Abstract
This paper reports the results of the chemical composition modeling for an atmospheric pressure DC air discharge with water cathode. The modeling was based on the combined solution of Boltzmann equation for electrons, equations of vibrational kinetics for ground states of N2, O2, H2O and NO molecules, equations of chemical kinetics and plasma conductivity equation. Calculations were carried out using experimental values of E/N and gas temperatures for the discharge currents range of 20–50 mA. The effect of H2O concentration on the plasma composition was studied. The main particles of plasma were shown to be O2(a1Δ, b1Σ), O(3P), NO, NO2, HNO3, H2O2 and OH. Effective vibrational temperatures of molecules were higher than gas temperature and they did not depend on the discharge current. Distribution functions on vibrational levels for N2, O2, H2O and NO ground states were non-equilibrium ones.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The large number of published works during the last year concerning the non-equilibrium plasma of atmospheric pressure in contact with solutions emphasizes the interest in this field of plasma physics and plasma chemistry [1]. Already several applications were successfully tested for such kind of plasmas. One of them is the wastewater purification from organic pollutants. To date, many devices were designed for these purposes; e.g. a corona discharge over the surface solution [2], a Contact Glow Discharge Electrolysis [3], a pulsed streamer discharge in solution [4, 5] and under solution [6], dielectric barrier discharge [7], gliding arc [8] and some other ones. In any case, plasma is a source of different kinds of active species. Some of them (e.g. O, ·OH, HO2 ·, ·H,O radicals and H2O2) can be formed in the gas phase followed by their penetration into the solution. Other particles react with water molecules at the gas-liquid interface resulting in non-equilibrium water dissociation under the action of ion-electron bombardment and VUV radiation. For understanding the mechanisms of the processes taking place in liquid it is necessary to know the active species concentrations in a gas phase and their change under variations of discharge parameters. Except for ozone concentration such data are rather limited. Independently on a discharge type the emission spectra show the radiation bands of excited states of N2, OH and NO and lines of atomic H and O at discharge in air or oxygen [1, 9]. But quantitative data are lacking. There are some studies where the OH radical concentrations in ground state were measured in gas phase for the atmospheric pressure DC discharge with aqueous cathode in an ambient air, He, Ar and N2 by a LIF method [10–12] and applying the absorption in UV region for Ar/H2O [13]. For the air it was discovered that OH concentration was about 101−–1016 cm−3 in the current range of 10–30 mA. It is safe to say that measurements of species densities in liquid plasmas are largely an unexplored area. For this reasons the modeling becomes an efficient method of plasma composition study. There are several studies devoted to modeling chemical composition for discharges containing additions of water vapor.
In study [14] a zero-dimension Global model was developed for the description of plasma chemical processes in the helium RF atmospheric pressure discharge with admixture of water molecules. The model considered 46 species and 555 reactions including reactions of excited states of atoms and molecules and positive and negative ions. Authors did not use any experimental data on plasma parameters. The input data for the calculation were electron density and initial composition of plasma-forming gas. The discharge power and therefore reduced field strength, E/N, were determined from the balance equations of charge particles. Gas temperature was assumed to be 300 K. The rate coefficients for processes of electron impact were determined by means of Boltzmann equation solution. Vibrational kinetics for H2O molecules was not taken into consideration.
In study [15] the zero-dimension modeling the chemical composition was carried out for an atmospheric pressure DBD in oxygen containing 2.3 % of water molecules. The species were O, O2, O3, OH, H, H2, H2O and H2O2 described by 37 reactions. The measured wave forms of voltage and discharge current were used for determination of E/N and current values averaged over impulse. The rate coefficients for electron impact and electron drift velocity were calculated from stationary Boltzmann equation taking into account electron collisions with O2 in a ground state only using averaged value of E/N. The electron densities were determined from plasma conductivity using averaged value of discharge current. Reactions of excited states were not considered as well as the vibrational kinetics of molecules.
The plasma chemistry processes were simulated in study [16] for an atmospheric pressure RF discharge (13.56 MHz) in helium containing admixtures of O2 and humid air. The O2 content was 0.5 % and humid air one was 0.05 %. The air composition included 10−2 % of CO2, 10−3 % of N2O, 10−5 % of NO2, and 10−6 % of NO. Water content in air was change in the range of 0–2.3 %. For the simulation authors used 0-D and 1-D Global models. The 0-D model included 1048 reactions and 59 species. The 1-D model included 184 reactions and 20 species. For the calculations of rate constants of electron impact Boltzmann equation was used. Both models shown good egreement.
