Abstract
Sophora davidii is an important leguminous scrub that is widely used for revegetation in the semiarid Loess Plateau and other arid valley areas of China, where it usually suffers drought stress. This study investigated the effects of arbuscular mycorrhizal (AM) fungi (Glomus mosseae and Glomus constrictum) and water stress on the growth and physiological performance of S. davidii seedlings under greenhouse pot conditions. Two soil water availability treatments (well-watered (WW) −0.10 MPa; water-stressed (WS) −0.86 MPa) were applied for 61 days. At the end of this experiment, G. mosseae and G. constrictum had colonized the roots of S. davidii seedlings. Water stress inhibited AM colonization, plant growth, chlorophyll concentration, gas exchange and chlorophyll fluorescence of S. davidii seedlings. Mycorrhizal seedlings had greater shoot dry weight, root dry weight, plant height, root length, instantaneous water use efficiency (iWUE), net photosynthetic rate (Pn), stomatal conductance (gs), maximal photochemical efficiency of PSII photochemistry (Fv/Fm), lower intercellular CO2 concentration and photochemical quenching values (qP), when compared with non-mycorrhizal seedlings under both WW and WS conditions. Furthermore, G. constrictum was found to be more efficient at improving the shoot and root mass, plant height, iWUE, Pn, gs, qP, and ΦPSII of S. davidii seedlings, when compared with G. mosseae under both WW and WS conditions. Our results demonstrate that AM Glomus symbiosis enhanced S. davidii seedling resistance by improving its growth and physiological performance under water stress conditions. This suggests that Glomus inoculation is a potential tool for enhancing outplanting performance of S. davidii in semiarid areas of China.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Drought is a major environmental problem in arid and semi-arid areas, and it is expected to increase in the near future as a consequence of anthropogenic disturbance and global climate change (Goicoechea et al. 2005; Hura et al. 2007). When these areas are subjected to water deficiency, soil fertility and structure can also be degraded, which limits the natural revegetation and makes the ecosystem more fragile and susceptible to further disturbances (Requena et al. 1997). Planting drought-tolerant legumes is a possible solution for the revegetation and the improvement of soil fertility in these areas, since they can provide nitrogen (N) to nutrient-deficient soils (Caravaca et al. 2003; Requena et al. 1997, 2001). Besides N fixation through symbiosis with bacteria, leguminous species are also associated with arbuscular mycorrhizal (AM) fungi (Marques et al. 2001).
AM fungi are found in almost all terrestrial ecosystems, where they form symbiotic relationships with the roots of approximately 80 % of all plant species (Smith and Read 1997). Plant-AM symbiosis can improve the performance of host plants (Aggangan et al. 2010; Bethlenfalvay et al. 1990; Goicoechea et al. 2005; Muthukumar and Udaiyan 2010; Ruiz-Lozano et al. 1995a, b). AM fungi can also alleviate water stress through a combination of physical, physiological and cellular effects on plants (Allen and Boosalis 1983; Ruiz-Lozano 2003; Requena et al. 1997, 2001; Wu et al. 2008b).
In recent years, the inoculation of leguminous plants with AM fungi before forestation under drought conditions has received considerable attentions (Caravaca et al. 2003; Marques et al. 2001; Requena et al. 1997, 2001). For example, Glomus coronatum and Glomus intraradices help Anthyllis cytisoides to establish and thrive in Mediterranean ecosystems by increasing growth and tissue N and P content (Requena et al. 1997).
Sophora davidii is a leguminous species native to East Asian temperate regions. It has been extensively planted in semiarid hill and gully areas of the Loess Plateau and other arid valley areas in China (Wu et al. 2008a). Plantations of S. davidii are used for windbreaks, sand fixation, water and soil conservation, and reclamation of barren land (Wu et al. 2008a). This small shrub has a high tolerance to drought, because it has a deep root system, small leaf area, and high leaf soluble sugars and K+ concentration (Li et al. 2009). S. davidii is well colonized by AM fungi, but there are few studies concerning the effects of AM fungi on the growth and physiological performance of S. davidii seedlings, especially under drought conditions (Feng et al. 2011).
