Abstract
We investigated the role of tetrapartite associations between an arbuscular mycorrhizal (AM) fungus (Glomus geosporum), phosphate solubilizing bacteria (Paenibacillus polymyxa), Frankia and Casuarina equisetifolia on growth, nutrient acquisition, nutrient utilization and seedling quality of C. equisetifolia. Seedlings of C. equisetifolia were grown in an Alfisol soil and inoculated with G. geosporum, P. polymyxa and Frankia either individually or in combinations. Inoculation of bioinoculants stimulated seedling growth, the efficiency of nutrient uptake and improved seedling quality. However, microbial inoculation generally reduced the efficiency of nutrient utilization in dry matter production (nutrient use efficiency). Inoculation of P. polymyxa or Frankia increased the extent of AM colonization, which resulted in the accumulation of the nutrients. Seedlings inoculated with Frankia and G. geosporum had more, and heavier nodules compared to seedlings inoculated with Frankia alone. Dual inoculation of microbes was more effective than individual inoculations. The growth response of seedlings to inoculation involving all the microbes was greater than the response to either individual or dual inoculations. The results of this study showed that the tetrapartite association could improve the growth, nutrient acquisition and seedling quality of C. equisetifolia under tropical nursery conditions.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Actinorhizal plants usually form root nodules in association with the nitrogen fixing actinomycete Frankia. Because of their nitrogen fixing ability, actinorrhizal plants can thrive in infertile soils. Actinorhizal plants also form association with arbuscular mycorrhizal (AM) fungi, thus forming a tripartite symbiosis, which enhances the success of these plants under poor soil conditions (Wheeler et al. 2000). Mycorrhizas improve the nutrient uptake especially phosphorus (P) of the host plants (Jakobsen 1999). The extraradical mycelia of the mycorrhizal fungi may act as extensions of the root systems (Rousseau et al. 1994) and have a high nutrient mobilizing potential (Lapeyrie et al. 1991). The uptake of nutrients like P and micronutrients facilitated by AM fungi can help actinorhizal nodulation and function under stress conditions.
Phosphate solubilizing bacteria (PSB) are beneficial microorganisms in the plant rhizosphere, as they solubilize bound phosphorus (P) and increase their availability for the plant (Rodrìguez and Fraga 1999). In addition, some reports have shown that PSB have a strong stimulatory impact on the growth of mycorrhizal fungi (Artursson et al. 2006). Paenibacillus polymyxa [previously Bacillus polymyxa (Ash et al. 1993)], is a common soil bacterium. Activities of PSB include soil P solubilization, production of antibiotics, auxins, cytokinins, chitinase, and hydrolytic enzymes, as well as promotion of increased soil porosity (Rodrìguez and Fraga 1999). All these activities could account for or contribute to plant growth promotion at various times and in various environments during the life cycle of a plant. The organic acids produced by P. polymyxa accelerate solubilization of bound phosphates, thereby enhancing their availability to plants. Therefore, it is expected that increased P availability due to the PSB might stimulate the interaction of AM fungi and Frankia on host plants. It is well established that symbiotic N2-fixation is highly dependent on the uptake of P, calcium and micronutrients by the host (Sanginga et al. 1989; Walker et al. 1993). This leads to the hypothesis that simultaneous inoculation of PSB along with AM fungi and Frankia would enhance seedling growth through enhanced mycorrhization and nodulation. Thus, an increase in seedling quality should occur in response to greater N and P availability to the host.
The rate of nutrient absorption and nutrient use efficiency (NuUE): the efficiency with which a nutrient is utilized to produce dry matter are the two factors that control the rate of plant growth in low soil nutrient conditions (Koide et al. 2000). The NuUE is known to vary with nutrient availability and mycorrhizal colonization (Koide et al. 2000) and a very few studies has assessed this for tree species (Muthukumar et al. 2001; Muthukumar and Udaiyan 2006).
Casuarina equisetifolia Forst. is a fast growing actinorrhizal tree extensively used for fuel, land reclamation, wind breaks on farms, erosion control, afforestation, wasteland development in tropics and subtropics (Subba Rao and Rodriguez Barrueco 1995). Although C. equisetifolia can associate with both ecto- and endo-mycorrhizal fungi, the endomycorrhizal association with AM fungi is more common in C. equisetifolia than ectomycorrhiza (Duponnois et al. 2003; He and Critchley 2008). Casuarina equisetifolia also respond spontaneously to nutrient deficiency by developing cluster roots under non-mycorrhizal conditions (Zaïd et al. 2003). There is some evidence indicating the importance of bioinoculants on C. equisetifolia growth under nursery (Vasanthakrishna et al. 1994; Rajendran et al. 2003) and field (Rajendran and Devaraj 2004) conditions. The objective of this study was to determine whether simultaneous inoculation of AM fungus, PBS and Frankia could enhance growth, nodulation and seedling quality of C. equisetifolia under tropical nursery conditions. Further, we intended to determine the effect of bioinoculants on the efficiency of nutrient uptake and nutrient use efficiency of C. equisetifolia as no previous studies have determined these for an actinorhizal plant species.
