2005; Alves-Rodrigues and Shao 2004; Calvo 2005; Landrum and Bone 2001).
Cereals are basic food stuffs of vegetable origin, also known as staple food, which are characterised mainly as sources of carbohydrates and proteins, and contain a relatively small amount of carotenoids compared to the majority of fruits and vegetables. However, the daily consumption of cereals and products derived from them by a large part of the population, especially in under-developed and developing countries, makes cereals a contributor of carotenoids which must be taken into consideration (Graham and Rosser 2000). Therefore, cereals can be considered ideal elements for use in biofortification strategies (Bai et al. 2011).
An essential step for the development of carotenoid biofortified crops is the complete characterization of the carotenoid profile contained in the vegetables, allowing this an adequate knowledge of their metabolism, which can be used for the selection and breeding of new cultivars. The success of such processes lies in the existence of a more or less extensive variation of carotenoid contents among different cereals. This variability is mainly due to three factors: (a) the genotype of the cereal, (b) the varietal selection, the pressure exerted by man and its domestication, and (c) the growing conditions and post-harvest storage and processing (industrial and domestic) practices. Regarding the last factor, the evaluation of such changes is an active area of study in food science research, which allows the prediction and selection of the optimal conditions for preserving these phytochemicals, driven by their nutritional properties as well as the applications derived from their natural role as being responsible for the colour of foods. Thermal treatments and others which include homogenisation and modification of the food matrix are factors which have a beneficial effect on the bioaccessibility of carotenoids since it facilitates their solubility. The term bioaccessibility is used to evaluate the amount of a nutrient that is released from a food during the digestion process. The bioaccessibility of lipophilic compounds, such as carotenoids, in natural foods is frequently fairly low and is constrained by various factors, particularly the degree of food processing and matrix composition. There are evidences suggesting that homogenisation and thermal treatment have positive effects on the bioaccessibility of these compounds. Therefore, foods such as cereals, which are normally consumed as processed foods, could present some advantages in comparison to fresh food.
Up to now, there have been several studies regarding the qualitative and quantitative composition of carotenoids in fruits and vegetables, but very few with respect to cereals and therefore studies which determine the carotenoid profile of these foodstuffs are necessary. Undoubtedly, investigations aimed at improving the bioaccessibility of carotenoids in cereals will benefit from such knowledge and it will aid in the information and recommendation provided to the consumer about the way of processing and consumption of these basic elements.
In the present manuscript, we revise the existing information about the composition and distribution of carotenoids present in cereals, highlighting factors which alter their profile, such as storage and processing. At the same time, the strategies for stimulating the carotenoid content in cereals, either by means of traditional breeding methods or by genetic manipulation, are also discussed.
Composition and distribution of carotenoids in cereals
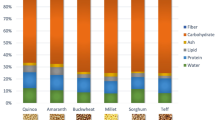
Cereals and their derivatives are good examples of foods that contain a complex mixture of minor components such as phenols, folates, vitamin E, phytic acid, sterols and carotenoids, some of them with antioxidants properties (McKevith 2004). In general, the content of bioactive compounds in cereals is mainly concentrated in the germ and bran portions and most of them are located in the aleurone layer. At the same time, the content in phytochemicals is subjected to important variations due to factors such as the type of cereal, the cultivar, cultivation conditions, stage in development, and storage and processing conditions (Fardet et al. 2008; Liu 2007;), with the greatest importance lying in the interactions between the genotype and the environment (Hidalgo et al. 2009; Lv et al. 2013; Van Hung and Hatcher 2011). The carotenoid profile in cereals is mainly composed of xanthophylls, with lutein as the most abundant, followed by zeaxanthin and β-cryptoxanthin in addition to carotenes such as α- and β-carotene in small amounts (Fig. 1). The largest concentration of carotenoids can be found in the embryo, although this part of the seed only represents 3–5 % of its total weight. On the contrary, the contribution of the endosperm, around 80–85 % of the cereal grain, is the most influential in the total content of carotenoids in the grain. The distribution of this carotenoid profile seems to vary among genotypes of the same type of cereal (Siebenhandl et al. 2007) and within the same grain since α-, β-carotene and zeaxanthin are concentrated in the bran and the germ, while lutein is distributed more homogeneously (Borrelli et al. 2008; Konopka et al. 2004; Ndolo and Beta 2013; Panfili et al. 2004). In relation to the composition of carotenoids in cereals, the bibliography is fairly scarce, especially for barley (Hordeum vulgare), rye (Secale cereale) and millet (Panicum miliaceum) (Choi et al. 2007; Kandlakunta et al. 2008; Mamatha et al. 2011).
In light of the fact that the level of carotenoids in cereals is low in comparison to fruits and vegetables, corn (Zea mays) of the yellow genotype may be cited as the cereal which presents the greatest levels (up to 63 μg/g) of these phytochemicals. Most of the cultivars present zeaxanthin as the major pigment but also show small amounts of β-cryptoxanthin α-carotene and β-carotene (Moros et al. 2002; Panfili et al. 2004). From the quantitative studies carried out on this cereal, a large variation in the contents of carotenoids has been determined among varieties (Berardo et al. 2004, 2009; Egesel et al. 2003; Kurilich and Juvik 1999; Quanckenbush et al. 1961), which serves as proof that genotype is the determining factor for this variety (Ibrahim and Juvik 2009; Menkir and Maziya-Dixon 2004), followed by storage and processing conditions (Burt et al. 2010; De Oliveira and Rodriguez-Amaya 2007; Scott and Eldridge 2005).
In the case of rice (Oryza sativa), it is obvious that the pigmented varieties are the ones which result in some interest from a quantitative point of view. In the grains of conventional rice the carotenoid content is concentrated almost exclusively in the bran and therefore the common practices of milling and stripping of the grain result in a drastic reduction in this component in the final product (Tan et al. 2005). Similar to most cereals, lutein is the main pigment (Belefant-Miller and Grace 2010), followed by zeaxanthin and β-carotene in crops with dark or black pigmentation (Frei and Becker 2005; Kim et al. 2010; Lamberts and Delcour 2008; Nakornriab et al. 2008). In some varieties of black rice from Thailand, the concentration of β-carotene found in its bran is significant (up to 40 μg/g) (Nakornriab et al. 2008).
A recent study carried out on glabrous canary seed (Phalaris canariensis L.) has shown a content of β-carotene ranging from 5.2 to 6.3 μg/g, which is the major pigment in this cereal (Li and Beta 2012). Although sorghum (Sorghum bicolor) has been considered one of the cereals with the least amount of carotenoids, the pioneer findings of Blessin et al. (1958) and Suryanarayana Rao et al. (1968) concerning varieties of yellow endosperm, as well as the recent studies of Kean et al. (2007, 2011), offer better perspectives for this cereal. These authors found a carotenoid content between 0.11 and 0.32 mg/kg, which although lower than the content corresponding to yellow corn, these data must not be overlooked, given the importance of this cereal in the diet of some populations (mainly in Asia and Africa).
Oats (Avena sativa) are also cited in the literature as one of the cereals with the lowest level of carotenoids, with contents of lutein and carotenes (α- and β-carotene) in the order of 0.20 mg/kg and 0.01 mg/kg, respectively (Panfili et al. 2004). Wheat (genus Triticum), along with corn and rice, are the most popular cereals due to their generalised consumption. This is the most consistently seen aspect in the literature, with special interest being paid to the study of their antioxidant profile and contents of healthy phytochemicals as well as their inter- and intra-varietal distribution (Adom et al. 2003, 2005; Di Silvestro et al. 2012; Lier and Lacroix 1974; Okarter et al. 2010; Velioglu et al. 1998; Zhou et al. 2004a, b; Zhou and Yu 2004a, b). Similar to the case of corn, the genetic variability among wheat genotypes concerning the contents of carotenoids, offers an interesting tool which can be effectively used in programs aimed at augmenting the contents of bioactive compounds with properties which are beneficial to health (Digesù et al. 2009; Leenhardt et al. 2006a).
Wheat varieties with diploid genome (AA), in particular the Einkorn wheat (Triticum monococcum), are considered those presenting the highest levels, in the order of approximately 2–4 times higher with respect to other wheat varieties. Einkorn also stands out as possessing the greatest antioxidant activity (Lavelli et al. 2009). Followed by these, tetraploid wheats (AABB) can be found and has been determined to contain the highest levels of carotenoids (around 5–6 µg/g) among the durum wheat cultivars (Triticum turgidum conv. durum), although others have been mentioned such as the ancestral Emmer (T. turgidum subsp. dicoccoides) with similar levels (approximately 3–5 µg/g). Finally, wheat varieties belonging to the hexaploid genome (AABBDD) are considered to have the lowest carotenoid content, with almost exclusive attention paid to the common bread wheat (T. aestivum), which shows an average content around 2 µg/g (Abdel-Aal et al. 2007; Hidalgo et al. 2006; Hidalgo and Brandolini 2008a; Serpen et al. 2008).