This study compared the growth, water status, gas-exchange, and chlorophyll concentration and fluorescense of non-mycorrhizal and Glomus inoculated S. davidii seedlings under well-watered and water-stressed conditions. The objective of this study was to assess whether AM Glomus inoculation and water stress affected the growth and physiological performance of S. davidii seedlings. Furthermore, we aimed to determine if Glomus inoculation improves the drought tolerance of S. davidii seedlings under greenhouse pot conditions.
Materials and methods
Experimental design
Treatments were a factorial combination of two factors: (1) mycorrhizal inoculation: i.e., Glomus mosseae (Nicol. and Gred.) Gredeman and Trappe, Glomus constrictum (Trappe) Gredeman and Trappe, and non-mycorrhizal control; (2) soil water availability, with two levels, i.e., well-watered (WW) and water-stressed (WS) conditions. Each of the six treatments had five replicates (pots) with a total of thirty pots (one seedling per pot). Pots were arranged in a complete randomized block design, and five blocks were applied in this study.
Soil and biological materials
The soil substrate used in this study was topsoil (5–20 cm) collected in Yangling, Shaanxi Province, China. The soil physicochemical properties were as follows: available N 33.61 mg kg−1, Olsen P 11.34 mg kg−1, available K 94.26 mg kg−1, organic matter 16.42 g kg−1, and pH 7.9 (1:5 soil:water ratio). Prior to being used in the experiments, soil was sieved through a 2 mm sieve, mixed with nursery substrates (sphagnum:perlite:vermiculite, 6:3:1, v/v/v) and fine sand (soil/nursery substrates/sand, 2:1:1, v/v/v), and autoclaved at 0.11 MPa and 121 °C for 2 h.
Seeds of S. davidii were collected in September 2009 from the Zhifanggou watershed of the Loess Plateau, Shaanxi Province, China (109°19′E, 36°51′N, and 1,010–1,431 m a.s.l.). Seeds were dried for 1 week under sunlight, after which plump and fully developed seeds were stored at room temperature (25 °C). Seeds of similar size were surface sterilized before sowing using 0.5 % NaClO for 20 min, washed four times with sterile distilled water, and then germinated on wet filter paper in Petri dishes at 28 °C. Ten days after sowing, each seedling was transplanted into a conical frustum plastic container (15 cm top diameter and 15 cm in depth) containing 1 kg of soil mixture.
Meanwhile, soil water content (SWC) in soil mixture was measured gravimetrically. Water weight (Wwater = Wfresh soil × SWC) and soil dry weight (Wdry soil = Wfresh soil − Wwater) of each pot were determined respectively. Ten pots were watered every day and allowed to drain freely until weight was constant for 1 week, the difference between this weight and Wdry soil was used to calculate field water capacity (FC) (Li et al. 2009). The SWC = 42.5 ± 0.2 % at field water content. The SWC was kept at 32.0 ± 1.2 and 10.8 ± 0.7% for the 75 and 25 % FC regeimes. Soil water potential was determined by a pressure plate apparatus (Richards 1941).
Both Glomus species were provided by the Institute of Plant Nutrition and Resources, Beijing Academy of Agriculture and Forestry Sciences, China. Mycorrhizal inocula consisted of rhizospheric soil, spores (spore density of 353–545 per 100 g dry soil), mycorrhizal hyphae, and infected root fragments (average 88 % colonization rate). Each pot was inoculated with a 30 g inoculum of the mycorrhizal treatment, or a non-mycorrhizal control treatment that consisted of 30 g of sterilized inoculum together with 10 ml of fungi-free filtrate meshing from the inoculum suspension. Mycorrhizal inocula were placed 5 cm below the S. davidii seedlings at the time of transplantation.
Growth conditions
Seedlings were grown in a greenhouse between May and October 2010 at a temperature of 18–30 °C, with 60–75 % relative humidity and a 10-h day/14-h night photoperiod. During the first 123 days (from May to August 2010), all inoculated and non-inoculated seedlings were allowed to grow in the above conditions (referred to as the growth phase), and the soil moisture in all pots was maintained at 75 % FC (−0.10 MPa of soil water potential). A 61 d water-stressed phase followed the growth phase between September and October 2010. In this phase, half of the pots were maintained in a well-watered (WW) condition at 75 % FC, whereas the reminder half were subjected to water-stress (WS) conditions at 25 % FC (−0.86 MPa of soil water potential) for a further 2 months (Wu et al. 2008b). Prior to water stress initiation, irrigation of the WS seedlings was suspended until the soil moisture reached 25 % FC. Aluminum foil covered the soil surface and the bottom of pots to limit water loss. Water loss was measured gravimetrically by weighing the pots, and the value of water loss in each pot was the difference between the Wfresh soil in 75 or 25 % FC and the actual Wfresh soil. Lost water was replaced with fresh distilled water each day at 18:00, in order to keep 75 and 25 % FC, respectively.