Materials and methods
Nursery site and soil preparation
The study was conducted at the tree nursery of the Botany Department, Bharathiar University, Coimbatore (11°01′ N and 96°93′ E, altitude 410 m a.s.l), Tamilnadu, India. The climate is monsoonal with an annual precipitation of 640 mm and a dry season between January and April. The maximum and minimum monthly temperatures are 31 and 21°C, respectively. Alfisol soil was used in this study. Topsoil (0–30 cm) of the soil was collected from a fallow field that had remained uncultivated for more than 7 years and represented areas where plantations are to be raised for protection purposes. The vegetation in this field was very sparse and dominated by grasses. Soil chemical analysis prior to experiments using standard procedures is presented in Table 1.
Assessment of indigenous microbial population
To assess the indigenous PSB populations in field soils, three 0.1-ml aliquots of soil diluted with sterile water (10−3–10−6) were spread on standard media for dilution plate counts for PSB (Pikovskaya 1948). The plates were incubated for 27°C for 3–5 days for PSB. The colonies that formed clear zones on the PSB medium were counted. The number of recorded PSB cells was expressed as log colony forming units (CFUs) per gram of soil. Infective propagules of indigenous AM fungi was assessed according to the Most Probable Number technique (Porter 1979).
Seed and inoculum
Casuarina equisetifolia seeds obtained from the Institute for Forest Genetics and Tree Breeding (IFGTB), Coimbatore, India, were scarified with 95% concentrated sulphuric acid for 2 min., rinsed thoroughly with running tap water for 30 min., soaked in distilled water overnight and sown in trays (30 × 22 × 6 cm) containing heat sterilized (121°C for 3 h) sand. The seedlings were maintained for 6 weeks. Two healthy and vigorously growing seedlings were transplanted into each black polythene bags (45 cm deep, 25 cm wide) containing 7.5 kg field soil. After establishment, it was thinned to one seedling per bag.
The AM fungus Glomus geosporum (Nicol. & Gerd.) Walker was selected as it has been reported to be associated with C. equisetifolia in different soil types of Tamilnadu, India (Sambandan et al. 1994). AM fungal inoculum consisted of soil containing spores and mycorrhizal roots from a pot culture of sorghum [Sorghum bicolor (L.) Moench.] colonized by G. geosporum (DBCC-72) and grown for 12 months. The AM fungal species originated from semi-arid grassland (Muthukumar and Udaiyan 2002). Five grams of AM fungal inoculum containing 258 propagules g−1 soil as assessed according to Porter (1979) was added to the planting hole at transplantation. Treatments not involving AM fungi received the same amount of sterile inoculum, which had been autoclaved at 121°C for 90 min., three times at regular intervals. Soil microbes in AM fungal inoculum were equalized across treatments by applying 25 ml ‘microbial cocktail’ to each bag. This ‘microbial cocktail’ was prepared by blending 100 g AM fungal inocula in 1 l deionized water and filtering it three times through a 25 μm sieve.
Frankia inoculum suspension was prepared from ca. 0.5 g of fresh nodule collected from C. equisetifolia trees growing in Bharathiar University Campus. The young nodule lobes were separated; surface sterilized (30% H2O2, 30 min.), rinsed in sterile distilled water and ground in a sterile mortar and pestle. The nodule suspension was made up to 500 ml with 2% sucrose solution (Reddell et al. 1988) prior to inoculation. Five ml of the nodule suspension (equivalent to 5 mg of nodule f. wt.) was pipetted around the roots of the seedlings. Treatments not involving Frankia received 5 ml of heat sterilized nodule suspension.
The PSB Paenibacillus polymyxa (isolate no. DBCC 005) was isolated and maintained as described earlier (Muthukumar et al. 2001). At transplantation 15 ml PSB inoculum grown in PSB medium without agar equivalent to 1.2 × 106 cells g−1 soil was pipetted into each bag specifying treatment. Seedlings not involving PSB received 15 ml of heat sterilized PSB inoculum.
Experimental design
There were seven treatments involving bioinoculants individually or in various combinations, and an uninoculated control. Each treatment consisted of ten replicates and measurements were made after destructive harvest at 60 and 120 days after transplantation (DAT). The experiment involved were 160 bags (8 × 10 × 2) set up in the nursery in a completely randomized design. The bags were rearranged every 15 days to ensure uniform growth conditions. Seedlings were watered to maintain field capacity and no nutrients were added. The maximum/minimum temperature range over the experiment period was 36°C/24°C, rainfall was 350 mm and relative humidity was between 58 and 70%.
Analyses
At harvest, the soil was washed from roots and a weighed portion of each root sample was preserved in formalin–acetic acid–alcohol (FAA) solution for the assessment of AM colonization. Shoot and roots were separated and oven dried at 70°C for 48 h for the determination of dry mass. The root samples were cleared in 2.5% KOH solution at 121°C for 30 min followed by acidification with 1.0% HCl and stained with 0.05% trypan blue in lactophenol (Koske and Gemma 1989). The extent of AM colonization was quantified according to the magnified intersection method (McGonigle et al. 1990). Infective propagules of AM fungi in the inoculum were determined by MPN method (Porter 1979).
Tissue nitrogen (N) in shoot and root samples was determined by the micro Kjeldahl digestion method using concentrated H2SO4 digest and selenium as catalyst. Total N was estimated using a Technicon Auto Analyser (Gedko International, UK). Tissue P concentration in seedling tissues was determined by the molybdenum blue method (Jackson 1971) using a Spectronic 20 electrophotocolorimeter after wet-ashing the plant samples in a nitric–sulphuric–perchloric acid mixture. Tissue potassium (K) was estimated by flame photometry (Davis 1962).
