Abstract
Kirtland’s warblers (Dendroica kirtlandii) persist in a naturally patchy environment of young, regenerating jack pine forests (i.e., 5–23 years old) created after wildfires and human logging activities. We examined how changing landscape structure from 26 years of forest management and wildfire disturbances influenced population size and spatial dispersion of male Kirtland’s warblers within their restricted breeding range in northern Lower Michigan, USA. The male Kirtland’s warbler population was six times larger in 2004 (1,322) compared to 1979 (205); the change was nonlinear with 1987 and 1994 identified as significant points of change. In 1987, the population trend began increasing after a slowly declining trend prior to 1987, and the rate of increase appeared to slow after 1994. Total amount of suitable habitat and the relative area of wildfire-regenerated habitat were the most important factors explaining population trend. Suitable habitat increased 149% primarily due to increasing plantations from forest management. The relative amount and location of wildfire-regenerated habitat modified the distribution of males among various habitat types, and the spatial variation in their abundance across the primary breeding range. These findings indicate that the Kirtland’s warbler male population shifted its use of habitat types temporally and spatially as the population increased and as the relative availability of habitats changed through time. We demonstrate that researchers and managers need to consider not only habitat quality, but the temporal and the spatial context of habitat availability and population levels when making habitat restoration decisions.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Explaining temporal trends in animal population abundance has important implications for management and conservation, especially for declining populations. Understanding how past population fluctuations were linked to changes in the landscape helps us understand current population levels and predict future population levels based on a landscape’s projected change. However, the spatiotemporal interaction between population dynamics and landscape change over an extended period of time and large geographical area has not been widely studied (Andren 1994; Flather and Sauer 1996).
Concern over declining area-sensitive neotropical bird populations has resulted in many theoretical and empirical studies incorporating regional and landscape-scale factors as predictors. Human land-use activities are increasingly subdividing previously continuous habitat into a patchy environment for many bird populations. Consequently, investigations have focused on the influence of patch geometry (i.e., size and isolation) on bird population demography and persistence within the context of habitat loss, habitat fragmentation, metapopulation and source-sink structure (Wiens 1994; Robinson et al. 1995; Villard et al. 1995; Schmiegelow et al. 1997; Trzcinski et al. 1999). Typically, a variety of population responses (e.g., presence, abundance, nest success) are measured at the patch, plot, or territory scale over a short time period because of the impracticality in long-term sampling over broad scales. However, inferring population change from results derived using patch or plot-centered studies in static landscapes is problematic (Doak and Mills 1994; Flather and Sauer 1996; Johnson et al. 2004). Empirical studies rarely relate long-term population fluctuations to dynamic landscapes where habitat varies in extent and arrangement through time in the focal species’ geographic range (Brown et al. 1995; Goldstein et al. 2003). Additionally, few empirical studies examine how habitat-specific demography interacts with landscape structure to affect population dynamics (Kadmon 1993).
Because declines are being recorded for many neotropical migrant birds that prefer disturbance-mediated early successional habitat (Brawn et al. 2001; Thompson and DeGraaf 2001), more attention is being directed towards the effects of spatial heterogeneity on these bird populations. Frequency and intensity of natural (e.g., fire) and human disturbances (e.g., forest management) establish the availability and arrangement of regenerating habitat spatially and temporally on the landscape (Pickett and Cadenasso 1995; Foster et al. 1998). Given the relatively short persistence time of early successional habitat on the landscape, a rapid shifting in habitat availability and quality may cause populations to use habitat differently, thereby influencing population distribution and abundance (Probst 1986; Pulliam and Danielson 1991; Kareiva and Wennergren 1995; Arthur et al. 1996). Studying a habitat specialist with a restricted distribution enhances our ability to identify habitat availability concurrently with population dispersion among habitat types, which will increase our understanding of the influence of large-scale landscape structure on populations.
The Kirtland’s warbler (Dendroica kirtlandii) is an example of an area-sensitive neotropical migrant that is a habitat specialist with a restricted breeding distribution. This species, an endangered neotropical migrant, breeds exclusively in young jack pine (Pinus banksiana) forests primarily in northern Lower Michigan on nutrient-poor, sandy sites in glacial outwash ecosystems (Mayfield 1960; Kashian et al. 2003), and limited areas in the Upper Peninsula of Michigan (Kepler et al. 1996; Probst et al. 2003). Because of this narrow range in suitable breeding habitat, potential breeding areas can be easily identified and monitored for singing males (Ryel 1981).
The entire known male Kirtland’s warbler breeding population in Michigan has been censused annually since 1971 after the previous decennial census indicated a 60% decline in the population (Walkinshaw 1983). Lack of suitable habitat was determined to be the ultimate factor limiting population numbers when brood parasitism by brown-headed cowbirds (Molothrus ater) was controlled (Byelich et al. 1976; Probst 1986; Kepler et al. 1996). Early-successional jack pine habitat, typically created after wildfires, declined on the landscape with greater fire suppression during the mid-century. To reverse the declining population trend, management agencies established an extensive habitat restoration program in 1981 that incorporated plantations and unburned, natural regeneration to supplement wildfire-regenerated habitat (Probst and Weinrich 1993; Kepler et al. 1996). By the 1990s, the composition of potential breeding habitat had become primarily plantation rather than wildfire-regenerated areas (Kashian et al. 2003; Probst et al. 2003) having the potential to alter how males were responding to landscape structure.