Plant measurements
Physiological parameters of S. davidii seedlings were measured at the end of the water stress phase, and each treatment had five plants for plant measurements. Relative chlorophyll concentration in the third expanded leaves was measured using a CM-1000 chlorophyll meter (Spectrum, Plainfield, Illinois, USA), according to the manufacturer’s instructions. Gas exchange parameters, including, net photosynthetic rate (Pn), stomatal conductance (gs), intercellular CO2 concentration (Ci) and transpiration rate (E), were determined for the third expanded leaves using a portable open flow gas exchange system LI-6400 (LI-COR, Inc., Lincoln, NE, USA) between 10:00 and 11:30 a.m. The photosynthetic photon flux density was 2,000 μmol m−2 s−1, the CO2 concentration was 350 cm3 m−3, the leaf temperature was 25.0 °C, and the air flow rate was 0.5 dm3 min−1. Instantaneous water use efficiency (iWUE) was calculated as the ratio of the net photosynthetic rate per transpiration rate.
Chlorophyll fluorescence of the third expanded leaves was measured at room temperature (25.0 °C) between 10:00 and 11:30 a.m. using a modulated plant fluorometer (Imaging-PAM, Heinz Walz GmbH, Germany), according to the manufacturer’s instructions. After darkening the leaves for 30 min, the minimum fluorescence (F0) and maximal fluorescence (Fm) yields were recorded for dark-adapted leaves, while the steady-state (Fs) and maximal (Fm′) fluorescence were determined for light-adapted leaves. The maximum fluorescence yield (Fm) was attained with a 3 s saturating pulse (2,000 μmol m−2 s−1), while the minimal fluorescence level in the light-adapted state (F0′) was determined by illuminating with a 3 s far-infrared light (5 μmol m−2 s−1). The maximum quantum yield of PSII (Fv/Fm, Fv = Fm − F0), the actual quantum yield of PSII (ΦPSII = (Fm′ − Fs)/Fm′), the nonphotochemical fluorescence quenching (NPQ = (Fm − Fm′)/Fm′) and the photochemical fluorescence quenching (qP = (Fm′ − Fs)/(Fm′ − Fo′)) were calculated (Maxwell and Johnson 2000).
After physiological measurements, shoots were cut 1 cm above the soil surface. Roots were gently pulled up from the pots, washed with tap water, any dirt and soil was carefully removed using fine-tip forceps (Aggangan et al. 2010). Plant height and root length were measured using a steel ruler. Root and shoot biomass were determined by oven drying at 70 °C for 72 h. Relative water content (RWC) was calculated according to Barrs and Weatherley (1962).
Root AM colonization was determined on 1 cm root sections after washing the intact roots in distilled water. Root sections were cleared for 15 min in 10 % KOH at 90 °C, bleached in alkaline hydrogen peroxide (3 ml NH4OH + 30 ml 10 % H2O2 + 60 ml H2O) for 20 min, acidified in 1 % HCl, and stained with 0.05 % (w/v) trypan blue in lactophenol, according to Phillips and Hayman (1970). AM colonization was determined using the gridline intersect method (Giovannetti and Mosse 1980).
Statistical analysis
Data were analyzed using a two-way ANOVA with AM inoculation and watering as main factors. Differences among treatment means were evaluated using Tukey’s multiple range test. All statistical analyses were performed using SPSS (version 13.0 for Windows, SPSS Inc., IL, USA).