This paper aims to estimate the active species composition for DC discharge of atmospheric pressure with distilled water cathode burning in an ambient air. As far as we know, plasma chemistry for that system was not analyzed yet.
Description of the Model
The model included Boltzmann equation for electrons, equations of vibration kinetics, chemical kinetics equations, and equation of plasma conductivity. The last equation was used for determination of electron density on the base of measured current density since the exact ionization mechanism is unknown.
The electron energy distribution function (EEDF) was obtained from the solution of the homogeneous Boltzmann equation using two term expansion in spherical harmonics. Collision integrals concerning charge particle formation were written as for usual inelastic processes. This is possible because the electron energy losses in these processes are negligible (less then 0.01 %) compared to the losses for other inelastic processes. It means that Boltzmann equation can be independently solved using charge balance equations. Collision integrals include the collisions of electrons with N2, O2, H2O, NO molecules in electronic ground state and with O(3P) atoms. The collisions of second kind with vibrationally excited molecules were taken into consideration as well. The cross-section sets for N2, O2, H2O, NO molecules and O(3P) atoms were taken from studies [17–21], respectively. Mathematical details of solution were described by us elsewhere [22, 23].
To determine the population of vibrational levels of N2, O2, H2O, NO molecules in ground state the equations system of quasi- stationary kinetics was solved. This system takes into consideration the single-quantum V–V, V–T exchange, e–v pumping and some other ones. These processes are listed in Table 1.
H2O molecule was described as three anharmonic oscillators—(100, 010, 001). The temperature dependencies of V–T rate constants K 01(T) for N2, O2, H2O, NO molecules were taken from studies [24–31] and V–V rate constants K 1001 (T) were taken from works [26, 28, 30–33]. The level rate constants were calculated using SSH generalized theory [24]. We did not use any approximations for the translational factor and the constant of V–T exchange for n → n−1 transition was calculated as follows:
where X e is dimensionless anharmonicity constant and Z tr is translational factor which was numerically calculated as
In this formula T g is gas temperature, \(\theta_{LT} = \frac{{\pi^{2} m_{AB} \omega_{0}^{2} }}{{2\alpha^{2} k}}\) is characteristic temperature for the first level, \(\theta_{LT}^{n} = \frac{{\pi^{2} m_{AB} \omega_{n}^{2} }}{{2\alpha^{2} k}}\)—characteristic temperature for the nth-level, \(\theta_{V} = \frac{{\hbar \omega_{1}^{AB} }}{k}\) is characteristic vibrational temperature for the first level, \(\theta_{V}^{n} = \frac{{\hbar \omega_{n}^{AB} }}{k}\)—characteristic vibrational temperature for the nth-level, k is the Boltzmann constant, m AB is reduced mass of the colliding particles, α is reverse radius in exponential repulsive potential of interaction, ω n is frequency for n → n−1 transition. The level V–V rate constants were calculated by the similar expressions [24].
The molecular parameters were taken from [34, 35] and α values are given in [36]. For the collision of H2O molecules with other molecules α values are unknown. For this reason they were estimated by the expression given in study [24]:
where I AB and I m are ionization potentials of colliding particles.
The equations of chemical kinetics included the set of reactions which are listed in the Table 2.
The two-body reactions were chosen on the basis of reaction sets proposed in studies [22, 93, 94] for modeling reaction mechanisms in a plasma of oxygen and air. These sets described the experimental data quite well for DC discharges. Three-body reactions were chosen according to study [53, 63]. Of course, we carried out the preliminary estimation of particles life times with respect to different reactions and some reactions were rejected. Specifically, we did not take into consideration the numerous reactions with ions of charge exchange and recombination types since the rates of such reactions are limited by the ionization rates. Under experimental values of E/N (18–28 Td) the ionization rates are less than rates of excitation and dissociation more than several orders of magnitude. For example, at E/N = 28 Td and water content of 0.05 % the ionization rate constant of O2(X) with electron impact is 9.1 × 10−19 cm3/s, the dissociation rate constant is 8.7 × 10−14 cm3/s, the rate constant of O2(a1Δ) excitation is 3.1 × 10−11 cm3/s. The ionization rate constants for N2 and H20 equal to 1.1 × 10−21 and 4.5 × 10−19 cm3/s, respectively. The molecules and atoms under study do not have metastable states able to provide ionization at collisions with ground states of particles. Thus, ion-molecular reactions did not practically influence on the concentrations of neutral species. Of course, the models proposed in studies [14, 16] include more reactions. But about more than half of them are ion-molecular reactions. These reactions necessary to take into account if the E/N values are unknown because E/N (or average electron energy, Te, for Global model) is determined from the equations of charge balance. But in our case the E/N was taken from experiment. Also, these reactions are important if we want to known the ion composition of plasma. But it was not the aim of given study.