The Kirtland’s warbler population, thus, consists of many loose aggregations of birds that continually shift throughout the landscape in response to regenerating habitat. The relationship between habitat amount and population size is, in part, the balance of newly created habitat versus old, declining habitat (Probst 1986) on the landscape, and the relative amounts of habitat types. The birds have different demographic rates among regenerating habitat types (Probst 1986; Probst and Hayes 1987; Bocetti 1994). Determining the degree to which differential habitat use and density influence the spatiotemporal population dynamics will be important to future predictions of population growth in relation to changing amounts and arrangement of habitat types (Pulliam et al. 1992; Morris 2003).
Using the historical results of the annual male Kirtland’s warbler official census (Ryel 1981; Probst et al. 2005) and other temporally referenced landscape data, the objectives of this study were to: (1) determine the male population trend from 1979 to 2004 to identify the timing and extent of population fluctuations; (2) determine the relative influence of habitat amount, arrangement, and differential male use of habitat types on population trend estimates; and (3) map the spatial dispersion of the population during identified time periods.
Methods
Study area
This research was primarily restricted to 23 established Kirtland’s warbler management areas (KWMAs) in northern Lower Michigan, USA containing the prescribed densities and arrangement of jack pine (i.e., patches have greater density than forestry plantations; rows planted in an opposing wave pattern). KWMAs are dispersed across an area approximately 137 km × 130 km covering 71,610 ha. Management areas varied in size between approximately 1,400 and 13,000 ha. Outwash sands (i.e., Grayling series) deposited by glacial meltwaters dominate these areas. These soils generally lack weatherable minerals, and are excessively well drained. Late-spring and fall freezes are common due to the area’s inland location and relatively high elevation contributing to the area’s unfavorable growing conditions (see Kashian et al. 2003). Few Kirtland’s warblers nest on lands outside of KWMAs. Habitat on other public and private lands usually lack the dense jack pine necessary to provide adequate canopy cover for nesting, are too small, or are not located on the well-drained soils required by this species’ habitat. The KWMAs are primarily surrounded by public or commercial forested lands that are managed for forest products, wildlife, and recreation.
Jack pine forests within KWMAs tend to be nearly pure, even-aged stands with scattered northern pin oak (Quercus ellipsoidalis), trembling and bigtooth aspen (Populus tremuloides and P. grandidentata), black cherry (Prunus serotina), and choke cherry (P. virginiana) (Probst 1988). Ground cover is a mixture of low shrubs (e.g., blueberry, (Vaccinium angustifolium), juneberry (Amelanchier spp.), sweetfern (Comptonia peregrina)), grasses, sedges, forbs and exposed bare ground (Byelich et al. 1976; Bocetti 1994; Probst and DonnerWright 2003).
Male abundance
Annual male population abundance from 1979 to 2004 was retrieved from the annual Kirtland’s warbler official census completed over its entire breeding range during the breeding season (Ryel 1981; Probst et al. 2005). Females do not sing. The official census was found to be a good index of the male population trend (Probst et al. 2005).
Defining the landscape structure
We merged geographic vector coverages of KWMAs maintained independently by the Huron-Manistee National Forests and the Michigan Department of Natural Resources. Lands within KWMAs are parceled into management stands for planning purposes. Only management stands typed as jack pine were included in the base coverage with the assumption that those stands contained the minimum cover and stem densities required for breeding habitat (see above). Each management stand in the coverage was attributed with area, year of origin (i.e., year planted or burned or harvested and left to naturally regenerate), and regeneration type (plantation, wildfire-regenerated, and unburned, natural regeneration). Stands were attributed with the number of males recorded during the Kirtland’s warbler official census from 1979 to 2004.
Areas outside KWMAs were added if ≥2 males used the area for ≥2 years; areas with <2 males were excluded from analysis. We assumed that the consecutive use of a patch indicated the patch contained the minimum cover and stem densities required for breeding. Included areas were typically on state and federal lands, but outside designated KWMAs, and were primarily forestry plantations (i.e., wider planting specification than that used for Kirtland’s warbler plantations) that typically had a large component of volunteer jack pine regeneration. Based on this definition, twenty-two patches totaling approximately 1,000 ha (1.6% of the landscape) and containing 35 males (0.2% of the cumulative total males) were not included in the base coverage. Federal and state stand management maps and 1992 digital orthoquads were used to recreate and digitize patch boundaries for these areas, and to reconcile past wildfire areas.
Because 2 year-old stock is used for planting federal and state lands, the year of origin for jack pine plantations was adjusted by 2 years to make the age structural components more similar between plantations and the other regeneration types. Adjacent management stands of the same regeneration type that had year of origin within 1 year were merged to form patches of similar age.