Results
AM colonization
AM colonization was not observed in the roots of non-inoculated seedlings, and all inoculated plants had been infected by Glomus species. AM colonization of seedlings inoculated with G. constrictum was 83.4 % under WW conditions and 74.6 % under WS conditions, while the G. mosseae colonization was 70.2 % under WW conditions and 62.8 % under WS conditions. AM colonization was significantly affected by water stress (F = 12.43, P < 0.01) and AM species (F = 7.68, P < 0.01) respectively, but not by the interaction between water stress and AM species (F = 0.46, P > 0.05).
Plant growth
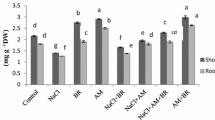
Water stress depressed shoot and root mass, plant height and root length in S. davidii seedlings (Table 1). Inoculated seedlings had higher shoot and root mass, plant height, and root length than non-inoculated seedlings. Furthermore, the two Glomus species had different capacities for stimulating seedling growth. The shoot and root mass and plant height in G. constrictum-inoculated seedlings were significantly higher than those in G. mosseae-inoculated seedlings. No significant difference in root length was found between Glomus species.
Water status and chlorophyll concentration
Water stress decreased RWC and relative chlorophyll concentration, but had no effect on iWUE in S. davidii leaves (Table 1, Fig. 1a). Mycorrhizal seedlings had a higher RWC compared with non-mycorrhizal seedlings under WS conditions, but not under WW conditions. iWUE was highest in G. constrictum-inoculated seedlings, and lowest in non-inoculated seedlings. Relative chlorophyll concentration was no significant differences among the three mycorrhizal treatments (single G. constrictum-, single G. mosseae- and non-inoculation).
Effects of Glomus mosseae and Glomus constrictum on the relative water content (RWC), maximum quantum yield of PSII (Fv/Fm), actual quantum yield of PSII (ΦPSII), and nonphotochemical quenching values (NPQ) in Sophora davidii leaves under well-watered (WW) and water-stressed (WS) conditions. FSWC, FAMF and FSWC × AMF represent the F-value of soil water conditions (SWC), AMF and SWC × AMF; Values are means ± SD; *P < 0.05, **P < 0.01, NS not significant; the same letter in each subfigure indicates no significant difference among treatments at P < 0.05 using Tukey’s test
Gas exchange
Water stress decreased the Pn, gs and E, and increased the Ci in S. davidii seedlings (Table 1). Mycorrhizal inoculation enhanced the Pn and gs, and reduced the Ci, but it had no influence on the E, when compared with the non-inoculation treatment. G. constrictum-inoculated seedlings had significantly higher Pn and gs than G. mosseae-inoculated seedlings. There were no significant differences in the Ci or E between G. constrictum- and G. mossea-inoculated seedlings.
Chlorophyll fluorescence
Water stress inhibited Fv/Fm, ΦPSII, NPQ, and qP in S. davidii seedlings. However, the negative effects of water stress on the Fv/Fm, ΦPSII, and NPQ were significantly higher in non-inoculated than in inoculated seedlings (significant interaction AM inoculation × watering condirtions) (Table 1, Fig. 1b, c, d). Except in the case of G. constrictum-inoculated seedlings under WW conditions, NPQ was not affected by AM inoculation. The Fv/Fm in G. constrictum-inoculated seedlings was significantly higher than that in G. mosseae-inoculated seedlings under WS conditions, but no notable difference in Fv/Fm was found between G. constrictum- and G. mosseae-inoculated seedlings under WW conditions. The ΦPSII in G. constrictum-inoculated seedlings was significantly higher than that in G. mosseae-inoculated seedlings under WW conditions, but no difference in ΦPSII was observed between G. constrictum- and G. mosseae-inoculated treatments under WS conditions.
Discussion
Water stress reduced AM colonization in S. davidii roots. This result is consistent with other pot-based experiments using Cucumis melo (Huang et al. 2011), Poncirus trifoliata (Wu et al. 2008b), Fragaria virginiana (Victoria and Borowicz 2010), and Oryza sativa (Ruiz-Sánchez et al. 2010). Water stress inhibits the germination of spores and the spread of hyphae in the soil after initial AM colonization, which might explain the negative effects of drought on AM colonization (Huang et al. 2011). Both Glomus species showed different development in S. davidii roots, with G. constrictum exhibiting a higher colonization than G. mosseae, irrespective of watering conditions. Higher AM colonization might better improve the growth of host plants by providing added direct transport channels for water and mineral nutrients under drought conditions (Ruiz-Lozano 2003).
