Abstract
Context
The classical theory of island biogeography explains loss of species in fragmented landscapes as an effect of remnant patch size and isolation. Recently this has been challenged by the habitat amount and habitat continuum hypotheses, according to which persistence in modified landscapes is related to total habitat amount rather than habitat configuration or the ability of species to use all habitats to varying degrees. Distinguishing between these theories is essential for effective conservation planning in modified landscapes.
Objective
Identify which factors of habitat type, amount and configuration predict the persistence of a keystone woodland specialist, the eastern bettong Bettongia gaimardi, in a fragmented landscape.
Method
In the Midlands region of Tasmania we carried out camera surveys at 62 sites in summer and winter. We included habitat and landscape features to model whether habitat amount or patch size and isolation influenced the presence of the eastern bettong, and to measure effects of habitat quality.
Results
Habitat amount within a 1 km buffer was a better predictor of occupancy than patch size and isolation. Occupancy was also affected by habitat quality, indicated by density of regenerating stems.
Conclusion
Our results support the habitat amount hypothesis as a better predictor of presence. For a species that is able to cross the matrix between remnant patches and utilise multiple patches, the island biogeography concept does not explain habitat use in fragmented landscapes. Our results emphasize the value of small remnant patches for conservation of the eastern bettong, provided those patches are in good condition.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Conversion of native vegetation for agriculture is a major and escalating threat to biodiversity globally (Tittensor et al. 2014; Newbold et al. 2015; Maxwell et al. 2016; Venter et al. 2016). Agricultural land covers 38% of the land surface of the earth (Kehoe et al. 2017). The conversion of land for agriculture has resulted in the widespread loss or reduction of contiguous native vegetation across all habitat types, including forests, grasslands and wetlands (Sodhi and Ehrlich 2010; Johnston 2013; Haddad et al. 2015). The socio-economic value of agricultural production (Venter et al. 2016) and growing human population will result in continuing expansion and intensification of agriculture, coupled with increased infrastructure and global human footprint (Lindenmayer and Fischer 2013; Laurance et al. 2014; Venter et al. 2016). Species that persist in largely cleared agricultural landscapes are often restricted to remnant fragments of habitat, which are smaller and may have undergone significant ecological modifications from their original state (Fahrig 2007; Fischer 2007). Understanding how biodiversity responds to the stressors of landscape modification, including decreased quality, fragmentation and loss of habitat, is important for managing persistence of biodiversity in agricultural landscapes.
Identifying features of fragmented agricultural landscapes that influence the persistence of the original native species is a significant research focus. Much effort has focussed on the effects of size and isolation of remnant patches of habitat (Haila 2002). This research was grounded in the equilibrium theory of island biogeography and the related theory of metapopulation ecology. These hypotheses assume that the target native species are unable to occupy the matrix that surrounds habitat remnants and predict that species richness increases with patch area and decreases with patch isolation (Fahrig 2002; Krauss et al. 2004; Laurance 2008). Patch size and isolation have indeed been reported to be important for species persistence, functional diversity and the retention of ecosystem functions in many taxa as seen in plants, invertebrates, amphibians and birds (Sahlin and Schroeder 2010; Sodhi and Ehrlich 2010; Mönkkönen et al. 2014; Munguía-Rosas and Montiel 2014; Magioli et al. 2015; Almeida-Gomes et al. 2016; Seahra et al. 2016).
Recent studies have questioned the relevance of this framework for species that are able to use multiple remnant patches of habitat. This is true of many species that are primarily dependent on forests and woodlands (Anderson et al. 2007; Ditmer et al. 2015; Ripperger et al. 2015). For such species, persistence in a given landscape may depend on the total amount and the quality of preferred habitat that remains, as distinct from the particular configuration of patches that determines the pattern of size and isolation (Fahrig 2013, 2017). There are two alternative hypotheses to explain species use of heterogeneous landscapes: the habitat amount and the habitat continuum hypotheses. The habitat amount hypothesis proposes the effects of fragmentation are due to a loss of available habitat. Species richness and abundance should increase with increasing total amount of habitat surrounding a site of interest, regardless of patch size and isolation. Instead, habitat amount should replace the effects of the latter (Fahrig 2013). This hypothesis has been supported by some empirical studies (Püttker et al. 2011; Melo et al. 2017; Seibold et al. 2017), but is not universal in its explanatory capacity. The amount of habitat explained species richness of small mammals in fragmented savannahs in Brazil, rather than patch size and isolation (Melo et al. 2017). Conversely, the density and species richness of plants in small fragments of grassland and birds in orchards was better explained by the island biogeography theory rather than by habitat amount (Bailey 2012; Lindgren and Cousins 2017; Torrenta and Villard 2017). Alternatively, the continuum hypothesis highlights all habitats can be used regardless of configuration. Instead, species distribution is driven by ecological processes (i.e.climate, latitude, food, shelter), and therefore occupancy occurs at a gradient within a landscape (Fischer et al. 2006). For example, species richness and composition of lizards gradually varied across a fragmented landscape, explained by a variety of variables such as climate, amount of food and space rather than site area or amount of cover (Fischer et al. 2005). Identifying which of the mechanisms posited by these theories is operating in fragmented landscapes is essential for management of biodiversity in such landscapes.