The 1980 Mack Lake Fire burned more than 24,000 ha over a diversity of physiographic areas causing jack pine regeneration to occur at different rates. Jack pine regenerated quicker in high-elevation areas compared to low-elevation areas resulting in various heights of trees and degrees of suitability within the burn apart from age (Walker et al. 2003). Based on these findings, the Mack Lake Burn area, after adjusting the year of origin for each management stand within the burn area, was separated into five patches based on jack pine growth rate and elevation (after Walker et al. 2003).
Defining suitable habitat
Whether a patch is suitable habitat within a given year depends on its age because of the changes in sampling height, understory, canopy cover, and lower live branch height (Probst 1988; Probst and Weinrich 1993). Suitable habitat was defined as jack pine patches 5–23 years old and ≥12 ha. This was the broadest definition possible based on the historical use of jack pine habitat by males (Probst and Weinrich 1993) and reflects how males used the habitat 1979 through 2004. The minimum patch size required for breeding has previously been reported as 32 ha (Mayfield 1960; Walkinshaw 1983); however, this was based on male use of the landscape prior to and during the 1980s, and did not incorporate forest openings. Reanalysis of male use based on our definition showed that males used patches as small as 12 ha from 1979 to 2004; specifically, 663 males (4.8% of all males) used 67 patches that were 12–32 ha in size. Nearly 9% of the habitat was composed of patches between 12 and 32 ha. Many of these patches were within larger complexes of suitably-aged jack pine habitat making these smaller areas more attractive to the warblers (Mayfield 1960; Walkinshaw 1983; Probst and Weinrich 1993).
Defining predictor variables
Population trends may be influenced not only by the total amount of suitable habitat, but by its composition if there is differential habitat use. Suitable habitat was subset into various habitat type-age categories to capture whether composition of the suitable habitat influenced the population trend (see Table 1). Suitable habitat was divided into three regeneration types: wildfire, plantation, and unburned, natural regeneration. Optimal habitat was defined as 8–15 year old wildfire-regenerated habitat based on higher productivity, and historical male preference for wildfire-regenerated habitat (Probst 1988; Probst and Weinrich 1993; Bocetti 1994). Optimal Kirtland’s warbler habitat (i.e., those stands with the highest density of warblers) has more than 7,500 stems per hectare, between 35% and 65% canopy cover, and is usually regenerated after wildfires in Lower Michigan (Probst 1988; Probst and Weinrich 1993). The 8–15 year old age class represents a time when singing males have been historically found at peak numbers within wildfire areas (Probst 1988). Young habitat was defined as 5–7 year old habitat of all regeneration types to describe the influx and availability of upcoming habitat on the landscape. This age category corresponds to a time when male numbers are typically low and building (Probst 1988). Marginal habitat was defined as 5–7 year old open, natural regeneration habitat (i.e., low stem density), because of reported lower densities and pairing success under habitat and age conditions relative to optimal breeding habitat (Probst and Hayes 1987).
The annual average patch size and density, and the average nearest neighbor distance to another occupied patch were measured (ArcMap GIS software, ESRI, Redlands, CA). The average nearest neighbor distance between patch centroids of occupied patches was calculated using the Average Nearest Neighbor Distance tool. Because the amount of suitable breeding habitat changed annually, patch density (number of patches per 1,000 ha suitable habitat), which is a scaled index of patch size, was used.
The distribution of individuals among various habitats may influence population dynamics (Pulliam 1988; Pidgeon et al. 2003). Probst and Weinrich (1993) proposed that the high proportion of males in marginal habitat from 1984 to 1989 would slow population growth. Thus, percentage of males in marginal habitat was included as an explanatory variable. Additionally, the percent of suitable habitat occupied by males was included as a more general index of male habitat use.
The amount of edge and core area is often measured in studies on neotropical migrant birds due to reported increased brood parasitism and nest predation with increasing patch edge and decreasing patch core area from forest fragmentation. During the time of this study, brown-headed cowbirds were trapped in breeding areas to minimize brood parasitism, so core/edge metrics were not included as explanatory variables.
Analytical approach
Generalized additive models (GAM) were used to estimate a nonparametric smoothed trend of the annual population count of singing males as a function of year and to the temporally-referenced explanatory covariates. In GAMs, the response is modeled as the additive sum of smoothed functions of predictor variables (Wood 2006). The smoothing procedure is incorporated into the model-fitting process so the data determines the nature of the relationships between the response and the set of explanatory variables rather than assuming some form of parametric relationship (Wood 2006).
GAMs were fit using the ‘mgcv’ package in the R software environment using a penalized thin plate regression spline basis to represent the smooth functions, which automatically selects the degree of smoothness of the functions (Wood 2006). The degree of smoothness is controlled by the smoothing parameter, which is calibrated by the degrees of freedom associated with the smoothing function. The smoothing parameter was set at 1.4 to correct over-fitting tendencies (Wood 2006, p. 224).
The number of singing males was modeled as a Poisson-distributed response variable with a log link function to year to determine the timing, length and trend of population fluctuations over the 26 years. To determine the uncertainty of the estimates, a 95% Bayesian confidence interval for the nonlinear functions was obtained from 100 simulations of the posterior distribution of the model coefficients (Wood 2006). A piecewise-regression model was used to determine significance of the change points identified visually from the fitted GAM population curve. These change points were used to divide the population growth curve into different periods.