Mycorrhizal symbiotic efficiency is often assessed in term of the growth status of host plants under various abiotic stress conditions (Huang et al. 2011; Ruiz-Lozano et al. 1995a, b). AM mycorrhization in the nursery improved the growth of S. davidii seedlings under water stress relative to non-mycorrhized plants. Similar results were reported for other leguminous plants, and indicates that these plants may have greater transplanting performance in degraded and harsh sites (Requena et al. 1997, 2001; Marques et al. 2001).
Water plays an essential role in physiological processes of plants. Plants need to ensure water uptake to be hydrated in order to survive under drought conditions (Kramer and Boyer 1995; Souza et al. 2010). In this study, the leaf RWC of mycorrhizal plants was higher than that in non-mycorrhizal plants, under WS conditions. This confirmed that AM symbiosis can clearly improve the water status of host plants subjected to water stress (Subramanian et al. 1995; Sánchez-Díaz et al. 1990). It is likely that the improved water status of mycorrhizal seedlings was attributed to AM hyphae, which act as extensions of the root system increasing water uptake capacity (Muthukumar and Udaiyan 2010; Ruiz-Lozano 2003). Faber et al. (1991) estimated that the water transport rates of AM hyphae ranged from 375 to 760 nl H2O h−1, which is an adequate level to meet the water demand of plants.
The two Glomus species decreased Ci and increased gs, irrespective of soil–water status. Plant photosynthesis often benefits from a higher Ci, but the reduction in Ci and the increase in gs indirectly suggests an increase in photosynthetic efficiency (Ruiz-Sánchez et al. 2010; Wu et al. 2008b). Compared to the non-AM seedlings, gs in G. mosseae-inoculated seedlings were increased by 24 %, and in G. mosseae-inoculated seedlings by 39 %, respectively. Some reports also suggest that the gas exchange rate of mycorrhizal plants is higher than that of non-mycorrhizal plants of similar size and nutrient status (Allen and Boosalis 1983; Augé et al. 1986; Bethlenfalvay et al. 1990; Huang et al. 2011; Ruiz-Lozano et al. 1995a, b). In order to keep their higher gs, plant leaves need to lower their thresholds of stomatal closure, mycorrhizal and non-mycorrhizal plants show different critical points of stomatal behaviour under water stress (Ruiz-Lozano 2003). For example, the leaf water potential of Glomus fasciculatum-inoculated wheat was about 0.2 MPa lower than that of non-mycorrhizal seedlings when stomata began to close (Allen and Boosalis 1983), while the leaf water potential at stomatal closure was 0.7 MPa lower in mycorrhizal roses compared with non-mycorrhizal Rosa hybrida L. cv. ‘Samantha’ (Augé et al. 1986).
Light energy absorbed by chlorophyll molecules can be partly used to drive photosynthesis (photochemistry), but it can also be dissipated through chlorophyll fluorescence (nonphotochemistry) (Maxwell and Johnson 2000). The two Glomus species significantly increased the Fv/Fm and ΦPSII in S. davidii seedlings, when compared with non-inoculated plants under water stress conditions. This demonstrated that AM inoculation under water stress conditions improved the energy cycling between the reaction centre and the chloroplast pool, and it enhanced the efficiency of excitation energy capture by chloroplasts (Wu et al. 2008b; Maxwell and Johnson 2000). The qP in the leaves of mycorrhizal S. davidii was clearly higher than that in the leaves of non-mycorrhizal S. davidii, irrespective of soil–water status, which suggested that both inoculations increased the photochemical capacity of PSII in light-adapted leaves and the steady-state fraction of oxidized PSII (Maxwell and Johnson 2000).
Glomus constrictum had a higher root colonization capacity, and increased plant growth and physiological performance of S. davidii seedlings more than did G. mosseae. This indicates that G. constrictum is a more efficient AM species than G. mosseae when colonizing S. davidii roots under water stress. According to our results, G. constrictum should preferently be used to inoculate the culture of S. davidii seedlings in the semiarid Loess Plateau of China. Similar results were reported for C. melo seedlings inoculated with different Glomus species in the same plateau, under WS conditions (Huang et al. 2011).