Quality of available habitat affects persistence of species in fragmented landscapes because the extent to which vegetation patches provide food and shelter will influence population vital rates, such as survival and reproduction, and therefore abundance at a local and landscape scale (Hanski 2015). While all patches in a landscape, including native remnants and the agricultural matrix, represent potential habitat (the habitat continuum hypothesis), habitat quality will vary greatly depending on a species’ habitat requirements, niche breadth and the extent of impacts that degrade the native remnants (Battin 2004; Mortelliti et al. 2010). Furthermore, some features of modified landscapes can act as barriers to movement (Ascensão et al. 2017) or cause elevated mortality during movement through the matrix (D’Amico et al. 2015; Ruiz-Capillas et al. 2015), and therefore limit the ability of species to gain access to remnant habitat.
In this study, we assess the pattern of occurrence in a fragmented agricultural region of a medium-sized marsupial, the eastern bettong (Bettongia gaimardi; Family Potoroidae). It is a solitary species, with an average home range of 0.9 km2 (Gardiner et al. in review). The eastern bettong is thought to be an ecosystem engineer because, like other potoroid marsupials, it modifies soil structure and fertility in the course of digging for its food (Fleming et al. 2014). It is also an ecological specialist, being largely dependent on sporocarps of ectomycorrhizal fungi that associate with eucalypts, acacias, and other sclerophyll woodland plants (Taylor 1992, 1993a). Potoroid marsupials are shown to be sensitive to patch size in fragmented landscapes (Bennett 1990), and have suffered major declines due to the impacts of invasive predators and habitat loss (Woinarski et al. 2004; Johnson 2006). Eastern bettongs have become extinct on the mainland of Australia but survive on the island state of Tasmania, where they are distributed across the drier eastern half of the island. Within this area, their occupancy of the landscape is quite heterogeneous and they have declined dramatically in parts of their range that have been fragmented for agriculture.
We focus our study on the Midlands bioregion of Tasmania, one of Australia’s Biodiversity Hotspots. It was originally dominated by native grasslands and dry sclerophyll woodlands, but more than 200 years of intensive agricultural production has resulted in conversion of approximately 90% of the original vegetation communities to pasture for sheep and cattle. Remnants are heavily modified by grazing and fire. Furthermore, the feral cat (Felis catus), is an abundant invasive predator across the state. It is a threat to native mammals, both as a predator and as the definitive primary host for the pathogen Toxoplasmosis gondii to which many marsupials have little resistance (Spencer et al. 2016; Doherty et al. 2017).
We use occupancy modelling to determine the landscape and local-scale attributes that influence presence of the eastern bettong in remnant woodland in the fragmented agricultural landscapes of the Tasmanian Midlands. In particular, we test whether patch size and isolation or habitat amount were more important in determining the species’ response to fragmentation. We considered the habitat continuum theory to not be as relevant to eastern bettongs, as they don’t extensively use the matrix as primary habitat. We also test whether disturbances are important in predicting the presence of bettongs. Road mortality from collisions with vehicles, pivot irrigation near water sources, and urban settlements are common in the Midlands bioregion. Lastly, as bettongs are prey for both native and invasive mammalian predators, we test whether the presence of predators—the Tasmanian devil (Sarcophilus harrisi), spotted tailed quoll (Dasyurus maculatus) and feral cat (Felis catus)—influence occupancy and detection of the eastern bettong.
Methods
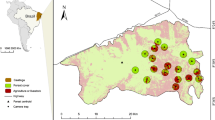
The Midlands bioregion is in the dry, lowland (below 500 m) parts of the catchments of the Macquarie and Derwent Rivers in central-east Tasmania, Australia. Native vegetation in the Midlands are now fragments in a sea of agriculture, with less than 10% of native woodland and less than 3% of native grassland remaining (Jones and Davidson 2016). We used camera traps (ReconyxTM PC800) to survey occupancy of mammals, including eastern bettongs, at a landscape scale across 7660 km2 of the Midlands bioregion of Tasmania. Two five-week surveys were carried out, one in the austral winter between June and August 2015, and the second survey in the summer between January and February 2016.
Site selection
To select sites based on these broad vegetation categories, we used and combined the vegetation classifications in the state government GIS vegetation layer: TASVEG 3.0 (http://dpipwe.tas.gov.au/conservation/development-planning-conservation-assessment/planning-tools/monitoring-and-mapping-tasmanias-vegetation-(tasveg)/tasveg-the-digital-vegetation-map-of-tasmania). Much of the remnant native vegetation of the Midlands is on private property, is fragmented to differing degrees, and is unevenly distributed across the landscape. To represent the variability in patch size and isolation, we stratified woodland patches into three size categories—small (20–100 ha), medium (100–200 ha) and large (> 200 ha). We then filtered the large number of sites in each category by habitat condition, presence of shrubby or grassy understorey, and selected equal numbers of sites from each patch size category. We differentiated native grassland from pasture sites by their dominant plant species. Native grasslands were dominated by at least 70% of native grass species including Poa spp, Themeda, Microlina and others. Pasture sites were chosen within paddocks dominated by introduced grass species. To provide independence for each habitat type sampled, sites were separated by a minimum of 1 km, a distance too large for a bettong to cross a matrix, however all of our sites were separated than more than 1 km. We collated a total of 18 variables considered a priori to be ecologically relevant to the study species (Fig. 1). A preliminary analysis revealed that bettongs were never detected at sites in pasture, and were detected only twice at native grassland sites, both of which were close to or surrounded by woodland. Given the low detections in pasture and grassland habitats, the sites that were in pasture and grassland were removed from the analysis, which was re-run using only the woodland sites (n = 62).