Nutrient–use efficiency (NuUE) was calculated according to Koide et al. (2000)
(dW, increase in plant d. wt.; dt, time difference).
Relative growth rate (RGR) was calculated (Williams 1946) using the formula:
(W1, intial plant d. wt.; W2, final plant d. wt.; t2 − t1, time interval).
The efficiency of nutrient uptake (ENuU) defined as the amount of nutrients absorbed per unit root mass (Gray and Schlesinger 1983) was calculated as follows;
Microbial inoculation effect (MIE) was calculated according to Muthukumar and Udaiyan (2006):
Seedling quality index (SQI) was calculated according to Dickson et al. (1960):
Statistical analysis
Analysis of Variance (ANOVA) was performed on all data to compare treatment effects and the influence of soil type and fertilizer application on treatments. Means were separated using Duncan’s Multiple Range Test (DMRT). Percentage data on mycorrhizal colonization were arcsine square root transformed prior to analysis. Pearson’s correlation analysis was used to assess the relationship between seedling growth and nutrient uptake, AM fungal colonization and components of nutrient efficiencies.
Results
Seedling growth and biomass
Seedlings inoculated with bioinoculants were taller and had increased stem girth and biomass both at 60 and 120 DAT (Table 2). Seedlings inoculated with bioinoculants were 11–48% taller compared to uninoculated seedlings at 60 DAT. Similarly, at 120 DAT bioinoculated seedlings were 14–48% taller than uninoculated seedlings. The stem girth of bioinoculated seedlings were respectively 4–56% and 6–55% higher compared to uninoculated seedlings at 60 and 120 DAT, respectively. Seedling biomass (shoot + root) of bioinocualted seedlings were 19–423 and 20–434% higher compared to uninoculated seedlings at 60 and 120 DAT, respectively. Bioinoculation significantly reduced the R/S ratio of the seedlings both at 60 and 120 DAT. However, seedlings inoculated with PSB or AM fungi along with Frankia recorded higher R/S ratios both at 60 and 120 DAT.
Growth rate
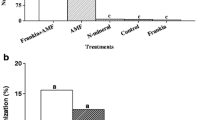
Relative growth rate of non-inoculated C. equisetifolia seedlings was 0.025 mg d−1, and ranged between 0.034 and 0.059 mg d−1 for individual inoculations, 0.063 and 0.100 mg d−1 for dual inoculated seedlings and 0.132 mg d−1 for multi-microbial inoculated seedlings (Fig. 1). Thus, multi-microbial inoculated seedlings had growth rates 65–356% higher compared to other inoculated seedlings and 453% higher compared to uninoculated control.
Relative growth rate of Casuarina equisetifolia seedlings inoculated with arbuscular mycorrhizal fungi (AM), Frankia (F), phosphate solubilizing bacteria (PSB) individually or in combinations as assessed after 120 days of growth. Bars bearing same letter(s) do not significantly differ according to Duncan’s Multiple Range Test (p < 0.05)
Seedling nutrients
Seedlings inoculated with bioinoculants had significantly higher concentration of nutrients in their tissues (Table 3). By the end of 120 DAT bioinocualted seedlings had 11–20% higher N, 50–192% higher P and 7–79% higher K in their shoots compared to uninoculated seedlings. Similarly, in roots the N, P and K concentration of bioinoculated seedlings were respectively 9–155%, 43–186% and 8–85% higher than uninoculated seedlings.
AM fungal colonization
Seedlings raised in natural soils had significantly low extent of their root colonized by AM fungi (Table 4). In contrast, bioinoculant inoculation significantly increased the extent of root length colonized by AM fungi and the inoculated seedlings had 2–6 fold higher colonization levels at 60 DAT and 1.5–4 fold higher colonization levels at 120 DAT compared to uninoculated seedlings.
Infective propagules of AM fungi
At 120 DAT, the infective propagules of AM fungi significantly varied among treatments (Fig. 2). Infective propagules were 55–130%, 144–193% and 210% higher in individual, dual and multimicrobial inoculations, respectively, compared to uninoculated control.
Infective propagules of arbuscular mycorrhizal (AM) fungi in Casuarina equisetifolia seedlings uninoculated (C) or inoculated with AM fungi (AM), Frankia (F), phosphate solubilizing bacteria (PSB) individually or in combinations as assessed after 120 days of growth. Bars bearing same letter(s) do not significantly differ according to Duncan’s Multiple Range Test (p < 0.05)
Nodulation
Seedlings inoculated with bioinoculants had more numerous and heavier nodules compared to uninoculated seedlings (Table 4). The nodule numbers were 8–84% and 11–89% higher in seedlings inoculated with bioinoculants at 60 and 120 DAT, respectively compared to uninoculated seedlings. Similarly, nodule dry weight of inoculated seedlings was 191–385% higher at 60 DAT and 28–307% higher at 120 DAT compared to uninoculated seedlings. A significant positive correlation existed between nodulation and the extent of AM fungal colonization (Table 6).