This study investigated the responses in terms of the growth, chlorophyll concentration, gas exchange, and chlorophyll fluorescence of S. davidii seedlings, when inoculated with two Glomus species under WW and WS conditions. In conclusion, Glomus-inoculated seedlings had higher performance under water stress conditions by improving the host growth and physiological status, and by alleviating the photoinhibition of S. davidii leaves. G. constrictum was found to be a more efficient fungus at improving the growth and physiological performance of S. davidii seedlings, compared with G. mosseae under WW and WS conditions. Re-establishing leguminous shrub communities is a key step in the revegetation of arid and semi-arid areas, because they are very valuable for restoring soil fertility and preventing erosion (Requena et al. 2001; Caravaca et al. 2003). The effectiveness of AM symbionts in improving the outplanting performance of certain leguminous plants has also been demonstrated in desertified Mediterranean areas and nutrient-deficient tropical forests in south-eastern Brazil (Caravaca et al. 2003; Requena et al. 1997, 2001; Marques et al. 2001). Revegetation strategies based on AM symbiont interactions (mycorrhiza-leguminous shrub) might also be considered in other arid and semi-arid ecosystems.
References
Aggangan NS, Moon HK, Han SH (2010) Growth response of Acacia mangium Willd. seedlings to arbuscular mycorrhizal fungi and four isolates of the ectomycorrhizal fungus Pisolithus tinctorius (Pers.) Coker and Couch. New For 39:215–230
Allen MF, Boosalis MG (1983) Effects of two species of VA mycorrhizal fungi on drought tolerance of winter wheat. New Phytol 93:67–76
Augé RM, Scheckel KA, Wample RL (1986) Greater leaf conductance of well-watered VA mycorrhizal rose plants is not related to phosphorus nutrition. New Phytol 103:107–116
Barrs HD, Weatherley PE (1962) A re-examination of the relative turgidity technique for estimating water deficits in leaves. Aust J Biol Sci 15:413–428
Bethlenfalvay GJ, Brown MS, Franson RL (1990) The Glycine-Glomus-Bradyrhizobium symbiosis, X relationships between leaf gas exchange and plant and soil water status in nodulated, mycorrhizal soybean under drought stress. Plant Physiol 94:723–728
Caravaca F, Alguacil MM, Figueroa D, Barea JM, Roldán A (2003) Re-establishment of Retama sphaerocarpa as a target species for reclamation of soil physical and biological properties in a semi-arid Mediterranean area. For Ecol Manage 182:48–58
Faber BA, Zasoski RJ, Munns DN, Shackel K (1991) A method for measuring hyphal nutrient and water uptake in mycorrhizal plants. Can J Bot 69:87–94
Feng XX, Tang M, Gong MG, Yu HX (2011) Spatial distribution of arbuscular mycorrhizal and glomalin in the rhizosphere of Sophora davidii on the Loess Plateau. J Northwest Sci Tech Univ Agric For (Nat Sci Ed) 39:96–102 (in Chinese)
Giovannetti M, Mosse B (1980) An evaluation of techniques for measuring vesicular arbuscular mycorrhizal infection in roots. New Phytol 84:489–500
Goicoechea N, Merino S, Sánchez-Díaz M (2005) Arbuscular mycorrhizal fungi can contribute to maintain antioxidant and carbon metabolism in nodules of Anthyllis cytisoides L. subjected to drought. J Plant Physiol 162:27–35
Huang Z, Zou ZR, He CX, He ZQ, Zhang ZB, Li JM (2011) Physiological and photosynthetic responses of melon (Cucumis melo L.) seedlings to three Glomus species under water deficit. Plant Soil 339:391–399
Hura T, Hura K, Grzesiak M, Rzepka A (2007) Effect of longterm drought stress on leaf gas exchange and fluorescence parameters in C3 and C4 plants. Acta Physiol Plant 29:103–113
Kramer PJ, Boyer JS (1995) Water relations of plants and soils. Academic Press, San Diego
Li FL, Bao WK, Wu N (2009) Effects of water stress on growth, dry matter allocation and water-use efficiency of a leguminous species, Sophora davidii. Agrofor Syst 77:193–201