Camera trapping and data handling
A total of 103 sites were selected to be geographically representative of the Midlands, including 62 sites in native woodland—the most common type of native vegetation remnant—20 sites in native grassland, and 21 sites in introduced pasture. Cameras were set in the middle of a patch, mounted on a vertical structure—a tree, fence post or wooden stake—approximately 1 m off the ground. A visual and an olfactory lure were placed 1.5 m in front of the camera, with the field of view of the camera targeted below the lure. The olfactory lure was a mixture of rolled oats, peanut butter, nut oil, sardines and tuna oil, placed on the ground in a PVC canister. The visual lure was a CD suspended from a tree at a height of 1 m, a method used to attract species attention (Algar et al. 2007). Cameras were set to take a rapid-fire sequence of 3 consecutive pictures with a ‘no delay’ setting when the infra-red sensor was triggered. Images were processed in ExifPro software (http://www.exifpro.com/). All animals captured in images were identified to species, if possible, and recorded with time of capture, for each survey night and site location. Images were used to construct a binary detection history; given low detections and lack of unique identifying features of bettongs we pooled detections. One or more detections within a week was scored as 1, and lack of detections were scored as 0. Images in which the species could not be confidently identified were discarded, as were false triggers. If cameras failed during the sampling period, the weeks of lost data were classed as ‘NA’, equivalent to not surveyed.
Landscape-scale variables
Landscape-scale variables were mapped and measured using TASVEG 3.0 in ArcGIS 10.2. These included patch size, patch isolation (Euclidean distance to closest woodland), distance to nearest drainage line or water body (wetland, farm dam), distance to nearest road, and the proportion of native woodland, native grassland, plantation (native restoration plantings; mostly Eucalyptus spp.), agriculture and urban area within a 1 km radius (buffer) of the camera site (Table 1). This buffer size is approximately equal to the typical home-range area of an eastern bettongs as reported by Taylor (1993b) and Gardiner et al. (in review).
Local-scale variables
Local-scale variables centred on the camera site were measured on 2 × 50 m transects that formed a cross with the camera site at the intersection (Table 1). Within the area of the transect, we estimated the percent cover of native vegetation in the over-storey (> 5 m), mid-storey (1–5 m) and shrub (0.2–1 m) layers. To assess how bettongs might use vegetation structure to detect and evade predators, we measured horizontal visibility (VVS) five meters from the camera at each cardinal point. Visibility was measured using a 1 m2 white sheet divided into units, held by an assistant and touching the ground. We recorded the percentage of the sighting sheet that was visible, i.e. not obscured by vegetation at each of the heights 0–25, 26–50, 51–75, 76–100 cm off the ground. For this study on bettongs, we used the average of the percentage measures at the 0–25 cm height, which is approximately the height of a bettong, at which it can visually survey its surroundings for danger. To provide an indicator of woodland health, we counted the total number of regenerating stems of Eucalyptus species (including seedlings, sprouts and saplings) and the total number of vegetation life forms (excluding invasive and perennial species) within 5 m of each transect. Greater numbers of regenerating stems have been suggested to indicate healthy woodland remnants with good recruitment (Bailey 2012). To assess the potential influence of predators on the occupancy of bettongs at a site, we included a matrix of detections for the spotted tail quoll and feral cat as a parameter. The Tasmanian devil was not included due to the significant decline of the species across the landscape.
Data handling and occupancy modelling
All analyses were run in R Studio version 3.2.1 using package ‘unmarked’ (Fiske and Chandler 2011). Occupancy was determined using a single-species, multi-season model while accounting for detection probability, as described by MacKenzie et al. (2003). We did not expect changes in colonisation and extinction within one year, therefore we kept these parameters constant throughout the analysis. Prior to modelling we performed a check of normality on all landscape and habitat variables and applied transformations (z- transformations for discrete variables and log transformations for continuous variables) if necessary. A check for collinearity was performed using variance inflation factors (VIF) and a visual pairwise comparison of variables. None of the variables showed high collinearity (VIF = > 3) and therefore all were retained for analysis.
First, we built and ranked models to determine factors that influenced detection probability of bettongs on any given night at a site. We ran models with null, single and combinations of the detection covariates including the age of the bait (starting at one day and increasing in daily increments until the end of the survey) season (winter and summer), and detections of quolls and cat at each survey while keeping occupancy constant. We ranked models using Akaike Information Criterion (AIC) and examined all models that were within 2 AIC units of the model with the lowest AIC value. Where there was more than one model in the candidate set, we chose the most parsimonious model (with the fewest parameters). The parameters in the best model were thereafter incorporated into all occupancy models.
