Abstract
Type 2 diabetes (T2D) is a metabolic disorder that can lead to memory impairment. T2D main features are insulin resistance and hyperglycemia. Physical exercise is a non-pharmacological intervention that can regulate glycemic levels and fight insulin resistance in T2D, but whether it influences memory has been discussed. There are 2 main types of physical exercise: aerobic exercise and resistance exercise. Here, we review about the consequences of different physical exercise protocols on memory in diabetic subjects and animal models of T2D. Physical exercise, aerobic or resistance training, most of the times, is a capable agent to prevent and treat memory loss on diabetic subjects and animal models of T2D. However, whether aerobic and resistance training combined improve memory in subjects with T2D remains controversial. Regarding animal models of T2D, aerobic and resistance training have been showed to be capable to prevent and treat memory loss. Acute and chronic protocols of exercise, generally, induce positive physiological responses and adaptations in T2D, such as a better glucose control. The ideal physical exercise protocol that will produce the best benefits to diabetic subjects and to animal models of T2D has not been described yet. A variety of combination between intensity, volume, frequency, and duration of the physical exercise protocol on future studies is necessary to both diabetic subjects and animal models of T2D to determine the best protocol that will induce more benefits on memory in T2D.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Type 2 diabetes (T2D) is a world health problem. T2D is a metabolic disorder and can lead to cognitive impairment (Bordier et al. 2014). Cognitive dysfunction and memory loss have been proven to be usual complications of T2D (Roriz-Filho et al. 2009). T2D issues with memory and cognition are more prevalent among the elderly (Bourdel-Marchasson et al. 2010). Cognition refers to the capacity of acquiring knowledge through thought and there are different perspectives to analyze the cognitive process (Stefano and Bianchini 2011), while memory refers to the brain capacity to encode, store and retrieve information (Atkinson and Shiffrin 1968). Besides this conceptual difference, cognition is often seen as part of the memory process. Changing lifestyle habits is the best way of preventing diabetes, such as through the adoption of performing exercise regularly (De Sousa et al. 2017; Roden 2012). Unveiling the epidemiological link between diabetes, cognitive decline, memory loss, dementia and lifestyle habits is an important public health challenge (Tuomilehto et al. 2001; Ferreira et al. 2018).
T2D main characteristic is insulin resistance, which occurs when there is the inhibition of the insulin receptors and/or its substrates, especially the insulin receptor substrate 2 (IRS-2), leading to hyperglycemia and hyperinsulinemia in the peripheral tissues (De Sousa 2018; De Sousa et al. 2020c). Insulin resistance can be induced by many different ways in animal models, such as using high-fat diet, drugs or using transgenic models (Busquets et al. 2019; De Sousa 2021). Ageing is considered to be one of the main risk factors for insulin resistance and T2D (Kitada et al. 2019). Insulin resistance occurs when there is a failure or reduced activation of phosphoidilinositol-3-kinase (PI3K) pathway (De Sousa 2017). Insulin hormone modulates synaptic plasticity, learning and memory, and if occurs any disturbances on insulin signaling pathway there would be an increased risk of developing cognitive decline and memory loss (Biessels and Kappelle 2005). Physical exercise can modulate the pattern of gene expression modifying several signaling pathways, and also can prevent and fight insulin resistance inhibiting cognitive function impairment and memory loss (Cassilhas et al. 2016; De Sousa 2017). Physical exercise is also capable to increase the phosphorylation of insulin receptors, which will favor the increasing of the activity of insulin pathways (De Sousa et al. 2020e). Physical exercise can inhibit negative changes in mood and behavior in humans (Cotman et al. 2007; Cassilhas et al. 2010) and animal models (Park et al. 2017; De Sousa et al. 2020d). However, little is known about the consequences of different physical exercise protocols on memory in human and animal models of T2D. Here, we review the consequences of physical exercise on memory in diabetic subjects and animal models of T2D.
Physical exercise
Aerobic exercise occurs when there is execution of exercises mainly with higher utilization of oxygen and recruitment of type I fibers, or slow twitch muscle fibers or red muscle fibers (Garber et al. 2011; Qaisar et al. 2016). Resistance exercise occurs when there is execution of exercises against any type of external resistance (i.e. individual's body mass, elastic resistance, water resistance, air resistance, etc.) with recruitment of type II fibers or fast twitch muscle fibers or white muscle fibers (Qaisar et al. 2016). Aerobic exercise enhances metabolic and cardiovascular functions, while resistance exercise enhances strength, and bone density (Soares and De Sousa 2013). Physical exercise can be performed at light, moderate, or high intensity presenting different results according to the intensity chosen (De Sousa 2017). Physical exercise can be also performed acute or chronically. It is considered to be acute exercise if the protocol is performed for no more than 4 weeks; sub-chronic between 4 and 8 weeks; and chronic exercise if the physical training is performed over 8 weeks.