These quantitative data indicate that the selection and domestication of cereal species carried out by man has derived in a wide variation of their carotenoid levels (both low and high) (Digesù et al. 2009). The case of durum wheat constitutes an example which has been extensively analysed due to the fact that the yellow colour of its semolina (also referred to as yellow pigment content; YPC, determined as lutein equivalents by the official method 125 of the International Association for Cereal Science and Technology and method 14–50 of the American Association of Cereal Chemists) is considered one of the principal criteria of quality of processed derived foods, mainly pasta (Blanco et al. 2011; Fratianni et al. 2005; Hentschel et al. 2002; Humphries et al. 2004). An inverse situation occurs with bread wheat destined to flour production, which presents lower levels of carotenoids due to the pressure exerted by industries motivated by a consumer preference for whiter flour for the further preparation of flour-containing products according to market demand.
The Triticeae tribe, which includes wheat, barley and rye species, is a series of polyploids which are closely related, being possible to generate fertile hybrid amphiploids among the different cultivated members of this tribe and its wild parents. Among the hybrid cereals, tritordeum, an innovative cereal with interesting properties and applications, must be pointed out. Tritordeum is a cereal which has been obtained from the cross-breeding between a wild barley, Hordeum chilense, with diploid genome (HchHch), and diploid and polyploid wheats (Martín and Chapman 1977). In addition to the development of amphiploids such as tritordeum, the use of H. chilense in the enhancement programs has also been focused on the inclusion of wheat with new characteristics of interest. Among the highlighted attributes, a high content of carotenoids, determined by 7Hch chromosome, stands out. The first fertile hybrids with the optimal characteristics for becoming a possible crop were only obtained following to the crossing of H. chilense with T. turgidum (a tetraploid durum wheat, with genomic denotation AABB), generating those denominated hexaploid tritordeums (2n = 6x = 42, HchHchAABB) which showed a low frequency of aneuploids, a wide variation in growth rate and an optimal level of fertility (Martín and Sanchez-Monge Laguna 1982).
Most of the estimations on the carotenoid content made on tritordeum did not provide detailed information about the individual composition of the pigment profile due to the lack of specificity of the traditional analytical methods employed for the characterisation of carotenoids in cereals. By means of estimations such as YPC, levels of carotenoids in the order of 5–6 times higher (in the range of 11–13 µg/g) have been found in tritordeum with respect to common wheat (2 µg/g) and 2–3 times higher with respect to durum wheat (5–6 µg/g) (Álvarez et al. 1995; Martín et al. 1999), which is comparable to the levels reported for Einkorn wheat. In an extensive study which evaluated a total of 35 primary lines of tritordeum together with their respective parental lineage, 27 H. chilense accessions and 19 durum wheat cultivars, the average levels of carotenes (µg/g of β-carotene equivalents, determined as YPC) for the amphiploid continued to be in the order of 2 times higher than durum wheat parent, but 3 times lower than the average of H. chilense (Álvarez et al. 1999). Among the advanced lines of tritordeum, HT621 was registered as an elite germplasm line due to its high content of carotenoid pigments (19 µg of β-carotene equivalents/g, YPC) (Ballesteros et al. 2005). Recent studies carried out in our laboratory (Atienza et al. 2007a; Mellado-Ortega and Hornero-Méndez 2012) have contributed substantially to the characterisation and quantification of individual carotenoid pigments in tritordeum. Similar to most cereals, lutein is also the major pigment (>85 %), with a high esterification degree (monoesters and diesters), along with minor quantities of β-carotene (Fig. 2). On average, the total carotenoid content of tritordeum (6.5 μg/g of fresh weight) was significantly higher, approximately eight times higher, when compared to durum wheat (0.7 μg/g of fresh weight). For the first time, the regioisomers of the monoesters and diesters of lutein in a cereals, in this case in tritordeum, have been isolated and characterized (Mellado-Ortega and Hornero-Méndez 2012) (Fig. 3). The fraction of monoesters is made up of the regioisomers, namely lutein-3′-O-linoleate, lutein-3-O-linoleate, lutein-3′-O-palmitate and lutein-3-O-palmitate, while the fraction of diesters is composed of two homoesters, lutein dilinoleate and lutein dipalmitate, and by the two regioisomers of a heterodiester, lutein-3′-O-linoleate-3-O-palmitate and lutein-3′-O-palmitate-3-O-linoleate. The esterification of lutein with only two different fatty acids (linoleic and palmitic acids) suggests a high-specificity degree for the in vivo process, which should be further investigated. The ability to distinguishing the regioisomers of mono- and diesters of lutein provides a powerful tool contributing to a better deciphering of this poorly known biochemical pathway, for which tritordeum grains could be used as an excellent plant model. Evidences for a preferential xanthophyll acyltransferase activity regarding the position (3 or 3′) and the acyl moiety are discussed. Further studies should be carried out in order to identify the acyltransferase enzymes (XAT) and the acyl donor molecules involved in the xanthophylls esterification process.
HPLC chromatograms corresponding to direct carotenoid extracts obtained from tritordeum and durum wheat. Peaks identity: 1 all-trans-zeaxanthin, 2 all-trans-lutein, 3 9-cis-lutein, 4 13-cis-lutein, 5 lutein monolinoleate, 6 lutein monopalmitate, 7 all-trans-α-carotene, 8 all-trans-β-carotene, 9 lutein dilinoleate, 10 lutein linoleatopalmitate, 11 lutein dipalmitate
Tritordeum is currently the subject of an intense breeding program being developed at the Institute of Sustainable Agriculture (CSIC; Cordoba, Spain) in order to optimise its use as a new cereal and its incorporation into the formulation of functional foods. Its cultivation areas are concentrated in Spain (Andalusia, Castilla and Catalonia), the south of Italy and the south of Portugal. In 2008 it was registered, under the name Aucan, the first line of tritordeum in the European Union Plant Variety Registration (CPVO), with other advanced lines in their final phases of development and evaluation. As a result of these efforts, at the beginning of 2013, the Spanish company Agrasys (www.agrasys.es) began to commercialise tritordeum flour under the brand name Vivagran®.
Cereal technology and its influence on the carotenoid contents in grains and derived products
Conservation and storage conditions, as well as different processing treatments, result in overall changes in the composition of foods, being this the main reason for the numerous studies carried out in the field of food technology which are crucial for determining some important sensorial and nutritional quality attributes (Kalt 2005; Mínguez-Mosquera et al. 1997; Nicoli et al. 1999; Rodríguez-Amaya 2003). In this way, it is important to distinguish between the effects of processing itself, and other environmental factors such as partial oxygen pressure, temperature, exposure to light and humidity as well as the interactions with other antioxidant and pro-oxidant molecules which are present (Lindley 1998; Nicoli et al. 1999). In the case of the carotenoids occurring in cereals, the storage of the natural and processed grain normally results in a decrease in the pigment contents, which is directly proportional to the processing time and the increase in the intensity of other variables such as temperature or the degradation process (Cristobal 1965; Weber 1987). It is interesting to note that the literature concerning this aspect is especially scarce since cereals are characterised as undergoing prolonged storage periods as part of their industrial and technological treatments. On the other hand, it is not easy to define a set of general guidelines in the behaviour of pigments in processes such as pealing, grinding, drying, roasting, fermentation, etc., due to the fact that positive, negative and even indifferent results can be found in the literature regarding the level of carotenoids present. In general, these processes would affect the internal structure of the cereal matrix and the processed product with substantial implications regarding accessibility and further bio-availability of the carotenoids.
The rate at which these changes occur has been a generalised subject of food science. The bibliography concerning the kinetics of carotenoid degradation and/or retention in foods is abundant as well as the disparity of the results which have often been contradictory (Mínguez-Mosquera et al. 1997). The profusion of such studies reflects the importance of carotenoid pigments for the food industry, not only from a nutritional point of view, but from a technological standpoint, with interest in not just quantifying losses in carotenoids but also analysing the conditions which provide their greater stability and subsequent retention. Colour is considered one of the main consumer acceptance criteria for processed foods. As mentioned before, in the flour and bread as well as the semolina and pasta this factor is important, especially for products derived from durum wheat. Therefore the research efforts in this field try to reproduce the processing and storage conditions of food, including cereals, with the aim of comparing different industrial treatments and monitoring the incidences of individual or joint factors such as water, temperature, light, oxygen and pH activities along with pro-oxidants and antioxidants (Fish and Davis 2003; Mínguez-Mosquera and Gandul-Rojas 1994; Ouchi et al. 2010; Saxena et al. 2012; Selim et al. 2000; Tsimidou 1997), allowing for an estimate of the shelf-life of foods and their bioactive compounds. Most of the studies have been carried out on vegetables such as carrots (Koca et al. 2007; Lemmens et al. 2010; Wagner and Warthesen 1995), tomato (Sharma and Le Maguer 1996; Tonon et al. 2007); peppers and pepper derived products (Carbonell et al. 1986), potatoes (Bechoff et al. 2010), and citrus fruit juices (Dhuique-Mayer et al. 2007; Zepka et al. 2009) where the authors commonly describe a degradation reaction which occurs according to zero or first order kinetics. However, the literature is quite limited in the particular case of cereals, probably due to the low level of pigments that these foods present and there are very few studies which carry out a detailed kinetic evaluation (Guzman-Tello and Cheftel 1990; Hidalgo and Brandolini 2008b; Mellado-Ortega 2013). Nevertheless, although such alterations have not been quantified, most authors agree in the fact that changes generally occur more rapidly at the beginning of processing and storage and as they progress the rate at which they are occurring diminishes (Burt et al. 2010; Quackenbush 1963).