Efficiency of nutrient uptake
Nutrient uptake per unit of root was higher for seedlings inoculated with bioinoculants (Table 5). The efficiency of nitrogen uptake (ENU) of multiple microbe-inoculated seedlings was 22–267% higher compared to other treatments including control. However, the efficiency of phosphorus uptake (EPU) of PSB and AM fungi inoculated seedlings was 52% higher than multiple microbe inoculated seedlings and 152–450% higher than other treatments. Likewise, the efficiency of potassium uptake (EKU) of PSB and AM fungi inoculated seedlings was 127–208% higher than other inoculations and 40% higher compared to multiple inoculation. Efficiency of nutrient uptake was correlated significantly and positively to nodulation, AM colonization, tissue nutrient content, and seedling biomass and growth rate (Table 6).
Nutrient use efficiencies
Microbial inoculations significantly altered the nutrient use efficiencies of C. equisetifolia seedlings (Fig. 3). Nitrogen use efficiency (NUE) and phosphorus use efficiency (PUE) of bioinocualted seedlings were significantly lower compared to uninoculated seedlings. The NUE of bioinoculants inoculated seedlings was 8–196% lower compared to uninoculated seedlings. Similarly, PUE of bioinoculants inoculated seedlings was 68–273% lower compared to uninoculated seedlings. Uninoculated seedlings recorded 2–54% higher KUE compared to microbial inoculated seedlings. The NUE (r = −0.940; p < 0.01; n = 8) and KUE (r = −0.808; p < 0.05; n = 8) were significantly and negatively correlated to relative growth rate.
Nutrient use efficiency of Casuarina equisetifolia seedlings uninoculated (C) or inoculated with arbuscular mycorrhizal fungi (A), Frankia (F), phosphate solubilizing bacteria (P) individually or in combinations (AF, AP, FP, AFP) as assessed after 120 days of growth. Bars bearing same letter(s) do not significantly differ according to Duncan’s Multiple Range Test (p < 0.05)
Microbial inoculation effect (MIE)
MIE of seedlings inoculated with all the microbes were 1.5–4.9 fold higher compared to seedlings inoculated with individual microbes and 1.1–1.4 fold higher compared to dual inoculated seedlings (Fig. 4).
Microbial inoculation effect in Casuarina equisetifolia inoculated with arbuscular mycorrhizal fungi (AM), Frankia (F), phosphate solubilizing bacteria (PSB) individually or in combinations as assessed after 120 days of growth. Bars bearing same letter(s) do not significantly differ according to Duncan’s Multiple Range Test (p < 0.05)
Seedling quality index (SQI)
Calculated SQI of multi-microbial inoculated seedlings were 92–461% higher compared to other inoculations and 528% higher compared to uninoculated seedlings (Fig. 5). SQI was significantly and positively correlated to the ENU and relative growth rate (r = 0.884; p < 0.01; n = 8).
Seedling quality index of Casuarina equisetifolia inoculated with arbuscular mycorrhizal fungi (AM), Frankia (F), phosphate solubilizing bacteria (PSB) individually or in combinations as assessed after 120 days of growth. Bars bearing same letter(s) do not significantly differ according to Duncan’s Multiple Range Test (p < 0.05)
Discussion
Bioinoculant application improved the growth of C. equisetifolia seedlings despite the general assumption that members of Casuarinaceae can thrive in soils of low fertility (Diem and Arahou 1996). This study further supports the positive response of C. equisetifolia seedlings in the nursery to bioinoculant application (Vasanthakrishna et al. 1994, 1995; Rajendran et al. 2003) and strengthens the microbial dependency of C. equisetifolia in soils of low fertility. In several studies (e.g., Ravikumar et al. 1997; Guissou et al. 1998; Gupta and Rahangdale 1999; Founoune et al. 2002; Gehring 2003; De Grandcourt et al. 2004; Lesueur and Duponnois 2005; Yamanaka et al. 2005) the plant growth promoting effects of microbes has been demonstrated in sterile substrates. However, the growth promoting effect in sterile soil may substantially differ from those in unsterile soil (Ortas 2003) possibly due to competition from resident microorganisms. Individual inoculation of G. geosporum or Frankia nodular suspensions improved growth of C. equisetifolia seedlings by 109 and 40%, respectively compared to uninoculated control. Similar results of lower intensities have been reported in other studies. Vasanthakrishna et al. (1994) and Rajendran et al. (2003) reported an increase in C. equisetifolia seedling dry weight of 3.45–39% in response to Glomus fasciculatum inoculation; and 3.45–25% in response to nodule suspension inoculation. However, the improved seedling growth in response to PSB inoculation contradicts the observation of Rajendran et al. (2003) where a reduction in seedling biomass (7%) has been reported due to PSB inoculation. Inoculation of Frankia along with G. geosporum resulted in better growth of C. equisetifolia than inoculation of Frankia alone. Several investigators have reported that dual inoculation of AM fungi and Frankia resulted in better growth of actinorhizal plants compared to individual inoculation with Frankia (Tian et al. 2002; Tiwari et al. 2003). We did not observe cluster roots in any of the root systems as cluster roots tend to occur under non-mycorrhizal condition (Zaïd et al. 2003).