It is known that the regular practice of physical exercise provides physiological benefits to subjects with T2D, such as better blood glucose and blood pressure control (De Sousa et al. 2020b). Animal models submitted to physical exercise training also reveal positive physiological effects, such as higher expression of peroxisome proliferator-activated receptor gamma coactivator 1-alpha (PGC-1α), reduction of inflammation and increment of mitochondrial membranes proteins (Botta et al. 2013). It has been showed that physical exercise can avoid the development of dementia in animal models of Alzheimer’s disease (Lourenco et al. 2019). Physical exercise also has showed benefits to subjects with mild cognitive impairment (Baker et al. 2010). However, if physical exercise leads to the development of a better memory or avoids memory loss or cognitive decline has been discussed (George and Hemachandra Reddy 2019).
Effects of physical exercise on memory in subjects with T2D
Espeland et al. evaluated non-demented men and women with T2D, aged 70–89 years, who were sedentary and who had functional limitations (Espeland et al. 2017). The physical exercise intervention consisted of walking, resistance training, and flexibility exercises. Each week, participants attended 2 center-based visits and perform home-based activity 3–4 times, with goals of 30 min of walking at moderate intensity, 10 min of primarily lower extremity strength training with ankle weights, 10 min of balance training, and large muscle group flexibility exercises. The intervention protocol lasted for 104 weeks. The cognitive function benefits occurred among participants with, but not without, diabetes. Physical exercise intervention improved the gait speed of older sedentary subjects with and without diabetes. Authors also suggest that the mechanisms through which physical activity affects physical and cognitive function in older adults may differ for subjects by diabetes status.
A recent study used aerobic and resistance training interventions combined to a weight loss plan and did not see any improvement in the memory of subjects with T2D (Stomby et al. 2017). This finding indicates that the usage of combined aerobic and resistance training to provide memory improvement, even when added to other interventions like a healthy diet, is still controversial. Further studies are necessary to determine where the combination of aerobic and resistance training are a good intervention to avoid dementia or improve memory in subjects with T2D.
Impressively, a study by Shellington et al. used a square-stepping exercise, what is considered to be a low-intensity program, and presented improvement of the executive function in subjects with T2D. We suggest that the usage of a memory task to realize the low-intensity program and the participants’ characteristics (men and women over 49 years with T2D and self-reported cognitive complaints) might have contributed to the improvement on memory. Another recent study evaluated an aerobic training intervention on men and women aged 50 to 75 years with T2D or metabolic syndrome (Olson et al. 2017). It is important to notice that the authors hypothesized that adopting and maintaining physical activity requires self-regulated behavior and repeatedly inhibiting habitual responses, such as playing video games or watching television and replacing them with healthy behaviors, such as going for a walk. Memory was improved in patients with T2D who exercised and got to self-regulate their behaviors by repeatedly inhibiting habitual responses.
Effects of physical exercise on memory in animal models of T2D
Animal models are mandatory to study acute and long-term consequences and molecular mechanisms in the brain (De Sousa et al. 2020e, d). A study by Cho et al., used OLETF rats, as a T2D model, and LETO rats as the control group (Cho et al. 2020). A vertical ladder, an equipment used to mimic resistance exercise, was used and the rats were trained to climb at 85° inclined position with weights corresponding initially to 50% of their body weights secured to their tails. At every trial extra 20 g was added. When the rat was able to climb 10 times with increasing weights, the session was considered complete. Exercise was used in combination with curcumin supplementation. Rats that exercised and received curcumin supplementation lost weight, improved cognition, glucose homeostasis and lipid profiles when compared with controls or just exercised rats.
On the other hand, the study of De Sousa et al. just used resistance training to reverse the cognitive decline seen in the rats induced to T2D by dexamethasone (0,5 mg/kg/day, i.p) (De Sousa et al. 2020a). The authors used a Tamaki machine, another equipment used to mimic resistance exercise, where rats were induced to perform squats by electrical stimulation. The training inhibited cognitive decline by stimulating higher activation of IRS-1 and reducing the activation of GSKβ. These findings together support the hypothesis that resistance training alone or combined with curcumin supplementation inhibit memory deficits in animal models of T2D.