Stability of carotenoids during the post-harvest storage of grains and flours
The storage process produces a loss in carotenoids mainly caused by oxidation, either of an enzymatic or non-enzymatic nature. The oxygen present in the medium is considered the major factor affecting the stability of carotenoids (Britton and Khachik 2009). Other modifications of the carotenoid molecule associated with these processes are the geometric isomerisation (cis/trans) produced by temperature and/or light and cause alterations in the composition rather than net losses in the pigments. The mechanisms of both processes have been extensively studied (El-Agamey and Mcgarvey 2008; Liaaen-Jensen and Lutnaes 2008).
While the oxidation of pigments in the storage of cereal grains comes mainly from oxygen, in processed foods such as flour the alteration of the matrix produces direct oxidative processes as well as those caused by enzymes which come into contact with carotenoids (Doblado-Maldonado et al. 2012). As previously mentioned, the results obtained in these studies depend on the conditions applied, mostly evaluating the effects of time and temperature applied during the treatments. The literature contains examples in which the effects of these two variables are clearly observed, which allow for the prediction of the consequences of these variables in the stability of the carotenoids present (Arya and Parihar 1981; Calucci et al. 2004; Farrington et al. 1981; Hidalgo and Brandolini 2008b; Nghia et al. 2006; Pinzino et al. 1999).
On occasion, in addition to storage, the experimental designs additionaly cover another factors corresponding to common practices in cereal technology (such as drying, grinding, shelling, etc.). This widens the study but at the same time difficult the analysis of the obtained results. Belefant-Miller and Grace (2010) evaluate the behaviour of carotenoids during the prolonged storage of rice which was previously shelled. A common practice in cereals prior to storage is the drying of the grains since the reduction in humidity prevents deterioration due to mould, acarus, etc., as well as a premature germination of the grain (Chelowski 1994; Jood and Kapoor 1994). Some authors, such as Burt et al. (2010) did not obtain a significant reduction in pigment content when comparing drying at a high temperature (90 °C) to the corresponding ambient temperature while maintain the rest of the storage conditions for corn. On the contrary, other studies have found significant differences in the pigment content which are attributed not only to the drying of the grain but also to duration of the process (Quackenbush 1963). In spite of this controversy, the optimal conditions for the preservation of cereals, and particularly to preserve the carotenoids present in them are always reached by employing an exhaustive method of water removal (dehydration) and/or storage at low temperatures.
Studies on the storage of grains and processed grains also allow for the evaluation of aspects relating to the post-carotenogenic metabolism such as esterification with fatty acids which mediate its accumulation and stability in these foods (Kaneko and Oyanagi 1995; Kaneko et al. 1995). The increase in the esterification of lutein as the storage of grain progresses, including storage under controlled temperature, is one aspect that has been characterised by several authors (Ahmad et al. 2013; Mellado-Ortega et al. 2015). This provides information about the ideal conditions for the storage of cereals which preserves the contribution of these phytochemicals. In addition, a specific evaluation has been applied in order to characterise the enzymatic systems involved in esterification of the xanthophylls during the storage of grains and flours of tritordeum and durum wheat. This has resulted in important differences attributed not only to the nature of esterification in each case, but also to the enzymatic regional selectivity of the lutein and fatty acid molecules participating in such reactions (Mellado-Ortega 2013). Regardless of the type of plant material, either grain or flour, the stability of the esterified and free pigment has been studied and it has been found that the degradation rate is always greater for the latter case. This provides valuable information which could be used in crop enhancement programs of cereals. In agreement with these results the studies aimed to increase the cereal contents in carotenoids should be based on the selection of varieties with greater concentrations of esterified xanthophylls with the objective of increasing their ability to store them within the grain tissues, along with improving their stability in the seeds.
On the other hand, the influence of thermal pre-treatments on the storage of flour has been studied to evaluate the generation of free radicals and their impact on endogenous antioxidants (Andersen et al. 2011), as well as the possible intervention of degrading enzymes (Rodriguez-Amaya 1997).
Effect of processing on the carotenoid contents of cereals
As explained above, the different processing techniques undoubtedly modify the carotenoid content present in the final product of processed cereals, due to, among other factors, an uneven distribution of the pigments in the different parts of the grain (Konopka et al. 2004; Zhou et al. 2004a, b). This has led to the modification and development of new processing techniques with the aim of preserving or stimulating the carotenoid contents along with other phytochemicals of nutritional importance. These emerging techniques are being incorporated into the traditional enhancement programs and genetic manipulation for obtaining bio-fortified grain crops (Fardet 2010; Hemery et al. 2007). Cereals are generally processed in two ways: dry fractioning followed by baking (at different temperature conditions, water contents and pressure) to produce product such as pasta, biscuits, breakfast cereal, etc., and fermentation, which produce alcoholic beverages. The production of the most popular cereal-derived products such as bread and baked goods include both treatments.
The fractionation and grinding processes of the cereal grains have allowed for the detailed analysis of carotenoids in the different layers and industrial fractions of the seeds (Ndolo and Beta 2013). The analyses of whole-grain products and those containing the germ (Kean et al. 2008), flour without bran and its formulations (Žilić et al. 2012), whole and shelled grains (Kean et al. 2011), grains, flour and semolina (Fares et al. 2008; Luterotti and Kljak 2010), are among the most common comparisons. One of the first studies carried out in this way was done by Blessin et al. (1963) on corn. These authors analysed the effect of manual and industrial processing on the pigment content found in different layers of the grain. Recently, Sellappan et al. (2009) has described the losses in β-carotene, iron and zinc in genetically modified rice varieties subjected to polishing and have estimated them to be higher than 70 %. Polishing is a common industrial practice for rice grains, which involves the near complete elimination of the aleurone layer and the embryo (Juliano 1994). The classification of the genotype of cereals, by means of the characterisation of its pigment contents due to specific formulations of the grains can be a good example of the use of these crop enhancement techniques. Descriptive analyses like those of Siebenhandl et al. (2007) contribute to this aspect, evaluating formulations of different bran particle sizes and flour for the classification and selection of diverse genotypes of wheat and barley. The size of the particle generated in these processes has a great influence from the technical and nutritional viewpoints. A reduction in the particle size facilitates the liberation of vitamins and other compounds from the outer layers of the grain (Fratianni et al. 2005; Kahlon et al. 1986; Zhou et al. 2004a, b). Zhou et al. (2004a, b) showed that micronizing the aleurone layer resulted in greater antioxidant activity compared to the non-micronized aleurone layers of bran and grain, possibly due to a greater availability of antioxidants.
The diverse thermal treatments which are applied with the objective of prolonging the shelf-life of processed cereal products have also been reproduced in the laboratory to determine their effect on carotenoid contents. Toasting the cereal seems to have a rather pronounced repercussion which causes a significant reduction in pigment contents. This was reported by De Oliveira and Rodriguez-Amaya (2007), who analysed a series of fresh and processed corn products and estimated the loss in zeaxanthin to be about 53 % after toasting the corn flour. Similar results have been described as a result of cooking rice (Lamberts and Delcour 2008). Scott and Eldridge (2005) performed a comparison of diverse thermal treatments and their effects on the carotenoid profiles of fresh, frozen and canned corn and found that the thermal treatments applied to corn prior to canning did not produce a relevant decrease in its carotenoid content. The freezing and scalding pre-treatments applied to cereals and vegetables in general may result in an increase in the bioavailability of carotenoids (Selman 1994). The heating by microwave of rice bran produced similar results without a reduction in the pigment levels which makes it a good alternative for favouring the stability of this cereal formulation (Abdul-Hamid et al. 2007). A more complex treatment consists of an extrusion, a multi-step treatment which is widely applied in the breakfast cereal industry. The mild conditions applied during the extrusion (high moisture content, short treatment time and low temperature) have a clearly positive effect on the retention of vitamins and carotenoids and the decrease in lipid peroxidation, among others (Cheftel 1986; Singh et al. 2007). Guzman-Tello and Cheftel (1990) studied the changes in the concentration of β-carotene under the most severe extrusion conditions of wheat flour and estimated losses ranging from 38 to 73 % for an applied temperature interval of 125–200 °C. Other treatments such as those involved in the malting process of barley are characterised for generating contradictory results concerning their effect on pigment levels. For example, Goupy et al. (1999) registered losses (of approximately 76 %) as well as gains in carotenoid contents in a study with different varieties of barley.