In the present study inoculation of G. geosporum improved the nutrient content of nodulated C. equisetifolia. This contrasts the observation of Yamanaka et al. (2005), where Gigaspora margarita failed to improve the P content of nodulated Alnus sieboldiana. The AM effect on nutrient uptake may be due to the well-known ability of the external mycelium to extend the soil volume that the plants explore for P and other nutrient uptake although they do not solubilize nutrients (Yamanaka et al. 2003). This is confirmed by an increased nutrient uptake per given unit of root in G. geosporum inoculated seedlings. Improved growth of C. equisetifolia in response to G. geosporum inoculation may be a result of increased nutrient content in addition to plant hormones produced by AM fungi (Barea and Azcón-Aguilar 1982) or induced by AM fungi (Allen et al. 1982). Mycorrhizal inoculation is known to induce changes in root morphology (Berta et al. 1995), and our findings show that G. geosporum inoculation increased root dry weights, thus confirming previous findings, where inoculation of G. intraradices increased root dry weights in Prunus cerasifera (Berta et al. 1995), and Allium cepa (Toro et al. 1997) by increasing root diameter. Therefore, mycorrhizal roots are more efficient in taking up soil nutrients than non-mycorrhizal roots (Stribley et al. 1980). Most of the observed changes in growth of the host plant in response to mycorrhizal inoculation are consequences of this increased uptake of P and other nutrients as shown in the present and other studies (Raju et al. 1990; Kothari et al. 1991).
In this study, nodule suspension inoculation increased the nodule number and mass in C. equisetifolia. A similar response to crushed nodule suspension inoculation has been reported for Casuarina (Mansour 2003; Rajendran et al. 2003; Rajendran and Devaraj 2004), Alnus (Wheeler et al. 1991) and Coriaria nepalensis (Tiwari et al. 2003). When seedlings were inoculated with Frankia and P. polymyxa along with G. geosporum, additional nodule material was formed as evidenced by increased nodule mass. This increased nodular material provided sufficient fixed N2 to support higher growth rates than seedlings inoculated with Frankia and PSB either individually or in combinations. Further nodule biomass correlated positively with seedling biomass. A similar positive correlation between seedling biomass and nodule biomass has been reported in Alpova diplophloeus (Yamanaka et al. 2003), red alder (Alnus rubra) and snowbrush (Ceanothus velutinus) (Rojas et al. 2001, 2002). These correlations confirm the view that symbiotic nitrogen fixation is dependent on host photosynthesis (Arnone and Gordon 1990). It has been suggested that phosphate plays an important role in the regulation of nodulation in actinorhizal species (Wall and Huss Danell 1996; Wheeler et al. 2000) and it is conceivable that nodulation of C. equisetifolia by Frankia may be particularly susceptible to the improvement in P nutrition which would follow enhanced colonization by AM fungi.
Inoculation of P. polymyxa alone significantly increased the P content of C. equisetifolia. This is in accordance with the studies carried out in lowly weathered soils with a low P fixation capacity where inoculation of PSB alone increased the plant P uptake by 25–73% in different plant species (Kucey and Leggett 1989; Singh and Singh 1993). Further, P. polymyxa behaved as mycorrhizal helper bacteria because it promoted root colonization by mycorrhizal fungi, confirming previous findings involving other mycorrhizal and PSB combinations (Garbaye 1994). The mechanisms by which these bacteria stimulate mycorrhizal colonization are poorly understood. However, specialized bacterial activities such as production of vitamins, amino acids, and hormones may be involved in these interactions (Garbaye 1994).
Dual inoculation of Glomus geosporum and P. polymyxa did change the P concentration of nodulated C. equisetifolia. Plants inoculated with PSB are expected to improve P concentration in the plants by increasing the soluble P in the soil. Osorio and Habte (2001) showed that Leucaena leucocephala inoculated with Glomus aggregatum and Mortierella sp., took up more P than those with AM fungi or bacteria separately. These types of observations have also been reported in neem (Azadirachta indica) and bamboo (Dendrocalamus strictus) (Muthukumar et al. 2001; Muthukumar and Udaiyan 2006). Azcón et al. (1976) showed that the improved P uptake of mycorrhizal plants resulted from the increased availability of the sparingly soluble forms of P to the plant roots by AM fungi. Several studies have shown that PSB interact with AM fungi by releasing phosphate ions in the soil, which causes a synergistic interaction that allows for better exploitation of poorly soluble P sources (Piccini and Azcón 1987; Pramanik and Singh 2004; Yamanaka et al. 2005). It is likely that the seedlings through AM fungi could more effectively take up the phosphate solubilized by the bacteria (Jeffries and Barea 1994). This was evidenced in the present study by the existence of strong correlations between the EPU, AM colonization and plant P.
The dry weight of multiple microbe-inoculated seedlings was significantly higher than seedlings inoculated with bioinoculants individually or dually. This increased biomass results from the one to three fold increases in growth rates of the seedlings inoculated with all the microbes over other inoculations. The increased growth rates of multiple microbe-inoculated seedlings could be the result of increased nutrient inflow rates possibly through AM fungi as suggested by increased ENuU values along with increased nodular efficiency and nutrient availability. This is further supported by the existence of a strong positive correlation between these variables.