Also, the reaction of H2O2 thermal dissociation was not included in reactions scheme. As it will be shown below the distribution of ground states of molecules on vibrational levels is not equilibrium. For this reason it is impossible to use the equilibrium dissociation rate constant and the correct determination of dissociation rate through dissociation continuum requires the solution of appropriate vibrational task. Unfortunately, we do not have appropriate data to carry out such calculation. That is why we assume that calculated concentrations of H2O2 can be overestimated.
Another disadvantage of our model is that we do not take into account the discharge heterogeneity. To consider the heterogeneity it is necessary to solve the continuity equations system for every component with appropriate boundary conditions and equation of heat conductivity. Unlike classical discharges which have holding walls this discharge does not have one. Therefore, in radial direction the boundary is infinity where the component concentration has to be zero. Additionally, it is necessary to have the diffusion coefficients for neutrals and mobility for ions which are unknown for such complex system. It is clear that the solution of this task is impossible in a reasonable time. Though, we suppose that zero-dimension model will be useful for estimation of concentrations of main chemically-active species. At least, the results obtained in study [16] at 1-D and 0-D simulations were very close. Some results on estimations of radial distribution influence of plasma parameters on the results of calculations will be shown below.
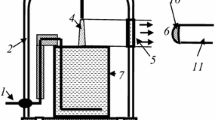
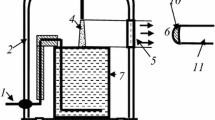
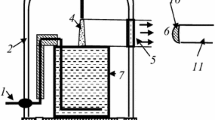
Modeling was carried out using experimental data obtained in our previous study [23]. In this work, the averaged values of gas temperatures, vibrational temperatures of N2(X), reduced electric field strengths (E/N) and diameters of plasma zone were obtained for the discharge current range of 20–50 mA. It was shown that the gas temperature (1,420 ± 90 K) and vibrational temperature (4,200 ± 100 K) did not depend on discharge current whereas E/N dropped from 28 up to 18 Td under the discharge current increase from 20 up to 50 mA. The diameter of positive column was linearly increased from 1.5 up to 2.5 mm in the same current range. Because the diameter was measured optically the real diameter can be a little more. Therefore, the electron density which was determined on conductivity can be somewhat overestimated. The solution volume (cathode) was 80 mL.
The system of equations was solved as follows:
-
1.
The Boltzmann equation was solved for a given E/N, gas temperature, discharge current density, initial composition of mixture (ground states of O2,N2 and H2O; O2:N2 ratio corresponds to air). Rate coefficients for the processes of electron impact as well as electron density were obtained.
-
2.
Using these values the equations of vibrational kinetics were solved. The relative accuracy of calculation was 1 %. Vibrational level distributions were determined.
-
3.
The composition of plasma was found by means of quasi- stationary equations of chemical kinetics which were solved iteratively. The relative accuracy of every concentration calculation was 1 %.
-
4.
Calculated concentrations and vibrational distributions we returned to point 1 and so on. The computational procedure was terminated when the particle concentrations change did not exceed the error limit of 1 %.
The main problem at the modeling was the lack of data on water molecules concentration in a gas phase. For this reason we used them as a given parameter.
Results and Discussion
The calculations showed that the best agreement between experiment and calculation for vibrational temperatures of N2(X) is achieved at the water content of 0.05 % (Fig. 1). The vibrational distributions are not equilibrium ones (Fig. 2).The vibrational temperatures for H2O, O2(X) and NO(X) determined on the population of first two levels are represented in Fig. 3. Vibrational temperatures for O2(X) and NO(X) are close to the gas ones (1,420 K) due to the fast V–T relaxation rate of these molecules on O(3P) atomic oxygen in comparison with V–V relaxation rates. The slight dependence of vibrational temperatures on the discharge current is related with slight dependence of excitation-de-excitation frequencies by electron impact for the first vibrational levels on the discharge current as well as with slight dependence of O(3P) concentration on discharge current (Fig. 4). The increase in a discharge current results in the increase in the electron densities, Ne, (Fig. 5). At the same time the rate constants, Kex, of electron impact are decreased with discharge current due to the dropping of E/N. As a result, the (Ne × Kex) product changes slightly.