The analyses carried out during the different steps involved in the processing of semolina and making of pasta indicate that it is during the kneading phase when the greatest decrease in carotenoid content is registered while the subsequent drying and maturation periods turn out to be the least aggressive (Fratianni et al. 2012; Hidalgo et al. 2010; Panfili et al. 2005). At the same time, important losses (up to 66 %) in carotenoids during the kneading phase are associated with the production of bread, biscuits and baked goods (Leenhardt et al. 2006b). Other authors, however, point out different production phases, especially the baking step as being influential in the reduction in pigments. In a study of the evolution of pigments during the production of bread from wheat flour and einkorn, Hidalgo et al. (2010) registered averaged losses of 47 % for the crust compared to 21 % for the centre of the bread. After comparing the two types of flour, einkorn was found to suffer the greatest loss in pigments. Degradation during kneading is governed by an enzymatic oxidation of the pigments which has led some authors to relate both aspects (degradative enzymatic activity and carotenoid loss) in the enhancement programs for the selection of optimal cereal genotypes for the bread making industry (Borrelli et al. 2003; Fu et al. 2013; Leenhardt et al. 2006a, b; Trono et al. 1999).
In the case of tritordeum, the properties of this new cereal make it one of the most suitable for bread applications, similar to common wheat, but on the contrary, less appropriate for formulations of semolina. As mentioned before, tritordeum flour has recently began to be commercialised with the aim of several applications such as bread, sandwich bread, biscuits, muffins and other cereal products (www.agrasys.es). In the sense, even though the elaboration process of bread and other products results in losses in carotenoid contents, as in any other cereal, tritordeum bread presents superior levels of lutein (6 times greater) than bread made from traditional wheat (according to the analyses carried out in our laboratory for the Agrasys company).
Applications of the biotechnology of carotenoids to cereal selection and breeding
Biotechnology, as a vegetable enhancement technique, applied to the increase in the carotenoid content in cereals has been used fundamentally on three species: corn, wheat and sorghum. In the case of rice the grains do not produce any carotenoid in the endosperm, however a new rice variety, Golden Rice, was developed at the beginning of this century by engineering grains to produce β-carotene (provitamin A) in order to help combating vitamin A deficiency in populations with rice-based traditional diets (this applies to countries such as India, Vietnam, Bangladesh, the Philippines, and Indonesia) (Paine et al. 2005; Ye et al. 2000). The relevance of these cereals and their impact on the nutrition of the populations of under-developed countries is reflected in biofortification programs such as the one of Harvestplus, belonging to the Consultive Group on International Agricultural Research (CGIAR) which is focused on the augmentation of zinc, iron and provitamin A contents in basic crops producing staple foods (http://www.harvestplus.org/).
The last developments in the exploration of the genomes of the main cereals such as wheat has been recently reported (Eversole et al. 2014), and has been made thanks to the existence of genomic models such as rice and sorghum (Mace and Jordan 2011; Paterson et al. 2009). A large part of the investigations has been based, in the first instance, on the study of the variability in the feature of being susceptible to traditional selection and breeding, in this case, for the enhancement of carotenoid content in a given population and in the second place on the ability to inherit such a trait (Clarke et al. 2006; Santra et al. 2005). Rice, sorghum and wheat genomes have shown diverse genetic importance for this characteristic as reflected in the existence of elite germplasm in these species (Chander et al. 2008; Ibrahim and Juvik 2009). Up to now, the study has been focused on the identification of QTLs (Quantitative Trait Loci) whose allelic variation is associated with a variation of quantitative trait such as the carotenoid content, which varies continuously. Numerous studies can be found which describe such locus and their relation to the phenotypic variation in the carotenoid contents of the endosperm of cereals (Chander et al. 2008; Patil et al. 2008; Pozniak et al. 2007; Salas-Fernandez et al. 2008; Wong et al. 2004). Studies at this level go beyond the justification of the overall carotenoid content and identify specific QTLs for certain carotenoids such as lutein and β-carotene. Salas-Fernandez et al. (2008) detected up to five QTLs for β-carotene content in sorghum located at the 1, 2, and 10 chromosomes. Howitt et al. (2009) identified three QTLs for lutein contents in the 3B, 5B and 7A chromosomes of durum wheat. More recently, Blanco et al. (2011) have mapped QTLs for β- and α-carotene in the chromosomes 2A, 3B and 7A.
In wheat, the most determinant QTLs for the pigment contents have been repeatedly mapped onto the 7A and 7B chromosomes, both on durum wheat and common wheat. The PSY1 codifying genes (phytoene synthase, EC 2.5.1.32) generally co-segregate with these QLTs. The allelic variations of these genes are numerous and their continuous appearance is very probable. This demonstrates a large number of genetic polymorphisms as well as many genomes to be explored (Ravel et al. 2013). One of the strategies in crop improvement is the search for new variation sources in related wild species such as Lophopyrum ponticum (Zhang et al. 2005) and H. chilense (Rodríguez-Suárez et al. 2011; Rodríguez-Suárez and Atienza 2012). The small grains of H. chilense are characterised as having a large amount of carotenoids and present at least two loci for the content of pigments located in the 2Hch and 7Hch chromosomes (Álvarez et al. 1998, 1999; Atienza et al. 2004, 2008). Knowledge about the chromosomal location was first studied by Álvarez et al. (1998) using additional lines of H. chilense in wheat. These authors located genes responsible for the level of pigments (determined as YPC) in H. chilense at chromosome 7 and mapped this feature onto the α arm of this chromosome. However, the possibility of an interaction between H. chilense genes and the durum wheat genes of tritordeum promoted a search for new locus. Subsequently, by means of the construction of the first genetic map of H. chilense, a new QTL entitled carot 1 was identified at chromosome 2 (Atienza et al. 2004; Hernández et al. 2001). Currently, the generation of maps of a wider range for these purposes is now available, and therefore there is a greater chance of finding genomic regions of interest in H. chilense (Rodríguez-Suárez et al. 2012).
This information constitute the first steps towards the development of a “Marker Assisted Selection” (MAS) program for the content of carotenoids in tritordeum, which is interesting for the identification and further transference of particular genes related with the carotenoid content from tritordeum to wheat lines subjected to improvement programs. In this way, the amphiploid tritordeum will also become a useful species for the enhancement of cereals acting as a bridge species between barley and wheat (Atienza et al. 2005, 2007a, b; Martín et al. 1999). The tritordeum lines, regardless of their ploidy level, show carotenoid contents which are higher than their wheat relatives (Atienza et al. 2007a). Therefore, the study of the genetic variation of this feature in tritordeum and its relation to the level of pigments of the parental species H. chilense and durum wheat has become a very active research topic. From these studies it appears that even though the genome Hch is clearly responsible for the level of pigments in tritordeum there can be interactions between the genetic backgrounds of both parental species that may not be obvious (Álvarez et al. 1999). Recently the first exhaustive analysis of the carotenoid profile of H. chilense was carried out, discovering that more than half of the lutein was esterified, with a similar esterification pattern to tritordeum. This corroborates the previous results and indicates that the esterification pattern of tritordeum should result from the genetic background of this wild barley (Mellado-Ortega and Hornero-Méndez 2015). Recent studies have demonstrated that lutein esterification in wheat and H. chilense is controlled by a loci located at the chromosomes 7D and 7Hch, respectively (Ahmad et al. 2015; Mattera et al. 2015).
The utility of the metabolic engineering to improve human nutrition reaches its best objectives in the basic foods field (staple foods) which have an important impact on the nutrition of under-developed populations. The new generation of crops with the Golden label such as Golden Potato or Golden Canola are increasing with time leading up to the well-known Golden Rice (Bai et al. 2011; Beyer 2010). The success of last case is encouraging similar studies and improvement programs with other species. The intense investigation from the first attempts made in rice by Burkhardt et al. (1997) to the most recent ones with the Golden Rice 2 generation (Paine et al. 2005) have provided stimulating ideas for this purpose. Another example is the introduction and expression of the y1 gene (gene coding for phytoene synthase in yellow endosperm corn) from corn into the hexaploid common wheat and its positive effect on increasing the carotenoid content in the endosperm (Cong et al. 2009). In the case of corn, the improved use of the γ-zein promotor (Marzábal et al. 1998), highly specific for the expression in endospermic tissue (super γ-zein), derived into an increase in carotenoids of up to 34 times with a preferential accumulation of β-carotene (Aluru et al. 2008). A more complex strategy was the one designed by Zhu et al. (2008) with the generation of an entire transgenic corn plant battery that resulted from the multiple combinations possible derived from a multi-gene transformation. More recently, the simultaneous increase in the vitamin contents (vitamins A, B and C) involving the simultaneous manipulation of three different metabolic routes (Naqvi et al. 2009), establishes corn as a likely crop to maximise carotenoid provitamin A contents, showing less restrictions than others such as rice or wheat (Wurtzel et al. 2012). The most recent Golden Rice investigations focus now on the study of the stability and transference of the transgenes for the selection and breeding of rice crops with a higher efficiency through introgression and other enhancement techniques (Datta et al. 2006, 2007). Other less studied grains such as sorghum and millet, but having a great impact on under-developed populations such as the African, are increasingly adapting to these techniques making its way into the biofortification mediated by genetic manipulation (O’Kennedy et al. 2006; www.grandchallenges.org/ImproveNutrition/Challenges/NutrientRichPlants/Pages/Sorghum.aspx).