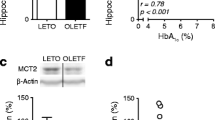
A recent study used Sprague–Dawley male rats and submitted them to 1 month of dietary high-fat diet (Shima et al. 2017). After one month a single dose of STZ (25 mg/kg) was given to induce insulin resistance on these rats. The T2D rats performed aerobic exercise at moderate-intensity on running wheel. Initially, animals were acclimatized for 4–5 days and trained to run on these wheels. The selected rats run 150 m/day (25–30 min), 5 days/week for 6 weeks. The authors indicated that aerobic exercise training could reverse diabetes-associated cognitive decline by reducing oxidative stress and inflammation in the brain of the rats induced to T2D. Finally, Shima et al., used the OLETF and LETO rats at 26 weeks of age to run on a treadmill at moderate intensity for 4 weeks (30 min/day, 5 days/week). The authors revealed that just 4 weeks of moderate-intensity aerobic exercise improves memory in a rat model of T2D. Glycogen levels were enhanced and MCT2 restored, which is a lactate transporter to neurons. Intriguingly, we did not find any studies in animal models of T2D that used the combination of the aerobic with the resistance training to evaluate memory.
Effects of physical exercise on memory in animal models and subjects with T2D: what have we learned?
Here, we have showed that just the animal studies evaluated molecular mechanisms of how aerobic and resistance training act on memory in T2D. To elucidate the molecular mechanisms by which memory is improved in animal models of T2D can be extremely relevant to science. Based on the results with the animal studies will allow us to investigate changes in the levels of specific proteins in different brain regions in humans. Thus, the molecular mechanisms through which physical exercise acts to prevent and treat cognitive decline and memory loss in T2D deserves further and deep investigation in diabetic subjects and also in animal models of T2D. Physical exercise seems to have, most of the times, positive effects on memory in T2D independently if it is evaluated in clinical studies or animal models. However, physical exercise protocols are not always capable of improving memory in diabetic individuals (Fig. 1).
Effects of physical exercise on memory in type 2 diabetes. Both types of physical exercise, aerobic and resistance training, improves memory and cognitive function in animal models of T2D. Most of the clinical studies also report beneficial effects of physical exercise to memory. However, it is not all physical exercise protocols that are capable of improving memory in diabetic individuals
There are many other aspects regarding the possibilities of doing research about the effects of physical exercise on memory in T2D, such as analyzing different populations through multiple techniques. For example, sex specific screening, evaluation and comparison is a must. It could be also subdivided into different age groups (60–65, 65–70, 70–75, 75–80, etc.). Using different techniques to study sex differences on different age groups would be extremely helpful, such as through case and cohort studies, randomized and clinical trials. Another example to be taken under consideration is previous conditions to T2D, such as being a smoker, alcohol consumption. Evaluating cellular and molecular mechanisms together with epidemiological studies on these different populations would bring greater light to better paths to fight T2D.
Conclusions
Physical exercise, aerobic or resistance training, most of the times, is a capable agent to prevent and treat memory loss on diabetic subjects and animal models of T2D. Wheter combined aerobic and resistance training can improve memory in subjects with T2D remains controversial. Regarding animal models of T2D, aerobic and resistance training are capable to prevent and treat memory loss. Acute and chronic protocols of exercise induce positive physiological responses and adaptations. Future studies combining both protocols, aerobic and resistance training, in animal models of T2D can contribute to find better ways to apply this combination to human studies, once the results with humans are still controversial. Different combinations between intensity, volume, frequency, and duration of the physical exercise protocols are necessary to future studies in both, diabetic subjects and animal models of T2D.
Data statement
Data sharing not applicable to this article as no datasets were generated or analysed during the current study.