Electron density was determined from the relation Ne = J/(e × V D ), where J is discharge current density, V D is electron drift velocity, e is electron charge. The dependence of electron density on the discharge current is related with dependences of electron drift velocity and diameter of positive column on discharge current. Electron drift velocity drops with the increase in a discharge current due to the decrease in E/N whereas diameter is increased. In spite of the increase in a discharge current the discharge current density drops. The change velocity of J and V D is different. As a result of common action of these two factors the specific dependence of electron density on discharge current is observed.
Considerable quantity of nitrogen oxides with various oxidation levels are formed in such plasma (Figs. 6, 7). Molecules of nitric acid (HNO3), nitrous acid (HNO2) and nitroxyl (HNO) are formed as well (Fig. 8). Nitrogen oxide (II) is dissolved poorly in water as well as N2O. But nitrogen oxide (IV) reacts with water fast forming nitric and nitrous acids: 2NO2 + H2O → HNO3 + HNO2. HNO3 is indefinitely dissolved in water. Therefore, it can be expected that the discharge action has to be accompanied by the appearance in a water of nitric acid and by the increase in solution acidity (the decrease in pH). Nitric acid is a strong oxidizer and it reacts easily with organics. The large NO concentration (order of magnitude higher than other particles) and low excitation threshold (~5.7 eV) has to result in the high emission intensity in short-wave UV region of spectra (200–271 nm) due to a radiation of γ-system of NO molecules (A2Σ → X2Π transition) as it was really observed in [23, 95].
The main oxygen-hydrogen particles were •OH, HO •2 radicals and hydrogen peroxide. Their concentrations are shown in Figs. 9 and 10. The orders of magnitude of •OH radical concentrations are close to those which were experimentally observed in study [10, 11] (1015–1016 cm−3). Although it is not so correct we added into reactions scheme the reaction of dissociation of H2O2 with equilibrium rate constant of 3.00 × 1014 × exp(−2,4430/T g )s−1 [51]. The result showed that H2O2 concentration could be overestimated by a factor of 1.5.
The main oxygen-containing species were atomic oxygen O(3P) and two lower metastable states of molecular oxygen O2(a1Δg) and O2(b1Σ +g ) (Fig. 11). Unlike DBD of atmospheric pressure, the concentration of ozone was very low whereas this of O(3P) was higher [96]. Thus, for DC discharge the O(3P) concentration is about 1015 cm−3 and for DBD-~1011 cm−3. For DC discharge the O3 concentration is about 1011 cm−3 and for DBD-~1016 cm−3. This is due to the change in the mechanism of ozone loss. For both discharges the main reaction of ozone formation is O(3P) + O2(X) + O2(X) → O3 + O2(X). For DBD the main reaction of ozone destruction is O(3P) + O3 → O2(X) + O2(X). In this reaction O(3P) atoms are not formed. For this reason, O(3P) atoms are transformed to ozone. For DC discharge the reaction O3 + O2(X) → O(3P) + 2O2(X) becomes the dominant due to the difference in gas temperature (~300 K for DBD and ~1,450 K for DC) leading to sharp increase in a rate constant of the reaction mentioned above. In this reaction O(3P) atoms appear again. Therefore, under the application of DC discharge for water processing it can be neglected with the ozone action on oxidation processes in a solution.
To check the influence of radial heterogeneity on the calculation results we acted by the following way.
At conditions under study the EEDF characteristic time of relaxation τ κ can be estimated as follows
where \(\bar{\varepsilon } \approx 1\) eV—electron average energy, m e —electron mass, σ m ≈ 10−15 cm2—momentum transfer cross section for electron collision with O2–N2 molecule, N = 4 × 1018cm−3—total particle concentration, \(\delta = (1.2 \times V_{D} /\bar{V})\)—average energy fraction losing with electron per one collision, \(\bar{V} = \sqrt {2\bar{\varepsilon } \times e/m_{e} } \approx 6 \times \,10^{7}\) cm/c—average electron velocity, V D ≈ 3 × 106cm/s—electron drift velocity.
The calculation gives the δ ≈ 0.06, τ r ≈ 6.9 × 10−11 s and \(\bar{V} \gg V_{D}\).
The EEDF characteristic length of relaxation λ is determined as
The calculation gives the λ = 2 × 10−4cm. Comparison this value with the radius of discharge (R ≈ 0.1 cm) shows that the EEDF is determined with the local value of E/N.
Typical values of diffusion coefficients, D, of neutral particles under the experimental conditions are ~6 cm2/s. It gives the characteristic diffusion time τ D = R 2/D ≈ 2 × 10−3s. The characteristic times of life of different species with respect to volume reactions are essentially shorter. Therefore, there is local balance of formation and loss processes.