We conducted the analysis in two steps. First, we constructed models specifically to test and compare the two hypotheses regarding response to landscape fragmentation that are relevant to eastern bettongs: patch size and isolation, and habitat amount. To test patch size and isolation, we built single models including patch area, and distance to nearest patch (isolation). To test habitat amount, we included the total amount of woodland within 1 km of the camera location, to ensure it incorporated the majority of a bettong’s home range. We also built models testing the total amount of other landscape and vegetation types within 1 km: total native vegetation, agriculture, plantation and urban area. Each model was compared and ranked to the null model (Table 2). The second step of the analysis was to include the variable within the best ranked model in all occupancy models. The rationale for including this variable, which was the amount of woodland habitat, was that initial analyses revealed such an over-riding importance of woodland habitat in describing the occupancy of bettongs that any influence of other variables was not described. We built models of single variables (in addition to the variable from the best-ranked model in the first step), followed by more complex models with combinations of variables, and ranked these using Akaike Information Criterion. Model fit to the data was assessed using McKenzie and Bailey goodness of fit within the AICModavg package (Mazerolle and Mazerolle 2017) using 1000 bootstrap samples to determine which model best-supported bettong presence in woodland habitats. We calculated the odds ratios (OR) for each variable to determine the association between presence and each of the variables (Supplementary Table 1).
Results
Eastern bettongs were only detected 67 times at a total of 17 of the 62 woodland sites over the two camera surveys. The detection model that included bait age was the best-performing model compared with models including single or combinations of season, feral cats, quolls and or the null model (Table 3). The probability of detecting a bettong decreased as bait age increased. A detection model including bait age was subsequently included in all occupancy models.
The amount of woodland habitat in a 1 km radius of the camera site was the only model of the eight single parameter models run in step one of the analysis that described the relationship of occupancy to habitat amount and distribution (Table 2). Occupancy of bettongs was positively related to amount of woodland habitat within that radius. All other models were at least 6 AIC values more than the best model suggesting that patch size and patch isolation were not as important in predicting presence as the amount of woodland (Fig. 2). Similarly the amount of pasture, plantation and native grassland within the buffer were not important predictors; all showed trends of decreasing presence of bettongs with increasing amount of pasture, plantation, urban settlement and native grassland within a 1 km buffer of the camera location.
When the amount of woodland was retained in all models, there were two competing models in the final candidate set (within 2 AICs). The top model, which carried more than 50% AIC model weight, included the amount of woodland in the 1 km buffer and the number of regenerating stems. The second model additionally included plant species richness and had an AIC weight of 30% (Table 4). These models both indicated that the predicted presence of eastern bettongs was likely to increase as each variable subsequently increased. The next model was separated from the candidate set by more than 4 AIC values, indicating little influence on bettong occupancy of predator detections or other factors of landscape disturbance and environmental characteristics.
Discussion
We tested whether the occupancy of a dietary and habitat specialist, the eastern bettong, was better explained by the amount of habitat in the landscape (habitat amount hypothesis) or the configuration of patches of habitat (island biogeography and metapopulation theory). Bettongs do not explicitly occupy the matrix, but rather use it to traverse between patches and so did not explicitly test the habitat continuum hypothesis. The key result of our study showed bettong occupancy to be strongly predicted by the amount of woodland habitat within a radius comparable to the size of individual home ranges. This result suggests that eastern bettongs respond to landscape alteration by incorporating sufficient habitat into their home range, even if they have to move across pasture to reach several patches of woodland. Our results are consistent with the habitat amount hypothesis of species’ response to landscape fragmentation. We did not find any effect on bettong occupancy of the size of patches of woodland or the degree of isolation of remnant patches independent of habitat amount, indicating that the classical theory of island biogeography (or metapopulation ecology) was not useful in predicting the effects of habitat loss on this species in this region. We also found that a measure of habitat quality—stem density, indicating both the ongoing tree regeneration in woodland patches and availability of food for bettongs—was a useful predictor of habitat quality. That is, in this disturbed and fragmented habitat, small and isolated patches of remnant woodland are valuable in supporting the persistence of the eastern bettong provided those patches remain in good condition.
Habitat fragmentation will affect species differently due to their different dietary requirements and movement patterns. Generalists have a broader niche breadth, and are more likely to persist in modified landscapes, while specialists or species in high trophic positions and rarer species are more at risk of local extinction (Doherty et al. 2018; Martinson and Fagan 2014). Patterns of patch-occupation of species will also differ according to their movement capacity. Low innate movement capacity can result in species being isolated to a patch, and therefore more likely to be influenced by habitat patch size and isolation. This could explain why some studies that have found stronger effects of patch size and isolation have been of plants and arthropods (Bullock et al. 2002; Ghazoul 2005; Evju and Sverdrup-Thygeson 2016; Haddad et al. 2017). Larger ranging species are more able to reach multiple patches of habitat within a landscape to find the resources they need (Gastón et al. 2016; Fattebert et al. 2017). The eastern bettong, while a woodland specialist species, is a mobile animal in which individuals have relatively large ranges for their size. GPS-tracking of eastern bettongs in this study area shows that bettong’s can occasionally cross small gaps between remnant woodland patches (Gardiner et al. in review). The classic metapopulation theory is therefore less applicable to species with movement patterns extending beyond single patches.