Uninoculated C. equisetifolia seedlings had the maximum NuUE, which indicates that these seedlings are under certain amount of resource limitation. Generally, plants respond to resource limitations by increasing the utilization efficiencies of the limiting resources (Chapin et al. 1987). Therefore, microbial inoculated C. equisetifolia seedlings with low NuUE are not inferior, since the quality index based on morphological qualities indicates them to be superior over uninoculated seedlings. The increased nutrient content of bioinoculated seedlings may be advantageous for these seedlings during out-planting as nutrient status of the seedlings influence resistance to various stresses (Ritchie 1984). Although AM fungi, PSB and Frankia naturally occur in soil, usually their numbers are not high enough to compete with other soil microbes commonly established in the rhizosphere. Thus, the amount of nutrients liberated or made available to seedlings by them is generally not sufficient for a substantial increase in situ seedling growth. Therefore, inoculation of seedlings by target microorganisms at a higher concentration than that normally found in the soil is necessary to take advantage of nutrient availability for better quality seedling production.
References
Allen MF, Moore TS, Christensen M (1982) Phytohormone changes in Bouteloua gracilis infected by vesicular-arbuscular mycorrhizae. II. Altered levels of gibberellin-like substances and abscisic acid in the host plant. Can J Bot 60:468–471
Arnone JA III, Gordon JC (1990) Effect of nodulation, nitrogen fixation and CO2 enrichment on the physiology, growth and dry mass allocation of seedlings of Alnus rubra Bong. New Phytol 116:55–66
Artursson V, Finlay RD, Jansson JK (2006) Interactions between arbuscular mycorrhizal fungi and bacteria and their potential for stimulating plant growth. Environ Microbiol 8:1–10
Ash C, Priest FG, Collins MD (1993) Molecular identification of rRNA group 3 bacilli using a PCR probe test. Proposal for the creation of a new genus Paenibacillus. Antonie van Leeuwenhoek 64:253–260
Azcón R, Barea JM, Hayman DS (1976) Utilization of rock phosphate in alkaline soils by plants inoculated with mycorrhizal fungi and phosphate-solubilizing bacteria. Soil Biol Biochem 8:135–138
Barea JM, Azcón-Aguilar C (1982) Production of plant growth-regulating substances by vesicular-arbuscular mycorrhizal fungus Glomus mosseae. Appl Environ Microbiol 43:810–813
Berta G, Trotta A, Fusconi A, Hooker JE, Munro M, Atkinson D, Giovannetti M, Morini S, Fortuna P, Tisserant B, Gianinazzi-Pearson V, Gianinazzi S (1995) Arbuscular mycorrhizal induced changes to plant growth and root system morphology in Prunus cerasifera. Tree Physiol 15:281–293
Chapin FS III, Bloom AJ, Field CB, Waring RH (1987) Plant responses to multiple environmental factors. Bioscience 37:49–56
Davis DJ (1962) Emission and absorption spectrochemical methods. In: Peach K, Tracey MV (eds) Modern methods of plant analysis. Springer, Heidelberg, pp 1–25
De Grandcourt A, Epron D, Montpied P, Louisanna E, Bèreau M, Garbaye J, Guehl JM (2004) Contrasting responses to mycorrhizal inoculation and phosphorus availability in seedlings of two tropical rainforest tree species. New Phytol 161:865–875
Dickson A, Leaf AL, Hosner JF (1960) Quality appraisal of white spruce and white pine seedling stock in forest nurseries. For Chronicle 36:10–13
Diem HG, Arahou M (1996) A review of cluster root formation: a primary strategy of Casuarinaceae to overcome soil nutrient deficiency. In: Pinyopusarek K, Turnbull JW, Midgley SJ (eds) Recent Casuarina research and development, Proc. of the 3rd Int. Casuarina Workshop. Da Nang, Vietnam, pp 51–58
Duponnois R, Diédhiou S, Chotte JL, Ourey SM (2003) Relative importance of the endomycorrhizal and (or) ectomycorrhizal associations in Allocasuarina and Casuarina genera. Can J Bot 49:281–287
Founoune H, Duponnois R, Bâ AM, El Bouami F (2002) Influence of the dual arbuscular endomycorrhizal/ectomycorrhizal symbiosis on the growth of Acacia holosericea (A. Cunn. ex G. Don) in glasshouse conditions. Ann For Sci 59:93–98
Garbaye J (1994) Helper bacteria: a new dimension to the mycorrhizal symbiosis. New Phytol 128:197–210
Gehring CA (2003) Growth responses to arbuscular mycorrhizal by rainforest seedlings vary with light intensity and tree species. Plant Ecol 167:127–139
Gray JT, Schlesinger WH (1983) Nutrient use by evergreen and deciduous shrubs in southern California. J Ecol 71:43–56
Guissou T, Bâ AM, Ouadba JM, Guinko S, Duponnois R (1998) Response of Parkia biglobosa (Jacq.) Benth, Tamarindus indica L. and Zizyphus mauritiana Lam. to arbuscular mycorrhizal fungi in a phosphorus-deficient sandy soil. Biol Fertil Soils 26:194–198