For the temperature profile we used the approximation
where T 0 is the temperature at discharge axis, T R = 300 K is the temperature at the discharge boundary.
This expression gives the average over discharge cross-section temperature which is measured as
Using this expression the temperature profile was calculated and on its base the E/N profile was calculated as well. For the electron density profile the following expression was applied
where N 0 e is electron density at discharge axis.
Then the model was solved for every point of discharge radius. Obtained results were averaged on discharge cross-section. Control computation showed that results of those calculations differed from calculation on 0-D model slightly. Thus, for the discharge current of 20 mA and water content of 0.2 % the concentration of O2(a1Δ) was 1.8 × 1014 cm−3 (1.7 × 1014 cm−3 on 0-D model), OH was 9 × 1014 cm−3 (1.3 × 1015 cm−3 on 0-D model), NO was 4.8 × 1016 cm−3 (4 × 1016 cm−3 on 0-D model).
The data obtained allow estimating the maximal rates which can be provided for the particles being formed in discharge under their penetration in a solution as follows. Flux particle density is equal to J = N × V T /4, where N is particle concentration, \(V_{T} = \sqrt {\frac{{8 \times k_{b} \times T_{g} }}{\pi \times m}}\) is thermal velocity and m is the particle mass. If all particles reaching the solution surface react into solution completely then average rate on volume can be expressed as \(\bar{W} = J \times S_{C} /V_{S}\), where S c is square of solution contact with discharge and Vs = 80 cm−3 is solution volume. The calculation gave the values of 1016, 6 × 1015 cm−3s−1 for •OH and H2O2, respectively for discharge current of 40 mA and water content of 0.05 %. These results can be compared with the data which were obtained in study [97] where the concentration of H2O2 was measured in a solution for the same set-up and for the same discharge conditions and H2O2 formation-loss was modeled. Results of the modeling showed that formation rates of H2O2 and •OH were more than ~1017 cm−3s−1. Therefore, the particles flux from plasma cannot provide the observed formation rates in liquid. And the main source of H2O2 and •OH should be the reactions of water molecules dissociation which are initiated under bombardment of liquid-gas interface by positive ions accelerated in a cathode voltage drop as it was implicitly postulated, for example, in study [4].
Concentration dependences on discharge current are defined by the two main reasons: results in the growth of electron densities (1) (Fig. 5) and in the decrease in E/N (2) taking place under the increase in discharge current. The first factor promotes to a growth of excitation frequency by electron impact. The second leads to the decrease in rate constants of electron impact. The degree of the constants decrease depends on the process threshold energy. For processes with the low threshold energy the increase in rate constants is slight and the first factor is the predominate one. For this reason the concentration of O2(a1Δg) (threshold is 0.98 eV for electron impact) is increased with discharge current while the O2(b1Σ +g ) concentration (threshold is 1.63 eV) barely depends on discharge current.
Of course, not all reactions in Table 2 are important for forming the composition of plasma. The accomplished calculations allow revealing the main reactions by comparing the rates of formation and loss. The knowledge of formation rates presents a separate interest since they determine the top possibilities of plasma with respect to generation of given kind of particles. The results of such analysis are listed in Table 3.
It is interesting to point out that the main processes of •OH formation are not the dissociation of H2O by electron impact but the following reactions: O(3P) + H2O → OH· + OH˙ and NO + HO ·2 → NO2 + ·OH.
The low values of obtained concentrations show that it is possible to solve the Boltzmann equation independently on the equations of chemical kinetics that take in account the electron collisions with N2(X) O2(X) and H2O molecules only. Nevertheless, super-elastic collisions of electrons with N2 vibrational excited molecules are also important.
Conclusions
On the basis of experimental data on parameters of an air atmospheric pressure DC discharge with water cathode the modeling chemical composition of plasma was carried out at the discharge current range of 20–50 mA. The main particles being formed in plasma were shown to be NO, HNO3, HNO, •OH, H2O2, O(3P) and O2(a1Δg). The dependencies of these particles concentrations on the discharge current are defined with the change in an electron concentration and in an EEDF. The specific feature of discharge is low concentration of ozone molecules and high vibrational temperature of N2 ground state. The comparison of estimated fluxes of active species from plasma onto gas-solution interface with available data on liquid phase allows to suppose that •OH, H2O2 molecules are formed as a result of ion bombardment of solution surface. High concentrations of HNO3 molecules in plasma have to lead to the formation of nitric acid solution and to the decrease in solution pH.