Each of the hypotheses considered in this study depicts the importance of fragmentation at different spatial scales. At one extreme, the metapopulation and island biogeography theory categorises landscapes into ‘habitat’ and ‘non-habitat’, disregarding the ability of individual species to use the spaces between habitat patches. On the other hand, the habitat continuum hypothesis suggests that all habitats are used by individual species, albeit at different intensities. Because landscapes are heterogeneous, no one hypothesis is likely to be applicable to all regions or species, as is evident from the varying support for each hypothesis in the literature. Also, as many of the different types of patterns of fragmentation are correlated (Evju and Sverdrup-Thygeson 2016), the habitat amount hypothesis can be considered as one component of a scale in measuring responses to habitat fragmentation. Hanski (2015) suggests the habitat amount hypothesis would be applicable only at small spatial scales and in regions that still retain much of their original habitat. Our results revealed a strong effect of habitat amount at large scales in an area that has lost most of its original woodland vegetation, in agreement with other large-scale studies (Melo et al. 2017; Seibold et al. 2017). The habitat amount hypothesis places importance on smaller fragments, which are often disregarded as being usable habitat. Our study indicates that smaller fragments can indeed make valuable contributions to the total amount of habitat accessible to a species. In such cases, the amount of available habitat can be achieved by restoring and or planting native vegetation around remnants. By removing the strict delineation of what is considered habitat, the habitat amount hypothesis can be an effective management tool in modified landscapes which still retain scattered remnants.
Habitat quality is a key factor in habitat selection, and therefore influences the presence of a species (Chandler and King 2011). The strongest indicator of woodland quality as habitat for bettongs was the total density of stems of over storey trees. High stem density indicates larger numbers of small stems, which in turn is an indication of continuing woodland regeneration and thus healthy woodland. Stem density also indicates availability of food for the eastern bettong. This species feeds mainly on the subterranean fruiting bodies of ectomycorrhizal (ECM) fungi (Johnson 1994b), and the abundance of bettongs in different habitats is positively related to mycorrhizal activity (Taylor 1992). ECM fungi are associated with the fine roots of trees and shrubs, especially with Eucalyptus and Acacia, which are the dominant canopy and mid-storey genera in our study area. Foraging activity of eastern bettongs is concentrated in areas where the density of stems is high, probably because this is related to higher biomass of fine roots, and therefore higher production of ECM fungi (Johnson 1994a). Apart from this effect of stem density, we found no relationships of vegetation cover or plant-species richness on the presence of bettongs.
Surprisingly, occupancy by eastern bettongs was not strongly related to detections of predators. Our camera study and current ongoing studies have recorded bettongs, feral cats and spotted-tailed quolls frequently using the same woodland. The Midlands has the highest recorded density of feral cats in Australia and their occupancy in the Midlands is best predicted by edge habitats between remnant woodland and pasture or grassland (R. Hamer, unpubl. data). Spotted-tailed quolls use the interior of woodland remnants (R. Hamer, unpubl. data) and both spotted-tailed quolls and Tasmanian devils are significant predators of medium sized mammals (Jones and Barmuta 1998; Pemberton et al. 2008; Andersen et al. 2017) and would probably prey on bettongs in shared habitat. Devils are currently very scarce due to severe population decline from facial tumour disease (Lazenby et al. 2018) The Midlands landscape supports high densities of European rabbits (Oryctolagus cuniculus) which are common prey for spotted-tailed quolls on mainland Australia (Belcher et al. 2007). It is possible that feral cats and spotted-tailed quolls focus on abundant alternative prey, including rabbits, black rats, and lizards. On the mainland of Australia, the eastern bettong has disappeared, primarily because of predation by red foxes (Woinarski et al. 2004; Johnson 2006). The red foxes have not been detected since 2009 (Sarre et al. 2013) and are likely no longer present in Tasmania.
The occupancy of eastern bettongs was not influenced by distance to infrastructure or water bodies. This could be attributed to the configuration of these features within the landscape. Most roads and water bodies are adjacent to expanses of pasture and the lack of detections of bettongs in pasture meant that bettongs were not present in close proximity to these landscape features. Species can be influenced by abiotic factors such as climate and soil type, as these directly influence mycorrhizal productivity. However it was unlikely these would be significant because there was not enough variation across the study site and within our study period.
The Midlands region of Tasmania is a National Biodiversity Hotspot and the focus of a restoration project to minimize and reverse the loss of biodiversity caused by the expansion of agriculture. Two large biodiversity corridors are being constructed across the Midlands to connect core the dry woodlands and forests from the Eastern Tiers to the Great Western Tiers (Jones and Davidson 2016). Existing remnant patches are being restored by managing grazing and fire and corridors planted following riverine systems to link remnants and core habitat. In both restoration and plantings, the full complement of tree, shrub and understorey species are being restored. Our results indicate that bettongs are likely to respond positively to these efforts as the total amount of habitat increases, the remnants are better connected and thus more accessible, and the regeneration (number of stems) improves habitat quality by increasing germination and survival of seedlings of the tree and understorey species which provide food for bettongs. We didn’t detect bettongs in any of the 23 restoration sites that were part of this survey. This can be explained by the current distribution and age of restoration plots. The new plantings, that are part of the recent biodiversity corridor construction, are too young to provide useful habitat to bettongs. Older restoration plots on farms have usually been established in extensively cleared areas, where they provide ecosystem services such as shelter for livestock in very open parts of the landscape. These restoration plots are thus often surrounded by pasture and grazing paddocks and are at a significant distance from woodland habitat. As a woodland specialist that plays an important functional role in ecosystem health, potentially promoting plant germination in diggings (Eldridge et al. 2015) and dispersing the spores of ectomycorrhizal fungi which provides nutrients of trees in low fertility soils, the bettong could itself enhance the success of restoration and recovery of degraded areas.
