Abstract
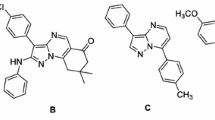
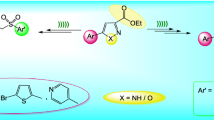
An environmentally friendly and mild Bischler cyclization was developed to access quinazolines with diverse substitution. Based on this method, a library of 53 quinazoline derivatives was prepared and tested in vitro for cytotoxicity and inhibition on T-cell and B-cell proliferation. Compounds 6b, 7b, 17b, 33, and 35 showed higher inhibitory activity on both T-cell and B-cell proliferations, with IC50 values of 6.16, 6.30, 5.43, 2.54, and 9.80 μM on T-cell, respectively. All the tested compounds showed no obvious cytotoxicity at 10 μM concentration. The preliminary structure–activity relationship was concluded revealing that 4-position is the key modification site for potent quinazoline immunosuppressive agent.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Italia JL, Bhardwaj V, Kumar MNVR (2006) Disease, destination, dose and delivery aspects of ciclosporin: the state of the art. Drug Discov Today 11: 846–854. doi:10.1016/j.drudis.2006.07.015
Sigal NH, Dumont FJ (1992) Cyclosporine-a, fk-506, and rapamycin—pharmacological probes of lymphocyte signal transduction. Annu Rev Immunol 10: 519–560. doi:10.1146/annurev.immunol.10.1.519
Clipstone NA, Crabtree GR (1992) Identification of calcineurin as a key signaling enzyme in lymphocyte-t activation. Nature 357: 695–697. doi:10.1038/357695a0
Wiederrecht G, Lam E, Hung S, Martin M, Sigal N (1993) The mechanism of action of fk-506 and cyclosporine-a. In: Allison AC (ed) Immunosuppressive and antiinflammatory drugs. Annals of the New York Academy of Sciences, New York, pp 9–19
O’Keefe SJ, O’Neill EA (1994) Cyclosporin A and FK-506: immunosuppression, inhibition of transcription and the role of calcineurin. Perspect Drug Discov Des 2: 85–102
Shaw T, Quan J, Totoritis MC (2003) B cell therapy for rheumatoid arthritis: the rituximab (anti-CD20) experience. Ann Rheum Dis 62: ii55–ii59. doi:10.1136/ard.62.suppl_2.ii55
Wai L-E, Fujiki M, Takeda S, Martinez OM, Krams SM (2008) Rapamycin, but not cyclosporine or FK506, alters natural killer cell function. Transplantation 85: 145–149. doi:10.1097/01.tp.0000296817.28053.7b
Smith JM, Nemeth TL, McDonald RA (2003) Current immunosuppressive agents: efficacy, side effects, and utilization. Pediatr Clin N Am 50: 1283–1288. doi:10.1016/s0031-3955(03)00121-4
Miller LW (2002) Cardiovascular toxicities of immunosuppressive agents. Am J Transplant 2: 807–818. doi:10.1034/j.1600-6143.2002.20902.x
Sheikh-Hamad D, Nadkarni V, Choi YJ, Truong LD, Wideman C, Hodjati R, Gabbay KH (2001) Cyclosporine A inhibits the adaptive responses to hypertonicity: a potential mechanism of nephrotoxicity. J Am Soc Nephrol 12: 2732–2741
Hojo M, Morimoto T, Maluccio M, Asano T, Morimoto K, Lagman M, Shimbo T, Suthanthiran M (1999) Cyclosporine induces cancer progression by a cell-autonomous mechanism. Nature 397: 530–534. doi:10.1038/17401
Liu G-B, Xu J-L, He C-C, Chen G, Xu Q, Xu H-X, Li J-X (2009) Synthesis and evaluation of a novel series of quinoline derivatives with immunosuppressive activity. Bioorg Med Chem 17: 5433–5441. doi:10.1016/j.bmc.2009.06.043
Guan J, Zhang Q, O’Neil M, Obaldia N, Ager A, Gerena L, Lin AJ (2005) Antimalarial activities of new pyrrolo 3,2-f quinazoline-1,3-diamine derivatives. Antimicrob Agents Chemother 49: 4928–4933. doi:10.1128/aac.49.12.4928-4933.2005
Alagarsamy V, Pathak US (2007) Synthesis and antihypertensive activity of novel 3-benzyl-2-substituted-3H- 1,2,4 triazolo 5,1-b quinazolin-9-ones. Bioorg Med Chem 15: 3457–3462. doi:10.1016/j.bmc.2007.03.007
Ma ZZ, Hano Y, Nomura T (2005) Luotonin A: a lead toward anti-cancer agent development. Heterocycles 65: 2203–2219
Bedi PMS, Kumar V, Mahajan MP (2004) Synthesis and biological activity of novel antibacterial quinazolines. Bioorg Med Chem Lett 14: 5211–5213. doi:10.1016/j.bmcl.2004.07.065
DeGrendele H (2003) Epidermal growth factor receptor inhibitors, gefitinib and erlotinib (Tarceva, OSI-774), in the treatment of bronchioloalveolar carcinoma. Clin Lung Cancer 5: 83–85
Liu G-B, Xu J-L, Geng M, Xu R, Hui R-R, Zhao J-W, Xu Q, Xu H-X, Li J-X (2010) Synthesis of a novel series of diphenolic chromone derivatives as inhibitors of NO production in LPS-activated RAW264.7 macrophages. Bioorg Med Chem 18: 2864–2871. doi:10.1016/j.bmc.2010.03.020