References
Bruggeman P, Leys C (2009) J Phys D Appl Phys 42(5):053001 28 pp
Grabowski LR, Van Veldhuizen EM, Pemen AJM, Rutgers WR (2006) Plasma Chem Plasma Process 26(1):3–17
Tomisawa S, Tesuka M (2007) Plasma Chem Plasma Process 27(4):486–495
Grymonpre DR, Sharma AK, Finney WC, Locke BR (2001) Chem Eng J 82(1–3):189–207
Malik MA, Schoenbach KH (2012) J Phys D Appl Phys 45(13):132001 5 pp
Lukes P, Locke BR (2005) J Phys D Appl Phys 38(22):4074–4081
Bobkova ES, Ivantsova NA, Grinevich VI, Rybkin VV (2012) Plasma Chem Plasma Process 32(1):97–107
Njoyin E, Ghogomu P, Laminsi S, Nzali S, Doubla A, Brisset JL (2009) Ind Eng Chem Res 48(22):9773–9780
Titov VA, Rybkin VV, Maximov AI, Choi H-S (2005) Plasma Chem Plasma Process 25(5):502–518
Li L, Nikiforov A, Xiong Q, Lu X, Taghizadeh L, Leys C (2012) J Phys D Appl Phys 45(12):125201
Nikiforov A, Li L, Xiong Q, Leys C, Lu XP (2011) Eur Phys J Appl Phys 56(2):4009–23013
Nikiforov A, Xiong Q, Britun N, Snyders R, Lu X, Leys C (2011) Proceedings of VI International Symposium on Theoretical and Applied Plasma Chemistry September 3–9 Ivanovo Russia ISUCT, pp 318–321
Ito H, Kano H (2008) Appl Phys Express 1:10601–10603
Liu DX, Bruggeman P, Iza F, Rong MZ, Kong MC (2010) Plasma Sources Sci Technol 19(2):025018 11 pp
Matsui Y, Takeushi N, Sasaki K, Hayashi R, Yasuoka K (2011) Plasma Sources Sci Technol 20(3):034015 11 pp
Mirakami T, Niemi K, Gans T, O’Connel D, Graham WG (2013) Plasma Sources Sci Technol 22(1):01503 29 pp
Gordiets BF, Ferreira CM, Guerra VL, Loureiro J, Nahorny J, Pagnon D, Touzeau Vialle M (1995) IEEE Trans Plasma Sci 23(23):750–768
Kajita S, Ushiroda S, Kondo Y (1990) J Phys D Appl Phys 67(9):4015–4023
Rybkin VV, Titov VA, Kholodkov IV (2008) Izv Vyssh Uchebn Zaved, Khim Khim Tekhnol 51(3):3–10 (in Russian)
Rybkin VV, Titov VA, Kholodkov IV (2009) Izv Vyssh Uchebn Zaved, Khim Khim Tekhnol 52(12):3–10 (in Russian)
Laher RR, Gilmore FR (1990) J Phys Chem Ref Data 19(1):277–304
Diamy A-M, Legrand J-C, Smirnov SA, Rybkin VV (2005) Contr Plasma Phys 45(1):5–21
Titov VA, Rybkin VV, Smirnov SA, Kulentsan AN, Choi H-S (2006) Plasma Chem Plasma Process 26(6):543–555
Chernyiy GG, Losev SA (eds) (1995) Physical-Chemical processes in gas dynamics. V. 1. Dynamics of physical-chemical processes in gas and plasma. MSU, Moscow (in Russian)
Nikitin EE, Osipov AI, Umanskiy SYa (1989) Translation-vibrational exchange under collision of homonuclear diatomic molecules. In: Smirnov BM (ed) Plasma chemistry. Atomizdat, Moscow (in Russian)
Capitelli M (ed) (1986) Nonequilibrium vibrational kinetics. Springer, Berlin
Polak LS, Slovetskiy DI, Urbas AD, Fedoseeva TV (1978) Relaxation measurements and excitation mechanisms of electron-vibrational levels of molecules in a glow discharge in nitrogen. In: Smirnov BM (ed) Plasma chemistry. Atomizdat, Moscow (in Russian)
Kiefer JH (1972) J Chem Phys 57(5):938–1956
Fortov VE (2005) Encyclopedia of low temperature plasma. Yanus, Moscow (in Russian)
Mnatsakanyan AK, Naidis GV (1985) High Temp (Teplofyzika Vysokikh Temperatur) 23(4):640–646 (in Russian)
Salnikov VA, Starik AM (1995) High Temp (Teplofyzika Vysokikh Temperatur) 33(1):121–133 (in Russian)