Conclusion
Our study shows habitat amount to be a more useful predictor of patch occupancy than patch size and isolation in a habitat specialist that is also sufficiently mobile to move between habitat patches. When a species is able to move across unsuitable habitat such as pasture to reach multiple patches, all remnants including smaller fragments contribute to the total amount of habitat available to the species. Habitat quality is also important and the use of these large and small remnants is increased as their quality improves. Where the species has disappeared due to the extent of habitat loss, increasing the amount of habitat and improving the quality of the remnants could be effective strategies for their persistence. Future work combining movement ecology and landscape ecology could further our understanding of the effective scale of management needed.
References
Algar D, Angus G, Williams M, Mellican A (2007) Influence of bait type, weather and prey abundance on bait uptake by feral cats (Felis catus) on Peron Peninsula, Western Australia. Conserv Sci West Aust 6(1):109
Almeida-Gomes M, Vieira MV, Rocha CFD, Metzger JP, De Coster G (2016) Patch size matters for amphibians in tropical fragmented landscapes. Biol Conserv 195:89–96
Andersen GE, Johnson CN, Barmuta LA, Jones ME (2017) Dietary partitioning of Australia’s two marsupial hypercarnivores, the Tasmanian devil and the spotted-tailed quoll, across their shared distributional range. PLoS ONE 12(11):e0188529
Anderson J, Rowcliffe JM, Cowlishaw G (2007) Does the matrix matter? A forest primate in a complex agricultural landscape. Biol Conserv 135(2):212–222
Ascensão F, Lucas PS, Costa A, Bager A (2017) The effect of roads on edge permeability and movement patterns for small mammals: a case study with Montane Akodont. Landscape Ecol 32(4):781–790
Bailey TG (2012) Eucalypt regeneration and ecological restoration of remnant woodlands in Tasmania. University of Tasmania, Australia
Battin J (2004) When good animals love bad habitats: ecological traps and the conservation of animal populations. Conserv Biol 18(6):1482–1491
Belcher CA, Nelson JL, Darrant JP (2007) Diet of the tiger quoll (Dasyurus maculatus) in south-eastern Australia. Aust J Zool 55(2):117–122
Bennett AF (1990) Habitat corridors and the conservation of small mammals in a fragmented forest environment. Landscape Ecol 4(2):109–122
Bullock JM, Moy I, Pywell RF, Coulson SJ, Nolan AM, Caswell H (2002) Plant dispersal and colonization processes at local and landscape scales. In: Bullock JM, Kenward R, Hails R (eds) Dispersal ecology. Blackwell Scientific, Malden, pp 279–302
Chandler R, King D (2011) Habitat quality and habitat selection of golden-winged warblers in Costa Rica: an application of hierarchical models for open populations. J Appl Ecol 48(4):1038–1047
D’Amico M, Román J, De los Reyes L, Revilla E (2015) Vertebrate road-kill patterns in Mediterranean habitats: who, when and where. Biol Conserv 191:234–242
Ditmer MA, Garshelis DL, Noyce KV, Laske TG, Iaizzo PA, Burk TE, Fieberg JR (2015) Behavioral and physiological responses of American black bears to landscape features within an agricultural region. Ecosphere 6(3):1–21
Doherty TS, Dickman CR, Johnson CN, Legge SM, Ritchie EG, Woinarski JCZ (2017) Impacts and management of feral cats Felis catus in Australia. Mamm Rev 47(2):83–97
Doherty TS, Driscoll DA (2018) Coupling movement and landscape ecology for animal conservation in production landscapes. Proc R Soc B 285:20172272
Eldridge DJ, Woodhouse JN, Curlevski NJA, Hayward M, Brown MV, Neilan BA (2015) Soil-foraging animals alter the composition and co-occurrence of microbial communities in a desert shrubland. ISME J 9:2671
Evju M, Sverdrup-Thygeson A (2016) Spatial configuration matters: a test of the habitat amount hypothesis for plants in calcareous grasslands. Landscape Ecol 31(9):1891–1902
Fahrig L (2002) Effect of habitat fragmentation on the extinction threshold: a synthesis. Ecol Appl 12(2):346–353
Fahrig L (2007) Non-optimal animal movement in human-altered landscapes. Funct Ecol 21(6):1003–1015
Fahrig L (2013) Rethinking patch size and isolation effects: the habitat amount hypothesis. J Biogeogr 40(9):1649–1663
Fahrig L (2017) Ecological responses to habitat fragmentation per se. Annu Rev Ecol Evol Syst 48(1):1–23
Fattebert J, Baubet E, Slotow R, Fischer C (2017) Landscape effects on wild boar home range size under contrasting harvest regimes in a human-dominated agro-ecosystem. Eur J Wildl Res 63(2):32
Fischer Lindenmayer (2007) Landscape modification and habitat fragmentation: a synthesis. Glob Ecol Biogeogr 16(3):265–280
Fischer J, Lindenmayer DB, Barry S, Flowers E (2005) Lizard distribution patterns in the Tumut fragmentation “Natural Experiment” in south-eastern Australia. Biol Conserv 123(3):301–315
Fischer J, Lindenmayer DB, Kaitala V (2006) Beyond fragmentation: the continuum model for fauna research and conservation in human-modified landscapes. Oikos 112(2):473–480