The relationship between the observed annual male population trend and explanatory variables were examined using GAMs (Table 1). The small sample of 26 years prevented more than two predictor variables being included in any one model. Thus, the candidate set of 11 models included each predictor variable singularly and several bivariate models (Table 1). Variables in the bivariate models were not highly correlated with the amount of habitat (r < 0.70). Partial deviance residual diagnostic plots were examined to check model assumptions. Model fit was determined based on the percentage deviance explained (a generalization of R 2). Candidate models were compared using the minimum generalized cross-validation score and the differences in Akaike Information Criteria scores corrected for small-sample size (AICc) over the set of candidate models (Burnham and Anderson 2002).
The location and relative abundance of the singing male population within patches was mapped to show their spatiotemporal distribution (i.e., clumping) across successive population periods identified from the GAM and breakpoint analyses.
Results
Change in landscape structure
The primary change on the landscape was the nearly 150% increase in the annual total area of suitable habitat from 1979 to 2004 (Fig. 1a). The amount of suitable habitat increased annually until 2002 when it began decreasing with the maturation of the early 1980s plantations and areas within the Mack Lake burn. As expected, an almost four-fold increase in plantation habitat accounted for the increase in suitable habitat, and comprised 77.1% of the total suitable breeding habitat by 2004 (Fig. 1a). By 1992, plantation habitat was nearly twice as abundant as wildfire habitat on the landscape, and by 2000, amount of plantation habitat was triple that of wildfire habitat. Even though wildfire habitat increased 52%, it composed only 18.2% of the suitable habitat by 2004 compared to approximately one-third from the 1980s to the mid-1990s (Fig. 1a). Wildfire habitat composed a higher percentage of the total suitable habitat from 1985 to 1991 primarily from four aging large wildfires with origins in the 1970s, and from emerging areas within the 1980 Mack Lake Burn. The amount and proportion of unburned, natural regeneration habitat declined to less than 5% of the total suitable habitat by 2002.
Annual trends in landscape metrics measured on Kirtland’s warbler’s potential breeding habitat (5–23 years old) within their primary breeding range, northern Lower Michigan, 1979–2004. Only jack pine patches within Kirtland’s warbler management areas ≥12 ha and patches used ≥2 years by more than two male Kirtland’s warblers if outside the management areas are included
In conjunction with the annual increase in suitable habitat, the number of suitable patches on the landscape increased (Fig. 1b). Furthermore, patch density declined 26.1% (Fig. 1b) reflecting increased patch size on the landscape (Fig. 1c). The mean size of plantation patches gradually increased from 40 to >60 ha by 2004. Number of wildfire-regenerated patches remained fairly constant (N = 30–34) (Fig. 1b) with the mean size varying from 110 to 210 ha, much larger than plantations (Fig. 1c). More wildfire patches occurred on the landscape in the mid-1980s and 1990 (N = 41–43) and in 1995 (N = 43) primarily from the 1980 Mack Lake Burn and the 1990 Stephan Bridge Burn, respectively. The average distance between occupied patches decreased 60% by 2004 from 3.5 to 2.3 km (Fig. 1d).
In 1994, occupied habitat increased from the relatively constant 30–34% range in the preceding decade to approximately 40% for the next decade (Fig. 1e). Males occupied one-half of the suitable habitat area from 1997 to 1999. Optimal habitat dropped below 10% of suitable habitat during 1984–1987 and 2001–2004 (Fig. 1f). During both periods, existing wildfire areas were aging, and there was an absence of any substantial amount of emerging wildfire areas. Wildfires originating in 1965–1975 were declining in suitability by 1984, and the extensive 1980 Mack Lake Burn was declining in suitability by the early 2000s. Prior to 1984, however, nearly 20% of suitable habitat was optimal (Fig. 1f). The period from 1984 to 1987 also saw the largest percent of suitable habitat composed of developing, young habitat (≥25%) (Fig. 1f), which were the earliest plantations under the expanded habitat management program begun in 1981.
Population temporal trends
Singing male Kirtland’s warblers increased from 205 to about six times that (1,322) in 2004. The annual number of males as a smoothed function of time (GAM with 6.26 e.d.f.) indicated a significant nonlinear increase over the 26 years (F = 226.7, P < 0.01; Fig. 2). The years 1987 and 1994 were identified as points of change in the population increase. Prior to 1987, the male population declined from 205 to 167 males (Probst and Weinrich 1993; Probst et al. 2003). After 1987, the male population began increasing, but the rate of increase slowed slightly around 1994 (Fig. 2).
Kirtland’s warbler male population curve using census data collected within their primary breeding range, northern Lower Michigan, 1979–2004. Solid curve represents the population curve from a generalized additive model with 6.26 estimated degrees of freedom. Dotted curve represents the 95% Bayesian confidence interval obtained by 100 simulations from the posterior distribution of the model coefficients (Wood 2006). Change point analysis identified 1987 and 1994 (dashed vertical line) as significant points of change in the gradient of the population increase
The model that included amount of suitable habitat and percentage of wildfire-regenerated habitat was the best model for the population trend given the models evaluated (Table 1). The second ranked model (based on AICc score) included the amount of suitable habitat and percent optimal habitat. However, the best model has an Akaike weight of 0.99 and is 314 times better than the second model in explaining the data.