Teng P, Liu H-L, Deng Z-S, Shi Z-B, He Y-M, Feng L-L, Xu Q, Li J-X (2011) Synthesis and biological evaluation of unique stereodimers of sinomenine analogues as potential inhibitors of NO production. Bioorg Med Chem 19: 3096–3104. doi:10.1016/j.bmc.2011.04.006
Connolly DJ, Cusack D, O’Sullivan TP, Guiry PJ (2005) Synthesis of quinazolinones and quinazolines. Tetrahedron 61: 10153–10202. doi:10.1016/j.tet.2005.07.010
Bischler A, Barad D (1892) Zur Kenntniss der pheomiazinderivte. Berichte 25: 3080–3097
Schofield K, Swain T, Theobald RS (1952) The preparation of some α ω-Di-quinazolinylalkanes. J Chem Soc 1924–1926
Ferrini S, Ponticelli F, Taddei M (2007) Covenient synthetic approach to 2,4-disubstituted quinazolines. Org Lett 9: 69–72. doi:10.1021/ol062540s
Schofield K (1954) Miscellaneous quinazoline derivatives. J Chem Soc 4034–4035
Chandregowda V, Rao GV, Reddy GC (2007) Convergent approach for commercial synthesis of gefitinib and erlotinib. Org Process Res Dev 11: 813–816. doi:10.1021/op700054p
Schnur RC, Arnold LD (1998) U.S. patent, US5747498
Sagiv-Barfi I, Weiss E, Levitzki A (2010) Design, synthesis, and evaluation of quinazoline T cell proliferation inhibitors. Bioorg Med Chem 18: 6404–6413. doi:10.1016/j.bmc.2010.07.004
Zhang L, Geng M, Teng P, Zhao D, Lu X, Li J-X (2012) Ultrasound-promoted intramolecular direct arylation in a capillary flow microreactor. Ultrason Sonochem 19: 250–256. doi:10.1016/j.ultsonch.2011.07.008
Mamaghani M, Loghmanifar A, Taati MR (2011) An efficient one-pot synthesis of new 2-imino-1,3-thiazolidin-4-ones under ultrasonic conditions. Ultrason Sonochem 18: 45–48. doi:10.1016/j.ultsonch.2010.05.009
Cravotto G, Cintas P (2006) Power ultrasound in organic systhesis: moving cavitational chemistry from academia to innovative and large-scale applications. Chem Soc Rev 35: 180–196. doi:10.1039/b503848k
Smits RA, Adami M, Istyastono EP, Zuiderveld OP, van Dam CME, de Kanter FJJ, Jongejan A, Coruzzi G, Leurs R, de Esch IJP (2010) Synthesis and QSAR of quinazoline sulfonamides as highly potent human histamine H-4 receptor inverse agonists. J Med Chem 53: 2390–2400. doi:10.1021/jm901379s
Thurmond RL, Gelfand EW, Dunford PJ (2008) The role of histamine H-1 and H-4 receptors in allergic inflammation: the search for new antihistamines. Nat Rev Drug Discov 7: 41–53. doi:10.1038/nrd2465
Verlinden BK, Niemand J, Snyman J, Sharma SK, Beattie RJ, Woster PM, Birkholtz L-M (2011) Discovery of novel alkylated (bis)urea and (bis)thiourea polyamine analogues with potent antimalarial activities. J Med Chem 54: 6624–6633. doi:10.1021/jm200463z
Ghiron C, Haydar SN, Aschmies S, Bothmann H, Castaldo C, Cocconcelli G, Comery TA, Di L, Dunlop J, Lock T, Kramer A, Kowal D, Jow F, Grauer S, Harrison B, La Rosa S, Maccari L, Marquis KL, Micco I, Nencini A, Quinn J, Robichaud AJ, Roncarati R, Scali C, Terstappen GC, Turlizzi E, Valacchi M, Varrone M, Zanaletti R, Zanelli U (2010) Novel alpha-7 nicotinic acetylcholine receptor agonists containing a urea moiety: identification and characterization of the potent, selective, and orally efficacious agonist 1 6-(4-fluorophenyl)pyridin-3-yl −3-(4-piperidin-1-ylbutyl) urea (SEN34625/WYE-103914). J Med Chem 53: 4379–4389. doi:10.1021/jm901692q
Jiang JB, Hesson DP, Dusak BA, Dexter DL, Kang GJ, Hamel E (1990) Synthesis and biological evaluation of 2-styrylquinazolin-4(3h)-ones, a new class of antimitotic anticancer agents which inhibit tubulin polymerization. J Med Chem 33: 1721–1728. doi:10.1021/jm00168a029
Cohen SB (2010) Targeting the B cell in rheumatoid arthritis. Best Pract Res Clin Rheumatol 24: 553–563. doi:10.1016/j.berh.2009.11.006
Hirohata S, Yanagida T, Kawai M, Kikuchi H (1999) Inhibition of human B cell activation by a novel nonsteroidal anti-inflammatory drug, indometacin farnesil. Immunopharmacology 44: 245–254. doi:10.1016/s0162-3109(99)00084-3
Chen H, Du X, Tang W, Zhou Y, Zuo J, Feng H, Li Y (2008) Synthesis and structure–immunosuppressive activity relationships of bakuchiol and its derivatives. Bioorg Med Chem 16: 2403–2411. doi:10.1016/j.bmc.2007.11.054
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Zhang, L., Gao, Z., Peng, C. et al. Ultrasound-promoted synthesis and immunosuppressive activity of novel quinazoline derivatives. Mol Divers 16, 579–590 (2012). https://doi.org/10.1007/s11030-012-9390-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11030-012-9390-1