Abbreviations
- AMD:
-
Age-related macular degeneration
- MAS:
-
Marker assisted selection
- PSY:
-
Phytoene synthase gene
- QTL:
-
Quantitative trait locus
- ROS:
-
Reactive oxygen species
- XAT:
-
Xanthophyll acyltransferase
- YPC:
-
Yellow pigment content
References
Abdel-Aal EM, Young JC, Rabalski I et al (2007) Identification and quantification of seed carotenoids in selected wheat species. J Cereal Sci 55:787–794
Abdul-Hamid A, Sulaiman RRR, Osman A et al (2007) Preliminary study of the chemical composition of rice milling fractions stabilized by microwave heating. J Food Comp Anal 20:627–637
Adom KK, Sorrells ME, Liu RH (2003) Phytochemical profiles and antioxidant activity of wheat varieties. J Agric Food Chem 51:7825–7834
Adom KK, Sorrells ME, Liu RH (2005) Phytochemicals and antioxidant activity of milled fractions of different wheat varieties. J Agric Food Chem 53:2297–2306
Agrasys. http://www.agrasys.es/es/index.html. Accessed Dec 2014
Ahmad FT, Asenstorfer RE, Soriano IR et al (2013) Effect of temperature on lutein esterification and lutein stability in wheat grain. J Cereal Sci 58:408–413
Ahmad FT, Mather DE, Law H-Y et al (2015) Genetic control of lutein esterification in wheat (Triticum aestivum L.) grain. J Cereal Sci 64:109–115
Ahmed SS, Lott MN, Marcus DM (2005) The macular xanthophylls. Surv Ophthalmol 50:183–193
Aluru M, Xu Y, Guo R et al (2008) Generation of transgenic maize with enhanced provitamin A content. J Exp Bot 59:3551–3562
Álvarez JB, Ballesteros J, Arriaga HO et al (1995) Rheological properties and baking performances of flours from hexaploid tritordeums. J Cereal Sci 23:291–299
Álvarez JB, Martín LM, Martín A (1998) Chromosomal localization of genes for carotenoid pigments using addition lines of Hordeum chilense in wheat. Plant Breed 117:287–289
Álvarez JB, Martín LM, Martín A (1999) Genetic variation for carotenoid pigment content in the amphiploid Hordeum chilense × Triticum turgidum conv. durum. Plant Breed 118:187–189
Alves-Rodrigues A, Shao A (2004) The science behind lutein. Toxicol Lett 150:57–83
Andersen ML, Erichsen HR, Skibsted LH et al (2011) Heat induced formation of free radicals in wheat flour. J Cereal Sci 54:494–498
Arya SS, Parihar DB (1981) Effect of moisture and temperature on storage changes in lipids and carotenoids of atta (wheat flour). Food Nahr 25:121–126
Atienza SG, Ramírez CM, Hernández P et al (2004) Chromosomal location of genes for carotenoid pigments in Hordeum chilense. Plant Breed 123:303–304
Atienza SG, Avila CM, Ramírez MC et al (2005) Application of near infrared reflectance spectroscopy to the determination of carotenoid content in tritordeum for breeding purposes. Aust J Agric Res 56:85–89
Atienza S, Ballesteros J, Martín A et al (2007a) Genetic variability of carotenoid concentration and degree of esterification among tritordeum (×Tritordeum Ascherson et Graebner) and durum wheat accessions. J Agric Food Chem 55:4244–4251
Atienza SG, Martín AC, Ramírez MC et al (2007b) Effects of Hordeum chilense cytoplasm on agronomic traits in common wheat. Plant Breed 126:5–8
Atienza SG, Martín A, Pecchioni N et al (2008) The nuclear-cytoplasmic interaction controls carotenoid content in wheat. Euphytica 159:325–331
Bai C, Twyman RM, Farré G et al (2011) A golden era-pro-vitamin A enhancement in diverse crops. In Vitro Cell Dev Biol Plant 47:205–221
Baker R, Günter C (2004) The role of carotenoids in consumer choice and the likely benefits from their inclusion into products for human consumption. Trends Food Sci Technol 15:484–488
Ballesteros JB, Ramírez MC, Martínez C et al (2005) Registration of HT621, a high carotenoid content tritordeum germplasm line. Crop Sci 45:2662–2663
Bechoff A, Dhuique-Mayer C, Dornier M et al (2010) Relationship between the kinetics of β-carotene degradation and formation of norisoprenoids in the storage of dried sweet potato chips. Food Chem 121:348–357
Belefant-Miller H, Grace SC (2010) Variations in bran carotenoid levels within and between rice subgroups. Plant Food Hum Nutr 65:358–363
Berardo N, Brenna OV, Amato A et al (2004) Carotenoids concentration among maize genotypes measured by near infrared reflectance spectroscopy (NIRS). Innov Food Sci Emerg Technol 5:393–398
Berardo N, Manzzinelli G, Valoti P et al (2009) Characterization of maize germplasm for the chemical composition of the grain. J Agric Food Chem 57:2378–2384
Beyer P (2010) Golden Rice and ‘Golden’ crops for human nutrition. New Biotechnol 27:478–481
Blanco A, Colasuonno P, Gadaleta A et al (2011) Quantitative trait loci for yellow pigment concentration and individual carotenoid compounds in durum wheat. J Cereal Sci 54:255–264
Blessin CW, VanEtten H, Wiebe R (1958) Carotenoid content of the grain from yellow endosperm-type sorghums. Cereal Chem 35:359–365
Blessin CW, Brecher JD, Dimler RJ (1963) Carotenoids of corn and sorghum: V. Distribution of xanthophylls and carotenes in hand-dissected and dry-milled fractions of yellow dent corn. Cereal Chem 40:582–590
Borrelli GM, De Leonardis AM, Fares C et al (2003) Effects of modified processing conditions on oxidative properties of semolina dough and pasta. Cereal Chem 80:225–231
Borrelli GM, De Leonardis AM, Platani C et al (2008) Distribution along durum wheat kernel of the components involved in semolina colour. J Cereal Sci 48:494–502
Britton G, Hornero-Méndez D (1997) Carotenoids and colour in fruit and vegetables. In: Tomás-Barberán FA, Robins RJ (eds) Phytochemistry of fruit and vegetables, Ch. 2. Clarendon Press, Oxford, pp 11–27
Britton G, Khachik F (2009) Carotenoids in food. In: Britton G, Liaaen-Jensen S, Pfander H (eds) Carotenoids, vol 5: nutrition and health. Birkhäuser Verlag, Basel, pp 45–66
Britton G, Liaaen-Jensen S, Pfander H (2009) Carotenoids, vol 5: nutrition and health. Birkhäuser Verlag, Basel
Burkhardt PK, Beyer P, Wünn J et al (1997) Transgenic rice (Oryza sativa) endosperm expressing daffodil (Narcissus pseudonarcissus) phytoene synthase accumulates phytoene, a key intermediate of provitamin A biosynthesis. Plant J 11:1071–1078
Burt A, Grainger C, Young J et al (2010) Impact of postharvest handling on carotenoid concentration and composition in high-carotenoid maize (Zea mays L.) kernels. J Agric Food Chem 58:8286–8292
Calucci L, Capocchi A, Galleschi L et al (2004) Antioxidants, free radicals, storage proteins, puroindolines, and proteolytic activities in bread wheat (Triticum aestivum) seeds during accelerated aging. J Agric Food Chem 52:4274–4281
Calvo MM (2005) Lutein: a valuable ingredient of fruit and vegetables. Crit Rev Food Sci 45:1–26
Carbonell JV, Piñaga F, Yusá V et al (1986) The dehydration of paprika with ambient and heated air and the kinetics of colour degradation during storage. J Food Eng 5:179–193
Chander S, Guo YQ, Yang XH et al (2008) Using molecular markers to identify two major loci controlling carotenoid contents in maize grain. Theor Appl Genet 116:223–233
Cheftel JC (1986) Nutritional effects of extrusion-cooking. Food Chem 20:263–283
Chelowski J (ed) (1994) Cereal grain. Mycotoxins, fungi and quality in drying and storage. Developments in food science. Elsevier, Amsterdam
Choi Y, Jeong H, Lee J (2007) Antioxidant activity of methanolic extracts from some grains consumed in Korea. Food Chem 103:130–138
Clarke FR, Clarke JM, McCaig TN et al (2006) Inheritance of yellow pigment concentration in seven durum wheat crosses. Can J Plant Sci 86:133–141
Cong L, Wang C, Chen L et al (2009) Expression of phytoene synthase 1 and carotene desaturase crtl genes result in an increase in the total carotenoids content in transgenic elite wheat (Triticum aestivum L.). J Agric Food Chem 57:8652–8660
Cristobal JAR (1965) Variation of the content of carotenes in cereals in relation to the time of storage. An Inst Invest Vet 15:85–92
Datta K, Rai M, Parkhi V et al (2006) Improved “Golden” rice and post-transgeneration enhancement of metabolic target products of carotenoids (β-carotene) in transgenic elite cultivars (IR64 and BR29). Curr Sci 91:935–939