The Kirtland’s warbler population was evidently tracking the increasing trend in suitable habitat on the landscape, but it was moderated to some degree by habitat origin. Prior to 1985, the percentage wildfire habitat was slightly declining as was the population trend (−18.5% change) until developing suitable habitat from the 1980 Mack Lake Burn became suitably-aged in 1985 (Fig. 1a). The population lagged behind this habitat increase and didn’t begin increasing numbers until after 1987 (Fig. 2). Additionally, optimal habitat defined by both age and origin from wildfire shows a declining trend from 1979 to 1987 (Fig. 1f).
The period 1988–1994 was characterized by a tripling in male warbler numbers (210% increase) over 7 years. Suitable habitat was composed of some of the largest relative amounts of wildfire habitat during this period (Fig. 1a). After 1994, the rate of population increase slowed, but still nearly doubled over the next 10 years (74.6% increase; Fig. 2). The percent of wildfire habitat began declining during this time (Fig. 1a). Areas of the 1980 Mack Lake Burn were becoming unsuitable, and the few developing wildfire areas were not large enough to substantially contribute to the overall amount of suitable habitat on the landscape.
Population spatial dispersion across the breeding range
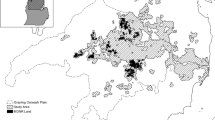
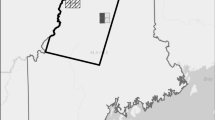
From 1979 to 1987, singing male Kirtland’s warblers occupied nearly one-third of the suitable habitat area (Fig. 1e), but occurred in relatively few patches each year (20–35 patches) (Fig. 3). Many occupied patches contained <10 males. Six maturing wildfire areas distributed across the species geographic range contained 70–80% of the male population (Fig. 3; Probst and Weinrich 1993). From 1979 to 1987, unburned, natural regeneration habitat held relatively more males than other years, but by 1986, males were beginning to redistribute out of this habitat into developing habitat (Probst and Weinrich 1993); from 1986 to 1989, 7.1–17.5% of males were found in the young habitat category. Young habitat during this time was composed primarily of the 1980 Mack Lake Burn area and plantations created during an accelerated habitat management program in 1981.
From 1988 to 1994, approximately 75% of males were found in wildfire habitat. Males declined in number in the older wildfire areas, but increased in number within the Mack Lake burn area and within the 1975 Bald Hill Burn (Fig. 4). Several larger plantations in the Damon Area began building in male numbers as well (Fig. 4). These areas had several orders of magnitude more males than other areas. Male Kirtland’s warblers broadened their geographic distribution into peripheral KWMAs, most notably eastward into the developing plantations in the Pine River KWMA (Fig. 4). By 1994, males shifted almost entirely out of natural regeneration habitat (2.5% males), and the percent of males found in plantation habitat slowly increased (21.2–51.7%) as the percent of males in wildfire habitat slowly declined (76.4–47.0%). Specifically, males went from using 12 plantation patches to 61, and 19 to 13 wildfire patches (Fig. 4) reflecting the shift in developing versus declining habitat types. By 1994, more males were occupying plantation rather than wildfire habitat for the first time.
From 1995 to 2004, male Kirtland’s warblers continued redistributing into plantations from wildfire habitat. About 85% of the males were found in plantation habitat by 2004. Males again used unburned, natural regenerated areas, increasing to almost 5% by 2004. With the increase in population size and subsequent use of plantations, the spatial dispersion of abundance became more evenly distributed throughout their lower breeding range. Males expanded their use of the northeastern and southernmost KWMAs (Fig. 5). Many patches had fewer than 100 males (Fig. 5). Interestingly, males began using areas outside KWMAs more frequently, particularly areas between the southern KWMAs.
Discussion
Analyzing a species’ spatiotemporal response to changing landscape structure identifies potential causes of population fluctuations important for long-term conservation, especially when applied throughout a species’ range. Our analyses regarding associations between landscape structure and the male Kirtland’s warbler population trend from 1979 to 2004 identified three periods of population growth. Total amount of suitable habitat and relative area of wildfire-regenerated habitat were the most important factors explaining the population trend. In addition, the relative amount and location of wildfire-regenerated habitat influenced the male Kirtland’s warbler distribution among various habitat types, and spatial dispersion across the landscape through time.
In simulated landscapes, amount of suitable habitat in the landscape rather than the spatial arrangement of the habitat explains more variation in population size (i.e., pure amount effect) (Andren 1994; Fahrig 1997; Flather and Bevers 2002). However, a review of empirical studies on bird populations in forest/agricultural landscapes (Mazerolle and Villard 1999) found patch characteristics (i.e., configuration) rather than habitat amount influenced bird responses and landscape predictors usually complemented patch characteristics. Fahrig (2003) contends that when habitat amount is not held constant or defined as is the case in many empirical studies, however, patch isolation becomes a function of reduced habitat amount rather than configuration as it is often depicted. In her review of empirical studies that controlled for habitat amount, habitat cover (i.e., loss) effects were much stronger than the configuration effects. However, recent literature suggests that statistical methods often used by empirical studies to control for the covariation between habitat amount and fragmentation (e.g., regression of residuals) can bias results towards habitat amount (Koper et al. 2007). Results from this empirical study where habitat amount varied temporally within defined management areas supports the relative importance of habitat amount in regulating regional bird population temporal abundance. Candidate models that included factors describing male habitat use and habitat arrangement such as distribution of males in marginal habitat, distance to the nearest occupied patch, and patch density were relatively unimportant to the population trend compared to total habitat amount.