Wysong IJ (1994) J Chem Phys 101(4):2800–2810
Frost MJ, Islam M, Smith IWM (1994) Can J Chem 72(3):606–611
Huber KP, Herzberg G (1979) Molecular spectra and molecular structure. IV. Constants of diatomic molecules. Litton Educational Publishing, Inc, NY
Glushko VP (ed) (1978) Thermodynamic properties of individual substances. Handbook. Nauka, Moscow (in Russian)
Radtsig AA, Smirnov BM (1980) Handbook on atomic and molecular physics. Atomizdat, Moscow (in Russian)
Eliasson B, Kogelschatz U (1986) Basic data for modeling of electrical discharges in gases: oxygen. Brown Boveri Research Report. KLR86-11C
Fehsenfeld FC, Albritton DL, Burt JA (1969) Can J Chem 47(10):1793–1795
Fehsenfeld FC, Ferguson EE, Schmeltekopf AL (1966) J Chem Phys 45(5):1844–1845
Kenner RD, Ogryzlo EA (1980) Int J Chem Kinet 12(7):502–508
Slanger TG, Black G (1979) J Chem Phys 70(7):3434–3443
Dvoryankin AN, Ibragimov LB, Kulagin YuA, Shelepin LA (1987) Mechanisms of electron relaxation in atomic-molecular media. In: Smirnov BM (ed) Plasma chemistry. Moscow, Energoatomizdat (in Russian)
Zinn J, Sutherland CD, Stone SN, Dunkan LM (1982) J Atmos Terr Phys 44(12):1143–1171
Young RA, Black G (1967) J Chem Phys 47(7):2311–2318
Smirnov BM (1982) Excited atoms. Energoizdat, Moscow (in Russian)
Slanger TG, Black G (1981) J Chem Phys 75(5):2247–2251
Slanger TG, Black G (1976) J Chem Phys 64(9):3763–3766
Atkinson R, Welge KH (1972) J Chem Phys 57(9):3689–3693
Ivanov VV, Klopovskii KS, Lopaev DV, Rakhimov AT, Rakhimova TV (2000) Plasma Phys Rep 26(11):980–990
Moore CE (1976) Selected tables of atomic spectra, atomic energy levels and multiplet tables—OI. Nat. Bur. Stand. US
Baulch DL, Cobos CJ, Cox RA, Esser C, Franec P, Just Th, Kerr JA, Pilling MJ, Troe J, Walker RN, Warnatz J (1992) J Phys Chem Ref Data 21(3):411–429
Peyrous R, Rignolet P, Held BJ (1989) J Appl Phys D Appl Phys 22(11):1658–1667
Chirokov A (2003) Self-organization of Microdischarges in DBD plasma. Master Thesis Dreaxel University
Tochikubo F, Ushida S, Watanabe T (2004) Jpn J Appl Phys 43(1):315–320
Smedt FD, Bui XV, Nguyen TL, Peeters J, Vereecken L (2005) J Phys Chem 109(1):2401–2409
Pontiga F, Soria C, Casrellanos A, Skalny JD (2002) Ozone Sci Eng 24(6):447–462
Baulch DL, Cox RA, Hampson RF, Kerr JA, Troe J (1980) J Phys Chem Ref Data 9(2):295–472
Filseth S, Stuhl F, Welge KH (1970) J Chem Phys 52(1):239–243
Prasad AN, Graggs JD (1965) Int J Electron 19(1):69–74
Aberth WH, Peterson JR (1960) Phys Rev 1(1):158–165
Olson RE, Peterson JR (1970) J Chem Phys 53(9):3351–3397
Akishev YuS, Deryugin AA, Karal’nik VB (1994) Plasma Phys Rep (Phyzika Plasmy) 20(6):571–584 (in Russian)
Kossyi IA, Kostinskiy IA, Matveev AA, Silakov VP (1994) Plasma chemical processes in non-equilibrium nitrogen-oxygen mixture. Proc Inst Gen Phys RAS 47:37–57 (in Russian)
Alexandrov NL (1978) Tech Phys (Zhurnal Tekhnicheskoi Fiziki) 48(7):1428–1431 (in Russian)
Kozlov SI, Vlaskov VA, Smirnova NV (1988) Space Res 26(5):738–745 (in Russian)
Smith K, Tomson R (1981) Numerical modeling gas lasers. Mir, Moscow (in Russian)
Krivonosova OE, Losev SA, Nalivaiyko VP (1987) Advisable data on rate constants of chemical reactions between molecules consisting from N and O atoms. In: Smirnov BM (ed) Plasma chemistry. Moscow, Energoatomizdat (in Russian)