Datta SK, Datta K, Parkhi V et al (2007) Golden rice: introgression, breeding, and field evaluation. Euphytica 154:271–278
De Oliveira G, Rodriguez-Amaya D (2007) Processed and prepared corn products as sources of lutein and zeaxanthin: compositional variation in the food chain. J Food Sci 72:S79–S85
Dhuique-Mayer C, Tbatou M, Carail M et al (2007) Thermal degradation of antioxidant micronutrients in citrus juice: kinetics and newly formed compounds. J Agric Food Chem 55:4209–4216
Di Silvestro R, Marotti LL, Bosi S et al (2012) Health-promoting phytochemicals of Italian common wheat varieties grown under low-input agricultural management. J Sci Food Agric 92:2800–2810
Digesù AM, Platani C, Cattivelli L et al (2009) Genetic variability in yellow pigment components in cultivated and wild tetraploid wheats. J Cereal Sci 50:210–218
Doblado-Maldonado AF, Pike OA, Sweley JC et al (2012) Key issues and challenges in whole wheat flour milling and storage. J Cereal Sci 56:119–126
Egesel CO, Wong JC, Lambert RJ et al (2003) Combining ability of maize inbreds for carotenoids and tocopherols. Crop Sci 43:818–823
El-Agamey A, McGarvey DJ (2008) Carotenoid radicals and radical ions. In: Britton G, Liaaen-Jensen S, Pfander H (eds) Carotenoids, vol 4: natural functions. Birkhäuser Verlag, Basel, pp 119–154
Eversole K, Feuillet C, Mayer KFX et al (2014) Slicing the wheat genome. Science 18:285–287
Fardet A (2010) New hypotheses for the health-protective mechanisms of whole-grain cereals: what is beyond fibre? Nutr Res Rev 23:65–134
Fardet A, Rock E, Rémésy C (2008) Is the in vitro antioxidant potential of whole-grain cereals and cereal products well reflected in vivo? J Cereal Sci 48:258–276
Fares C, Codianni P, Nigro F et al (2008) Processing and cooking effects on chemical, nutritional and functional properties of pasta obtained from selected emmer genotypes. J Sci Food Agric 88:2435–2444
Farrington FF, Warwick MJ, Shearer G (1981) Changes in the carotenoids and sterol fractions during the prolonged storage of wheat flour. J Sci Food Agric 32:948–950
Fernandez-Orozco R, Gallardo-Guerrero L, Hornero-Méndez D (2013) Carotenoid profiling in tubers of different potato (Solanum sp) cultivars: accumulation of carotenoids mediated by xanthophyll esterification. Food Chem 141:2864–2872
Fish WW, Davis AR (2003) The effects of frozen storage conditions on lycopene stability in watermelon tissue. J Agric Food Chem 51:3582–3585
Fratianni A, Irano M, Panfili G et al (2005) Estimation of color of durum wheat. Comparison of WSB, HPLC, and reflectance colorimeter measurements. J Agric Food Chem 53:2373–2378
Fratianni A, Di Criscio T, Mignogna R et al (2012) Carotenoids, tocols and retinols evolution during egg pasta-making processes. Food Chem 131:590–595
Frei M, Becker K (2005) Fatty acids and all-trans-β-carotene are correlated in differently colored rice landraces. J Sci Food Agric 85:2380–2384
Fu BX, Schlichting L, Pozniak CJ et al (2013) Pigment loss from semolina to dough: rapid measurement and relationship with pasta colour. J Cereal Sci 57:560–566
Goupy P, Hugues M, Boivin P, Amiot MJ (1999) Antioxidant composition and activity of barley (Hordeum vulgare) and malt extracts and of isolated phenolic compounds. J Sci Food Agric 79:1625–1634
Graham RD, Rosser JM (2000) Carotenoids in staple foods: their potential to improve human nutrition. Food Nutr Bull 21:404–409
Grand Challenges in Global Health. www.grandchallenges.org/ImproveNutrition/Challenges/NutrientRichPlants/Pages/Sorghum.aspx. Accessed May 2015
Guzman-Tello R, Cheftel JC (1990) Colour loss during extrusion cooking of β-carotene-wheat flour mixes as an indicator of the intensity of thermal and oxidative processing. Int J Food Sci Technol 25:420–434
Harvestplus. Breeding crops for better nutrition. http://www.harvestplus.org/. Accessed May 2015
Hemery Y, Rouau X, Lullien-Pellerin V et al (2007) Dry processes to develop wheat fractions and products with enhanced nutritional quality. J Cereal Sci 46:327–347
Hentschel V, Kranl K, Hollmann J et al (2002) Spectrophotometric determination of yellow pigment content and evaluation of carotenoids by high-performance liquid chromatography in durum wheat grain. J Agric Food Chem 50:6663–6668
Hernández P, Dorado G, Prieto P et al (2001) A core genetic map of Hordeum chilense and comparisons with maps of barley (Hordeum vulgare) and wheat (Triticum aestivum). Theor Appl Genet 102:1259–1264
Hidalgo A, Brandolini A (2008a) Protein, ash, lutein and tocols distribution in einkorn (Triticum monococcum L. ssp. monococcum) seed fractions. Food Chem 107:444–448
Hidalgo A, Brandolini A (2008b) Kinetics of carotenoids degradation during the storage of einkorn (Triticum monococcum L. ssp. monococcum) and bread wheat (Triticum aestivum L. ssp. aestivum) flours. J Agric Food Chem 56:11300–11305
Hidalgo A, Brandolini A, Pompei C et al (2006) Carotenoids and tocols of einkorn wheat (Triticum monococcum ssp. monococcum L.). J Cereal Sci 44:182–193
Hidalgo A, Brandolini A, Ratti S (2009) Influence of genetic and environmental factors on selected nutritional traits of Triticum monococcum. J Agric Food Chem 57:6342–6348
Hidalgo A, Brandolini A, Pompei C (2010) Carotenoids evolution during pasta, bread and water biscuit preparation from wheat flours. Food Chem 121:746–751
Howitt CA, Pogson BJ (2006) Carotenoid accumulation and function in seeds and non-green tissues. Plant, Cell Environ 29:435–445
Howitt CA, Cavanagh CR, Bowerman AF et al (2009) Alternative splicing, activation of cryptic exons and amino acid substitutions in carotenoid biosynthetic genes are associated with lutein accumulation in wheat endosperm. Funct Integr Genomics 9:363–376
Humphries JM, Graham RD, Mares DJ (2004) Application of reflectance colour measurement to the estimation of carotene and lutein content in wheat and triticale. J Cereal Sci 40:151–159
Ibrahim K, Juvik J (2009) Feasibility for improving phytonutrient content in vegetable crops using conventional breeding strategies: case study with carotenoids and tocopherols in sweet corn and broccoli. J Agric Food Chem 57:4636–4644
ICC (International Association for Cereal Science and Technology) (1990) Method 152. Determination of the yellow pigment content of durum wheat semolina and flour. In: Standard methods of the International Association for Cereal Science and Technology. Verlag Moritz Schäfer, Detmold
Jood S, Kapoor AC (1994) Vitamin contents of cereal grains as affected by storage and insect infestation. Plant Food Hum Nutr 46:237–243
Juliano BO (ed) (1994) Rice: chemistry and technology, 2nd ed. EUA, St. Paul, pp 17–160 and 647–680
Kahlon TS, Chow FI, Hoefer JL et al (1986) Bioavailability of vitamins A and E as influenced by wheat bran and bran particle size. Cereal Chem 63:490–493
Kalt W (2005) Effects of production and processing factors on major fruit and vegetable antioxidants. J Food Sci 70:R11–R19
Kandlakunta B, Rajendran A, Thingnganing L (2008) Carotene content of some common (cereals, pulses, vegetables, spices and condiments) and unconventional sources of plant origin. Food Chem 106:85–89
Kaneko S, Oyanagi A (1995) Varietal differences in the rate of esterification of endosperm lutein during the storage of wheat seeds. Biosci Biotechnol Biochem 59:2312–2313
Kaneko S, Nagamine T, Yamada T (1995) Esterification of endosperm lutein with fatty acids during the storage of wheat seeds. Biosci Biotechnol Biochem 59:1–4
Kean EG, Ejeta G, Hamaker BR et al (2007) Characterization of carotenoid pigments in mature and developing kernels of selected yellow-endosperm sorghum varieties. J Agric Food Chem 55:2619–2626
Kean EG, Hamaker BR, Ferruzzi MG (2008) Carotenoid bioaccessibility from whole grain and degermed maize meal products. J Agric Food Chem 56:9918–9926
Kean EG, Bordenave N, Ejeta G et al (2011) Carotenoid bioaccesibility from whole grain and decorticated yellow endosperm sorghum porridge. J Cereal Sci 54:450–459