The redistribution of the male population across the landscape with the location and aging of their preferred wildfire habitat suggests that patch quality is an important determinant of population spatial dispersion at coarse scales. With et al. (1997) using neutral models found that increasing habitat area increased connectivity causing populations to become more randomly distributed across the landscape once habitat amount is above a critical threshold on the landscape; however, species with habitat affinities for an uncommon habitat type retain a patchy distribution. Additionally, when the uncommon habitat type had the highest quality, based on a higher carrying capacity, the population became more aggregated across the landscape. Our results support these findings. Due to the species’ habitat specificity, the Kirtland’s warbler population retained a patchy distribution across the landscape despite increasing habitat. This pattern, however, was enhanced by the spatially disjunct nature of the management areas.
The influence of wildfire habitat was most evident temporally when areas within the large Mack Lake Burn of 1980 became suitable around 1987; this increase in habitat corresponded with an increase in the Kirtland’s warbler population. Further, males spatially redistributed from aging wildfire areas into the developing areas within Mack Lake Burn (this study; Probst and Weinrich 1993). The male population change in 1994 appeared due to a combination of plantation and wildfire habitat. Specifically, males began dispersing into plantation habitat when wildfire-regenerated habitat began declining near 1994. For the first time, most males were found in plantation habitat in 1994 and afterwards.
This shift of males out of their historically preferred wildfire habitat and into plantations may explain the slowing of the population increase if demographic rates between the two habitat types differ. From 1990 to 1992, Bocetti (1994) found that the mean number of young fledged per nest attempt was comparable between plantation and wildfire habitat of different ages, but fewer nests were initiated in plantation habitat due to fewer females per male as a result of more unmated males in plantations and greater polygyny in wildfire habitat. In addition, there was a slightly lower mean clutch size in plantations than wildfire habitat. She concluded plantations were acting as source habitats even though they produced slightly fewer young than wildfire areas (Bocetti 1994), but it is difficult to attribute small productivity differences to habitat type only. Plantations were typically smaller than wildfires areas (Donner 2007), so patch size may be influencing demographics as well.
The importance of patch-age distribution and the percent of males in marginal habitat to population growth were not evident in this study. Probst and Weinrich (1993) predicted that population growth would slow after 1992 because of the declining suitability of the Bald Hill Burn and the high proportion of males in marginal habitat in 1984 and 1987. Additionally, the authors predicted that the high proportion of males in wildfire areas from 1988 to 1991 might increase annual productivity so that males would fully occupy areas that were suitable from 1990 to 1993. Our study found that the population began slowing several years later than predicted. In addition, not until 1994 did males begin occupying a higher proportion of suitable habitat. However, our results agreed with the predicted patterns of a slowing population, and that the population would occupy more of the available landscape (Probst and Weinrich 1993). The difference in predicted years between the studies was likely due to the age definition of suitable habitat. Probst and Weinrich (1993) used a more restrictive definition of suitable habitat for their predictions (8–20 years for wildfire habitat and 10–20 years for plantation and unburned, natural regeneration habitat). As a result, the timing of events would differ and the predicted annual amount of suitable habitat on the landscape would be less compared to our study.
Males’ preference for using wildfire habitat may reflect the number or quality of suitable nesting (territory) sites available within these areas. Comparing a subset of wildfire and plantation areas, Bocetti (1994) found wildfire sites had greater jack pine density, more openings, and greater woody debris, which are all important local characteristics for nesting and foraging (Probst 1988; Bocetti 1994). This information, along with habitat differences in pairing success (Probst and Hayes 1987) and demographics (Bocetti 1994), suggests there may be a difference in the number and quality of sites available for the warblers among the regeneration types. Theories of habitat selection (Fretwell and Lucas 1970; Pulliam and Danielson 1991; Brown et al. 1995) predict males should move into lesser-quality sites as higher-quality sites are filled. When the Kirtland’s warbler population was at low levels in the late 1980s and early 1990s, individuals were using the poorer quality unburned, natural regeneration areas, presumably because the aging higher-quality wildfire habitat was near saturation (Probst and Weinrich 1993). Once additional wildfire habitat was available, the population redistributed into these areas and essentially stopped using unburned, natural regeneration habitat (Probst and Weinrich 1993). At relatively higher population levels (late 1990s), males began using this poorer-quality habitat again. Optimal wildfire habitat was declining in suitability due to succession, and plantation habitat was potentially near saturation during this period. The effect of male distribution in poorer-quality habitat on the population’s temporal patterns in abundance, however, was not evident. The proportion of males found in unburned, natural regeneration habitat at high population levels in the late 1990s (around 6%) was potentially too low to effectively slow the population growth compared to when nearly 15% of males were using unburned, natural regeneration in the early 1980s during low population sizes.