References
Atkinson RC, Shiffrin RM (1968) Human Memory: A proposed system and its control processes BT - The Psychology of Learning and Motivation. Psychol Learn Motiv 2:89–195
Baker LD, Frank LL, Foster-Schubert K et al (2010) Effects of aerobic exercise on mild cognitive impairment: A controlled trial. Arch Neurol 67:71–79. https://doi.org/10.1001/archneurol.2009.307
Biessels GJ, Kappelle LJ (2005) Increased risk of Alzheimer’s disease in Type II diabetes : insulin resistance of the brain or insulin-induced amyloid pathology? Proteins Dis 1041–1044
Bordier L, Doucet J, Boudet J, Bauduceau B (2014) Update on cognitive decline and dementia in elderly patients with diabetes. Diabetes Metab 40:331–337. https://doi.org/10.1016/j.diabet.2014.02.002
Botta A, Laher I, Beam J et al (2013) Short Term Exercise Induces PGC-1α, Ameliorates Inflammation and Increases Mitochondrial Membrane Proteins but Fails to Increase Respiratory Enzymes in Aging Diabetic Hearts. PLoS One 8 https://doi.org/10.1371/journal.pone.0070248
Bourdel-Marchasson I, Lapre E, Laksir H, Puget E (2010) Insulin resistance, diabetes and cognitive function: Consequences for preventative strategies. Diabetes Metab 36:173–181. https://doi.org/10.1016/j.diabet.2010.03.001
Busquets O, Ettcheto M, Eritja À et al (2019) c-Jun N-terminal Kinase 1 ablation protects against metabolic-induced hippocampal cognitive impairments. J Mol Med 97:1723–1733. https://doi.org/10.1007/s00109-019-01856-z
Cassilhas RC, Antunes HKM, Tufik S, de Mello MT (2010) Mood, Anxiety, and Serum IGF-1 in Elderly Men Given 24 Weeks of High Resistance Exercise. Percept Mot Skills 110:265–276. https://doi.org/10.2466/pms.110.1.265-276
Cassilhas RC, Tufik S, De Mello MT (2016) Physical exercise, neuroplasticity, spatial learning and memory. Cell Mol Life Sci 73:975–983. https://doi.org/10.1007/s00018-015-2102-0
Cho JA, Park SH, Cho J et al (2020) Exercise and curcumin in combination improves cognitive function and attenuates ER stress in diabetic rats. Nutrients 12:1–14. https://doi.org/10.3390/nu12051309
Cotman CW, Berchtold NC, Christie L-A (2007) Exercise builds brain health: key roles of growth factor cascades and inflammation. Trends Neurosci 30:464–472. https://doi.org/10.1016/j.tins.2007.06.011
De Sousa RAL (2017) Brief report of the effects of the aerobic, resistance, and high-intensity interval training in type 2 diabetes mellitus individuals Diabetes mellitus. Int J Diabetes Dev Ctries 38:138–145. https://doi.org/10.1007/s13410-017-0582-1
De Sousa RAL, Torres YS, Figueiredo CPCP et al (2017) Consequences of gestational diabetes to the brain and behavior of the offspring. An Acad Bras Cienc 90:2279–2291. https://doi.org/10.1590/0001-3765201720170264
De Sousa RAL (2018) Gestational diabetes is associated to the development of brain insulin resistance in the offspring. Int J Diabetes Dev Ctries 39:408–416. https://doi.org/10.1007/s13410-018-0618-1
De Sousa RAL, Caria ACI, De Jesus Silva FM et al (2020a) High-intensity resistance training induces changes in cognitive function, but not in locomotor activity or anxious behavior in rats induced to type 2 diabetes. Physiol Behav 223:1–7. https://doi.org/10.1016/j.physbeh.2020.112998
De Sousa RAL, Hagenbeck KF, Arsa G, Pardono E (2020b) Moderate / high resistance exercise is better to reduce blood glucose and blood pressure in middle-aged diabetic subjects. Rev Bras Educ Física e Esporte 34:165–175
De Sousa RAL, Harmer AR, Freitas DA et al (2020c) An update on potential links between type 2 diabetes mellitus and Alzheimer’s disease. Mol Biol Rep. https://doi.org/10.1007/s11033-020-05693-z
De Sousa RAL, Peixoto MFD, Leite HR et al (2020d) Neurological consequences of exercise during prenatal Zika virus exposure to mice pups. Int J Neurosci 1https://doi.org/10.1080/00207454.2020.1860970
De Sousa RAL, Rodrigues CM, Mendes BF et al (2020e) Physical exercise protocols in animal models of Alzheimer ’s disease: a systematic review. Metab Brain Dis 1–11. https://doi.org/10.1007/s11011-020-00633-z
De Sousa RAL (2021) Animal models of gestational diabetes: characteristics and consequences to the brain and behavior of the offspring. Metab Brain Dis 2014–2019