Kim JK, Lee SY, Chu SM et al (2010) Variation and correlation analysis of flavonoids and carotenoids in Korean pigmented rice (Oryza sativa L.) cultivars. J Agric Food Chem 58:12804–12809
Koca N, Burdurlu HS, Karadeniz F (2007) Kinetics of color changes in dehydrated carrots. J Food Eng 78:449–455
Konopka I, Kozirok W, Rotkiewicz D (2004) Lipids and carotenoids of wheat grain and flour and attempt of correlating them with digital image analysis of kernel surface and cross-sections. Food Res Int 37:429–438
Kurilich AC, Juvik JA (1999) Quantification of carotenoid and tocopherol antioxidants in Zea mays. J Agric Food Chem 47:1948–1955
Lamberts L, Delcour JA (2008) Carotenoids in raw and parboiled brown and milled rice. J Agric Food Chem 56:11914–11919
Landrum JT, Bone RA (2001) Lutein, zeaxanthin, and the macular pigment. Arch Biochem Biophys 385:28–40
Lavelli V, Hidalgo A, Pompei C et al (2009) Radical scavenging activity of einkorn (Triticum monococcum L. subsp. monococcum) wholemeal flour and its relationship to soluble phenolic and lipophilic antioxidant content. J Cereal Sci 49:319–321
Leenhardt F, Lyan B, Rock E et al (2006a) Genetic variability of carotenoid concentration, and lipoxygenase and peroxidase activities among cultivated wheat species and bread wheat varieties. Eur J Agron 25:170–176
Leenhardt F, Lyan B, Rock E et al (2006b) Wheat lipoxygenase activity induces greater loss of carotenoids than vitamin E during breadmaking. J Agric Food Chem 54:1710–1715
Lemmens L, De Vleeschouwer K, Moelants KRN et al (2010) β-Carotene isomerization kinetics during thermal treatments of carrot puree. J Agric Food Chem 58:6816–6824
Li W, Beta T (2012) An evaluation of carotenoid levels and composition of glabrous canaryseed. Food Chem 133:782–786
Liaaen-Jensen S, Lutnaes BF (2008) E/Z isomers and isomerization. In: Britton G, Liaaen-Jensen S, Pfander H (eds) Carotenoids, vol 4: natural functions. Birkhäuser Velag, Basel, pp 15–36
Lier JB, Lacroix LJ (1974) Carotenoids of durum wheat: induced high pigment levels obtained by treatment of the growing plant with chlorophenylthiotriethylamine (CPTA) hydrochloride. Cereal Chem 51:188–194
Lindley MG (1998) The impact of food processing on antioxidants in vegetable oils, fruits and vegetables. Trends Food Sci Tech 9:336–340
Liu RH (2007) Whole grain phytochemicals and health. J Cereal Sci 46:207–219
Luterotti S, Kljak K (2010) Spectrophotometric estimation of total carotenoids in cereal grain products. Acta Chim Slov 57:781–787
Lv J, Lu Y, Niu Y et al (2013) Effect of genotype, environment, and their interaction on phytochemical compositions and antioxidant properties of soft winter wheat flour. Food Chem 138:454–462
Mace ES, Jordan DR (2011) Integrating sorghum whole genome sequence information with a compendium of sorghum QTL studies reveals uneven distribution of QTL and of gene-rich regions with significant implications for a crop improvement. Theor Appl Genet 123:169–191
Maiani G, Periago-Castón MJ, Catasta G et al (2009) Carotenoids: actual knowledge on food sources, intakes, stability and bioavailability and their protective role in humans. Mol Nutr Food Res 53:S194–S218
Mamatha B, Sangeetha R, Baskaran V (2011) Provitamin-A and xanthophyll carotenoids in vegetables and food grains of nutritional and medicinal importance. Int J Food Sci Tech 46:315–323
Martín A, Chapman V (1977) A hybrid between Hordeum chilense and Triticum aestivum. Cereal Res Commun 5:365–368
Martín A, Sanchez-Monge Laguna E (1982) Citology and morphology of the amphiploid Hordeum chilense × Triticum turgidum conv. durum. Euphytica 31:261–267
Martín A, Álvarez JA, Martín LM et al (1999) The development of tritordeum: a novel cereal for food processing. J Cereal Sci 30:85–95
Marzábal P, Busk PK, Ludevid MD et al (1998) The bifactorial endosperm box of γ-zein gene: characterisation and function of the Pb3 and GZM cis-acting elements. Plant J 16:41–52
Mattera G, Cabrera A, Hornero-Méndez D et al (2015) Lutein esterification in wheat endosperm is controlled by the homoeologous group 7, and is increased by the simultaneous presence of chromosomes 7D and 7Hch from Hordeum chilense. Crop Pasture Sci. doi:10.1071/CP15091
McKevith B (2004) Nutricional aspects of cereals. Nutr Bull 29:111–142
Mellado-Ortega E (2013) Biosynthesis, accumulation and stability of carotenoids in cereals. Comparative study of durum wheat (Triticum turgidum) and tritordeum (Hordeum chilense × T. turgidum conv. durum). PhD Thesis Dissertation, University of Seville, Spain
Mellado-Ortega E, Hornero-Méndez D (2012) Isolation and identification of lutein esters, including their regioisomers, in tritordeum (×Tritordeum Ascherson et Graebner) grains: evidence for a preferential xanthophyll acyltransferase activity. Food Chem 135:1344–1352
Mellado-Ortega E, Hornero-Méndez D (2015) Carotenoid profiling of Hordeum chilense grains: the parental proof for the origin of the high carotenoid content and esterification pattern of tritordeum. J Cereal Sci 62:15–21
Mellado-Ortega E, Atienza SG, Hornero-Méndez D (2015) Carotenoid evolution during postharvest storage of durum wheat (Triticum turgidum conv. durum) and tritordeum (×Tritordeum Ascherson et Graebner) grains. J Cereal Sci 62:134–142
Menkir A, Maziya-Dixon B (2004) Influence of genotype and environment on β-carotene content of tropical yellow-endosperm maize genotypes. Maydica 49:313–318
Mínguez-Mosquera MI, Gandul-Rojas B (1994) Mechanism and kinetics of carotenoid degradation during the processing of green table olives. J Agric Food Chem 42:1551–1554
Mínguez-Mosquera MI, Jarén-Galán M, Gandul-Rojas B, Hornero-Méndez D, Garrido-Fernández J, Gallardo-Guerrero L (eds) (1997) Clorofilas y carotenoides en tecnología de alimentos. Servicio de Publicaciones de la Universidad de Sevilla, Sevilla
Moros EE, Darnoko D, Cheryan M, Perkins EG, Jerrell J (2002) Analysis of xanthophylls in corn by HPLC. J Agric Food Chem 50:5787–5790
Nakornriab M, Sriseadka T, Wongpornchai S (2008) Quantification of carotenoid and flavonoid components in brans of some Thai black rice cultivars using supercritical fluid extraction and high-performance liquid chromatography-mass spectrometry. J Food Lipids 15:488–503
Naqvi S, Zhu C, Farre G et al (2009) Transgenic multivitamin corn through biofortification of endosperm with three vitamins representing three distinct metabolic pathways. Proc Natl Acad Sci USA 106:7762–7767
Ndolo VU, Beta T (2013) Distribution of carotenoids in endosperm, germ, and aleurone fractions of cereal grain kernels. Food Chem 139:663–671
Nghia PT, Liem DT, Hai TV et al (2006) Effect of storage conditions on total carotenoid content in golden rice grains. Omonrice 14:18–27
Nicoli MC, Anese M, Parpinel M (1999) Influence of processing on the antioxidant properties of fruit and vegetables. Trends Food Sci Technol 10:94–100
Nishino H (1997) Cancer prevention by natural carotenoids. J Cell Biochem 27:86–91
O’Kennedy MM, Grootbooma A, Shewry PR (2006) Harnessing sorghum and millet biotechnology for food and health. J Cereal Sci 44:224–235
Okarter N, Liu C, Sorrells M et al (2010) Phytochemical content and antioxidant activity of six diverse varieties of whole wheat. Food Chem 119:249–257
Olson JA (1989) Biological actions of carotenoids. J Nutr 119:94–95
Ouchi A, Aizawa K, Iwasaki Y et al (2010) Kinetic study of the quenching reaction of singlet oxygen by carotenoids and food extracts in solution. Development of a singlet oxygen absorption capacity (SOAC) assay method. J Agric Food Chem 58:9967–9978
Paine JA, Shipton CA, Chaggner S et al (2005) Improving the nutritional value of Golden Rice through increased pro-vitamin A content. Nat Biotechnol 23:482–487
Panfili G, Fratianni A, Distaam M (2004) Improved normal-phase high-performance liquid chromatography procedure for the determination of carotenoids in cereals. J Agric Food Chem 52:6373–6377
Panfili G, Fratianni A, Irano M (2005) Change in tocochromanol and carotenoid content during technological processes of cereals. Tec Molit 5:493–498