The observed spatiotemporal population patterns occurred in the absence of significant brown-headed cowbird nest parasitism, which severely limited the population before this 26-year study period. An interagency cowbird control effort was implemented in 1972 to control nest parasitism after productivity was reportedly reduced by 60% to only 0.8 fledglings per nest (cf. Kepler et al. 1996). If cowbird control is reduced or eliminated, there is the potential for reduced productivity. Habitat restoration without considering factors influencing demographic rates may not be sufficient to recover or sustain populations (Fahrig 2001; Schrott et al. 2005).
Kirtland’s warbler managers’ integration of a landscape perspective during early habitat restoration plans (Byelich et al. 1976; Probst 1988) effectively set up a long-term empirical study on population response to changing landscape structure. Results show the population was responding temporally to the amount of habitat on the landscape, but males were also responding spatially and temporally to the type of habitat available and relative amounts of preferred habitat (i.e., patch quality). These findings have important implications for managing bird populations with specific habitat affinities, and emphasize the importance of temporal variability in landscape structure towards structuring populations. Specifically, when population and habitat amount are at low levels, adding habitat, especially habitat that is more preferred or of higher quality, may be especially important for increasing the population size or strategically dispersing the population. In contrast, when habitat amount and population size are at relatively high levels and above a habitat amount threshold, the availability and location of higher quality habitat may not be as important as providing ample amounts of suitable habitat to maintain designated management levels. Our study demonstrates that researchers and managers need to consider not only habitat quality, but the temporal and spatial context of habitat availability and population levels when making habitat restoration decisions.
References
Andren H (1994) Effects of habitat fragmentation on birds and mammals in landscapes with different proportions of suitable habitat: a review. Oikos 71:355–366
Arthur SM, Manly BGJ, McDonald LL, Garner GW (1996) Assessing habitat selection when availability changes. Ecology 77:215–227
Bocetti C (1994) Density, demography, and mating success of Kirtland’s warblers in managed and natural habitats. Dissertation, Ohio State University
Brawn JD, Robinson SK, Thompson FR III (2001) The role of disturbance in the ecology and conservation of birds. Annu Rev Ecol Syst 32:251–276
Brown JH, Mehlman DW, Stevens GC (1995) Spatial variation in abundance. Ecology 76:2028–2043
Burnham KP, Anderson DR (2002) Model selection and multimodal inference: a practical information-theoretic approach, 2nd edn. Springer, New York
Byelich J, DeCapita ME, Irvine GW, Radtke RE, Johnson NI, Jones WR, Mayfield H, Mahalak WJ (1976) Kirtland’s warbler recovery plan. USDI Fish and Wildlife Service, Twin Cities
Doak DF, Mills LS (1994) A useful role for theory in conservation. Ecology 75:615–626
Donner DM (2007) Spatiotemporal response of the male Kirtland’s warbler population to changing landscape structure over 26 years. PhD Dissertation. University of Wisconsin – Madison, Madison, WI, 116 p
Fahrig L (1997) Relative effects of habitat loss and fragmentation on population extinction. J Wildl Manage 61:603–610
Fahrig L (2001) How much habitat is enough? Biol Conserv 100:65–74
Fahrig L (2003) Effects of habitat fragmentation on biodiversity. Annu Rev Ecol Evol Syst 34:487–515
Flather CH, Bevers M (2002) Patchy reaction-diffusion and population abundance: the relative importance of habitat amount and arrangement. Am Nat 159:40–56
Flather CH, Sauer JR (1996) Using landscape ecology to test hypotheses about large-scale abundance patterns in migratory birds. Ecology 77:28–35
Foster DR, Knight DH, Franklin JF (1998) Landscape patterns and legacies resulting from large, infrequent forest disturbances. Ecosystems 1:497–510
Fretwell SD, Lucas HL (1970) On territorial behaviour and other factors influencing habitat distribution in birds. Acta Biotheor 19:16–36
Goldstein MI, Corson MS, Lacher TE Jr, Grant WE (2003) Managed forests and migratory bird populations: evaluating spatial configurations through simulation. Ecol Modell 162:155–175
Johnson CJ, Boyce MS, Mulders R, Gunn A, Gau RJ, Cluff HD, Case RL (2004) Quantifying patch distribution at multiple spatial scales: applications to wildlife-habitat models. Landsc Ecol 20:869–882
Kadmon R (1993) Population dynamic consequences of habitat heterogeneity: an experimental study. Ecology 74:816–825
Kareiva P, Wennergren U (1995) Connecting landscape patterns to ecosystem and population processes. Nature 373:299–302
Kashian DM, Barnes BV, Walker WS (2003) Landscape ecosystems of northern Lower Michigan and the occurrence and management of the Kirtland’s warbler. For Sci 49:140–159