Gupta N, Rahangdale R (1999) Response of Albizzia lebbeck and Dalbergia sissoo towards dual inoculation of Rhizobium and arbuscular mycorrhizal fungi. Indian J Exp Biol 37:1005–1011
He XH, Critchley C (2008) Frankia nodulation, mycorrhization and interactions between Frankia and mycorrhizal fungi in Casuarina plants. In: Varma A (ed) Mycorrhiza 3: state of the art, genetics and molecular biology, eco function, biotechnology, ecophysiology, structure and systematics. Springer-Verlag, Berlin, Germany, pp 767–781
Jackson ML (1971) Soil chemical analysis. Prentice Hall, New Delhi
Jakobsen I (1999) Transport of phosphorus and carbon in arbuscular mycorrhizas. In: Varma A, Hock B (eds) Mycorrhiza: structure, function, molecular biology and biotechnology, 2nd edn. Springer-Verlag, Berlin, Germany, pp 305–332
Jeffries P, Barea JM (1994) Biogeochemical cycling and arbuscular mycorrhizas in the sustainability of plant–soil systems. In: Gianinazzi S, Schüepp H (eds) Impact of arbuscular mycorrhizas on sustainable agriculture and natural ecosystems. Birkhäuser Verlag, Basel, Switzerland, pp 101–115
Koide RT, Goff MD, Dickie IA (2000) Component growth efficiencies of mycorrhizal and nonmycorrhizal plants. New Phytol 148:163–168
Koske RE, Gemma JN (1989) A modified procedure for staining roots to detect VA mycorrhizas. Mycol Res 92:486–488
Kothari SK, Marschner H, Romheld V (1991) Contribution of VAM hyphae in acquisition phosphorus and zinc by maize grown in a calcareous soil. Plant Soil 131:177–185
Kucey RMN, Leggett ME (1989) Microbial mediated increases in plant available phosphorus. Adv Agron 42:199–228
Lapeyrie F, Ranger J, Vairelles D (1991) Phosphate-solubilizing activity of ectomycorrhizal fungi in vitro. Can J Bot 69:342–346
Lesueur D, Duponnois R (2005) Relations between rhizobial nodulation and root colonization of Acacia crassicarpa provinces by an arbuscular mycorrhizal fungus Glomus intraradices Schenck and Smith or an ectomycorrhizal fungus Pisolithus tinctorius Coker & Couch. Ann For Sci 62:467–474
Mansour SR (2003) Improving wood and biomass production of some Casuarina species through symbiotic association in Egypt. NFT News 6:1–2
McGonigle TP, Miller MH, Evans DG, Fairchild GL, Swan JA (1990) A method which gives an objective measure of colonization of roots by vesicular–arbuscular mycorrhizal fungi. New Phytol 115:495–501
Muthukumar T, Udaiyan K (2002) Arbuscular mycorrhizal fungal composition in semi-arid soils of Western Ghats, southern India. Curr Sci 82:624–628
Muthukumar T, Udaiyan K (2006) Growth of nursery-grown bamboo inoculated with arbuscular mycorrhizal fungi and plant growth promoting rhizobacteria in two soil types with and without fertilizer application. New For 31:469–485
Muthukumar T, Udaiyan K, Rajeshkannan V (2001) Response of neem (Azadirachta indica A. Juss) to indigenous arbuscular mycorrhizal fungi, phosphate-solubilizing and symbiotic nitrogen-fixing bacteria under tropical nursery conditions. Biol Fertil Soils 34:417–426
Olsen SR, Cole CE, Watanabe FS, Dean LA (1954) Estimation of available phosphorus in soil by extraction with sodium bicarbonate. USDA Circ 939:1–9
Ortas I (2003) Effect of selected mycorrhizal inoculation on phosphorus sustainability in sterile and non-sterile soils in the Harran Plain in South Anatolia. J Plant Nutr 26:1–17
Osorio NW, Habte M (2001) Synergistic influence of an arbuscular mycorrhizal fungus as a P solubilizing fungus on growth and P uptake of Leucaena leucocephala in an Oxisol. Arid Land Res Manage 15:263–274
Piccini DF, Azcón R (1987) Effect of phosphate-solubilizing bacteria and vesicular–arbuscular mycorrhiza (VAM) on the utilization of bayoran rock phosphate by alfalfa plants using a sand vermiculate medium. Plant Soil 101:45–50
Pikovskaya RI (1948) Mobilization of phosphorus in soil in connection with vital activity of some microbial-species. Mikrobiologia 17:362–370
Piper CS (1950) Soil and plant analysis. Interscience Publications, New York
Porter WM (1979) The most probable number method for enumerating infective propagules of vesicular–arbuscular mycorrhizal fungi in soil. Aust J Soil Res 17:515–519
Pramanik K, Singh RK (2004) Effect of levels and mode of phosphorus and biofertilizers on chickpea (Cicer arietinum) under dry land conditions. Indian J Agron 48:294–296
Rajendran K, Devaraj P (2004) Biomass and nutrient distribution and their return of Casuarina equisetifolia inoculated with biofertilizers in farm land. Biomass Bioenergy 26:235–249
Rajendran K, Sugavanam V, Devaraj P (2003) Effect of microbial inoculation on quality seedling production of Casuarina equisetifolia. J Trop For Sci 15:82–96
Raju PS, Clark RB, Ellis JR (1990) Effects of species of VAM fungi on growth and mineral uptake of sorghum at different temperatures. Plant Soil 121:165–170