Marques MS, Pagano M, Scotti MRMML (2001) Dual inoculation of a woody legume (Centrolobium tomentosum) with rhizobia and mycorrhizal fungi in south-eastern Brazil. Agrofor Syst 52:107–117
Maxwell K, Johnson GN (2000) Chlorophyll fluorescence—a practical guide. J Exp Bot 51:659–668
Muthukumar T, Udaiyan K (2010) Growth response and nutrient utilization of Casuarina equisetifolia seedlings inoculated with bioinoculants under tropical nursery conditions. New For 40:101–118
Phillips JM, Hayman DS (1970) Improved procedure for clearing roots and staining parasitic and vesicular-arbuscular mycorrhizal fungi for rapid assessment of infection. Trans Br Mycol Soc 55:158–161
Requena N, Jimenez I, Toro M, Barea JM (1997) Interactions between plant-growth-promoting rhizobacteria (PGPR), arbuscular mycorrhizal fungi and Rhizobium spp. in the rhizosphere of Anthyllis cytisoides, a model legume for revegetation in Mediterranean semi-arid ecosystems. New Phytol 136:667–677
Requena N, Pérez-Solís E, Azcón-Aguilar C, Jeffries P, Barea JM (2001) Management of indigenous plant-microbe symbioses aids restoration of desertified ecosystems. Appl Environ Microbiol 67:495–498
Richards LA (1941) A pressure-membrane extraction apparatus for soil solution. Soil Sci 51:377–386
Ruiz-Lozano JM (2003) Arbuscular mycorrhizal symbiosis and alleviation of osmotic stress. New perspectives for molecular studies. Mycorrhiza 13:309–317
Ruiz-Lozano JM, Azcón R (1995) Hyphal contribution to water uptake in mycorrhizal plants as affected by the fungal species and water status. Physiol Plant 95:472–478
Ruiz-Lozano JM, Gómez M, Azcón R (1995) Influence of different Glomus species on the time-course of physiological plant responses of lettuce to progressive drought stress periods. Plant Sci 110:37–44
Ruiz-Sánchez M, Aroca R, Muñoz Y, Polón R, Ruiz-Lozano JM (2010) The arbuscular mycorrhizal symbiosis enhances the photosynthetic efficiency and the antioxidative response of rice plants subjected to drought stress. J Plant Physiol 167:862–869
Sánchez-Díaz M, Pardo M, Antolín M, Peña J, Aguirreolea J (1990) Effect of water stress on photosynthetic activity in the Medicago-Rhizobium-Glomus symbiosis. Plant Sci 71:215–221
Smith SE, Read DJ (1997) Mycorrhizal symbiosis, 2nd edn. Academic Press, London
Souza BD, Meiado MV, Rodrigues BM, Santos MG (2010) Water relations and chlorophyll fluorescence responses of two leguminous trees from the Caatinga to different watering regimes. Acta Physiol Plant 32:235–244
Subramanian KS, Charest C, Dwyer LM, Hamilton RI (1995) Arbuscular mycorrhizas and water relations in maize under drought stress at tasseling. New Phytol 129:643–650
Victoria A, Borowicz (2010) The impact of arbuscular mycorrhizal fungi on strawberry tolerance to root damage and drought stress. Pedobiologia 53:265–270
Wu FZ, Bao WK, Li FL, Wu N (2008a) Effects of drought stress and N supply on the growth, biomass partitioning and water-use efficiency of Sophora davidii seedlings. Environ Exp Bot 63:248–255
Wu QS, Xia RX, Zou YN (2008b) Improved soil structure and citrus growth after inoculation with three arbuscular mycorrhizal fungi under drought stress. Eur J Soil Biol 44:122–128
Acknowledgments
This study was funded by the Key Project of the National Natural Science Foundation of China (30730073, 31170567), the Program for Changjiang Scholars and Innovative Research Team in the University of China (IRT1035) and the Doctoral Program of Higher Education of China (20100204110033, 20110204130001). We also thank anonymous reviewers for their valuable suggestion to enhance the manuscript and Dr Duncan E. Jackson for improving the English language used in this manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gong, M., Tang, M., Chen, H. et al. Effects of two Glomus species on the growth and physiological performance of Sophora davidii seedlings under water stress. New Forests 44, 399–408 (2013). https://doi.org/10.1007/s11056-012-9349-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11056-012-9349-1