Kepler CB, Irvine GW, DeCapita ME, Weinrich J (1996) The conservation management of Kirtland’s warbler Dendroica kirtlandii. Bird Conserv Int 6:11–22
Koper N, Schmiegelow FKA, Merrill EH (2007) Residuals cannot distinguish between ecological effects of habitat amount and fragmentation: implications for the debate. Landsc Ecol 22:811–820
Mayfield HF (1960) The Kirtland’s warbler. Cranbrook Institute of Science, Bloomfield Hills, 242 pp
Mazarolle MJ, Villard M (1999) Patch characteristics and landscape context as predictors of species presence and abundance: a review. Ecoscience 6:117–124
Morris DW (2003) Toward an ecological synthesis: a case for habitat selection. Oecologia 136:1–13
Pickett STA, Cadenasso ML (1995) Landscape ecology: spatial heterogeneity in ecological systems. Science 269:331–340
Pidgeon AM, Radeloff VC, Mathews NE (2003) Landscape-scale patterns of black-throated sparrow (Amphispiza bilineata) abundance and nest success. Ecol Appl 13:530–542
Probst JR (1986) A review of factors limiting the Kirtland’s warbler on its breeding grounds. Am Midl Nat 116:87–100
Probst JR (1988) Kirtland’s warbler breeding biology and habitat management. In: Hoekstra JW, Capp J (eds) Integrating forest management for wildlife and fish. USDA Gen. Tech. Report NC-122. U.S. North Central Research Station, St. Paul, pp 28–35
Probst JR, DonnerWright DM (2003) Fire and shade effects on ground cover structure in Kirtland’s warbler habitat. Am Midl Nat 149:320–334
Probst JR, Hayes JP (1987) Pairing success of Kirtland’s warblers in marginal vs. suitable habitat. Auk 104:234–241
Probst JR, Weinrich J (1993) Relating Kirtland’s warbler population to changing landscape composition and structure. Landsc Ecol 8:257–271
Probst JR, Donner DM, Bocetti C, Sjogren S (2003) Population increase in Kirtland’s warbler and summer range expansion to Wisconsin and Michigan’s Upper Peninsula, USA. Oryx 37:365–373
Probst JR, Donner DM, Worland M, Weinrich J, Huber P, Ennis KR (2005) Comparing census methods for the endangered Kirtland’s warbler. J Field Ornithol 76:50–60
Pulliam HR (1988) Sources, sinks and population regulation. Am Nat 132:652–661
Pulliam HR, Danielson BJ (1991) Sources, sinks, and habitat selection: a landscape perspective on population dynamics. Am Nat 132:S50–S66
Pulliam HR, Dunning JB, Liu J (1992) Population dynamics in complex landscapes: a case study. Ecol Appl 2:165–177
Robinson SK, Thompson FR III, Donovan TM, Whitehead DR, Faaborg J (1995) Regional forest fragmentation and the nesting success of migratory birds. Science 267:1987–1990
Ryel LA (1981) The fourth decennial census of Kirtland’s warbler, 1981. Jack-Pine Warbler 59:93–95
Schmiegelow FKA, Machtans CS, Hannon SJ (1997) Are boreal birds resilient to forest fragmentation? An experimental study of short-term community responses. Ecology 78:1914–1932
Schrott GR, With KA, King AW (2005) Demographic limitations of the ability of habitat restoration to rescue declining populations. Conserv Biol 19:1181–1193
Thompson FR III, DeGraaf RM (2001) Conservation approaches for woody, early successional communities in the eastern United States. Wildl Soc Bull 29:483–494
Trzcinski MK, Fahrig L, Merriam G (1999) Independent effects of forest cover and fragmentation on the distribution of forest breeding birds. Ecol Appl 9:586–593
Villard M, Merriam G, Maurer BA (1995) Dynamics in subdivided populations of neotropical migratory birds in a fragmented temperate forest. Ecology 76:27–40
Walker WS, Barnes BV, Kashian DM (2003) Landscape ecosystems of the Mack Lake burn, northern Lower Michigan, and the occurrence of the Kirtland’s warbler. For Sci 49:119–139
Walkinshaw LH (1983) Kirtland’s warbler: the natural history of an endangered species. Cranbrook Inst Sci Bull, vol 58. Bloomfield Hill, Michigan
Wiens JA (1994) Habitat fragmentation: island v landscape perspectives on bird conservation. Ibis 137:S97–S104
With KA, Gardner RH, Turner MG (1997) Landscape connectivity and population distributions in heterogeneous environments. Oikos 78:151–169
Wood SN (2006) Generalized additive models: an introduction with R. Chapman and Hall/CRC, Florida
Acknowledgments
This study was supported by the U.S. Forest Service Northern Research Station. We thank Dean Anderson, Carol Bocetti, Dave Ewert, Eric Gustafson, Steven Sjogren, Jerry Weinrich, and two anonymous reviewers for providing valuable comments on the manuscript. We are also indebted to Elaine Carlson and Keith Kintigh with the Michigan Department of Natural Resources, and Phil Huber with the Huron-Manistee National Forest for their guidance and assistance with historical data.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Donner, D.M., Probst, J.R. & Ribic, C.A. Influence of habitat amount, arrangement, and use on population trend estimates of male Kirtland’s warblers. Landscape Ecol 23, 467–480 (2008). https://doi.org/10.1007/s10980-008-9208-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10980-008-9208-9