Ravikumar R, Ananthakrishnan G, Appasamy T, Ganapathi A (1997) Effect of endomycorrhizae (VAM) on bamboo seedling growth and biomass productivity. For Ecol Manage 98:205–208
Reddell P, Rosbrook PA, Bowen GO, Gwale D (1988) Growth response in Casuarina cunninghamiana plantings inoculation with Frankia. Plant Soil 108:76–86
Ritchie GA (1984) Assessing seedling quality. In: Duryea ML, Landis TD (eds) Forest nursery manual: production of bareroot seedlings. Martinus Nijhoff/Dr W. Junk, The Hague, pp 243–259
Rodrìguez H, Fraga R (1999) Phosphate solubilizing bacteria and their role in plant growth promotion. Biotechnol Adv 17:319–339
Rojas NS, Li CY, Perry DA, Ganio LM (2001) Frankia and nodulation of red alder and snowbrush grown on soils from Douglas-fir forests in the H.J. Andrews Experimental Forest of Oregon. Appl Soil Ecol 17:141–149
Rojas NS, Perry DA, Li CY, Ganio LM (2002) Interactions among soil biology, nutrition, and performance of actinorhizal plant species in the H.J. Andrews Experimental Forest of Oregon. Appl Soil Ecol 19:13–26
Rousseau JVD, Sylvia DM, Fox AJ (1994) Contribution of ectomycorrhiza to the potential nutrient-absorbing surface of pine. New Phytol 128:639–644
Sambandan K, Kannan K, Raman N (1994) Vesicular-arbuscular mycorrhizae of Casuarina equisetifolia Forst. in four different soil types in Tamil Nadu. Indian For 120:510–514
Sanginga N, Danso SKA, Bowen GD (1989) Nodulation and growth response of Allocasuarina and Casuarina species to phosphorus fertilization. Plant Soil 118:125–132
Singh HP, Singh TA (1993) The interaction of rockphosphate, Bradyrhizobium, vesicular-arbuscular mycorrhizae and phosphate-solubilizing microbes on soybean grown in a sub-Himalayan mollisol. Mycorrhiza 4:37–43
Stribley DP, Tinker PB, Rayner JH (1980) Relation of internal phosphorus concentration and plant weight in plants infected by vesicular-arbuscular mycorrhizas. New Phytol 86:261–266
Subba Rao NS, Rodriguez Barrueco C (1995) Casuarinas. Oxford and IBH, New Delhi, India
Tian C, He X, Zhong Y, Chen J (2002) Effects of VA mycorrhizae and Frankia dual inoculation on growth and nitrogen fixation of Hippophae tibetana. For Ecol Manage 170:307–312
Tiwari M, Singh SP, Tiwari A, Sundriyal RC (2003) Effect of symbiotic associations on growth of host Coriaria nepalensis and its facilitative impact on oak and pine seedlings in Central Himalaya. For Ecol Manage 184:141–147
Toro M, Azcón R, Barea JM (1997) Improvement of arbuscular mycorrhiza development by inoculation of soil with phosphate-solubilizing rhizobacteria to improve rock phosphate bioavailability (32P) and nutrient cycling. Appl Environ Microbiol 63:4408–4412
Vasanthakrishna M, Bagyaraj DJ, Nirmalnath PJ (1994) Responses of Casuarina equisetifolia to inoculation with Glomus fasciculatum and/or Frankia. For Ecol Manage 68:399–402
Vasanthakrishna M, Bagyaraj DJ, Nirmalnath PJ (1995) Selection of efficient VAM fungi for Casuarina equisetifolia second screening. New For 121:157–162
Walker RB, Chowdappa P, Gessel SP (1993) Major element deficiencies in Casuarina equisetifolia. Fert Res 34:127–133
Wall LG, Huss Danell K (1996) Phosphorus and nitrogen fixation modulate auto regulation of nodulation in Alnus incana-Frankia symbiosis. Plant Physiol 111:315
Wheeler CT, Hollingsworth MK, Hooker JE, McNeill JD, Mason WI, Moffat AJ, Sheppard LJ (1991) The effect of inoculation with either cultured Frankia or crushed nodules on nodulation and growth of Alnus rubra and Alnus glutinosa seedlings in forest nurseries. For Ecol Manage 43:153–166
Wheeler CT, Tilak M, Scrimgeour CM, Hooker JE, Handley LL (2000) Effects of symbiosis with Frankia and arbuscular mycorrhizal fungus on the natural abundance of 15N in four species of Casuarina. J Exp Bot 343:287–297
Williams RF (1946) The physiology of plant growth with special reference to the concept of net assimilation rate. Ann Bot 10:41–72
Yamanaka T, Li CY, Bormann BT, Okabe H (2003) Tripartite associations in an alder: effects of Frankia and Alpova diplophloeus on the growth, nitrogen fixation and mineral acquisition of Alnus tenuifolia. Plant Soil 254:179–186
Yamanaka T, Akama A, Li CY, Okabe H (2005) Growth, nitrogen fixation and mineral acquisition of Alnus sieboldiana after inoculation of Frankia together with Gigaspora margarita and Pseudomonas putida. J For Sci 10:21–26
Zaïd EH, Arahou M, Diem HG, Morabet RE (2003) Is Fe deficiency rather than P deficiency the cause of cluster root formation in Casuarina species? Plant Soil 248:229–235
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Muthukumar, T., Udaiyan, K. Growth response and nutrient utilization of Casuarina equisetifolia seedlings inoculated with bioinoculants under tropical nursery conditions. New Forests 40, 101–118 (2010). https://doi.org/10.1007/s11056-009-9186-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11056-009-9186-z