Fiske I, Chandler R (2011) Unmarked: an R package for fitting hierarchical models of wildlife occurrence and abundance. J Stat Softw 43(10):1–23
Fleming PA, Anderson H, Prendergast AS, Bretz MR, Valentine LE, Hardy GES (2014) Is the loss of australian digging mammals contributing to a deterioration in ecosystem function? Mamm Rev 44(2):94–108
Gastón A, Blázquez-Cabrera S, Garrote G, Mateo-Sánchez MC, Beier P, Simón MA, Saura S (2016) Response to agriculture by a woodland species depends on cover type and behavioural state: insights from resident and dispersing Iberian lynx. J Appl Ecol 53(3):814–824
Ghazoul J (2005) Pollen and seed dispersal among dispersed plants. Biol Rev 80(3):413–443
Haddad NM, Brudvig LA, Clobert J, Davies KF, Gonzalez A, Holt RD, Cook WM (2015) Habitat fragmentation and its lasting impact on Earth’s ecosystems. Sci Adv 1(2):e1500052
Haddad NM, Gonzalez A, Brudvig LA, Burt MA, Levey DJ, Damschen EI (2017) Experimental evidence does not support the habitat amount hypothesis. Ecography 40(1):48–55
Haila Y (2002) A conceptual genealogy of fragmentation research: from island biogeography to landscape ecology. Ecol Appl 12(2):321–334
Hanski I (2015) Habitat fragmentation and species richness. J Biogeogr 42(5):989–993
Johnson C (1994a) Distribution of feeding activity of the Tasmanian bettong (Bettongia gaimardi) in relation to vegetation patterns. Wildl Res 21(3):249–255
Johnson CN (1994b) Nutritional ecology of a mycophagous marsupial in relation to production of hypogeous fungi. Ecology 75(7):2015–2021
Johnson C (2006) Australia’s mammal extinctions: a 50000 year history. Cambridge University Press, Cambridge
Johnston CA (2013) Wetland losses due to row crop expansion in the Dakota Prairie Pothole Region. Wetlands 33(1):175–182
Jones ME, Barmuta LA (1998) Diet overlap and relative abundance of sympatric dasyurid carnivores: a hypothesis of competition. J Anim Ecol 67(3):410–421
Jones ME, Davidson N (2016) Applying an animal-centric approach to improve ecological restoration. Restor Ecol 24(6):836–842
Kehoe L, Romero-Muñoz A, Polaina E, Estes L, Kreft H, Kuemmerle T (2017) Biodiversity at risk under future cropland expansion and intensification. Nat Ecol Evol 1(8):1129
Krauss J, Klein A-M, Steffan-Dewenter I, Tscharntke T (2004) Effects of habitat area, isolation, and landscape diversity on plant species richness of calcareous grasslands. Biodivers Conserv 13(8):1427–1439
Laurance WF (2008) Theory meets reality: how habitat fragmentation research has transcended island biogeographic theory. Biol Conserv 141(7):1731–1744
Laurance WF, Sayer J, Cassman KG (2014) Agricultural expansion and its impacts on tropical nature. Trends Ecol Evol 29(2):107–116
Lazenby BT, Tobler MW, Brown WE, Hawkins CE, Hocking GJ, Hume F, Thalmann S (2018) Density trends and demographic signals uncover the long-term impact of transmissible cancer in Tasmanian devils. J Appl Ecol 55(3):1368–1379
Lindenmayer DB, Fischer J (2013) Habitat fragmentation and landscape change: an ecological and conservation synthesis. Island Press, Washington, DC
Lindgren JP, Cousins SAO (2017) Island biogeography theory outweighs habitat amount hypothesis in predicting plant species richness in small grassland remnants. Landsc Ecol 32:1895–1906
MacKenzie DI, Nichols JD, Hines JE, Knutson MG, Franklin AB (2003) Estimating site occupancy, colonization, and local extinction when a species is detected imperfectly. Ecology 84:2200–2207
Magioli M, Ribeiro MC, Ferraz KMPMB, Rodrigues MG (2015) Thresholds in the relationship between functional diversity and patch size for mammals in the Brazilian Atlantic Forest. Anim Conserv 18(6):499–511
Martinson HM, Fagan WF (2014) Trophic disruption: a meta-analysis of how habitat fragmentation affects resource consumption in terrestrial arthropod systems. Ecol Lett 17(9):1178–1189
Maxwell SL, Fuller RA, Brooks TM, Watson JE (2016) Biodiversity: the ravages of guns, nets and bulldozers. Nature 536(7615):143–145
Mazerolle MJ, Mazerolle MMJ (2017) Package ‘AICcmodavg’
Melo GL, Sponchiado J, Cáceres NC, Fahrig L (2017) Testing the habitat amount hypothesis for South American small mammals. Biol Conserv 209:304–314
Mönkkönen M, Rajasärkkä A, Lampila P (2014) Isolation, patch size and matrix effects on bird assemblages in forest reserves. Biodivers Conserv 23(13):3287–3300
Mortelliti A, Amori G, Boitani L (2010) The role of habitat quality in fragmented landscapes: a conceptual overview and prospectus for future research. Oecologia 163(2):535–547
Munguía-Rosas MA, Montiel S (2014) Patch size and isolation predict plant species density in a naturally fragmented forest. PLoS ONE 9(10):e111742
Newbold T, Hudson LN, Hill SL, Contu S, Lysenko I, Senior RA, Day J (2015) Global effects of land use on local terrestrial biodiversity. Nature 520(7545):45