Espeland MA, Lipska K, Miller ME et al (2017) Effects of physical activity intervention on physical and cognitive function in sedentary adults with and without diabetes. J Gerontol A Biol Sci Med Sci 72:861–866. https://doi.org/10.1093/gerona/glw179
Ferreira LSS, Fernandes CS, Vieira MNN, De-Felice FG (2018) Insulin resistance in Alzheimer’s disease. Front Neurosci 12:1–11. https://doi.org/10.1016/j.trsl.2016.12.005
Garber CE, Blissmer B, Deschenes MR et al (2011) Quantity and quality of exercise for developing and maintaining cardiorespiratory, musculoskeletal, and neuromotor fitness in apparently healthy adults: Guidance for prescribing exercise. Med Sci Sports Exerc 43:1334–1359. https://doi.org/10.1249/MSS.0b013e318213fefb
George EK, Hemachandra Reddy P (2019) Can healthy diets, regular exercise, and better lifestyle delay the progression of dementia in elderly individuals? J Alzheimer’s Dis 72:S37–S58. https://doi.org/10.3233/JAD-190232
Kitada M, Ogura Y, Monno I, Koya D (2019) Sirtuins and Type 2 Diabetes: Role in Inflammation, Oxidative Stress, and Mitochondrial Function. Front Endocrinol (Lausanne) 10:1–12. https://doi.org/10.3389/fendo.2019.00187
Lourenco MV, Frozza RL, de Freitas GB et al (2019) Exercise-linked FNDC5/irisin rescues synaptic plasticity and memory defects in Alzheimer’s models. Nat Med 165–175.https://doi.org/10.1038/s41591-018-0275-4
Olson EA, Mullen SP, Raine LB et al (2017) Integrated social- and neuro-cognitive model of physical activity behavior in older aduts with metabolic disease. Physiol Behav 176:139–148. https://doi.org/10.1016/j.physbeh.2017.03.040
Park H, Lee J, Cho H et al (2017) Physical exercise ameliorates mood disorder-like behavior on high fat diet- induced obesity in mice. Psychiatry Res 250:71–77. https://doi.org/10.1016/j.psychres.2017.01.012
Qaisar R, Bhaskaran S, Van Remmen H (2016) Muscle fiber type diversification during exercise and regeneration. Free Radic Biol Med 98:56–67. https://doi.org/10.1016/j.freeradbiomed.2016.03.025
Roden M (2012) Exercise in type 2 diabetes: to resist or to endure? Diabetologia 55:1235–1239. https://doi.org/10.1007/s00125-012-2513-5
Roriz-Filho JS, Sá-Roriz TM, Rosset I et al (2009) (Pre)diabetes, brain aging, and cognition. Biochim Biophys Acta - Mol Basis Dis 1792:432–443. https://doi.org/10.1016/j.bbadis.2008.12.003
Shima T, Matsui T, Jesmin S et al (2017) Moderate exercise ameliorates dysregulated hippocampal glycometabolism and memory function in a rat model of type 2 diabetes. Diabetologia 60:597–606. https://doi.org/10.1007/s00125-016-4164-4
Soares FHR, de Sousa MBC (2013) Different types of physical activity on inflammatory biomarkers in women with or without metabolic disorders: a systematic review. Women Health 53:298–316. https://doi.org/10.1080/03630242.2013.782940
Stefano F, Bianchini F (2011) On the historical dynamics of cognitive science: a view from the periphery. Early mechanisms and new Ideas, the search for a theory of cognition
Stomby A, Otten J, Ryberg M et al (2017) A paleolithic diet with and without combined aerobic and resistance exercise increases functional brain responses and hippocampal volume in subjects with type 2 diabetes. Front Aging Neurosci 9:1–10. https://doi.org/10.3389/fnagi.2017.00391
Tuomilehto J, Lindstrom J, Eriksson JG et al (2001) Prevention of type 2 diabetes mellitus by changes in lifestyle among subjects with impaired glucose tolerance. N Engl J Med 344:1343–1350
Funding
Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), Brazil—Finance Code 001.
Author information
Authors and Affiliations
Contributions
RALS wrote the manuscript; performed the literature research; analyzed and critically discussed the data; RCC analyzed and critically discussed the data; ACIC analyzed and critically discussed the data. The authors read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Conflicts of interest
The author declares no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
De Sousa, R.A.L., Improta-Caria, A.C. & Cassilhas, R.C. Effects of physical exercise on memory in type 2 diabetes: a brief review. Metab Brain Dis 36, 1559–1563 (2021). https://doi.org/10.1007/s11011-021-00752-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-021-00752-1