Paterson AH, Bowers JE, Bruggmann R et al (2009) The sorghum bicolor genome and the diversification of grasses. Nature 457:551–556
Patil RM, Oak MD, Tamhankar SA et al (2008) Mapping and validation of a major QTL for yellow pigment content on 7AL in durum wheat (Triticum turgidum L. ssp. durum). Mol Breeding 21:485–496
Pinzino C, Nanni B, Zandomeneghi M (1999) Aging, free radicals, and antioxidants in wheat seeds. J Agric Food Chem 47:1333–1339
Pozniak CJ, Knox RE, Clarke FR et al (2007) Identification of QTL and association of a phytoene synthase gene with endosperm colour in durum wheat. Theor Appl Genet 114:525–537
Quackenbush FW (1963) Corn carotenoids: effects of temperature and moisture on losses during storage. Cereal Chem 40:266–269
Quanckenbush F, Firch J, Rabourn W et al (1961) Composition of corn. Analysis of carotenoids in corn grain. J Agric Food Chem 9:132–135
Ravel C, Dardevet M, Leenhardt F et al (2013) Improving the yellow pigment content of bread wheat flour by selecting the three homoeologous copies of Psy1. Mol Breed 31:87–99
Rodriguez-Amaya DB (1997) Carotenoids and food preparation: the retention of provitamin a carotenoids in prepared, processed, and stored foods. Opportunities for Micronutrient Intervention (OMNI), Arlington, USA
Rodríguez-Amaya DB (2003) Food carotenoids: analysis, composition and alterations during storage and processing of foods. Forum Nutr 56:35–37
Rodríguez-Suárez C, Atienza SG (2012) Hordeum chilense genome, a useful tool to investigate the endosperm yellow pigment content in the Triticeae. BMC Plant Biol 12:200
Rodríguez-Suárez C, Giménez MJ, Ramírez MC et al (2011) Exploitation of nuclear and cytoplasm variability in Hordeum chilense for wheat breeding. Plant Genet Resour 9:313–316
Rodríguez-Suárez C, Giménez MJ, Gutiérrez N et al (2012) Development of wild barley (Hordeum chilense)-derived DArT markers and their use into genetic and physical mapping. Theor Appl Genet 124:713–722
Salas-Fernandez MG, Hamblin MT, Li L et al (2008) Quantitative trait loci analysis of endosperm color and carotenoid content in sorghum grain. Crop Sci 48:1732–1743
Santra M, Rao VS, Tamhankar SA (2003) Modification of AACC procedure for measuring β-carotene in early generation durum wheat. Cereal Chem 80:130–131
Santra M, Santra DK, Rao VS et al (2005) Inheritance of β-carotene concentration in durum wheat (Triticum turgidum L. ssp. durum). Euphytica 144:215–221
Saxena A, Maity T, Raju PS et al (2012) Degradation kinetics of colour and total carotenoids in jackfruit (Artocarpus heterophyllus) bulb slices during hot air drying. Food Bioprocess Technol 5:672–679
Scott C, Eldridge A (2005) Comparison of carotenoid content in fresh, frozen and canned corn. J Food Comp Anal 18:551–559
Selim K, Tsimidou M, Biliaderis CG (2000) Kinetic studies of degradation of saffron carotenoids encapsulated in amorphous polymer matrices. Food Chem 71:199–206
Sellappan K, Datta K, Parkhi V et al (2009) Rice caryopsis structure in relation to distribution of micronutrients (iron, zinc, β-carotene) of rice cultivars including transgenic indica rice. Plant Sci 177:557–562
Selman JD (1994) Vitamin retention during blanching of vegetables. Food Chem 49:137–147
Serpen A, Gökmen V, Karagöz A et al (2008) Phytochemical quantification and total antioxidant capacities of Emmer (Triticum dicoccon Schrank) and Einkorn (Triticum monococcum L.) wheat landraces. J Agric Food Chem 56:7285–7292
Sharma SK, Le Maguer M (1996) Kinetics of lycopene degradation in tomato pulp solids under different processing and storage conditions. Food Res Int 29:309–315
Siebenhandl S, Grausgruber H, Pellegrini N et al (2007) Phytochemical profile of main antioxidants in different factions of purple and blue wheat, and black barley. J Agric Food Chem 55:8541–8547
Singh S, Gamlath S, Wakeling L (2007) Nutritional aspects of food extrusion: a review. Int J Food Sci Tech 42:916–929
Suryanarayana Rao K, Rukmini C, Mohan VS (1968) Carotene content of some yellow-endosperm varieties of sorghum. Indian J Agric Sci 38:368–372
Tan J, Baisakh N, Oliva N et al (2005) The screening of rice germplasm, including those transgenic rice lines which accumulate β-carotene in their polished seeds, for their carotenoid profile. Int J Food Sci Technol 40:563–569
Tonon RV, Baroni AF, Hubinger MD (2007) Osmotic dehydration of tomato in ternary solutions: influence of process variables on mass transfer kinetics and an evaluation of the retention of carotenoids. J Food Eng 82:509–517
Trono D, Pastore D, Di-Fonzo N (1999) Carotenoid dependent inhibition of durum wheat lipoxygenase. J Cereal Sci 29:99–102
Tsimidou M (1997) Kinetic studies of saffron (Crocus sativus L.) quality deterioration. J Agric Food Chem 45:2890–2898
Van Hung P, Hatcher DW (2011) Ultra-performance liquid chromatography (UPLC) quantification of carotenoids in durum-wheat: influence of genotype and environment in relation to the colour of yellow alkaline noodles (YAN). Food Chem 125:1510–1516
Velioglu YS, Mazza G, Gao L et al (1998) Antioxidant activity and total phenolics in selected fruits, vegetables, and grain products. J Agric Food Chem 46:4113–4117
Wagner LA, Warthesen JJ (1995) Stability of spray-dried encapsulated carrot carotenes. J Food Sci 60:1048–1053
Weber EJ (1987) Carotenoids and tocols of corn grain determinate by HPLC. J Am Oil Chem Soc 8:1129–1134
Wong JC, Lambert RJ, Wurtzel ET et al (2004) QTL and candidate genes phytoene synthase and ζ-carotene desaturase associated with the accumulation of carotenoids in maize. Theor Appl Genet 108:349–359
Wurtzel ET, Cuttriss A, Vallabhaneni R (2012) Maize provitamin A carotenoids, current resources, and future metabolic engineering challenges. Front Plant Sci 3:29
Ye X, Al-Babili S, Klöti A, Zhang J, Lucca P, Beyer P, Potrykus I (2000) Engineering the provitamin A (beta-carotene) biosynthetic pathway into (carotenoid-free) rice endosperm. Science 287:303–305
Zepka LQ, Borsarelli CD, Azevedo MA et al (2009) Thermal degradation kinetics of carotenoids in a cashew apple juice model and its impact on the system color. J Agric Food Chem 57:7841–7845
Zhang W, Lukaszewski AJ, Kolmer J et al (2005) Molecular characterization of durum and common wheat recombinant lines carrying leaf rust resistence (Lr19) and yellow pigment (Y) genes from Lophopyrum ponticum. Theor Appl Genet 111:573–582
Zhou K, Yu L (2004a) Antioxidant properties of bran extracts from Trego wheat grown at different locations. J Agric Food Chem 52:1112–1117
Zhou K, Yu L (2004b) Effects of extraction solvent on wheat bran antioxidant activity estimation. Lebensm Wiss Technol 37:717–721
Zhou K, Laux JJ, Yu L (2004a) Comparison of swiss red wheat grain fractions for their antioxidant properties. J Agric Food Chem 52:1118–1123
Zhou K, Su L, Yu L (2004b) Phytochemicals and antioxidant properties in wheat bran. J Agric Food Chem 52:6108–6114
Zhu C, Naqvi S, Breitenbach J et al (2008) Combinatorial genetic transformation generates a library of metabolic phenotypes for the carotenoid pathway in maize. Proc Natl Acad Sci USA 105:18232–18237
Žilić S, Serpen A, Akillioğlu G et al (2012) Distribution of phenolic compounds, yellow pigments and oxidative enzymes in wheat grains and their relation to antioxidant capacity of bran and debranned flour. J Cereal Sci 56:562–568
Acknowledgments
This work was supported by funding from the Ministerio de Ciencia e Innovación (Spanish Government, Projects AGL2010-14850/ALI and AGL2014-53195R). EMO was the recipient of a JAE-Predoctoral grant (CSIC) co-financed by the ESF. Authors are members of the IBERCAROT Network, funded by CYTED (ref. 112RT0445).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Mellado-Ortega, E., Hornero-Méndez, D. Carotenoids in cereals: an ancient resource with present and future applications. Phytochem Rev 14, 873–890 (2015). https://doi.org/10.1007/s11101-015-9423-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11101-015-9423-3