Jannuzzi MP, Jeffries JB, Kaufman F (1982) Chem Phys Lett 87(6):570–574
Piper LG, Caledonia GE, Kennelaly JP (1981) J Chem Phys 75(6):2847–2853
Silakov VP (1990) Mechanism of supporting the long-lived plasma in molecular nitrogen at high pressure. Preprint of Moscow Engineering Physical Institute N 010-90 M (in Russian)
Slovetskiy DI (1980) Mechanisms of chemical reactions in non-equilibrium plasma. Mir, Moscow (in Russian)
Piper LG (1982) J Chem Phys 77(5):2373–2377
Young RA, Black G, Slanger TG (1970) J Chem Phys 51(1):116–121
Piper LG (1987) J Chem Phys 87(3):1625–1629
Yaron M, Von Engel A, Vidaud PH (1976) Chem Phys Lett 37(1):159–161
Didyukov AI, Kulagin YuA, Shelepin LA, Yarygina VN (1989) Quantum Electroniks 16(5):892–904 (in Russian)
O’Brien RJ, Myers GH (1970) J Chem Phys 53(10):3832–3835
Yau AW, Shepherd GG (1979) Planet Space Sci 27(4):481–490
Husain D, Mitra SK, Young AN (1974) J Chem Soc Faradey Trans Part II 70(10):1721–1731
Delcroix JL, Ferreira CV, Ricard F (1973) Proceedings of the XI International Conference on Phenomena in Ionized Gases: Invited papers. Prague. 301
Baulch DL, Cox RA, Crutzen PJ (1982) J Phys Chem Ref Data 11(2):327–496
Dmitrieva IK, Zenevich VA (1984) Russian J Phys Chem B Focus Phys (Zhurnal Khimicheskoi Fiziki) 3(8):1075–1080 (in Russian)
Pancheshnyi SV, Starikovskaia SM, Starikovskii AYu (1998) Chem Phys Lett 294(6):523–527
DeMore WB, Sander SP, Golden DM, Hampson RF, Kurylo MJ, Howard CJ, Ravishankara AR, Kolb CE, Molina MJ (1997) Chemical kinetics and photochemical data for use in stratospheric modeling. Evaluation number 12. JPL Publication 97–4
Tsang W, Herron JT (1991) J Phys Chem Ref Data 20(4):609–663
Atkinson R, Baulch DL, Cox RA, Hampson RF Jr, Kerr JA, Troe J (1992) J Phys Chem Ref Data 21(6):1125–1568
Hipples H, Krasteva N, Nasterlack S, Striebel F (1999) J Chem Phys A 110(14):6781–6788
Gauthier MJE, Snelling DR (1975) J Photochem 4(1):27–50
Smirnov BM (1978) Negative ions. Atomizdat, Moscow (in Russian)
Su M-C, Kumaran SS, Lim KP, Michael JV, Wagner AF, Hardling LB, Fang D-C (2002) J Phys Chem A 106(36):8261–8270
Smith CA, Molina LT, Lamb JJ, Molina MJ (1984) Int J Chem Kinet 16(1):41–55
Bates DR, Moffett RJ (1966) Proc R Soc Lond A 291(1424):1–8
Gordiets B, Ferreira CM, Nahorny J, Pagnon D, Touzeau M, Vialle M (1996) J Phys D Appl Phys 29(4):1021–1031
Smirnov SA, Rybkin VV, Kholodkov IV, Titov VA (2002) High Temp 40(3):323–330
Staack D, Farouk B, Gursol A, Fridman A (2005) Plasma Sources Sci Tenchnol 14(4):700–711
Eliasson B, Kogelschatz U (1991) IEEE Trans Plasma Sci 19(2):309–323
Bobkova ES, Shikova TG, Grinevich VI, Rybkin VV (2012) High Energy Chem 46(1):56–59
Acknowledgments
This study was supported by the RFBR Grant, Project Number 14-02-01113 A.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bobkova, E.S., Smirnov, S.A., Zalipaeva, Y.V. et al. Modeling Chemical Composition for an Atmospheric Pressure DC Discharge in Air with Water Cathode by 0-D model. Plasma Chem Plasma Process 34, 721–743 (2014). https://doi.org/10.1007/s11090-014-9539-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11090-014-9539-z