Pemberton D, Gales S, Bauer B, Gales R, Lazenby B, Medlock K (2008) The diet of the Tasmanian devil, Sarcophilus harrisii, as determined from analysis of scat and stomach contents. In: Papers and Proceedings of the Royal Society of Tasmania, vol 142, pp. 13–22
Püttker T, Bueno AA, De Barros CD, Sommer S, De Pardini R (2011) Immigration rates in fragmented landscapes—empirical evidence for the importance of habitat amount for species persistence. PLoS ONE 6(11):e27963
Ripperger SP, Kalko EK, Rodríguez-Herrera B, Mayer F, Tschapka M (2015) Frugivorous bats maintain functional habitat connectivity in agricultural landscapes but rely strongly on natural forest fragments. PLoS ONE 10(4):e0120535
Ruiz-Capillas P, Mata C, Malo JE (2015) How many rodents die on the road? Biological and methodological implications from a small mammals’ roadkill assessment on a Spanish motorway. Ecol Res 30(3):417–427
Sahlin E, Schroeder LM (2010) Importance of habitat patch size for occupancy and density of aspen-associated saproxylic beetles. Biodivers Conserv 19(5):1325–1339
Sarre SD, MacDonald AJ, Barclay C, Saunders GR, Ramsey DSL (2013) Foxes are now widespread in Tasmania: DNA detection defines the distribution of this rare but invasive carnivore. J Appl Ecol 50(2):459–468
Seahra SE, Yurkonis KA, Newman JA (2016) Species patch size at seeding affects diversity and productivity responses in establishing grasslands. J Ecol 104(2):479–486
Seibold S, Bässler C, Brandl R, Fahrig L, Förster B, Heurich M, Müller J (2017) An experimental test of the habitat-amount hypothesis for saproxylic beetles in a forested region. Ecology 98(6):1613–1622
Sodhi NS, Ehrlich PR (2010) Conservation biology for all. Oxford University Press, Oxford
Spencer PBS, Yurchenko AA, David VA, Scott R, Koepfli KP, Driscoll C, O’brien SJ, Menotti-Raymond M (2016) The population origins and expansion of feral cats in Australia. J Hered 107(2):104–114
Taylor RJ (1992) Distribution and abundance of fungal sporocarps and diggings of the Tasmanian bettong, Bettongia gaimardi. Aust J Ecol 17(2):155–160
Taylor RJ (1993a) Habitat requirements of the Tasmanian bettong (Bettongia gaimardi), a mycophagous marsupial. Wildl Res 20(5):699–710
Taylor RJ (1993b) Home range, nest use and activity of the Tasmanian bettong, Bettongia gaimardi. Wildl Res 20(1):87–95
Tittensor DP, Walpole M, Hill SL, Boyce DG, Britten GL, Burgess ND, Baumung R (2014) A mid-term analysis of progress toward international biodiversity targets. Science 346(6206):241–244
Torrenta R, Villard MA (2017) A test of the habitat amount hypothesis as an explanation for the species richness of forest bird assemblages. J Biogeogr 44:1791–1801
Venter O, Sanderson EW, Magrach A, Allan JR, Beher J, Jones KR, Levy MA (2016) Sixteen years of change in the global terrestrial human footprint and implications for biodiversity conservation. Nat Commun 7:12558
Woinarski JCZ, Risler J, Kean L (2004) Response of vegetation and vertebrate fauna to 23 years of fire exclusion in a tropical Eucalyptus open forest, Northern Territory, Australia. Aust Ecol 29(2):156–176
Acknowledgements
We thank Greening Austral Tasmania Inc. (GA), Bush Heritage Australia (BHA), Tasmania Land Conservancy (TLC) and the Tasmanian Department of Primary Industry, Parks, Water and Environment (DPIPWE) for helping with appropriate contacts and guidance in selecting sampling sites. In particular, we would like to thank Neil Davidson and Sebastian Burgess (GA) for working closely with us on the ecological restoration project in the Midlands, Matt Appleby (BHA) for his extensive help and knowledge of the Midlands, and Oberon Carter (DPIPWE) for running the GIS-based site selection algorithm. The project was funded by an Australian Research Council (ARC) Linkage Scheme grant (LP130100949). Christopher Johnson was supported on an ARC Australian Professorial Fellowship and Menna Jones on an ARC Future Fellowship (FT100100031). Thanks to the Holsworth Research Endowment for providing crucial funding for covering fieldwork costs. Greatest acknowledgements to PhD students: Rowena Hamer, Glen Bain, and Mauel Ruiz for aiding in fieldwork. Finally. Dr. Peter Gardiner for an outsider’s perspective and to the reviewers and editor for comments contributing to the completion of the manuscript.
Author information
Authors and Affiliations
Contributions
RG, MJ, CJ, and RH conceived the ideas and designed methodology. RG and RH collected and analysed the data. RG, CJ and MJ wrote the manuscript. All authors contributed to the final approval for publication.
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Gardiner, R., Bain, G., Hamer, R. et al. Habitat amount and quality, not patch size, determine persistence of a woodland-dependent mammal in an agricultural landscape. Landscape Ecol 33, 1837–1849 (2018). https://doi.org/10.1007/s10980-018-0722-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10980-018-0722-0